Epigenetic Biomarkers as a New Diagnostic Tool in Bladder Cancer—From Early Detection to Prognosis
Abstract
1. Introduction
1.1. Epidemiology
1.2. Etiology
1.3. Treatment
1.4. Prognosis
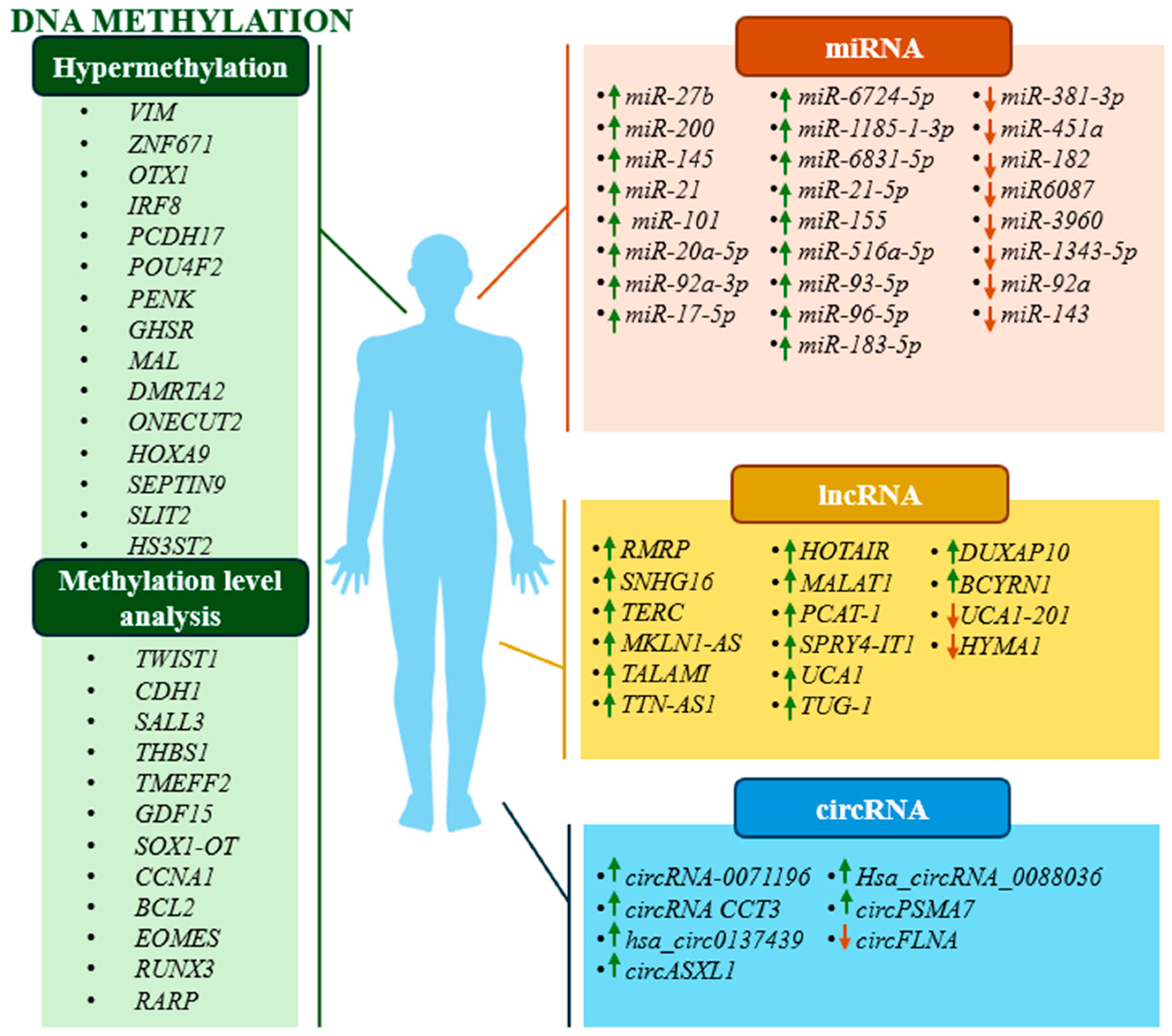
2. Methodology
3. Diagnosis of Bladder Cancer
3.1. Classic Approach
3.2. Epigenetic Biomarkers in the Diagnosis of BC
3.3. Available Tests Based on Analysis of Epigenetic Changes for BC Diagnosis
3.4. Epigenetic Biomarkers in BC—Cohort Studies
3.5. Potential Epigenetic Biomarkers in BC (Bioinformatic Analyses and Cell Line Studies)
4. Epigenetic Biomarkers in Clinical Prognosis of BC
4.1. DNA Methylation Biomarkers—Cohort Studies
4.2. ncRNA Biomarkers—Cohort Studies
5. Strength and Limitations
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2024, 74, 229–263. [Google Scholar] [CrossRef] [PubMed]
- Freedman, N.D.; Silverman, D.T.; Hollenbeck, A.R.; Schatzkin, A.; Abnet, C.C. Association between smoking and risk of bladder cancer among men and women. JAMA 2011, 306, 737–745. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer. List of Classifications by Cancer Sites with Sufficient or Limited Evidence in Humans. IARC Monographs Volumes 1–136a. Available online: https://monographs.iarc.who.int/wp-content/uploads/2019/07/Classifications_by_cancer_site.pdf (accessed on 5 July 2024).
- Dobruch, J.; Oszczudłowski, M. Bladder cancer: Current challenges and future directions. Medicina 2021, 57, 749. [Google Scholar] [CrossRef] [PubMed]
- Henning, G.M.; Barashi, N.S.; Smith, Z.L. Advances in biomarkers for detection, surveillance, and prognosis of bladder cancer. Clin. Genitourin. Cancer 2021, 19, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Kluth, L.A.; Black, P.C.; Bochner, B.H.; Catto, J.; Lerner, S.P.; Stenzl, A.; Sylvester, R.; Vickers, A.J.; Xylinas, E.; Shariat, S.F. Prognostic and prediction tools in bladder cancer: A comprehensive review of the literature. Eur. Urol. 2015, 68, 238–253. [Google Scholar] [CrossRef]
- Gilyazova, I.; Enikeeva, K.; Rafikova, G.; Kagirova, E.; Sharifyanova, Y.; Asadullina, D.; Pavlov, V. Epigenetic and Immunological Features of Bladder Cancer. Int. J. Mol. Sci. 2023, 24, 9854. [Google Scholar] [CrossRef]
- Lobo, N.; Afferi, L.; Moschini, M.; Mostafid, H.; Porten, S.; Psutka, S.P.; Gupta, S.; Smith, A.B.; Williams, S.B.; Lotan, Y. Epidemiology, screening, and prevention of bladder cancer. Eur. Urol. Oncol. 2022, 5, 628–639. [Google Scholar] [CrossRef]
- Dyrskjøt, L.; Hansel, D.E.; Efstathiou, J.A.; Knowles, M.A.; Galsky, M.D.; Teoh, J.; Theodorescu, D. Bladder cancer. Nat. Rev. Dis. Primers 2023, 9, 58. [Google Scholar] [CrossRef]
- Hurst, C.D.; Alder, O.; Platt, F.M.; Droop, A.; Stead, L.F.; Burns, J.E.; Burghel, G.J.; Jain, S.; Klimczak, L.J.; Lindsay, H.; et al. Genomic subtypes of non-invasive bladder cancer with distinct metabolic profile and female gender bias in KDM6A mutation frequency. Cancer Cell 2017, 32, 701–715.e7. [Google Scholar] [CrossRef]
- Ségal-Bendirdjian, E.; Geli, V. Non-canonical roles of telomerase: Unraveling the imbroglio. Front. Cell Dev. Biol. 2019, 7, 332. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, N.; Rinaldetti, S.; Cheikh, B.B.; Zhou, Q.; Hass, E.P.; Jones, R.T.; Joshi, M.; LaBarbera, D.V.; Knott, S.R.V.; Cech, T.R.; et al. TRIM28 is a transcriptional activator of the mutant TERT promoter in human bladder cancer. Proc. Natl. Acad. Sci. USA 2021, 118, e2102423118. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, E.; L’hôte, C.G.; Kennedy, W.; Tomlinson, D.C.; Knowles, M.A. Mutant fibroblast growth factor receptor 3 induces intracellular signaling and cellular transformation in a cell type-and mutation-specific manner. Oncogene 2009, 28, 4306–4316. [Google Scholar] [CrossRef] [PubMed]
- Hurst, C.D.; Cheng, G.; Platt, F.M.; Castro, M.A.; Marzouka, N.-A.S.; Eriksson, P.; Black, E.V.; Alder, O.; Lawson, A.R.; Lindskrog, S.V.; et al. Stage-stratified molecular profiling of non-muscle-invasive bladder cancer enhances biological, clinical, and therapeutic insight. Cell Rep. Med. 2021, 2, 100472. [Google Scholar] [CrossRef] [PubMed]
- Alexandrov, L.B.; Nik-Zainal, S.; Wedge, D.C.; Aparicio, S.A.; Behjati, S.; Biankin, A.V.; Bignell, G.R.; Bolli, N.; Borg, A.; Borresen-Dale, A.L.; et al. Erratum: Signatures of mutational processes in human cancer (Nature (2013) 500 (415–421). Nature 2013, 502, 258. [Google Scholar] [CrossRef]
- Robertson, A.G.; Kim, J.; Al-Ahmadie, H.; Bellmunt, J.; Guo, G.; Cherniack, A.D.; Hinoue, T.; Laird, P.W.; Hoadley, K.A.; Akbani, R.; et al. Comprehensive molecular characterization of muscle-invasive bladder cancer. Cell 2017, 171, 540–556.e25. [Google Scholar] [CrossRef]
- Li, Q.; Damish, A.W.; Frazier, Z.; Liu, D.; Reznichenko, E.; Kamburov, A.; Bell, A.; Zhao, H.; Jordan, E.J.; Gao, S.P.; et al. ERCC2 helicase domain mutations confer nucleotide excision repair deficiency and drive cisplatin sensitivity in muscle-invasive bladder cancer. Clin. Cancer Res. 2019, 25, 977–988. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.V.; Hurst, C.D.; Knowles, M.A. Oncogenic FGFR3 gene fusions in bladder cancer. Hum. Mol. Genet. 2013, 22, 795–803. [Google Scholar] [CrossRef]
- Huan, J.; Grivas, P.; Birch, J.; Hansel, D.E. Emerging roles for mammalian target of rapamycin (mTOR) complexes in bladder cancer progression and therapy. Cancers 2022, 14, 1555. [Google Scholar] [CrossRef] [PubMed]
- Goriki, A.; Seiler, R.; Wyatt, A.W.; Contreras-Sanz, A.; Bhat, A.; Matsubara, A.; Hayashi, T.; Black, P.C. Unravelling disparate roles of NOTCH in bladder cancer. Nat. Rev. Urol. 2018, 15, 345–357. [Google Scholar] [CrossRef]
- Li, S.; Xin, K.; Pan, S.; Wang, Y.; Zheng, J.; Li, Z.; Liu, X.; Liu, B.; Xu, Z.; Chen, X. Blood-based liquid biopsy: Insights into early detection, prediction, and treatment monitoring of bladder cancer. Cell. Mol. Biol. Lett. 2023, 28, 28. [Google Scholar] [CrossRef]
- O’Brien, J.; Hayder, H.; Zayed, Y.; Peng, C. Overview of microRNA biogenesis, mechanisms of actions, and circulation. Front. Endocrinol. 2018, 9, 402. [Google Scholar] [CrossRef] [PubMed]
- Robinson, E.K.; Covarrubias, S.; Carpenter, S. The how and why of lncRNA function: An innate immune perspective. Biochim. Biophys. Acta (BBA)—Gene Regul. Mech. 2020, 1863, 194419. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.L.; Yang, L. Regulation of circRNA biogenesis. RNA Biol. 2015, 12, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Tsukahara, T. A view of pre-mRNA splicing from RNase R resistant RNAs. Int. J. Mol. Sci. 2014, 15, 9331–9342. [Google Scholar] [CrossRef]
- Geng, Y.; Jiang, J.; Wu, C. Function and clinical significance of circRNAs in solid tumors. J. Hematol. Oncol. 2018, 11, 98. [Google Scholar] [CrossRef]
- Poletajew, S.; Krajewski, W.; Kaczmarek, K.; Kopczyński, B.; Stamirowski, R.; Tukiendorf, A.; Zdrojowy, R.; Słojewski, M.; Radziszewski, P. The learning curve for transurethral resection of bladder tumour: How many is enough to be independent, safe and effective surgeon? J. Surg. Educ. 2020, 77, 978–985. [Google Scholar] [CrossRef]
- Morales, A.; Eidinger, D.; Bruce, A.W. Intracavitary Bacillus Calmette-Guerin in the treatment of superficial bladder tumors. J. Urol. 1976, 116, 180–182. [Google Scholar] [CrossRef]
- Babjuk, M.; Burger, M.; Capoun, O.; Cohen, D.; Compérat, E.M.; Escrig, J.L.; Gontero, P.; Liedberg, F.; Masson-Lecomte, A.; Mostafid, A.H.; et al. European Association of Urology guidelines on non–muscle-invasive bladder cancer (Ta, T1, and carcinoma in situ). Eur. Urol. 2022, 81, 75–94. [Google Scholar] [CrossRef]
- Huncharek, M.; Geschwind, J.F.; Witherspoon, B.; McGarry, R.; Adcock, D. Intravesical chemotherapy prophylaxis in primary superficial bladder cancer: A meta-analysis of 3703 patients from 11 randomized trials. J. Clin. Epidemiol. 2000, 53, 676–680. [Google Scholar] [CrossRef]
