Nailfold Capillaroscopy Changes in Patients with Idiopathic Inflammatory Myopathies
Abstract
1. Introduction
2. Materials and Methods
2.1. Inclusion Criteria
- Diagnosed with an IIM per the 2017 EULAR/ACR classification criteria.
2.2. Exclusion Criteria
- Overlap syndrome with other autoimmune diseases (e.g., systemic sclerosis, systemic lupus erythematosus (SLE), Sjögren’s syndrome).
- Other causes of myopathy (e.g., endocrine, metabolic, infectious, neuropathic, or drug-induced).
- Absence of NFC.
3. Results
3.1. Clinical and Laboratory Findings
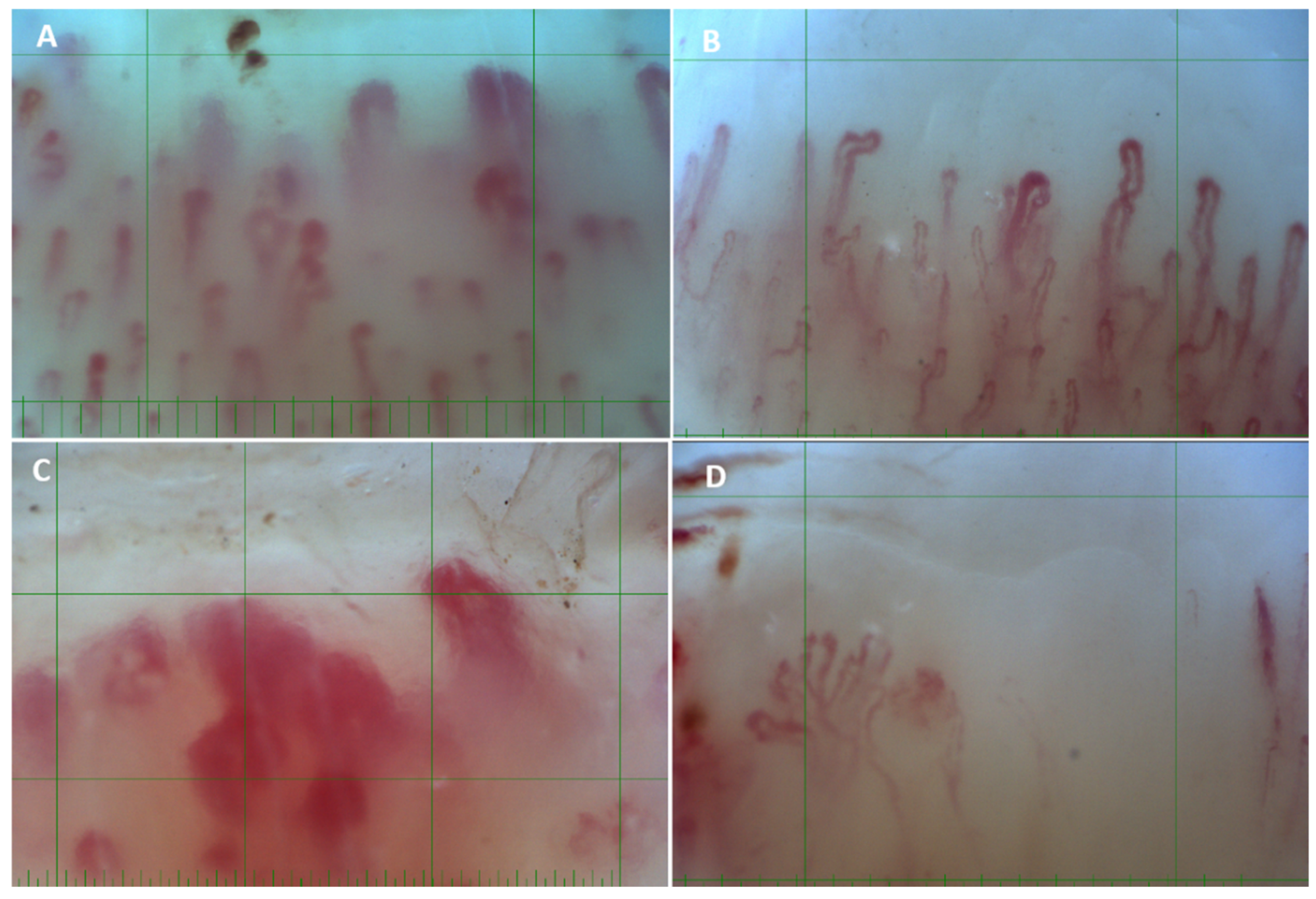
3.2. Capillaroscopic Findings
3.3. Association between NFC and Clinical/Laboratory Findings
4. Discussion
5. Limitation of This Study
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bohan, A.; Peter, J.B. Polymyositis and dermatomyositis (first of two parts). N. Engl. J. Med. 1975, 292, 344–347. [Google Scholar] [CrossRef] [PubMed]
- Damoiseaux, J.; Vulsteke, J.B.; Tseng, C.W.; Platteel, A.C.M.; Piette, Y.; Shovman, O.; Bonroy, C.; Hamann, D.; De Langhe, E.; Musset, L.; et al. Autoantibodies in idiopathic inflammatory myopathies: Clinical associations and laboratory evaluation by mono- and multispecific immunoassays. Autoimmun. Rev. 2019, 18, 293–305. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, I.E.; Tjarnlund, A.; Bottai, M.; Werth, V.P.; Pilkington, C.; de Visser, M.; Alfredsson, L.; Amato, A.A.; Barohn, R.J.; Liang, M.H.; et al. 2017 European league against rheumatism/American College of Rheumatology Classification Criteria for Adult and Juvenile Idiopathic Inflammatory Myopathies and Their Major Subgroups. Arthritis Rheumatol. 2017, 69, 2271–2282. [Google Scholar] [CrossRef] [PubMed]
- Smith, V.; Herrick, A.L.; Ingegnoli, F.; Damjanov, N.; De Angelis, R.; Denton, C.P.; Distler, O.; Espejo, K.; Foeldvari, I.; Frech, T.; et al. Standardisation of nailfold capillaroscopy for the assessment of patients with Raynaud’s phenomenon and systemic sclerosis. Autoimmun. Rev. 2020, 19, 102458. [Google Scholar] [CrossRef] [PubMed]
- Cutolo, M.; Sulli, A.; Pizzorni, C.; Accardo, S. Nailfold videocapillaroscopy assessment of microvascular damage in systemic sclerosis. J. Rheumatol. 2000, 27, 155–160. [Google Scholar]
- Piette, Y.; Reynaert, V.; Vanhaecke, A.; Bonroy, C.; Gutermuth, J.; Sulli, A.; Cutolo, M.; Smith, V. Standardised interpretation of capillaroscopy in autoimmune idiopathic inflammatory myopathies: A structured review on behalf of the EULAR study group on microcirculation in Rheumatic Diseases. Autoimmun. Rev. 2022, 6, 103087. [Google Scholar] [CrossRef]
