Oral Dexmedetomidine Achieves Superior Effects in Mitigating Emergence Agitation and Demonstrates Comparable Sedative Effects to Oral Midazolam for Pediatric Premedication: A Systematic Review and Meta-Analysis of Randomized Controlled Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol
2.2. Data and Literature Sources
2.3. Study Selection
2.4. Data Extraction
2.5. Assessment of Methodological Quality
2.6. Statistical Analysis
3. Results
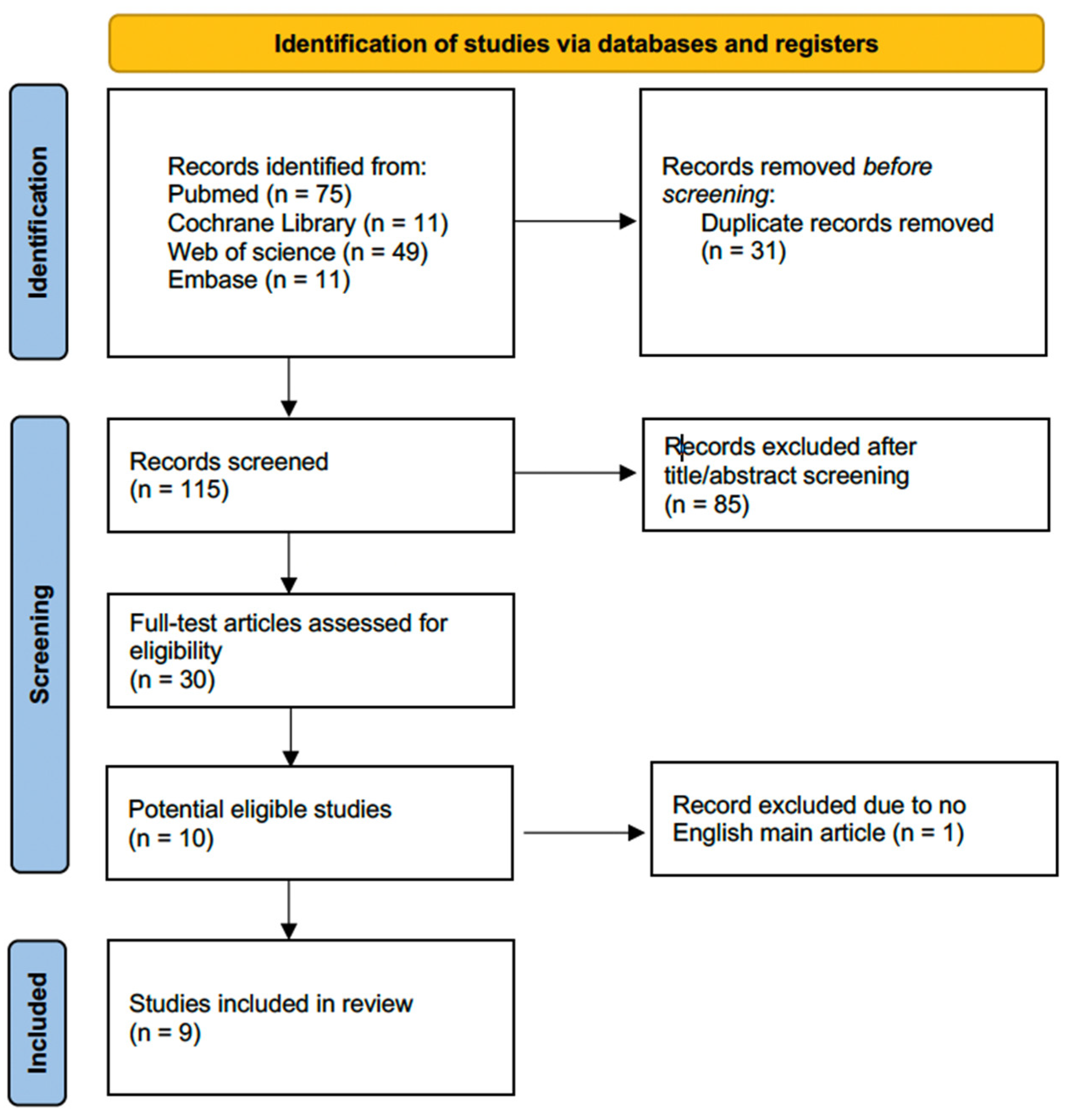
3.1. Study Selection
3.2. Study Characteristics and Patient Populations
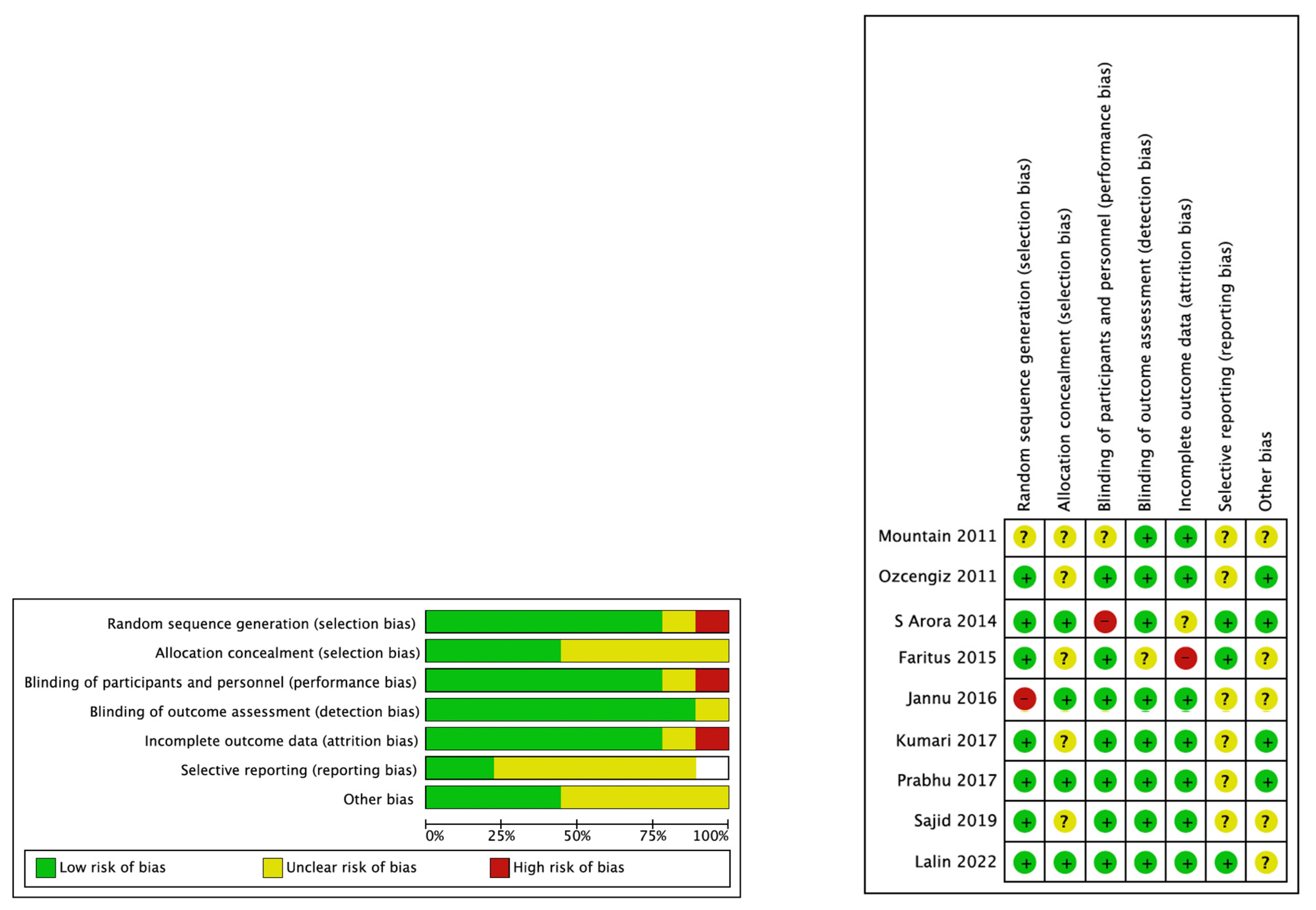
3.3. Quality of the Included Studies
3.4. Meta-Analysis Results
3.4.1. Incidence of Satisfactory Sedation
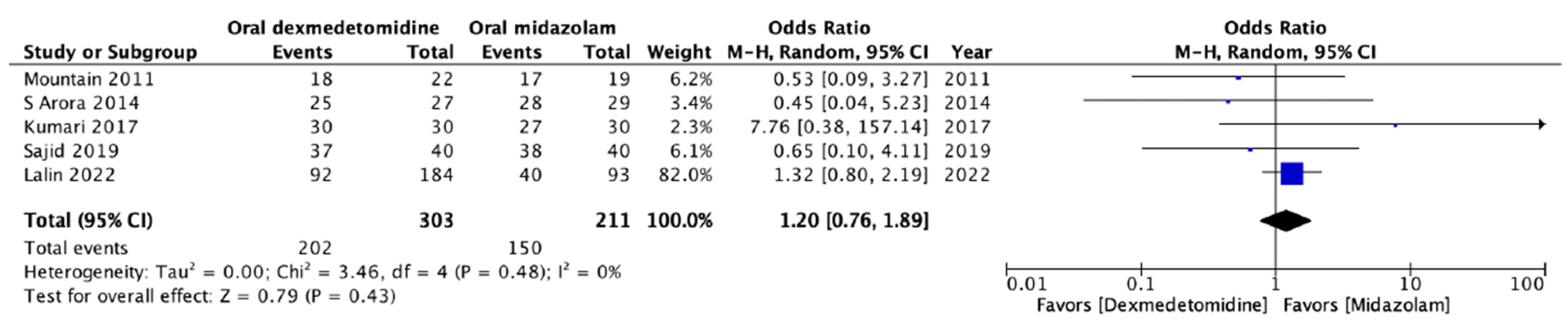
3.4.2. Incidence of Satisfactory Sedation during Parental Separation
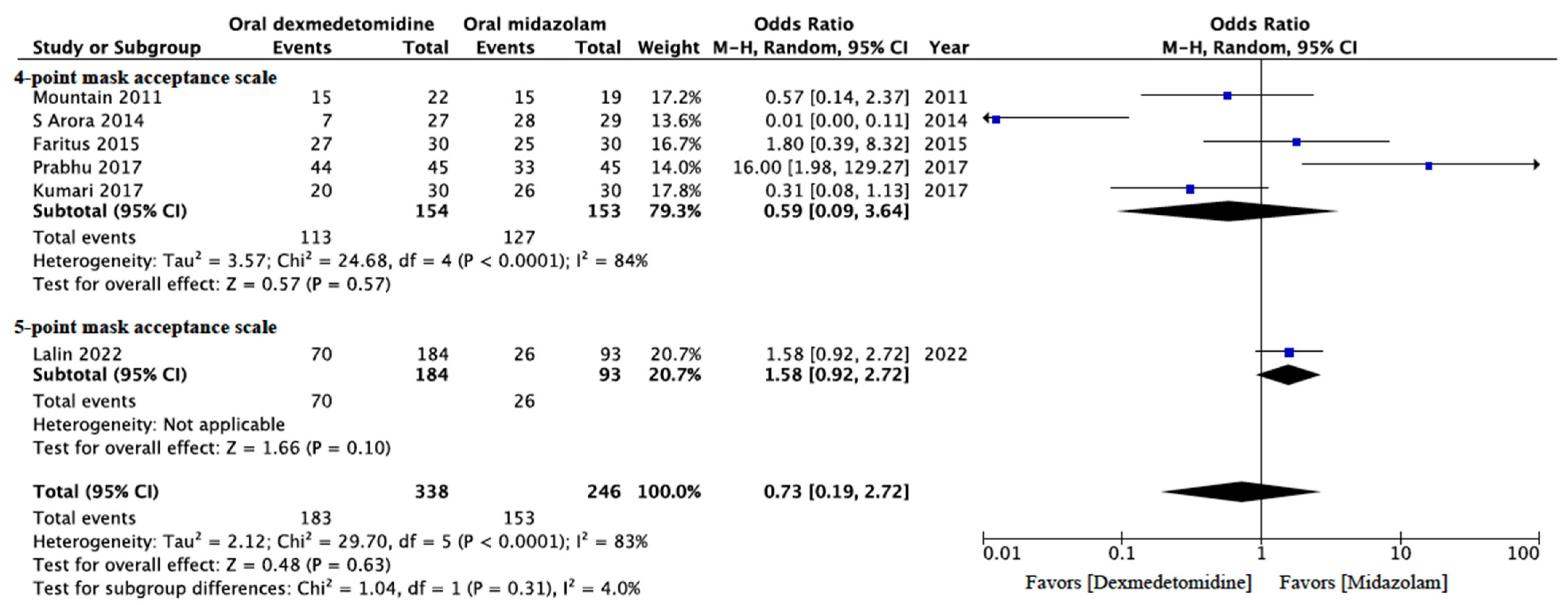
3.4.3. Incidence of Satisfactory Sedation during Mask Induction
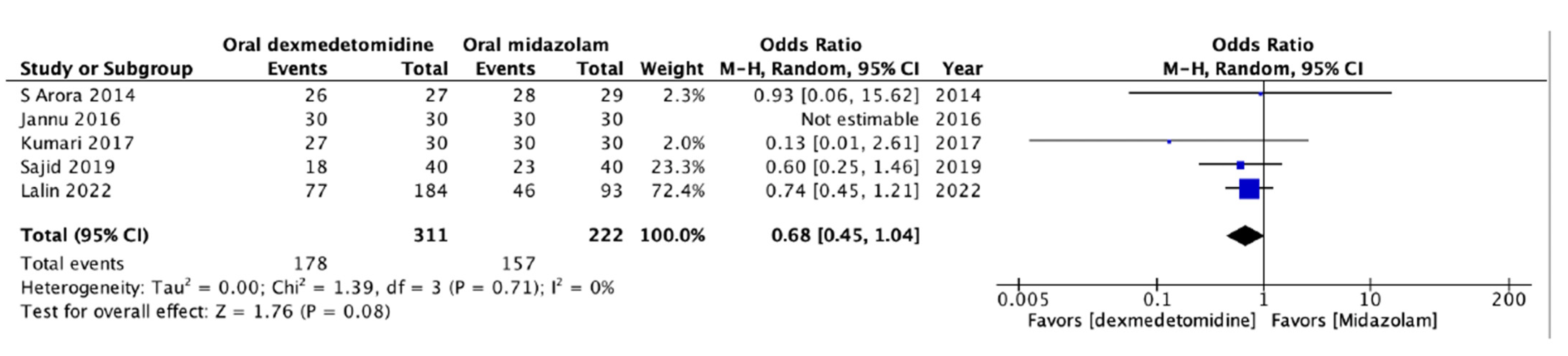
3.4.4. Incidence of Emergence Agitation
3.5. Adverse Effects
3.6. GRADE Findings
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kanaya, A. Emergence agitation in children: Risk factors, prevention, and treatment. J. Anesth. 2016, 30, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Dave, N.M. Premedication and Induction of Anaesthesia in paediatric patients. Indian J. Anaesth. 2019, 63, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, A.; Kain, Z.N.; Larsson, P.; Lönnqvist, P.; Wolf, A.R. The place of premedication in pediatric practice. Paediatr. Anesth. 2009, 19, 817–828. [Google Scholar] [CrossRef]
- Abdallah, C.; Hannallah, R. Premedication of the child undergoing surgery. Middle East J. Anaesthesiol. 2011, 21, 165–174. [Google Scholar] [PubMed]
- Mamuda, A.; Gudaji, M.I. Effect of oral midazolam in preventing postoperative emergence agitation in children. Sahel Med. J. 2019, 22, 1–7. [Google Scholar] [CrossRef]