- Witjes, J.A.; Bruins, H.M.; Cathomas, R.; Compérat, E.M.; Cowan, N.C.; Gakis, G.; Hernández, V.; Espinós, E.L.; Lorch, A.; Neuzillet, Y.; et al. European Association of Urology guidelines on muscle-invasive and metastatic bladder cancer: Summary of the 2020 guidelines. Eur. Urol. 2021, 79, 82–104. [Google Scholar] [CrossRef]
- Compérat, E.; Amin, M.B.; Cathomas, R.; Choudhury, A.; De Santis, M.; Kamat, A.; Stenzl, A.; Thoeny, H.C.; Witjes, J.A. Current best practice for bladder cancer: A narrative review of diagnostics and treatments. Lancet 2022, 400, 1712–1721. [Google Scholar] [CrossRef] [PubMed]
- De Santis, M.; Bellmunt, J.; Mead, G.; Kerst, J.M.; Leahy, M.; Maroto, P.; Gil, T.; Marreaud, S.; Daugaard, G.; Skoneczna, I.; et al. Randomized phase II/III trial assessing gemcitabine/carboplatin and methotrexate/carboplatin/vinblastine in patients with advanced urothelial cancer who are unfit for cisplatin-based chemotherapy: EORTC study 30986. J. Clin Oncol. 2012, 30, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Loriot, Y.; Necchi, A.; Park, S.H.; Garcia-Donas, J.; Huddart, R.; Burgess, E.; Fleming, M.; Rezazadeh, A.; Mellado, B.; Varlamov, S.; et al. Erdafitinib in locally advanced or metastatic urothelial carcinoma. N. Engl. J. Med. 2019, 381, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Powles, T.; Rosenberg, J.E.; Sonpavde, G.P.; Loriot, Y.; Durán, I.; Lee, J.L.; Matsubara, N.; Vulsteke, C.; Castellano, D.; Wu, C.; et al. Enfortumab vedotin in previously treated advanced urothelial carcinoma. N. Engl. J. Med. 2021, 384, 1125–1135. [Google Scholar] [CrossRef] [PubMed]
- Hoimes, C.J.; Flaig, T.W.; Milowsky, M.I.; Friedlander, T.W.; Bilen, M.A.; Gupta, S.; Srinivas, S.; Merchan, J.R.; McKay, R.R.; Petrylak, D.P.; et al. Enfortumab vedotin plus pembrolizumab in previously untreated advanced urothelial cancer. J. Clin. Oncol. 2023, 41, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Tagawa, S.T.; Balar, A.V.; Petrylak, D.P.; Kalebasty, A.R.; Loriot, Y.; Fléchon, A.; Jain, R.K.; Agarwal, N.; Bupathi, M.; Barthelemy, P.; et al. TROPHY-U-01: A phase II open-label study of sacituzumab govitecan in patients with metastatic urothelial carcinoma progressing after platinum-based chemotherapy and checkpoint inhibitors. J. Clin. Oncol. 2021, 39, 2474–2485. [Google Scholar] [CrossRef]
- Pan, S.; Zhan, Y.; Chen, X.; Wu, B.; Liu, B. Bladder cancer exhibiting high immune infiltration shows the lowest response rate to immune checkpoint inhibitors. Front. Oncol. 2019, 9, 1101. [Google Scholar] [CrossRef]
- Zhou, B.; Guo, R. Integrative analysis of genomic and clinical data reveals intrinsic characteristics of bladder urothelial carcinoma progression. Genes 2019, 10, 464. [Google Scholar] [CrossRef]
- Matuszczak, M.; Kiljańczyk, A.; Salagierski, M. A liquid biopsy in bladder Cancer—The current Landscape in urinary biomarkers. Int. J. Mol. Sci. 2022, 23, 8597. [Google Scholar] [CrossRef]
- Chatterjee, D.; Mou, S.I.; Sultana, T.; Hosen, M.I.; Faruk, M.O. Identification and validation of prognostic signature genes of bladder cancer by integrating methylation and transcriptomic analysis. Sci. Rep. 2024, 14, 368. [Google Scholar] [CrossRef]
- Cancer.gov. Available online: https://www.cancer.gov/types/bladder/survival (accessed on 27 April 2023).
- Lucca, I.; de Martino, M.; Klatte, T.; Shariat, S.F. Novel biomarkers to predict response and prognosis in localized bladder cancer. Urol. Clin. 2015, 42, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Martinez, V.G.; Munera-Maravilla, E.; Bernardini, A.; Rubio, C.; Suarez-Cabrera, C.; Segovia, C.; Lodewijk, I.; Dueñas, M.; Martínez-Fernández, M.; Paramio, J.M. Epigenetics of bladder cancer: Where biomarkers and therapeutic targets meet. Front. Genet. 2019, 10, 1125. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Li, G.; Wu, S. Advances in diagnosis and therapy for bladder cancer. Cancers 2022, 14, 3181. [Google Scholar] [CrossRef] [PubMed]
- Veeratterapillay, R.; Gravestock, P.; Nambiar, A.; Gupta, A.; Aboumarzouk, O.; Rai, B.; Vale, L.; Heer, R. Time to turn on the blue lights: A systematic review and meta-analysis of photodynamic diagnosis for bladder cancer. Eur. Urol. Open Sci. 2021, 31, 17–27. [Google Scholar] [CrossRef]
- Yafi, F.A.; Brimo, F.; Steinberg, J.; Aprikian, A.G.; Tanguay, S.; Kassouf, W. Prospective analysis of sensitivity and specificity of urinary cytology and other urinary biomarkers for bladder cancer. In Urologic Oncology: Seminars and Original Investigations; Elsevier: Amsterdam, The Netherlands, 2015; pp. 66.e25–66.e31. [Google Scholar]
- Tomiyama, E.; Fujita, K.; Hashimoto, M.; Uemura, H.; Nonomura, N. Urinary markers for bladder cancer diagnosis: A review of current status and future challenges. Int. J. Urol. 2024, 31, 208–219. [Google Scholar] [CrossRef]
- Schmidbauer, J.; Remzi, M.; Klatte, T.; Waldert, M.; Mauermann, J.; Susani, M.; Marberger, M. Fluorescence cystoscopy with high-resolution optical coherence tomography imaging as an adjunct reduces false-positive findings in the diagnosis of urothelial carcinoma of the bladder. Eur. Urol. 2009, 56, 914–919. [Google Scholar] [CrossRef]
- Li, Q.; Tang, J.; He, E.; Li, Y.; Zhou, Y.; Wang, B. Differentiation between high-and low-grade urothelial carcinomas using contrast enhanced ultrasound. Oncotarget 2017, 8, 70883. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stav, K.; Leibovici, D.; Goren, E.; Livshitz, A.; Siegel, Y.I.; Lindner, A.; Zisman, A. Adverse effects of cystoscopy and its impact on patients’ quality of life and sexual performance. Isr. Med. Assoc. J. 2004, 6, 474–478. [Google Scholar]
- Witjes, J.A.; Morote, J.; Cornel, E.B.; Gakis, G.; van Valenberg, F.J.P.; Lozano, F.; Sternberg, I.A.; Willemsen, E.; Hegemann, M.L.; Paitan, Y.; et al. Performance of the bladder EpiCheck™ methylation test for patients under surveillance for non–muscle-invasive bladder cancer: Results of a multicenter, prospective, blinded clinical trial. Eur. Urol. Oncol. 2018, 1, 307–313. [Google Scholar] [CrossRef]
- Feber, A.; Dhami, P.; Dong, L.; de Winter, P.; Tan, W.S.; Martínez-Fernández, M.; Paul, S.S.; Hynes-Allen, A.; Rezaee, S.; Gurung, P.; et al. UroMark—A urinary biomarker assay for the detection of bladder cancer. Clin. Epigenetics 2017, 9, 8. [Google Scholar] [CrossRef]
- Pharo, H.D.; Jeanmougin, M.; Ager-Wick, E.; Vedeld, H.M.; Sørbø, A.K.; Dahl, C.; Larsen, L.K.; Honne, H.; Brandt-Winge, S.; Five, M.; et al. BladMetrix: A novel urine DNA methylation test with high accuracy for detection of bladder cancer in hematuria patients. Clin. Epigenetics 2022, 14, 115. [Google Scholar] [CrossRef] [PubMed]
- Piatti, P.; Chew, Y.C.; Suwoto, M.; Yamada, T.; Jara, B.; Jia, X.Y.; Guo, W.; Ghodoussipour, S.; Daneshmand, S.; Ahmadi, H.; et al. Clinical evaluation of Bladder CARE, a new epigenetic test for bladder cancer detection in urine samples. Clin. Epigenetics 2021, 13, 84. [Google Scholar] [CrossRef] [PubMed]
- Bang, B.R.; Zhong, J.; Oh, T.J.; Lee, J.Y.; Seo, Y.; Woo, M.A.; Lim, J.S.; Na, Y.G.; Song, K.H.; Shin, J.H.; et al. EarlyTect BCD, a Streamlined PENK Methylation Test in Urine DNA, Effectively Detects Bladder Cancer in Patients with Hematuria. J. Mol. Diagn. 2024, 26, 613–623. [Google Scholar] [CrossRef]
- Steinbach, D.; Kaufmann, M.; Hippe, J.; Gajda, M.; Grimm, M.O. High Detection Rate for Non–Muscle-Invasive Bladder Cancer Using an Approved DNA Methylation Signature Test. Clin. Genitourin. Cancer 2020, 18, 210–221. [Google Scholar] [CrossRef]
- Harsanyi, S.; Novakova, Z.V.; Bevizova, K.; Danisovic, L.; Ziaran, S. Biomarkers of bladder cancer: Cell-free DNA, epigenetic modifications and non-coding RNAs. Int. J. Mol. Sci. 2022, 23, 13206. [Google Scholar] [CrossRef]
- Fang, Q.; Zhang, X.; Nie, Q.; Hu, J.; Zhou, S.; Wang, C. Improved urine DNA methylation panel for early bladder cancer detection. BMC Cancer 2022, 22, 237. [Google Scholar] [CrossRef] [PubMed]
- Hentschel, A.E.; Beijert, I.J.; Bosschieter, J.; Kauer, P.C.; Vis, A.N.; Lissenberg-Witte, B.I.; Moorselaar, R.J.A.; Steenbergen, R.D.M.; Nieuwenhuijzen, J.A. Bladder cancer detection in urine using DNA methylation markers: A technical and prospective preclinical validation. Clin. Epigenetics 2022, 14, 19. [Google Scholar] [CrossRef]