- Maricq, H.R.; LeRoy, E.C.; D’Angelo, W.A.; Medsger, T.A., Jr.; Rodnan, G.P.; Sharp, G.C.; Wolfe, J.F. Diagnostic potential of in vivo capillary microscopy in scleroderma andrelated disorders. Arthritis Rheumatol. 1980, 23, 183–189. [Google Scholar] [CrossRef]
- Grassi, W.; Del Medico, P. (Eds.) Atlas of Capillaroscopy, 1st ed.; EDRA: Perignano, Italy, 2004; pp. 234–235. [Google Scholar]
- Barth, Z.; Witczak, B.N.; Flatø, B.; Koller, A.; Sjaastad, I.; Sanner, H. Assessment of Microvascular Abnormalities by Nailfold Capillaroscopy in Juvenile Dermatomyositis After Medium- to Long-Term Followup. Arthritis Care Res. 2018, 70, 768–776. [Google Scholar] [CrossRef]
- Mugii, N.; Hasegawa, M.; Matsushita, T.; Hamaguchi, Y.; Horie, S.; Yahata, T.; Inoue, K.; Someya, F.; Fujimoto, M.; Takehara, K. Association between nail-fold capillary findings and disease activity in dermatomyositis. Rheumatology 2011, 50, 1091–1098. [Google Scholar] [CrossRef]
- Christen-Zaech, S.; Seshadri, R.; Sundberg, J.; Paller, A.S.; Pachman, L.M. Persistent association of nailfold capillaroscopy changes and skin involvement over thirty-six months with duration of untreated disease in patients with juvenile dermatomyositis. Arthritis Rheumatol. 2008, 58, 571–576. [Google Scholar] [CrossRef]
- Cutolo, M.; Sulli, A.; Secchi, M.E.; Olivieri, M.; Pizzorni, C. The contribution of capillaroscopy to the differential diagnosis of connective autoimmune diseases. Best Pract. Res. Clin. Rheumatol. 2007, 21, 1093–1108. [Google Scholar] [CrossRef] [PubMed]
- Shenavandeh, S.; Rashidi, F. Nailfold capillaroscopy changes with disease activity in patients with inflammatory myositis including overlap myositis, pure dermatomyositis, and pure polymyositis. Reumatologia 2022, 60, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Manfredi, A.; Sebastiani, M.; Cassone, G.; Pipitone, N.; Giuggioli, D.; Colaci, M.; Salvarani, C.; Ferri, C. Nailfold capillaroscopic changes in dermatomyositis and polymyositis. Clin. Rheumatol. 2015, 34, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Kubo, S.; Todoroki, Y.; Nakayamada, S.; Nakano, K.; Satoh, M.; Nawata, A.; Satoh, Y.; Miyagawa, I.; Saito, K.; Smith, V.; et al. Significance of nailfold videocapillaroscopy in patients with idiopathic inflammatory myopathies. Rheumatology 2019, 58, 120–130. [Google Scholar] [CrossRef]