- Nair, S.; Wolf, A. Emergence delirium after paediatric anaesthesia: New strategies in avoidance and treatment. BJA Educ. 2018, 18, 30–33. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Latif, M.S.; Putland, A.J.; McCluskey, A.; Meadows, D.P.; Remington, S.A.M. Oral midazolam premedication for day case breast surgery, a randomised prospective double-blind placebo-controlled study. Anaesthesia 2001, 56, 990–994. [Google Scholar] [PubMed]
- Hall, J.E.; Uhrich, T.D.; Barney, J.A.; Arain, S.R.; Ebert, T.J. Sedative, amnestic, and analgesic properties of small-dose dexmedetomidine infusions. Anesth. Analg. 2000, 90, 699–705. [Google Scholar] [CrossRef]
- Khan, Z.P.; Ferguson, C.N.; Jones, R.M. alpha-2 and imidazoline receptor agonists. Their pharmacology and therapeutic role. Anaesthesia 1999, 54, 146–165. [Google Scholar] [CrossRef]
- Weerink, M.A.S.; Struys, M.M.R.F.; Hannivoort, L.N.; Barends, C.R.M.; Absalom, A.R.; Colin, P. Clinical Pharmacokinetics and Pharmacodynamics of Dexmedetomidine. Clin. Pharmacokinet. 2017, 56, 893–913. [Google Scholar] [CrossRef]
- Anttila, M.; Penttilä, J.; Helminen, A.; Vuorilehto, L.; Scheinin, H. Bioavailability of dexmedetomidine after extravascular doses in healthy subjects. Br. J. Clin. Pharmacol. 2003, 56, 691–693. [Google Scholar] [CrossRef]
- Peng, K.; Wu, S.-R.; Ji, F.-H.; Li, J. Premedication with dexmedetomidine in pediatric patients: A systematic review and meta-analysis. Clinics 2014, 69, 777–786. [Google Scholar] [CrossRef]
- Arora, S.; Saini, K.; Bhardwaj, N. A comparative evaluation of midazolam, clonidine and dexmedetomidine as oral premedicants in children: A double blind randomized clinical trial. Anaesth. Pain Intensive Care 2014, 18, 355–360. [Google Scholar]