- Jiang, Y.H.; Liu, Y.S.; Wei, Y.C.; Jhang, J.F.; Kuo, H.C.; Huang, H.H.; Chan, M.W.Y.; Lin, G.L.; Cheng, W.C.; Lin, S.C.; et al. Hypermethylation Loci of ZNF671, IRF8, and OTX1 as Potential Urine-Based Predictive Biomarkers for Bladder Cancer. Diagnostics 2024, 14, 468. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Jiang, G.; Zhang, N.; Liu, S.; Lin, X.; Perschon, C.; Zheng, S.L.; Ding, Q.; Wang, X.; Na, R.; et al. HOXA9, PCDH17, POU4F2, and ONECUT2 as a urinary biomarker combination for the detection of bladder cancer in Chinese patients with hematuria. Eur. Urol. Focus 2020, 6, 284–291. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, J.; Ruan, W.; Huang, M.; Wang, C.; Wang, H.; Jiang, Z.; Wang, S.; Liu, Z.; Liu, C.; et al. Urine DNA methylation assay enables early detection and recurrence monitoring for bladder cancer. J. Clin. Investig. 2020, 130, 6278–6289. [Google Scholar] [CrossRef]
- Zhang, C.; Xu, X.; Wang, T.; Lu, Y.; Lu, Z.; Wang, T.; Pan, Z. Clinical performance and utility of a noninvasive urine-based methylation biomarker: TWIST1/Vimentin to detect urothelial carcinoma of the bladder. Sci. Rep. 2024, 14, 7941. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Chao, H.; Deng, H.; Yu, Z.; Zhao, R.; Huang, L.; Gong, Y.; Zhu, Y.; Wang, Q.; Li, F.; et al. A novel and sensitive DNA methylation marker for the urine-based liquid biopsies to detect bladder cancer. BMC Cancer 2022, 22, 510. [Google Scholar] [CrossRef] [PubMed]
- Ruan, W.; Chen, X.; Huang, M.; Wang, H.; Chen, J.; Liang, Z.; Zhang, J.; Yu, Y.; Chen, S.; Xu, S.; et al. A urine-based DNA methylation assay to facilitate early detection and risk stratification of bladder cancer. Clin. Epigenetics 2021, 13, 91. [Google Scholar] [CrossRef]
- Guo, R.Q.; Xiong, G.Y.; Yang, K.W.; Zhang, L.; He, S.M.; Gong, Y.Q.; He, Q.; Li, X.Y.; Wang, Z.C.; Bao, Z.Q.; et al. Detection of urothelial carcinoma, upper tract urothelial carcinoma, bladder carcinoma, and urothelial carcinoma with gross hematuria using selected urine-DNA methylation biomarkers: A prospective, single-center study. In Urologic Oncology: Seminars and Original Investigations; Elsevier: Amsterdam, The Netherlands, 2018; pp. 342.e15–342.e23. [Google Scholar]
- Roperch, J.P.; Grandchamp, B.; Desgrandchamps, F.; Mongiat-Artus, P.; Ravery, V.; Ouzaid, I.; Roupret, M.; Phe, V.; Ciofu, C.; Tubach, F.; et al. Promoter hypermethylation of HS3ST2, SEPTIN9 and SLIT2 combined with FGFR3 mutations as a sensitive/specific urinary assay for diagnosis and surveillance in patients with low or high-risk non-muscle-invasive bladder cancer. BMC Cancer 2016, 16, 704. [Google Scholar] [CrossRef]
- Dahmcke, C.M.; Steven, K.E.; Larsen, L.K.; Poulsen, A.L.; Abdul-Al, A.; Dahl, C.; Guldberg, P. A prospective blinded evaluation of urine-DNA testing for detection of urothelial bladder carcinoma in patients with gross hematuria. Eur. Urol. 2016, 70, 916–919. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Tian, Y.; Xu, H. Improved Noninvasive Bladder Cancer Diagnosis using Urine Sediments and Novel DNA Methylation Biomarker Panels. Clin. Lab. 2016, 62, 327–336. [Google Scholar] [CrossRef]
- Wang, J.; Peng, X.; Li, R.; Liu, K.; Zhang, C.; Chen, X.; Huang, G.; Zhao, L.; Chen, Z.; Lai, Y. Evaluation of serum miR-17-92 cluster as noninvasive biomarkers for bladder cancer diagnosis. Front. Oncol. 2021, 11, 795837. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Wei, X.; Qu, X.; Zhu, Y. Potential clinical application of microRNAs in bladder cancer. J. Biomed. Res. 2024, 38, 289–306. [Google Scholar] [CrossRef]
- Yu, Z.; Lu, C.; Lai, Y. A serum miRNAs signature for early diagnosis of bladder cancer. Ann. Med. 2023, 55, 736–745. [Google Scholar] [CrossRef]
- Suarez-Cabrera, C.; Estudillo, L.; Ramón-Gil, E.; Martínez-Fernández, M.; Peral, J.; Rubio, C.; Lodewijk, I.; de Bernardo, A.M.; Garcia-Escudero, R.; Villacampa, F.; et al. BlaDimiR: A urine-based miRNA score for accurate bladder cancer diagnosis and follow-up. Eur. Urol. 2022, 82, 663–667. [Google Scholar] [CrossRef]
- El-Shal, A.S.; Shalaby, S.M.; Abouhashem, S.E.; Elbary, E.H.A.; Azazy, S.; Rashad, N.M.; Sarhan, W. Urinary exosomal microRNA-96-5p and microRNA-183-5p expression as potential biomarkers of bladder cancer. Mol. Biol. Rep. 2021, 48, 4361–4371. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Lu, Z.; Liu, C.; Meng, Y.; Ma, Y.; Zhao, W.; Liu, J.; Yu, J.; Chen, J. miRNA-96 suppresses KRAS and functions as a tumor suppressor gene in pancreatic cancer. Cancer Res. 2010, 70, 6015–6025. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Fu, H.; Xu, C.; Tie, Y.; Xing, R.; Zhu, J.; Qin, Y.; Sun, Z.; Zheng, X. miR-183 inhibits TGF-β1-induced apoptosis by downregulation of PDCD4 expression in human hepatocellular carcinoma cells. BMC Cancer 2010, 10, 354. [Google Scholar] [CrossRef]
- Myatt, S.S.; Wang, J.; Monteiro, L.J.; Christian, M.; Ho, K.K.; Fusi, L.; Dina, R.E.; Brosens, J.J.; Ghaem-Maghami, S.; Lam, E.W. Definition of microRNAs that repress expression of the tumor suppressor gene FOXO1 in endometrial cancer. Cancer Res. 2010, 70, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Shi, X.; Li, H.; Hui, J.; Liu, R.; Chen, Z.; Lu, Y.; Tan, W. Urinary Exosomal miRNAs as biomarkers of bladder Cancer and experimental verification of mechanism of miR-93-5p in bladder Cancer. BMC Cancer 2021, 21, 1293. [Google Scholar] [CrossRef]
- Matsuzaki, K.; Fujita, K.; Jingushi, K.; Kawashima, A.; Ujike, T.; Nagahara, A.; Ueda, Y.; Tanigawa, G.; Yoshioka, I.; Ueda, K.; et al. MiR-21-5p in urinary extracellular vesicles is a novel biomarker of urothelial carcinoma. Oncotarget 2017, 8, 24668. [Google Scholar] [CrossRef]
- Chen, X. MiR-101 acts as a novel bio-marker in the diagnosis of bladder carcinoma. Medicine 2019, 98, e16051. [Google Scholar] [CrossRef]
- Lu, C.; Lin, S.; Wen, Z.; Sun, C.; Ge, Z.; Chen, W.; Li, Y.; Zhang, P.; Wu, Y.; Wang, W.; et al. Testing the accuracy of a four serum microRNA panel for the detection of primary bladder cancer: A discovery and validation study. Biomarkers 2024, 29, 276–284. [Google Scholar] [CrossRef]
- Grimaldi, A.M.; Lapucci, C.; Salvatore, M.; Incoronato, M.; Ferrari, M. Urinary miRNAs as a diagnostic tool for bladder cancer: A systematic review. Biomedicines 2022, 10, 2766. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, X.; Zhang, D.; Yu, Y.; Cai, L.; Zhang, C. Long non-coding RNA urothelial carcinoma–associated 1 as a tumor biomarker for the diagnosis of urinary bladder cancer. Tumor Biol. 2017, 39, 1010428317709990. [Google Scholar] [CrossRef]
- Zhan, Y.; Du, L.; Wang, L.; Jiang, X.; Zhang, S.; Li, J.; Yan, K.; Duan, W.; Zhao, Y.; Wang, L.; et al. Expression signatures of exosomal long non-coding RNAs in urine serve as novel non-invasive biomarkers for diagnosis and recurrence prediction of bladder cancer. Mol. Cancer 2018, 17, 142. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Wang, R.; Han, C.; Wang, Z.; Jin, X. A panel of urinary long non-coding RNAs differentiate bladder cancer from urocystitis. J. Cancer 2020, 11, 781. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zhang, X.; Wang, X.; Sun, R.; Li, Y.; Li, J.; Quan, W.; Yao, Y.; Hou, Y.; Li, D.; et al. The clinical value of rapidly detecting urinary exosomal lncRNA RMRP in bladder cancer with an RT-RAA-CRISPR/Cas12a method. Clin. Chim. Acta 2024, 562, 119855. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Shang, A.; Sun, Z.; Gao, Y.; Huang, J.; Ping, Y.; Chang, W.; Gu, C.; Sun, J.; Ji, P.; et al. Urinary exosomal long noncoding RNA TERC as a noninvasive diagnostic and prognostic biomarker for bladder urothelial carcinoma. J. Immunol. Res. 2022, 2022, 9038808. [Google Scholar] [CrossRef]
- Yang, Y.; Li, J.; Yao, W.; Zou, G.; Ye, X.; Mo, Q. Diagnostic value of urine cyclic RNA-0071196 for bladder urothelial carcinoma. BMC Urol. 2024, 24, 88. [Google Scholar] [CrossRef]