- Russo, R.A.; Katsicas, M.M. Clinical characteristics of children with Juvenile Systemic Sclerosis: Follow-up of 23 patients in a single tertiary center. Pediatr. Rheumatol. Online J. 2007, 5, 6. [Google Scholar] [CrossRef]
- Ganczarczyk, M.L.; Lee, P.; Armstrong, S.K. Nailfold capillary microscopy in polymyositis and dermatomyositis. Arthritis Rheumatol. 1988, 31, 116–119. [Google Scholar] [CrossRef]
- Shenavandeh, S.; Zarei Nezhad, M. Association of nailfold capillary changes with disease activity, clinical and laboratory findings in patients with dermatomyositis. Med. J. Islam. Repub. Iran 2015, 29, 233. [Google Scholar]
- Sebastiani, M.; Triantafyllias, K.; Manfredi, A.; González-Gay, M.A.; Palmou-Fontana, N.; Cassone, G.; Drott, U.; Delbrück, C.; Rojas-Serrano, J.; Bertolazzi, C.; et al. Nailfold Capillaroscopy Characteristics of Antisynthetase Syndrome and Possible Clinical Associations: Results of a Multicenter International Study. J. Rheumatol. 2019, 46, 279–284. [Google Scholar] [CrossRef]
- Bergman, R.; Sharony, L.; Schapira, D.; Nahir, M.A.; Balbir-Gurman, A. The handheld dermatoscope as a nail-fold capillaroscopic instrument. Arch. Dermatol. 2003, 139, 1027–1030. [Google Scholar] [CrossRef]
- Ingegnoli, F.; Zeni, S.; Gerloni, V.; Fantini, F. Capillaroscopic observations in childhood rheumatic diseases and healthy controls. Clin. Exp. Rheumatol. 2005, 23, 905–911. [Google Scholar]
- Nascif, A.K.; Terreri, M.T.; Len, C.A.; Andrade, L.E.; Hilario, M.O. Inflammatory myopathies in childhood: Correlation between nailfoldcapillaroscopy findings and clinical and laboratory data. J. Pediatr. 2006, 82, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Pavlov-Dolijanovic, S.; Damjanov, N.; Stojanovic, R.; Vujasinovic Stupar, N.; Stanisavljevic, D. Scleroderma pattern of nailfold capillary changes as predictive value for the development of a connective tissue disease: A follow-up study of 3029 patients with primary Raynaud’s phenomenon. Rheumatol. Int. 2012, 32, 3039–3045. [Google Scholar] [CrossRef] [PubMed]
| DM n (%) | PM n (%) | ADM n (%) | JDM n (%) | |
|---|---|---|---|---|
| 37 | 35 | 13 | 5 | |
| Age | 51.76 ± 12.88 | 50.46 ± 13.53 | 51.45 ± 14.64 | 10.42 ± 3.91 |
| Female | 24 (64.9) | 28 (80) | 10 (76.9) | 3 (60) |
| Muscle weakness | 37 (100) | 35 (100) | 0 | 4 (80) |
| Gottron’s sign | 27 (73) | 0 | 10 (76.9) | 4 (80) |
| Heliotrope rash | 21 (56.8) | 0 | 9 (69.2) | 4 (80) |
| Periungual erythema | 13 (35.1) | 0 | 6 (46.2) | 3 (60) |
| Mechanic hand | 13 (35.1) | 12 (34.3) | 2 (15.4) | 0 |