- Jannu, V.; Mane, R.; Dhorigol, M.; Sanikop, C. A comparison of oral midazolam and oral dexmedetomidine as premedication in pediatric anesthesia. Saudi J. Anaesth. 2016, 10, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, N.; Kumari, S.; Usha, G.; Talwar, V.; Gupta, P. Comparison of Oral Clonidine, Oral Dexmedetomidine, and Oral Midazolam for Premedication in Pediatric Patients Undergoing Elective Surgery. Anesth. Essays Res. 2017, 11, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, T.; Sajid, B.; Jumaila, M. A comparison of oral dexmedetomidine and oral midazolam as premedicants in children. J. Anaesthesiol. Clin. Pharmacol. 2019, 35, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Lalin, D.; Singh, S.; Thakur, V. A comparison of midazolam, dexmedetomidine 2μg/kg and dexmedetomidine 4μg/kg as oral premedication in children, a randomized double-blinded clinical triall. Acta Anaesthesiol. Belg. 2022, 73, 45–51. [Google Scholar] [CrossRef]
- Lee, S.-J.; Sung, T.-Y. Emergence agitation: Current knowledge and unresolved questions. Korean J. Anesthesiol. 2020, 73, 471–485. [Google Scholar] [CrossRef]
- Prabhu, M.K.; Mehandale, S.G. Comparison of oral dexmedetomidine versus oral midazolam as premedication to prevent emergence agitation after sevoflurane anaesthesia in paediatric patients. Indian J. Anaesth. 2017, 61, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Özcengiz, D.; Gunes, Y.; Ozmete, O. Oral melatonin, dexmedetomidine, and midazolam for prevention of postoperative agitation in children. J. Anesth. 2011, 25, 184–188. [Google Scholar] [CrossRef]
- Mountain, B.W.; Smithson, L.; Cramolini, M.; Wyatt, T.H.; Newman, M. Dexmedetomidine as a pediatric anesthetic premedication to reduce anxiety and to deter emergence delirium. AANA J. 2011, 79, 219–224. [Google Scholar]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; DeBeer, H.; et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64, 383–394. [Google Scholar] [CrossRef] [PubMed]
