Treatment Sequencing and Independent Outcomes of First- and Second-Line Chemotherapy in a Retrospective Series of Patients with Biliary Tract Cancer
Abstract
1. Introduction
2. Methods
3. Results
3.1. mOS According to Treatment Sequencing
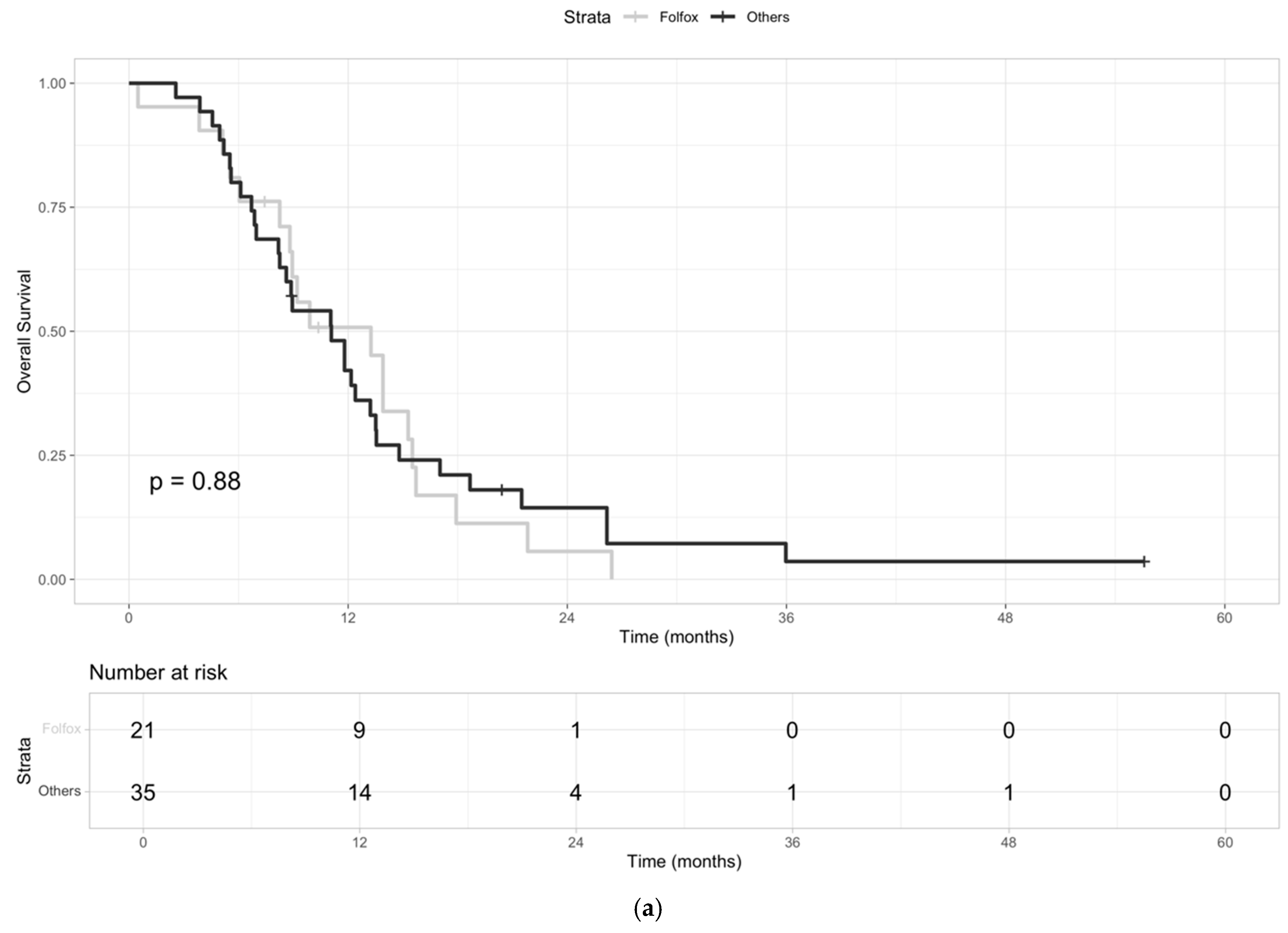
3.2. mOS and mPFS According to First-Line Treatment
3.3. mOS and mPFS According to Second-Line Treatment
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BTC= biliary tract cancers |
| CTX = chemotherapy |
| GBC = gallbladder cancer |
| GemCis = Gemcitabine/Cisplatin |
| GemOx = Gemcitabine/Oxaliplatin |
| iCCA = intrahepatic cholagiocarcinoma |
| pCCA = perihilar cholagiocarcinoma |
| dCCA = distal cholagiocarcinoma |
| KM = Kaplan–Meier |
| mOS = median overall survival |
| mPFS = median progression-free survival |
| mo = months |
| RT = radiotherapy |
| vs = versus |
References
- Van Dyke, A.L.; Shiels, M.S.; Jones, G.S.; Pfeiffer, R.M.; Petrick, J.L.; Beebe-Dimmer, J.L.; Koshiol, J. Biliary Tract Cancer Incidence and Trends in the United States by Demographic Group, 1999–2013. Cancer 2019, 125, 1489–1498. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Tavolari, S.; Brandi, G. Cholangiocarcinoma: Epidemiology and Risk Factors. Liver Int. 2019, 39, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Tariq, N.-A.; McNamara, M.G.; Valle, J.W. Biliary Tract Cancers: Current Knowledge, Clinical Candidates and Future Challenges. Cancer Manag. Res. 2019, 11, 2623–2642. [Google Scholar] [CrossRef]
- Cillo, U.; Fondevila, C.; Donadon, M.; Gringeri, E.; Mocchegiani, F.; Schlitt, H.J.; Ijzermans, J.N.M.; Vivarelli, M.; Zieniewicz, K.; Olde Damink, S.W.M.; et al. Surgery for Cholangiocarcinoma. Liver Int. 2019, 39, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Doherty, B.; Nambudiri, V.E.; Palmer, W.C. Update on the Diagnosis and Treatment of Cholangiocarcinoma. Curr. Gastroenterol. Rep. 2017, 19, 2. [Google Scholar] [CrossRef]
- Valle, J.; Wasan, H.; Palmer, D.H.; Cunningham, D.; Anthoney, A.; Maraveyas, A.; Madhusudan, S.; Iveson, T.; Hughes, S.; Pereira, S.P.; et al. Cisplatin plus Gemcitabine versus Gemcitabine for Biliary Tract Cancer. N. Engl. J. Med. 2010, 362, 1273–1281. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.-Y.; Ruth, H.A.; Qin, S.; Chen, L.-T.; Okusaka, T.; Vogel, A.; Kim, J.W.; Suksombooncharoen, T.; Ah, L.M.; Kitano, M.; et al. Durvalumab plus Gemcitabine and Cisplatin in Advanced Biliary Tract Cancer. NEJM Evid. 2022, 1, EVIDoa2200015. [Google Scholar] [CrossRef]