- Song, Z.; Zhang, Q.; Zhu, J.; Yin, G.; Lin, L.; Liang, C. Identification of urinary hsa_circ_0137439 as a potential biomarker and tumor regulator of bladder cancer. Neoplasma 2020, 67, 137–146. [Google Scholar] [CrossRef]
- Mamdouh, S.; Sherif, H.; Romeih, M.; Elesaily, K. Urine micro-RNA signature as a potential non-invasive diagnostic biomarker in bladder cancer. Asian Pac. J. Cancer Prev. APJCP 2023, 24, 121. [Google Scholar]
- Zhang, X.F.; Zhang, X.Q.; Chang, Z.X.; Wu, C.C.; Guo, H. microRNA-145 modulates migration and invasion of bladder cancer cells by targeting N-cadherin. Mol. Med. Rep. 2018, 17, 8450–8456. [Google Scholar] [CrossRef]
- Zhang, H.H.; Qi, F.; Cao, Y.H.; Zu, X.B.; Chen, M.F. Expression and clinical significance of microRNA-21, maspin and vascular endothelial growth factor-C in bladder cancer. Oncol. Lett. 2015, 10, 2610–2616. [Google Scholar] [CrossRef]
- Piao, X.M.; Jeong, P.; Kim, Y.H.; Byun, Y.J.; Xu, Y.; Kang, H.W.; Ha, Y.S.; Kim, W.T.; Lee, J.Y.; Woo, S.H.; et al. Urinary cell-free microRNA biomarker could discriminate bladder cancer from benign hematuria. Int. J. Cancer 2019, 144, 380–388. [Google Scholar] [CrossRef]
- Usuba, W.; Urabe, F.; Yamamoto, Y.; Matsuzaki, J.; Sasaki, H.; Ichikawa, M.; Takizawa, S.; Aoki, Y.; Niida, S.; Kato, K.; et al. Circulating miRNA panels for specific and early detection in bladder cancer. Cancer Sci. 2019, 110, 408–419. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Zhao, H.; Qian, X.; Qiu, J. MiR-20a in cell-free urine as a potential diagnostic biomarker for non-muscle invasive bladder cancer: A Chinese population-based study. Int. J. Clin. Exp. Med. 2018, 11, 209–216. [Google Scholar]
- Urquidi, V.; Netherton, M.; Gomes-Giacoia, E.; Serie, D.J.; Eckel-Passow, J.; Rosser, C.J.; Goodison, S. A microRNA biomarker panel for the non-invasive detection of bladder cancer. Oncotarget 2016, 7, 86290. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, Y.; Liu, X.; Fang, A.; Wang, J.; Yang, Y.; Wang, L.; Du, L.; Wang, C. Direct quantitative detection for cell-free miR-155 in urine: A potential role in diagnosis and prognosis for non-muscle invasive bladder cancer. Oncotarget 2016, 7, 3255. [Google Scholar] [CrossRef] [PubMed]
- Motawi, T.K.; Rizk, S.M.; Ibrahim, T.M.; Ibrahim, I.A.R. Circulating microRNAs, miR-92a, miR-100 and miR-143, as non-invasive biomarkers for bladder cancer diagnosis. Cell Biochem. Funct. 2016, 34, 142–148. [Google Scholar] [CrossRef]
- Liu, C.; Xu, P.; Shao, S.; Wang, F.; Zheng, Z.; Li, S.; Liu, W.; Li, G. The value of urinary exosomal lncRNA SNHG16 as a diagnostic biomarker for bladder cancer. Mol. Biol. Rep. 2023, 50, 8297–8304. [Google Scholar] [CrossRef]
- Bian, B.; Li, L.; Ke, X.; Chen, H.; Liu, Y.; Zheng, N.; Zheng, Y.; Ma, Y.; Zhou, Y.; Yang, J.; et al. Urinary exosomal long non-coding RNAs as noninvasive biomarkers for diagnosis of bladder cancer by RNA sequencing. Front. Oncol. 2022, 12, 976329. [Google Scholar] [CrossRef]
- Sarfi, M.; Abbastabar, M.; Khalili, E. Increased expression of urinary exosomal LnCRNA TUG-1 in early bladder cancer. Gene Rep. 2021, 22, 101010. [Google Scholar] [CrossRef]
- Luo, L.; Xie, Q.; Wu, Y.; Li, P.; Qin, F.; Liao, D.; Wang, K. Circular RNA CCT3 is a unique molecular marker in bladder cancer. BMC Cancer 2023, 23, 977. [Google Scholar] [CrossRef]
- Tang, G.; Xie, W.; Qin, C.; Zhen, Y.; Wang, Y.; Chen, F.; Du, Z.; Wu, Z.; Zhang, B.; Shen, Z.; et al. Expression of circular RNA circASXL1 correlates with TNM classification and predicts overall survival in bladder cancer. Int. J. Clin. Exp. Pathol. 2017, 10, 8495. [Google Scholar]
- Cheng, H.; Liu, Y.; Chen, G. Identification of potential DNA methylation biomarkers related to diagnosis in patients with bladder cancer through integrated bioinformatic analysis. BMC Urol. 2023, 23, 135. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Liu, X.; Yang, A.; Fu, W.; Yin, F.; Zeng, X. Associations of tumor suppressor SPARCL1 with cancer progression and prognosis. Oncol. Lett. 2017, 14, 2603–2610. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Zhang, H.; Ge, W.; Liu, X.; Loera, S.; Chu, P.; Chen, H.; Peng, J.; Zhou, L.; Yu, S.; et al. Secreted protein acidic and rich in cysteines-like 1 suppresses aggressiveness and predicts better survival in colorectal cancers. Clin. Cancer Res. 2012, 18, 5438–5448. [Google Scholar] [CrossRef]
- Esposito, I.; Kayed, H.; Keleg, S.; Giese, T.; Sage, E.H.; Schirmacher, P.; Friess, H.; Kleeff, J. Tumor-suppressor function of SPARC-like protein 1/Hevin in pancreatic cancer. Neoplasia 2007, 9, 8–17. [Google Scholar] [CrossRef]
- Turtoi, A.; Musmeci, D.; Naccarato, A.G.; Scatena, C.; Ortenzi, V.; Kiss, R.; Murtas, D.; Patsos, G.; Mazzucchelli, G.; Pauw, E.D.; et al. Sparc-like protein 1 is a new marker of human glioma progression. J. Proteome Res. 2012, 11, 5011–5021. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Wang, Y.X.; Shen, A.; Yang, X.Q.; Liang, C.C.; Huang, R.J.; Jian, R.; An, N.; Xiao, Y.L.; Wang, L.S.; et al. Construction of a gene model related to the prognosis of patients with gastric cancer receiving immunotherapy and exploration of COX7A1 gene function. Eur. J. Med. Res. 2024, 29, 180. [Google Scholar] [CrossRef]
- Shen, H.; Zhang, L.; Zhou, J.; Chen, Z.; Yang, G.; Liao, Y.; Zhu, M. Epidermal growth factor-containing fibulin-like extracellular matrix protein 1 (EFEMP1) acts as a potential diagnostic biomarker for prostate cancer. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2017, 23, 216. [Google Scholar] [CrossRef]
- Fu, S.; Luan, T.; Jiang, C.; Huang, Y.; Li, N.; Wang, H.; Wang, J. miR-3622a promotes proliferation and invasion of bladder cancer cells by downregulating LASS2. Gene 2019, 701, 23–31. [Google Scholar] [CrossRef]
- Xu, X.H.; Sun, J.M.; Chen, X.F.; Zeng, X.Y.; Zhou, H.Z. MicroRNA-494-3p facilitates the progression of bladder cancer by mediating the KLF9/RGS2 axis. Kaohsiung J. Med. Sci. 2022, 38, 1070–1079. [Google Scholar] [CrossRef]
- Ren, W.; Hu, J.; Li, H.; Chen, J.; Ding, J.; Zu, X.; Fan, B. miR-616-5p promotes invasion and migration of bladder cancer via downregulating NR2C2 expression. Front. Oncol. 2021, 11, 762946. [Google Scholar] [CrossRef]
- Lv, X.Y.; Ma, L.; Chen, J.F.; Yu, R.; Li, Y.; Yan, Z.J.; Cheng, Y.; Ma, Q. Knockdown of DUXAP10 inhibits proliferation and promotes apoptosis in bladder cancer cells via PI3K/Akt/mTOR signaling pathway. Int. J. Oncol. 2017, 52, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; German, P.; Bai, S.; Barnes, S.; Guo, W.; Qi, X.; Lou, H.; Liang, J.; Jonasch, E.; Mills, G.B.; et al. The PI3K/AKT pathway and renal cell carcinoma. J. Genet. Genom. 2015, 42, 343–353. [Google Scholar] [CrossRef] [PubMed]
- Arima, J.; Yoshino, H.; Fukumoto, W.; Kawahara, I.; Saito, S.; Li, G.; Fukuda, I.; Lizasa, S.; Mitsuke, A.; Sakaguchi, T.; et al. LncRNA BCYRN1 as a Potential Therapeutic Target and Diagnostic Marker in Serum Exosomes in Bladder Cancer. Int. J. Mol. Sci. 2024, 25, 5955. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Shi, G.; Li, Q.; Li, W.; Zhou, H. Long noncoding RNA XIST participates in bladder cancer by downregulating p53 via binding to TET1. J. Cell. Biochem. 2019, 120, 6330–6338. [Google Scholar] [CrossRef]
- Li, H.J.; Gong, X.; Li, Z.K.; Qin, W.; He, C.X.; Xing, L.; Zhou, X.; Zhao, D.; Cao, H.L. Role of long non-coding RNAs on bladder cancer. Front. Cell Dev. Biol. 2021, 9, 672679. [Google Scholar] [CrossRef]
- Zhou, X.; Xiao, L.; Meng, F.; Zuo, F.; Wu, W.; Li, G.; Han, F.; Peng, G.; Shen, H. GAS6-AS1 drives bladder cancer progression by increasing MMP7 expression in a ceRNA-and RBP-dependent manner. Transl. Oncol. 2024, 48, 102065. [Google Scholar] [CrossRef]