- Faritus, S.Z.; Khazaee-Koohpar, M.; Ziyaeifard, M.; Mehrabanian, M.J. Oral dexmedetomidine versus midazolam as anesthetic premedication in children undergoing congenital heart surgery. Anesthesiol. Pain Med. 2015, 5, e25032. [Google Scholar] [CrossRef] [PubMed]
- Coté, C.J.; Wilson, S. Guidelines for Monitoring and Management of Pediatric Patients Before, During, and After Sedation for Diagnostic and Therapeutic Procedures: Update 2016. Pediatrics 2016, 138, e20161212. [Google Scholar] [CrossRef] [PubMed]
- Singh, C.; Pandey, R.K.; Saksena, A.K.; Chandra, G. A comparative evaluation of analgo-sedative effects of oral dexmedetomidine and ketamine: A triple-blind, randomized study. Paediatr. Anaesth. 2014, 24, 1252–1259. [Google Scholar] [CrossRef]
- Ng, K.T.; Sarode, D.; Lai, Y.S.; Teoh, W.Y.; Wang, C.Y. The effect of ketamine on emergence agitation in children: A systematic review and meta-analysis. Pediatr Anesth. 2019, 29, 1163–1172. [Google Scholar] [CrossRef] [PubMed]
- Rao, Y.; Zeng, R.; Jiang, X.; Li, J.; Wang, X. The Effect of Dexmedetomidine on Emergence Agitation or Delirium in Children After Anesthesia—A Systematic Review and Meta-Analysis of Clinical Studies. Front. Pediatr. 2020, 8, 329. [Google Scholar] [CrossRef]
- Sahyoun, C.; Krauss, B. Clinical implications of pharmacokinetics and pharmacodynamics of procedural sedation agents in children. Curr. Opin. Pediatr. 2012, 24, 225–232. [Google Scholar] [CrossRef]
- Iirola, T.; Ihmsen, H.; Laitio, R.; Kentala, E.; Aantaa, R.; Kurvinen, J.-P.; Scheinin, M.; Schwilden, H.; Schüttler, J.; Olkkola, K. Population pharmacokinetics of dexmedetomidine during long-term sedation in intensive care patients. Br. J. Anaesth. 2012, 108, 460–468. [Google Scholar] [CrossRef]
- Sanfilippo, F.L.V.L.; La Via, L.; Tigano, S.; Morgana, A.; La Rosa, V.; Astuto, M. Trial Sequential Analysis: The evaluation of the robustness of meta-analyses findings and the need for further research. EuroMediterranean Biomed. J. 2021, 16, 104–107. [Google Scholar]