- Kelley, R.K.; Ueno, M.; Yoo, C.; Finn, R.S.; Furuse, J.; Ren, Z.; Yau, T.; Klümpen, H.-J.; Chan, S.L.; Ozaka, M.; et al. Pembrolizumab in Combination with Gemcitabine and Cisplatin Compared with Gemcitabine and Cisplatin Alone for Patients with Advanced Biliary Tract Cancer (KEYNOTE-966): A Randomised, Double-Blind, Placebo-Controlled, Phase 3 Trial. Lancet 2023, 401, 1853–1865. [Google Scholar] [CrossRef] [PubMed]
- Lamarca, A.; Palmer, D.H.; Wasan, H.S.; Ross, P.J.; Ma, Y.T.; Arora, A.; Falk, S.; Gillmore, R.; Wadsley, J.; Patel, K.; et al. Second-Line FOLFOX Chemotherapy versus Active Symptom Control for Advanced Biliary Tract Cancer (ABC-06): A Phase 3, Open-Label, Randomised, Controlled Trial. Lancet Oncol. 2021, 22, 690–701. [Google Scholar] [CrossRef] [PubMed]
- Javle, M.; Borad, M.J.; Azad, N.S.; Kurzrock, R.; Abou-Alfa, G.K.; George, B.; Hainsworth, J.; Meric-Bernstam, F.; Swanton, C.; Sweeney, C.J.; et al. Pertuzumab and Trastuzumab for HER2-Positive, Metastatic Biliary Tract Cancer (MyPathway): A Multicentre, Open-Label, Phase 2a, Multiple Basket Study. Lancet Oncol. 2021, 22, 1290–1300. [Google Scholar] [CrossRef] [PubMed]
- Subbiah, V.; Lassen, U.; Élez, E.; Italiano, A.; Curigliano, G.; Javle, M.; de Braud, F.; Prager, G.W.; Greil, R.; Stein, A.; et al. Dabrafenib plus Trametinib in Patients with BRAFV600E-Mutated Biliary Tract Cancer (ROAR): A Phase 2, Open-Label, Single-Arm, Multicentre Basket Trial. Lancet Oncol. 2020, 21, 1234–1243. [Google Scholar] [CrossRef] [PubMed]
- Abou-Alfa, G.K.; Sahai, V.; Hollebecque, A.; Vaccaro, G.; Melisi, D.; Al-Rajabi, R.; Paulson, A.S.; Borad, M.J.; Gallinson, D.; Murphy, A.G.; et al. Pemigatinib for Previously Treated, Locally Advanced or Metastatic Cholangiocarcinoma: A Multicentre, Open-Label, Phase 2 Study. Lancet Oncol. 2020, 21, 671–684. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Macarulla, T.; Javle, M.M.; Kelley, R.K.; Lubner, S.J.; Adeva, J.; Cleary, J.M.; Catenacci, D.V.; Borad, M.J.; Bridgewater, J.; et al. Ivosidenib in IDH1-Mutant, Chemotherapy-Refractory Cholangiocarcinoma (ClarIDHy): A Multicentre, Randomised, Double-Blind, Placebo-Controlled, Phase 3 Study. Lancet Oncol. 2020, 21, 796–807. [Google Scholar] [CrossRef] [PubMed]