- Yang, J.; Qi, M.; Fei, X.; Wang, X.; Wang, K. Hsa_circRNA_0088036 acts as a ceRNA to promote bladder cancer progression by sponging miR-140-3p. Cell Death Dis. 2022, 13, 322. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Wang, L.; Shi, Z.; Zhu, A.; Zhang, G.; Hong, Z.; Cheng, C. Circular RNA circFLNA inhibits the development of bladder carcinoma through microRNA miR-216a-3p/BTG2 axis. Bioengineered 2021, 12, 11376–11389. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.; Ma, X.; Ying, Y.; Liu, Z.; Tang, Y.; Shu, X.; Sun, J.; Wu, Y.; Lu, D.; Wang, X.; et al. N6-methyladenosine-modified CircPSMA7 enhances bladder cancer malignancy through the miR-128–3p/MAPK1 axis. Cancer Lett. 2024, 585, 216613. [Google Scholar] [CrossRef]
- Oliver, J.; Garcia-Aranda, M.; Chaves, P.; Alba, E.; Cobo-Dols, M.; Onieva, J.L.; Barragan, I. Emerging noninvasive methylation biomarkers of cancer prognosis and drug response prediction. In Seminars in Cancer Biology; Academic Press: Cambridge, MA, USA, 2022; pp. 584–595. [Google Scholar]
- Yoon, H.Y.; Kim, Y.J.; Kim, J.S.; Kim, Y.W.; Kang, H.W.; Kim, W.T.; Yun, S.J.; Ryu, K.H.; Lee, S.C.; Kim, W.J. RSPH9 methylation pattern as a prognostic indicator in patients with non-muscle invasive bladder cancer. Oncol. Rep. 2016, 35, 1195–1203. [Google Scholar] [CrossRef][Green Version]
- Zhan, L.; Zhang, B.; Tan, Y.; Yang, C.; Huang, C.; Wu, Q.; Zhang, Y.; Chen, X.; Zhou, M.; Shu, A. Quantitative assessment of the relationship between RASSF1A gene promoter methylation and bladder cancer (PRISMA). Medicine 2017, 96, e6097. [Google Scholar] [CrossRef] [PubMed]
- Shivakumar, M.; Lee, Y.; Bang, L.; Garg, T.; Sohn, K.A.; Kim, D. Identification of epigenetic interactions between miRNA and DNA methylation associated with gene expression as potential prognostic markers in bladder cancer. BMC Med. Genom. 2017, 10, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Fang, L.; Zang, Y.; Xu, Z. Identification of core genes and key pathways via integrated analysis of gene expression and DNA methylation profiles in bladder cancer. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2018, 24, 3024. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, J.; Xu, M.; Zhu, T. Exploring prognostic DNA methylation genes in bladder cancer: A comprehensive analysis. Discov. Oncol. 2024, 15, 331. [Google Scholar] [CrossRef] [PubMed]
- Takagi, K.; Naruse, A.; Akita, K.; Muramatsu-Maekawa, Y.; Kawase, K.; Koie, T.; Horie, M.; Kikuchi, A. CALN1 hypomethylation as a biomarker for high-risk bladder cancer. BMC Urol. 2022, 22, 176. [Google Scholar] [CrossRef]
- Chen, J.Q.; Salas, L.A.; Wiencke, J.K.; Koestler, D.C.; Molinaro, A.M.; Andrew, A.S.; Seigne, J.D.; Karagas, M.R.; Kelsey, K.T.; Christensen, B.C. Immune profiles and DNA methylation alterations related with non-muscle-invasive bladder cancer outcomes. Clin. Epigenetics 2022, 14, 14. [Google Scholar] [CrossRef]
- Zhang, C.; Shen, K.; Zheng, Y.; Qi, F.; Luo, J. Genome-wide screening of abberant methylated drivers combined with relative risk loci in bladder cancer. Cancer Med. 2020, 9, 768–782. [Google Scholar] [CrossRef]
- Kim, C.; Oh, S.; Im, H.; Gim, J. Unveiling Bladder Cancer Prognostic Insights by Integrating Patient-Matched Sample and CpG Methylation Analysis. Medicina 2024, 60, 1175. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, J.; Zhang, Q.; Liang, Y.; Du, Y.; Wang, G. Identification of prognostic biomarkers for bladder cancer based on DNA methylation profile. Front. Cell Dev. Biol. 2022, 9, 817086. [Google Scholar] [CrossRef]
- Koukourikis, P.; Papaioannou, M.; Georgopoulos, P.; Apostolidis, I.; Pervana, S.; Apostolidis, A. A Study of DNA Methylation of Bladder Cancer Biomarkers in the Urine of Patients with Neurogenic Lower Urinary Tract Dysfunction. Biology 2023, 12, 1126. [Google Scholar] [CrossRef]
- Azzouzi, M.; El Ahanidi, H.; Alaoui, C.H.; Chaoui, I.; Benbacer, L.; Tetou, M.; Hassan, I.; Bensaid, M.; Oukabli, M.; Ameur, A.; et al. Evaluation of DNA methylation in promoter regions of hTERT, TWIST1, VIM and NID2 genes in Moroccan bladder cancer patients. Cancer Genet. 2022, 260, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Yin, J.; Dai, Y.; Guan, Y.; Chen, P.; Chen, Y.; Huang, C.; Lu, Y.J.; Zhang, L.; Song, D. A novel CpG methylation risk indicator for predicting prognosis in bladder cancer. Front. Cell Dev. Biol. 2021, 9, 642650. [Google Scholar] [CrossRef]
- Zhou, Q.; Chen, Q.; Chen, X.; Hao, L. Bioinformatics analysis to screen DNA methylation-driven genes for prognosis of patients with bladder cancer. Transl. Androl. Urol. 2021, 10, 3604. [Google Scholar] [CrossRef]
- Mitash, N.; Agnihotri, S.; Tiwari, S.; Agrawal, V.; Mandhani, A. MicroRNA-21 could be a molecular marker to predict the recurrence of nonmuscle invasive bladder cancer. Indian J. Urol. 2017, 33, 283–290. [Google Scholar] [PubMed]
- Chen, X.; Wu, B.; Xu, Z.; Li, S.; Tan, S.; Liu, X.; Wang, K. Downregulation of miR-133b predict progression and poor prognosis in patients with urothelial carcinoma of bladder. Cancer Med. 2016, 5, 1856–1862. [Google Scholar] [CrossRef]
- Zeng, W.; Zhu, J.F.; Liu, J.Y.; Li, Y.L.; Dong, X.; Huang, H.; Shan, L. miR-133b inhibits cell proliferation, migration and invasion of esophageal squamous cell carcinoma by targeting EGFR. Biomed. Pharmacother. 2019, 111, 476–484. [Google Scholar] [CrossRef]
- Avgeris, M.; Tsilimantou, A.; Levis, P.K.; Tokas, T.; Sideris, D.C.; Stravodimos, K.; Ardavanis, A.; Scorilas, A. Loss of GAS5 tumour suppressor lncRNA: An independent molecular cancer biomarker for short-term relapse and progression in bladder cancer patients. Br. J. Cancer 2018, 119, 1477–1486. [Google Scholar] [CrossRef]
- Jiao, R.; Jiang, W.; Wei, X.; Zhang, M.; Zhao, S.; Huang, G. Clinicopathological significance and prognosis of long noncoding RNA SNHG16 expression in human cancers: A meta-analysis. BMC Cancer 2020, 20, 662. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Luo, Y.; He, W.; Zhao, Y.; Kong, Y.; Liu, H.; Zhong, G.; Li, Y.; Li, J.; Huang, J.; et al. Exosomal long noncoding RNA LNMAT2 promotes lymphatic metastasis in bladder cancer. J. Clin. Investig. 2020, 130, 404–421. [Google Scholar] [CrossRef]
- Lin, G.; Sheng, H.; Xie, H.; Zheng, Q.; Shen, Y.; Shi, G.; Ye, D. circLPAR1 is a novel biomarker of prognosis for muscle-invasive bladder cancer with invasion and metastasis by miR-762. Oncol. Lett. 2019, 17, 3537–3547. [Google Scholar] [CrossRef]
- Liu, J.; Liu, T.; Wang, X.; He, A. Circles reshaping the RNA world: From waste to treasure. Mol. Cancer 2017, 16, 58. [Google Scholar] [CrossRef] [PubMed]
- Andrew, A.S.; Karagas, M.R.; Schroeck, F.R.; Marsit, C.J.; Schned, A.R.; Pettus, J.R.; Armstrong, D.A.; Seigne, J.D. MicroRNA Dysregulation and Non-Muscle–Invasive Bladder Cancer Prognosis. Cancer Epidemiol. Biomark. Prev. 2019, 28, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, K.; Seki, N.; Matsushita, R.; Yonemori, M.; Yoshino, H.; Nakagawa, M.; Enokida, H. Tumour-suppressive miRNA-26a-5p and miR-26b-5p inhibit cell aggressiveness by regulating PLOD2 in bladder cancer. Br. J. Cancer 2016, 115, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Borkowska, E.M.; Konecki, T.; Pietrusiński, M.; Borowiec, M.; Jabłonowski, Z. MicroRNAs which can prognosticate aggressiveness of bladder cancer. Cancers 2019, 11, 1551. [Google Scholar] [CrossRef]
- Juracek, J.; Stanik, M.; Vesela, P.; Radova, L.; Dolezel, J.; Svoboda, M.; Slaby, O. Tumor expression of miR-34a-3p is an independent predictor of recurrence in non–muscle-invasive bladder cancer and promising additional factor to improve predictive value of EORTC nomogram. In Urologic Oncology: Seminars and Original Investigations; Elsevier: Amsterdam, The Netherlands, 2019; pp. 184.e1–184.e7. [Google Scholar]
- Ding, Z.S.; He, Y.H.; Deng, Y.S.; Peng, P.X.; Wang, J.F.; Chen, X.; Zhao, P.Y.; Zhou, X.F. MicroRNA-34a inhibits bladder cancer cell migration and invasion and upregulates PTEN expression. Oncol. Lett. 2019, 18, 5549–5554. [Google Scholar] [CrossRef]
- Yang, L.; Sun, H.F.; Guo, L.Q.; Cao, H.B. MiR-10a-5p: A promising biomarker for early diagnosis and prognosis evaluation of bladder cancer. Cancer Manag. Res. 2021, 13, 7841–7850. [Google Scholar] [CrossRef] [PubMed]
- Bogale, D.E. The roles of FGFR3 and c-MYC in urothelial bladder cancer. Discov. Oncol. 2024, 15, 295. [Google Scholar] [CrossRef]