- De Cassai, A.; Pasin, L.; Boscolo, A.; Salvagno, M.; Navalesi, P. Trial sequential analysis: Plain and simple. Korean J. Anesthesiol. 2021, 74, 363–365. [Google Scholar] [CrossRef]

| Study * | Country | Jadad Score | n | Dexmedetom-idine (Dose) | Midazolam (Dose) | Outcomes | Age (Years) | Type of Surgery | ASA | Anesthesia | Timing of Premedication |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mountain (2011) [21] | United States | 5 | 41 | 4 μg/kg | 0.5 mg/kg | Parental separation anxiety scale (PSAS) a Mask acceptance scale (MAS) b Pediatric anesthesia emergence delirium scale (PAEDS) c | 1–6 | Dental restoration procedure | I, II | Sevoflurane, O2, and N2O | 30 min |
| Ozcengiz (2011) [20] | Turkey | 5 | 100 | 2.5 μg/kg | 0.5 mg/kg | Hemodynamic variables Emergence agitation scale (EAS) d | 3–9 | Oesophagealdilatation procedure | I, II | Sevoflurane 8%, N2O 50%, and O2 | 40–45 min |
| Arora (2014) [13] | India | 5 | 85 | 4 μg/kg | 0.5 mg/kg | 4-point sedation scale e Parental separation anxiety scale (PSAS) a Mask acceptance scale (MAS) b | 1–4 | Elective urogenital procedure | I, II | Sevoflurane followed by isoflurane | 30 min for midazolam 60 min for dexmedetomi-dine |
| Faritus (2015) [25] | Iran | 4 | 60 | 2 μg/kg | 0.5 mg/kg | Ramsay sedation scale (not included because of unclear data analysis) Mask acceptance scale (MAS) b | 2–12 | Congenital heart surgery | III | Sevoflurane | 45 min |
| Jannu (2016) [14] | India | 5 | 60 | 4 μg/kg | 0.75 mg/kg | 4-point sedation scale f Parental separation anxiety scale (not included due to insufficient data) 5-point mask acceptance scale g (not included due to insufficient data) Emergence agitation h | 1–7 | Elective minor lowerabdominal surgery | I, II | Sevoflurane, O2, and N2O | 40 min |