- Palloni, A.; Bisello, S.; Maggio, I.; Massucci, M.; Galuppi, A.; Di Federico, A.; Rizzo, A.; Ricci, A.D.; Siepe, G.; Morganti, A.G.; et al. The Potential Role of Adjuvant Chemoradiotherapy in Patients with Microscopically Positive (R1) Surgical Margins after Resection of Cholangiocarcinoma. Curr. Oncol. 2023, 30, 4754–4766. [Google Scholar] [CrossRef] [PubMed]
- Amit, U.; Shagun, M.; Plastaras, J.P.; Metz, J.M.; Karasic, T.B.; Lubas, M.J.; Ben-Josef, E. Clinical Outcomes and Risk Stratification in Unresectable Biliary Tract Cancers Undergoing Radiation Therapy. Radiat. Oncol. 2024, 19, 102. [Google Scholar] [CrossRef] [PubMed]
- Okusaka, T.; Nakachi, K.; Fukutomi, A.; Mizuno, N.; Ohkawa, S.; Funakoshi, A.; Nagino, M.; Kondo, S.; Nagaoka, S.; Funai, J.; et al. Gemcitabine Alone or in Combination with Cisplatin in Patients with Biliary Tract Cancer: A Comparative Multicentre Study in Japan. Br. J. Cancer 2010, 103, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Choi, I.S.; Kim, K.H.; Lee, J.H.; Suh, K.J.; Kim, J.-W.; Park, J.H.; Kim, Y.J.; Kim, J.-S.; Kim, J.H.; Kim, J.W. A Randomised Phase II Study of Oxaliplatin/5-FU (mFOLFOX) versus Irinotecan/5-FU (mFOLFIRI) Chemotherapy in Locally Advanced or Metastatic Biliary Tract Cancer Refractory to First-Line Gemcitabine/Cisplatin Chemotherapy. Eur. J. Cancer 2021, 154, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Neuzillet, C.; Casadei-Gardini, A.; Brieau, B.; Vivaldi, C.; Brandi, G.; Tougeron, D.; Filippi, R.; Vienot, A.; Silvestris, N.; Pointet, A.-L.; et al. Fluropyrimidine Single Agent or Doublet Chemotherapy as Second Line Treatment in Advanced Biliary Tract Cancer. Int. J. Cancer 2020, 147, 3177–3188. [Google Scholar] [CrossRef] [PubMed]
- Hyung, J.; Kim, I.; Kim, K.-P.; Ryoo, B.-Y.; Jeong, J.H.; Kang, M.J.; Cheon, J.; Kang, B.W.; Ryu, H.; Lee, J.S.; et al. Treatment with Liposomal Irinotecan Plus Fluorouracil and Leucovorin for Patients with Previously Treated Metastatic Biliary Tract Cancer: The Phase 2b NIFTY Randomized Clinical Trial. JAMA Oncol. 2023, 9, 692–699. [Google Scholar] [CrossRef] [PubMed]