- Yin, X.H.; Jin, Y.H.; Cao, Y.; Wong, Y.; Weng, H.; Sun, C.; Deng, J.H.; Zeng, X.T. Development of a 21-miRNA signature associated with the prognosis of patients with bladder cancer. Front. Oncol. 2019, 9, 729. [Google Scholar] [CrossRef]
- Yerukala Sathipati, S.; Tsai, M.J.; Shukla, S.K.; Ho, S.Y.; Liu, Y.; Beheshti, A. MicroRNA signature for estimating the survival time in patients with bladder urothelial carcinoma. Sci. Rep. 2022, 12, 4141. [Google Scholar] [CrossRef]
- Zhu, Q.L.; Zhan, D.M.; Chong, Y.K.; Ding, L.; Yang, Y.G. MiR-652-3p promotes bladder cancer migration and invasion by targeting KCNN3. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 8806–8812. [Google Scholar]
- Chen, C.; Zheng, H.; Luo, Y.; Kong, Y.; An, M.; Li, Y.; He, W.; Gao, B.; Zhao, Y.; Huang, J.; et al. UMOylation promotes extracellular vesicle–mediated transmission of lncRNA ELNAT1 and lymph node metastasis in bladder cancer. J. Clin. Investig. 2021, 131, e146431. [Google Scholar] [CrossRef] [PubMed]
- Liang, T.; Xu, F.; Wan, P.; Zhang, L.; Huang, S.; Yang, N.; Wang, Y. Malat-1 expression in bladder carcinoma tissues and its clinical significance. Am. J. Transl. Res. 2021, 13, 3555. [Google Scholar] [PubMed]
- Zheng, H.; Chen, C.; Luo, Y.; Yu, M.; He, W.; An, M.; Gao, B.; Kong, Y.; Ya, Y.; Lin, Y.; et al. Tumor-derived exosomal BCYRN1 activates WNT5A/VEGF-C/VEGFR3 feedforward loop to drive lymphatic metastasis of bladder cancer. Clin. Transl. Med. 2021, 11, e497. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, Y.; Fujita, K.; Netto, G.J.; Nonomura, N. Clinical application of TERT promoter mutations in urothelial carcinoma. Front. Oncol. 2021, 11, 705440. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Zhao, N.; Zhang, H.; Xie, D. Circular RNA CircHIPK3 promotes gemcitabine sensitivity in bladder cancer. J. Cancer 2020, 11, 1907. [Google Scholar] [CrossRef]
- Zhang, J.X.; Lu, J.; Xie, H.; Wang, D.P.; Ni, H.E.; Zhu, Y.; Ren, L.H.; Meng, X.X.; Wang, R.L. circHIPK3 regulates lung fibroblast-to-myofibroblast transition by functioning as a competing endogenous RNA. Cell Death Dis. 2019, 10, 182. [Google Scholar] [CrossRef]
- Su, Y.; Feng, W.; Shi, J.; Chen, L.; Huang, J.; Lin, T. circRIP2 accelerates bladder cancer progression via miR-1305/Tgf-β2/smad3 pathway. Mol. Cancer 2020, 19, 23. [Google Scholar] [CrossRef]
- Zhu, J.; Luo, Y.; Zhao, Y.; Kong, Y.; Zheng, H.; Li, Y.; Gao, B.; Ai, L.; Huang, H.; Huang, J.; et al. circEHBP1 promotes lymphangiogenesis and lymphatic metastasis of bladder cancer via miR-130a-3p/TGFβR1/VEGF-D signaling. Mol. Ther. 2021, 29, 1838–1852. [Google Scholar] [CrossRef]
- Zhenhai, Z.; Qi, C.; Shuchao, Z.; Zhongqi, W.; Xue, S.; Zhijun, G.; Zhijie, M.; Jianmin, L.; Beibei, L.; Yuanyuan, G. MiR-205-3p suppresses bladder cancer progression via GLO1 mediated P38/ERK activation. BMC Cancer 2023, 23, 956. [Google Scholar] [CrossRef]
- Hao, Y.; Zhu, Y.; Sun, F.; Xu, D.; Wang, C. MicroRNA-30c-5p arrests bladder cancer G2/M phase and suppresses its progression by targeting PRC1-mediated blocking of CDK1/Cyclin B1 axis. Cell. Signal. 2023, 110, 110836. [Google Scholar] [CrossRef]
- Awadalla, A.; Abol-Enein, H.; Hamam, E.T.; Ahmed, A.E.; Khirallah, S.M.; El-Assmy, A.; Mostafa, S.A.; Babalghith, A.O.; Ali, M.; Abdel-Rahim, M.; et al. Identification of epigenetic interactions between miRNA and gene expression as potential prognostic markers in bladder cancer. Genes 2022, 13, 1629. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Sang, Y.; Liu, Z.; Shao, J. Negative correlation between circular RNA SMARC5 and MicroRNA 432, and their clinical implications in bladder cancer patients. Technol. Cancer Res. Treat. 2021, 20, 15330338211039110. [Google Scholar] [CrossRef]
- Spagnuolo, M.; Costantini, M.; Ferriero, M.; Varmi, M.; Sperduti, I.; Regazzo, G.; Cicchillitti, L.; Mendez, A.B.D.; Cigliana, G.; Pompeo, V.; et al. Urinary expression of let-7c cluster as non-invasive tool to assess the risk of disease progression in patients with high grade non-muscle invasive bladder Cancer: A pilot study. J. Exp. Clin. Cancer Res. 2020, 39, 68. [Google Scholar] [CrossRef] [PubMed]
- Braicu, C.; Buiga, R.; Cojocneanu, R.; Buse, M.; Raduly, L.; Pop, L.A.; Chira, S.; Budisan, L.; Jurj, A.; Ciocan, C.; et al. Connecting the dots between different networks: miRNAs associated with bladder cancer risk and progression. J. Exp. Clin. Cancer Res. 2019, 38, 433. [Google Scholar] [CrossRef]
- Chen, H.; Xu, L.; Wang, L. Expression of miR-182 and Foxo3a in patients with bladder cancer correlate with prognosis. Int. J. Clin. Exp. Pathol. 2019, 12, 4193. [Google Scholar] [PubMed]
- Yang, K.; Tang, H.; Ding, M.; Guo, Y.; Kai, K.; Xiao, J.; Shen, Y.; Miao, S.; Zhou, R. Expression of miR-195 and MEK1 in patients with bladder cancer and their relationship to prognosis. Int. J. Clin. Exp. Pathol. 2019, 12, 843. [Google Scholar]
- Li, D.; Hao, X.; Song, Y. An integrated analysis of key microRNAs, regulatory pathways and clinical relevance in bladder cancer. OncoTargets Ther. 2018, 11, 3075–3085. [Google Scholar] [CrossRef]
- Martins, E.P.; Vieira de Castro, J.; Fontes, R.; Monteiro-Reis, S.; Henrique, R.; Jerónimo, C.; Costa, B.M. Relevance of HOTAIR rs920778 and rs12826786 Genetic Variants in Bladder Cancer Risk and Survival. Cancers 2024, 16, 434. [Google Scholar] [CrossRef]
- Novikova, E.L.; Kulakova, M.A. There and back again: Hox clusters use both DNA strands. J. Dev. Biol. 2021, 9, 28. [Google Scholar] [CrossRef]
- Xia, W.; Chen, C.; Zhang, M.R.; Zhu, L.N. LncRNA PCAT6 aggravates the progression of bladder cancer cells by targeting miR-513a-5p. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 9908–9914. [Google Scholar]
- Zhang, S.; Du, L.; Wang, L.; Jiang, X.; Zhan, Y.; Li, J.; Yan, K.; Duan, W.; Zhao, Y.; Wang, L.; et al. Evaluation of serum exosomal Lnc RNA-based biomarker panel for diagnosis and recurrence prediction of bladder cancer. J. Cell. Mol. Med. 2019, 23, 1396–1405. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Miao, P.; Ming, Y.; Tao, J.; Shen, H. Circ-ZFR promotes progression of bladder cancer by upregulating WNT5A via sponging miR-545 and miR-1270. Front. Oncol. 2021, 10, 596623. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Chen, R.X.; Wei, W.S.; Li, Y.H.; Feng, Z.H.; Tan, L.; Chen, J.W.; Yuan, G.J.; Chen, S.L.; Guo, S.J.; et al. PRMT5 Circular RNA Promotes Metastasis of Urothelial Carcinoma of the Bladder through Sponging miR-30c to Induce Epithelial–Mesenchymal Transition. Clin. Cancer Res. 2021, 27, 2664. [Google Scholar] [CrossRef] [PubMed]

| Authors | Test Name | Target Biomarker | Method | Study Material | Diagnostic Performance | Study Group (n) | Race/ Nationality |
|---|---|---|---|---|---|---|---|
| Bang et al. (2024) [56] | EarlyTest BCD | PENK | qPCR | Voided urine | SN = 81% SP = 91.5% | 210 (21 BC, 189 non-BC based on cystoscopy) | Korean, American |
| Pharo et al. (2022) [54] | BladMetrix | 8 methylated gene regions | ddPCR | Urinary exfoliated cell DNA | SN = 92.1% SP = 93.3% | 273 (with gross haematuria) (93 BC) | European |
| Piatti et al. (2021) [55] | Bladder CARE | DNA methylation in TRNA-Cys, SIM2, and NKX1 | Methylation-sensitive restriction enzymes with qPCR | Urine | SN = 93.5% SP = 92.6% | 213 (77 BC, 136 non-BC) | Caucasian, Asian, African American, Hispanic |
| Steinbach et al. (2020) [57] | GynTect | DNA methylation in ASTN1, DLX1, ITGA4, RXFP3, SOX17, and ZNF671 | GynTect Assay | Urine | SN = 60% SP = 96.7% | 70 (40 BC, 15 BPH, 15 Urolithiasis) | German |
| Witjes et al. (2018) [52] | Bladder EpiCheck | 15 methylated gene regions | qPCR | Urinary exfoliated cell DNA | SN = 68.2% SP = 88% | 353 (UC) | Caucasian |
| Feber et al. (2017) [53] | UroMark | 150 CpG loci methylation | Targeted bisulfite sequencing | Urinary exfoliated cell DNA | SN = 98% SP = 97% | 274 (107 BC, 167 non-BC) | English |
| Authors | Study Biomarker | Methylation Status | Study Material | Diagnostic Performance | Study Group (n) | Race/ Nationality |