| Kumari (2017) [15] | India | 5 | 90 | 4 μg/kg | 0.5 mg/kg | 3-point sedation scale i Parental separation anxiety scale (PSAS) a Mask acceptance scale (MAS) b | 4–12 | Elective ophthalmic surgery | I,II | Halothane | 30 min |
| Prabhu (2017) [19] | India | 5 | 90 | 4 μg/kg | 0.5 mg/kg | Mask acceptance scale (MAS) b Emergence agitation (PAEDS) a | 1–10 | Elective surgery | I,II | Sevoflurane | 45 min |
| Sajid (2019) [16] | India | 5 | 80 | 4 μg/kg | 0.5 mg/kg | 5-point sedation scale j Parental separation anxiety scale (PSAS) a Intravenous acceptability score Pediatric anesthesia emergence delirium scale (PAEDS) c | 1–6 | Elective herniotomy | I | Isoflurane | 40 min |
| Lalin (2022) [17] | India | 5 | 279 | 2 or 4 μg/kg | 0.5 mg/kg | 4-point sedation scale f Parental separation anxiety scale (PSAS) a 5-point mask acceptance scale g Postoperative agitation score (not included due to insufficient data) | 1–7 | Pyeloplasty, hernia surgery, urethral surgery, ureteral reimplant, hypospadias | I,II | Sevoflurane +/− caudal block | 45 min |
| Certainty Assessment | Number of Patients | Effect | Certainty | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Number of Studies | Study Design | Risk of Bias | Inconsistency | Indirectness | Impression | Dexmedetomidine | Midazolam | Odds Ratio (95% CI) | |
| Incidence of satisfactory sedation | |||||||||
| 5 | RCT | not serious | not serious | not serious | not serious | 311 | 222 | 0.68 (0.45–1.04) | High |
| Incidence of satisfactory sedation during parent separation | |||||||||
| 5 | RCT | not serious | not serious | not serious | not serious | 303 | 211 | 1.20 (0.76–1.89) | High |
| Incidence of satisfactory sedation during mask acceptance | |||||||||
| 6 | RCT | not serious | serious a | not serious | not serious | 338 | 246 | 0.73 (0.19–2.72) | Moderate |