| Standard group First line: Gemcitabine/Cisplatin Second line: mFOLFOX | n 20 (35.7%) |
| Age | Median 60 years (42–74) |
| Sex | Male = 11 (55%) |
| Female = 9 (45%) | |
| Control group All other treatment sequencing in first line and second line | n 36 (64.3%) |
| Age | Median 62 years (29–78) |
| Sex | Male = 19 (52.8%) |
| Female = 17 (47.2%) | |
| First line | |
| Gemcitabine/Cisplatin | n 10 (27.8%) |
| Gemcitabine/Oxaliplatin | n 25 (69.4%) |
| mFolfox | n 1 (2.8%) |
| Second line | |
| Gemcitabine/Capecitabine | n 16 (44.4%) |
| mFOLFIRI | n 6 (16.7%) |
| Gemcitabine | n 6 (16.7%) |
| Capecitabine | n 2 (5.6%) |
| TKI anti-FGFR | n 2 (5.6%) |
| Sorafenib | n 1 (2.8%) |
| mAb anti-HER2 | n 1 (2.8%) |
| Carboplatin/Gemcitabine | n 1 (2.8%) |
| mFOLFOX | n 1 (2.8%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frega, G.; Palloni, A.; Deiana, C.; Rizzo, A.; Ricci, A.D.; Brandi, G. Treatment Sequencing and Independent Outcomes of First- and Second-Line Chemotherapy in a Retrospective Series of Patients with Biliary Tract Cancer. J. Clin. Med. 2024, 13, 7262. https://doi.org/10.3390/jcm13237262
Frega G, Palloni A, Deiana C, Rizzo A, Ricci AD, Brandi G. Treatment Sequencing and Independent Outcomes of First- and Second-Line Chemotherapy in a Retrospective Series of Patients with Biliary Tract Cancer. Journal of Clinical Medicine. 2024; 13(23):7262. https://doi.org/10.3390/jcm13237262
Chicago/Turabian StyleFrega, Giorgio, Andrea Palloni, Chiara Deiana, Alessandro Rizzo, Angela Dalia Ricci, and Giovanni Brandi. 2024. "Treatment Sequencing and Independent Outcomes of First- and Second-Line Chemotherapy in a Retrospective Series of Patients with Biliary Tract Cancer" Journal of Clinical Medicine 13, no. 23: 7262. https://doi.org/10.3390/jcm13237262
APA StyleFrega, G., Palloni, A., Deiana, C., Rizzo, A., Ricci, A. D., & Brandi, G. (2024). Treatment Sequencing and Independent Outcomes of First- and Second-Line Chemotherapy in a Retrospective Series of Patients with Biliary Tract Cancer. Journal of Clinical Medicine, 13(23), 7262. https://doi.org/10.3390/jcm13237262