|---|---|---|---|---|---|---|
| Zhang et al. (2024) [64] | TWIST1, VIM | Methylation level analysis | Urine cell sediment | SN = 78% SP = 83% | 231 (77 BC, 81 other urological malignancies, 19 benign disease, 26 UTUC, 28 healthy) | China |
| Jiang et al. (2024) [61] | ZNF671, OTX1, IRF8 | Hypermethylation | Voided urine | SN = 75% SP = 90.9% | 114 (61 BC, 53 non-BC) | Taiwan |
| Fang et al. (2022) [59] | PCDH17, POU4F2, PENK | Hypermethylation | Urine cell sediment | SN = 87% SP = 97% | 207 (107 BC, 100 non-BC) | China |
| Hentschel et al. (2022) [60] | GHSR, MAL | Hypermethylation | Urine pellet | SN = 80% SP = 93% | 208 (108 BC, 34 benign haematuria, 43 other benign urological conditions, 23 healthy) | Dutch |
| Deng et al. (2022) [65] | DMRTA2 | Hypermethylation | Urine | SN = 82.9% SP = 92.5% | 520 (79 BC, 107 other malignancies, 22 benign tumours of bladder8 recurring cancers, 304 healthy | China |
| Ruan et al. (2021) [66] | ONECUT2, VIM | Hypermethylation | Urine | Cohort 1: SN = 88.1% SP = 89.7% Cohort 2: SN = 91.2% SP = 85.7% | Cohort 1: 98 (patients suspected of BC) (59 BC, 39 non-BC); Cohort 2: 174 (haematuria patients) (34 BC, 140 non-BC) | China |
| Wu et al. (2020) [62] | ONECUT2, HOXA9, PCDH17, POU4F2 | Hypermethylation | Urine | SN = 90.5% SP = 73.2% | 111 (53 BC, 58 non-BC) | China |
| Chen et al. (2020) [63] | OTX1, SOX1-OT | Methylation level analysis | Urine | SN = 91.7% SP = 77.3% | 175 (109 BC, 66 benign diseases) | China |
| Guo et al. (2018) [67] | VIM, CDH1, SALL3, THBS1, TMEFF2, GDF15 | Methylation level analysis | Voided urine | SN = 89% SP = 74% | 473 (217 UC, 256 controls) | China |
| Roperch et al. (2016) [68] | SEPTIN9, SLIT2 | Hypermethylation | Urine | SN = 91% SP = 71.4% | 272 (167 NMIBC, 105 controls) | France |
| HS3ST2, SEPTIN9, SLIT2 | Hypermethylation | Urine | SN = 90.4% SP = 75.2% | 272 (167 NMIBC, 105 controls) | France | |
| HS3ST2, SEPTIN9 | Hypermethylation | Urine | SN = 90.4% SP = 72.4% | 272 (167 NMIBC, 105 controls) | France | |
| Dahmcke et al. (2016) [69] | CCNA1, ONECUT2, BCL2, EOMES, SALL3, VIM | Methylation level analysis | Urine | SN = 89.9% SP = 88.6% | 475 (99 BC, 376 controls) | Denmark |
| Wang et al. (2016) [70] | RUNX3, RARP | Methylation level analysis | Urine | SN = 96.6% SP = 88.9% | 139 (112 BC, 10 healthy, 17 glandular cystitis) | China |
| Authors | Study Biomarker | Biomarker Change | Biomarker Targets | Study Material | Diagnostic Performance | Study group (n) | Race/ Nationality |
|---|---|---|---|---|---|---|---|
| miRNAs | |||||||
| Lu et al. (2024) [82] | miR-221-5p, miR-181a-5p, miR-15a-5p, miR-222-3p | Aberrantly expressed | Not specified | Serum | SN = 82.1% SP = 85.7% | 224 (112 BC, 112 controls) | China |
| Yu et al. (2023) [73] | miR-27b, miR-381-3p, miR-451a | Overexpression: miR-27b Underexpression: miR-381-3p, miR-451a | SMAD4, FOXO1 | Serum | SN = 86.7% SP = 77.4% | 224 (112 BC, 112 healthy) | China |
| Mamdouh et al. (2023) [91] * Zhang et al. (2018) [92] ** Zhang et al. (2015) [93] | miR-200 | Overexpression | Not specified | Urine and tissue | Tissue: SN = 93.3% SP = 100% Urine: SN = 62.2% SP = 100% | 136 (111 BC, 25 healthy) | Egyptian |
| miR-145 | N-cadherin * | Tissue: SN = 80% SP = 100% Urine: SN = 78.4% SP = 91.7% | |||||
| miR-21 | maspin, VEGF-C ** | Tissue: SN = 73.3% SP = 80% Urine: SN = 83.3% SP = 100% | |||||
| SurezCabrera et al. (2022) [74] | miR-145, miR-182 | Overexpression (miR-145), Underexpression (miR-182) | FCS1 | Urine | SN = 93% SP = 86% | 82 (40 BC, 42 controls) | European |
| Wang et al. (2021) [71] | miR-20a-5p, miR-92a-3p, miR-17-5p | Overexpression | p21, PTEN | Serum | SN = 90.4% SP = 94.4% | 164 (74BC, 90 healthy) | China |
| El-Shal et al. (2021) [75] | miR-96-5p, miR-183-5p | Overexpression | FOXO, KRAS PDCD4 | Voided urine | miR-96 alone: SN = 80.4% SP = 91.8% miR-183 alone: SN = 78.4% SP = 81.6% both combined: SN = 88.2% SP = 87.8% | 100 (51 BC, 21 benign bladder lesions, 28 healthy) | Egyptian |
| Lin et al. (2021) [79] | miR-516a-5p, miR-93-5p | Overexpression | miR-516a-5p (not specified) miR-93-5p: PEDF, EGFR, FoxO pathway, PI3K-Akt pathway, BTG2 | Midstream urine | miR-93-5p alone: SN = 74.1% SP = 90.2% miR-516a-5p alone: SN = 72.9% SP = 89.9% both combined: SN = 85.2% SP = 82.4% | 104 (53 BC, 51 healthy) | China |
| Piao et al. (2019) [94] | miR-6124, miR-4511 | Aberrantly expressed | Not specified | urine | SN = 91.5% SP = 76.2% (ratio miR-6124 to miR-4511) | 543 (326 BC, 174 haematuria, non-BC pyuria) | Republic of Korea |
| Chen (2019) [81] | miR-101 | Underexpression | Not specified | serum | SN = 82% SP = 80.9% | 232 (122 BC, 110 healthy) | China |
| Usuba et al. (2018) [95] | miR-6087, miR-6724-5p, miR-3960, miR-1343-5p, miR-1185-1-3p miR-6831-5p, miR-4695-5p combined | miR-4695-5p: no significant change Underexpression: miR6087, miR-3960, miR-1343-5p Overexpression: miR-6724-5p, miR-1185-1-3p, miR-6831-5p | Not specified | serum | SN = 95% SP = 87% | 972 (392 BC, 100 non-BC, 480 other cancers) | Japan |
| Huang et al. (2018) [96] | miR-20a | Overexpression | Not specified | Urine | SN = 72.1% SP = 87.5% | 166 (80 NMIBC, 86 healthy) | China |
| Matsuzaki et al. (2017) [80] | miR-21-5p | Overexpression | Not specified | Urine | SN = 75% SP = 98% | 60 (24 controls, 36 UC) | Japan |
| Urquidi et al. (2016) [97] | 25 miRNAs combined | Aberrantly expressed | Not specified | Midstream urine | SN = 87% SP = 100% | 121 (61 cases, 60 controls) | USA |
| Zhang et al. (2016) [98] | miR-155 | Overexpression | APC, VHL, PIK3R1, MLH1 | Voided urine | SN = 80.2% SP = 84.6% | 324 (162 NIMBC, 86 cystitis, 76 healthy) | China |
| Motawi et al. (2016) [99] | miR-143, miR-92a | Underexpression | Not specified | Plasma | SN = 94.3%, SP = 86.6% | 132 (70BC, 62 healthy) | Egyptian |
| lncRNAs | |||||||
| Gao et al. (2024) [87] | RMRP | Overexpression | Not specified | Urine | SN = 83% SP = 70% (RT-qPCR) SN = 95% SP = 92.5% (RT-RAA- CRISPR/Cas12a) | 339 (229 BC, 110 benign lesions) | China |
| Liu et al. (2023) [100] | SNHG16 | Overexpression | possibly Wnt/β-catenin pathway | Urine | SN = 61.9% SP = 83.3% | 84 (42 BC, 42 Healthy) | China |
| Chen et al. (2022) [88] | TERC | Overexpression | Not specified | Urine | SN = 78.7% SP = 77.8% | 152 (89 BLCA, 63 Healthy) | China |
| Bian et al. (2022) [101] | MKLN1-AS | Overexpression | Not specified | Urine | SN = 79.1% SP = 67.4% | 92 (46 BC, 46 Controls) | China |
| TALAM1 | Not specified | SN = 90.1% SP = 55.8% | |||||
| TTN-AS1 | Not specified | SN = 76.7% SP = 76.7% | |||||
| UCA1 | PI3K-Akt-mTOR pathway, GLS2, HMGB1, p21 | SN = 90.7% SP = 90.7% | |||||
| Sarfi et al. (2021) [102] | TUG-1 | Overexpression | Not specified | Voided urine | SN = 76.7% SP = 77.8% | 40 (30 NMIBC, 10 Controls) | Iran |
| Yu et al. (2020) [86] | UCA-1-201, HOTAIR, HYMA1, MALAT1 | Overexpression: HOTAIR, MALAT1 Underexpression: UCA-1-201, HYMA1 | Not specified | Urine | SN = 93.3% SP = 96.7% | 120 (60 Urocystitis, 60 NMIBC) | China |
| Zhan et al. (2018) [85] | MALAT1, PCAT-1, SPRY4-IT1 | Overexpression | Not specified | Urine | SN = 62.5% SP = 85.6% | 208 (104 BC, 104 healthy) | China |
| other ncRNAs | |||||||
| Yang et al. (2024) [89] | circRNA-0071196 | Overexpression | CIT, miR-19b-3p, | Urine | SN = 87.5% SP = 85% | 70 (40 BUC, 30 non-BUC) | China |
| Luo et al. (2023) [103] | circRNA CCT3 | Overexpression | PP2A, miR-135a-5p | Plasma | SN = 86.1% SP = 60% | 125 (85 BC, 40 healthy) | China |
| Song et al. (2020) [90] | hsa_circ0137439 | Overexpression | miR-142-5p | Urine | SN = 87.9 SP = 80.1% (BC vs. Controls) SN = 88.6% SP = 73.5% (NMIBC vs. MIBC) | 146 (62 NMIBC, 54 MIBC, 30 controls) | China |
| Tang et al. (2017) [104] | circASXL1 | Overexpression | Not specified | Tumour tissue | SN = 68.6% SP = 76.9% | 61 pairs of tumour tissue and adjacent normal mucosa | China |
| Authors | Study Biomarker | Methylation Status | Study Material | Assessed Study Endpoints (Associated Change) | Study Group (n) | Race/ Nationality |