| Incidence of emergence agitation | |||||||||
| 5 | RCT | not serious | seriou a | not serious | not serious | 162 | 159 | 0.16 (0.06–0.44) | Moderate |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jen, C.-K.; Lu, K.-C.; Chen, K.-W.; Lu, Y.-R.; Huang, I.-T.; Huang, Y.-C.; Huang, C.-J. Oral Dexmedetomidine Achieves Superior Effects in Mitigating Emergence Agitation and Demonstrates Comparable Sedative Effects to Oral Midazolam for Pediatric Premedication: A Systematic Review and Meta-Analysis of Randomized Controlled Studies. J. Clin. Med. 2024, 13, 1174. https://doi.org/10.3390/jcm13041174
Jen C-K, Lu K-C, Chen K-W, Lu Y-R, Huang I-T, Huang Y-C, Huang C-J. Oral Dexmedetomidine Achieves Superior Effects in Mitigating Emergence Agitation and Demonstrates Comparable Sedative Effects to Oral Midazolam for Pediatric Premedication: A Systematic Review and Meta-Analysis of Randomized Controlled Studies. Journal of Clinical Medicine. 2024; 13(4):1174. https://doi.org/10.3390/jcm13041174
Chicago/Turabian StyleJen, Chun-Kai, Kuo-Ching Lu, Kuan-Wen Chen, Yun-Ru Lu, I-Tao Huang, Yu-Chen Huang, and Chun-Jen Huang. 2024. "Oral Dexmedetomidine Achieves Superior Effects in Mitigating Emergence Agitation and Demonstrates Comparable Sedative Effects to Oral Midazolam for Pediatric Premedication: A Systematic Review and Meta-Analysis of Randomized Controlled Studies" Journal of Clinical Medicine 13, no. 4: 1174. https://doi.org/10.3390/jcm13041174
APA StyleJen, C.-K., Lu, K.-C., Chen, K.-W., Lu, Y.-R., Huang, I.-T., Huang, Y.-C., & Huang, C.-J. (2024). Oral Dexmedetomidine Achieves Superior Effects in Mitigating Emergence Agitation and Demonstrates Comparable Sedative Effects to Oral Midazolam for Pediatric Premedication: A Systematic Review and Meta-Analysis of Randomized Controlled Studies. Journal of Clinical Medicine, 13(4), 1174. https://doi.org/10.3390/jcm13041174