|---|---|---|---|---|---|---|
| Kim et al. (2024) [133] | PTK2 | hypermethylation | Tissue | OS (↓) | BC patients (n = 275) healthy donors (n = 10) | Republic of Korea |
| Zhang et al. (2024) [129] | AHNAK | hypermethylation | Tissue | OS (↓) | BC patients (n = 812) | China |
| Koukourikis et al. (2023) [135] | DAPK | hypermethylation | Urine | OS (↓) | BC patients (n = 414) healthy donors (n = 10) | Greece |
| El Azzouzi et al. (2022) [136] | TWIST1 | hypermethylation | Tissue | PFS (↓) | BC patients (n = 70) | Morocco |
| Takagi et al. (2022) [130] | CALN1 | hypomethylation | Tissue | PFS (↓) | BC patients (n = 82) | Japan |
| Zhang et al. (2022) [134] | FASLG, PRKCZ | hypermethylation | Tissue | PFS (↓) | BC patients (n = 408) healthy donors (n = 14) | China |
| Chen et al. (2022) [131] | BLCAP | hypomethylation | Peripheral blood | PFS (↓) OS (↓) | BC patients (n = 603) | USA |
| Guo et al. (2021) [137] | TNFAIP8L3 | hypomethylation | Tissue | PFS (↓) OS (↓) | BC patients (n = 357) | China |
| Zhou et al. (2021) [138] | PTPRN2 | hypomethylation | Tissue | OS (↓) | BC patients (n = 399) | China |
| Guo et al. (2021) [137] | APC | hypermethylation | Tissue | PFS (↓) | BC patients (n = 357) | China |
| Zhang et al. (2020) [128] | KRT8 | hypomethylation | Tissue | OS (↓) | BC patients (n = 41) healthy donors (n = 35) | China |
| Zhang et al. (2018) [128] | CDH1 | hypomethylation | Tissue | OS (↓) | BC patients (n = 167) healthy donors (n = 13) | China |
| Zhan et al. (2017) [126] | RASSF1A | hypermethylation | Tissue | PFS (↓) OS (↓) | BC patients (n = 389) | China |
| Shivakumar et al. (2017) [127] | NACC2 | hypomethylation | Tissue | OS (↓) | BC patients (n = 403) | USA |
| Yoon et al. (2016) [125] | RSPH9 | hypermethylation | Tissue | PFS (↓) | BC patients (n = 128) healthy donors (n = 8) | Republic of Korea |
| Authors | Study Biomarker | Biomarker Change | Biomarker Targets | Study Material | Assessed Study Endpoints (Associated Change) | Study Group (n) | Race/ Nationality |
|---|---|---|---|---|---|---|---|
| miRNAs | |||||||
| Zhenhai et al. (2023) [165] | miR-205-3p | Downregulated | GLO1 | Tissue | PFS (↓) OS (↓) | BC patients (n = 35) | China |
| Hao et al. (2023) [166] | miR-30c-5p | Downregulated | PRC1 | Tissue | OS (↓) | BC patients (n = 445) | China |
| Awadalla et al. (2022) [167] | miR-138 | Downregulated | HIF1α | Tissue | CSS (↓) | BC patients (n = 157) | Egypt |
| Awadalla et al. (2022) [167] | miR-let-7a | Downregulated | WNT7A | Tissue | CSS (↓) | BC patients (n = 157) | Egypt |
| Yerukala et al. (2022) [155] | miR-652-5p | Upregulated | KCNN3 | Tissue | OS (↓) | BC patients (n = 106) | USA |
| Zhang et al. (2021) [168] | miR-432 | Downregulated | SMARCA5 | Tissue | OS (↓) | BC patients (n = 156) | China |
| Yang et al. (2021) [152] Borkowska et al. (2019) [149] | miR-10a-5p | Upregulated | FGFR3 | Plasma | OS (↓) | BC patients (n = 88) healthy donors (n = 36) | China |
| Andrew et al. (2019) [147] | miR-26b-5p | Downregulated | PLOD2 | Tissue | PFS (↓) | BC patients (n = 231) | Lebanon |
| Spagnuolo et al. (2020) [169] | miR-let-7c-5p | Upregulated | HRAS | Urine | PFS (↓) | BC patients (n = 57) healthy donors (n = 20) | Italy |
| Borkowska et al. (2019) [149] | miR-21-5p | Upregulated | TP53 | Tissue | OS (↓) | BC patients (n = 55) healthy donors (n = 30) | Poland |
| Braicu et al. (2019) [170] | miR-143 | Downregulated | TP53 | Tissue | OS (↓) | BC patients (n = 409) healthy donors (n = 19) | Romania |
| Chen et al. (2019) [171] | miR-182 | Upregulated | FOXO3a | Tissue | OS (↓) | BC patients (n = 60) healthy donors (n = 20) | China |
| Juracek et al. (2019) [150] Ding et al. (2019) [151] | miR-34a-3p | Downregulated | PTEN | Tissue | OS (↓) | BC patients (n = 78) | Czech Republic |
| Yin et al. (2019) [154] | miR-185 | Upregulated | ITGB5 | Tissue | OS (↓) | BC patients (n = 408) healthy donors (n = 19) | China |
| Yang et al. (2019) [172] | miR-195 | Downregulated | MEK1 | Tissue | OS (↓) | BC patients (n = 80) healthy donors (n = 30) | China |
| Li et al. (2018) [173] | miR-145 | Upregulated | CDK4 | Tissue | OS (↓) | BC patients (n = 127) | China |
| Mitash et al. (2017) [139] | miR-21 | Upregulated | PTEN | Tissue | RFS (↓) | BC patients (n = 31) | India |
| Zhang et al. (2016) [98] | miR-155 | Upregulated | APC | Urine | RFS (↓) PFS (↓) | BC patients (n = 162) healthy donors (n = 86) | China |
| Chen et al. (2016) [140] | miR-133b | Downregulated | EGFR | Tissue | PFS (↓) OS (↓) | BC patients (n = 146) | China |
| lncRNAs | |||||||
| Martins et al. (2024) [174] Novikova et al. (2021) [175] | lncRNA-HOX | Upregulated | HOXD | Peripheral blood | OS (↓) | BC patients (n = 106) healthy donors (n = 199) | Portugal |
| Chen et al. (2022) [88] | lncRNA-TERC | Upregulated | TERT | Tissue | OS (↓) | BC patients (n = 89) healthy donors (n = 63) | China |
| Chen et al. (2021) [157] | lncRNA-ELNAT1 | Upregulated | UBC9 | Urine | OS (↓) | BC patients (n = 242) | China |
| Liang et al. (2021) [158] | lncRNA-MALAT1 | Upregulated | MDM2 | Tissue | OS (↓) | BC patients (n = 90) | China |
| Zheng et al. (2021) [159] | lncRNA-BCYRN1 | Upregulated | WNT5A | Tissue | OS (↓) | BC patients (n = 210) | China |
| Jiao et al. (2020) [143] | lncRNA-SNHG16 | Upregulated | CCL5 | Tissue | OS (↓) | BC patients (n = 1148) | China |
| Chen et al. (2020) [144] | lncRNA-LNMAT2 | Upregulated | PROX1 | Tissue | OS (↓) | BC patients (n = 266) | China |
| Xia et al. (2020) [176] | lncRNA-PCAT6 | Upregulated | MIR513A1 | Tissue | OS (↓) | BC patients (n = 21) healthy donors (n = 21) | China |
| Zhang et al. (2019) [177] | lncRNA-UBC1 | Upregulated | PRC2 | Serum | RFS (↓) | BC patients (n = 260) healthy donors (n = 260) | China |
| Avgeris et al. (2018) [142] | lncRNA-GAS5 | Downregulated | CDK6 | Tissue | PFS (↓) | BC patients (n = 176) | Greece |
| other ncRNAs | |||||||
| Luo et al. (2023) [103] | circRNA-CCT3 | Upregulated | PP2A | Urine | RFS (↓) OS (↓) | BC patients (n = 85) healthy donors (n = 40) | China |
| Luo et al. (2021) [178] | circRNA-ZFR | Upregulated | WNT5A | Tissue | OS (↓) | BC patients (n = 60) | China |
| Chen et al. (2021) [179] | circRNA-PRMT5 | Upregulated | SNAIL1 | Tissue | OS (↓) | BC patients (n = 119) | China |
| Zhu et al. (2021) [164] | circRNA-EHBP1 | Upregulated | TGFβR1 | Tissue | OS (↓) | BC patients (n = 186) | China |
| Xie et al. (2020) [161] Zhang et al. (2019) [162] | circRNA-HIPK3 | Downregulated | SOX4 | Tissue | OS (↓) | BC patients (n = 68) | China |
| Su et al. (2020) [163] | circRNA-RIP2 | Downregulated | SMAD3 | Tissue | OS (↓) | BC patients (n = 58) | China |
| Lin et al. (2019) [145] | circRNA-LPAR1 | Downregulated | WNT5A | Tissue | DSS (↓) | BC patients (n = 125) | China |
| Tang et al. (2017) [104] | circRNA-ASXL1 | Upregulated | TP53 | Tissue | OS (↓) | BC patients (n = 61) | China |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaszek, N.; Bogdanowicz, A.; Siwiec, J.; Starownik, R.; Kwaśniewski, W.; Mlak, R. Epigenetic Biomarkers as a New Diagnostic Tool in Bladder Cancer—From Early Detection to Prognosis. J. Clin. Med. 2024, 13, 7159. https://doi.org/10.3390/jcm13237159
Jaszek N, Bogdanowicz A, Siwiec J, Starownik R, Kwaśniewski W, Mlak R. Epigenetic Biomarkers as a New Diagnostic Tool in Bladder Cancer—From Early Detection to Prognosis. Journal of Clinical Medicine. 2024; 13(23):7159. https://doi.org/10.3390/jcm13237159
Chicago/Turabian StyleJaszek, Natalia, Alicja Bogdanowicz, Jan Siwiec, Radosław Starownik, Wojciech Kwaśniewski, and Radosław Mlak. 2024. "Epigenetic Biomarkers as a New Diagnostic Tool in Bladder Cancer—From Early Detection to Prognosis" Journal of Clinical Medicine 13, no. 23: 7159. https://doi.org/10.3390/jcm13237159
APA StyleJaszek, N., Bogdanowicz, A., Siwiec, J., Starownik, R., Kwaśniewski, W., & Mlak, R. (2024). Epigenetic Biomarkers as a New Diagnostic Tool in Bladder Cancer—From Early Detection to Prognosis. Journal of Clinical Medicine, 13(23), 7159. https://doi.org/10.3390/jcm13237159