| Raynaud’s phenomena | 30 (81.1) | 14 (40) | 10 (76.9) | 4 (80) |
| ILD | 14 (37.8) | 12 (34.3) | 8 (61.5) | 0 |
| Arthralgia | 21 (56.8) | 24 (68.6) | 7 (53.8) | 3 (60) |
| Arthritis | 12 (32.4) | 15 (42.9) | 4 (30.8) | 2 (40) |
| ANA | 18 (48.6%) | 17 (51.4) | 6 (46.2) | 2 (40) |
| Anti Jo1 | 12 (32.4%) | 16 (45.7) | 4 (30.8) | 1 (20) |
| Myositis blot | 15/23 (Mi2 3, PL7 2, PL12 4, Ro52 4, U1RNP 2) | 15/21, PL7 5, PL12 2, EJ 1, Ro52 7 | 5/12 (PL7 1, Pl12 1, Mi2 1, Ro52 1, MDA5 1) | 0 |
| Feature | Variable | CON. | DM | PM | ADM | JDM | |||
|---|---|---|---|---|---|---|---|---|---|
| N (%) | N (%) | * p | N (%) | * p | N (%) | * p | N (%) | ||
| Mean capillary density | Reduced | 1 (2.8) | 23 (62.1) | <0.001 | 12 (34.3) | 0.006 | 11 (84.6) | <0.001 | 2 (40) |
| Sign. reduced | 0 | 3(8.1) | 1 (2.8) | 1 (7.7) | 2 (40) | ||||
| Morphology | Abnormal | 5 (14.3) | 31 (83.8) | <0.001 | 16 (45.7) | 0.008 | 12 (92.3) | <0.001 | 5 (100) |
| Dimension | Enlarged | 1 (2.8) | 28 (75.7) | <0.001 | 1 (2.8) | 0.421 | 9 (69.2) | <0.001 | 4 (80) |
| Giant | 0 | 25 (67.6) | 1 (2.8) | 9 (69.2) | 1 (20) | ||||
| Micro- hemorr. | Rare | 4 (11.4) | 19 (51.4) | <0.001 | 5 (14.3) | 0.721 | 7 (53.8) | 0.03 | 4 (80) |
| Frequent | 0 | 7 (18.9) | 0 | 0 | 0 | ||||
| SBVP | Present | 1 (2.8) | 3 (8.1) | 0.325 | 18 (51.4) | <0.001 | 0 | 0.781 | 2 (40) |
| NFC pattern | Normal | 32 (91.4) | 1 (2.7) | <0.001 | 17 (48.6) | <0.001 | 0 | <0.001 | 0 |
| Nonspecific | 3 (8.6) | 6 (16.2) | 17 (48.6) | 3 (23.1) | 1 | ||||
| SD pattern | 0 | 30 (81.1) | 1 (2.9) | 10 (76.9) | 4 | ||||
| DM n (%) | PM | ADM | JDM | |
|---|---|---|---|---|
| Early pattern | 12 (32.4%) | 1 (2.9%) | 2 (15.4%) | 1 (20%) |
| Active pattern | 15 (40.5%) | 0 | 7 (53.8%) | 2 (40%) |
| Late pattern | 3 (8.1%) | 0 | 1 (7.7%) | 1 (20%) |
| Capillaroscopy Pattern | p Value | ||||||
|---|---|---|---|---|---|---|---|
| Normal | Nonspecific Pattern | Scleroderma Pattern | |||||
| N | % | N | % | N | % | ||
| Sex (F) | 15 | 83.3% | 19 | 70.4% | 30 | 66.7% | 0.417 |
| Smoking | 8 | 44.4% | 9 | 33.3% | 16 | 35.6% | 0.733 |
| Muscle weakness | 18 | 100.0% | 24 | 88.9% | 35 | 77.8% | 0.056 |
| Gottron’s sign | 0 | 0.0% | 5 | 18.5% | 36 | 80.0% | <0.001 |
| Heliotrope rash | 1 | 5.6% | 6 | 22.2% | 27 | 60.0% | <0.001 |
| Periungual erythema | 0 | 0.0% | 2 | 7.4% | 20 | 44.4% | <0.001 |
| Mechanic hand | 2 | 11.1% | 12 | 44.4% | 13 | 28.9% | 0.056 |
| Raynaud’s phenomena | 6 | 33.3% | 18 | 66.7% | 38 | 84.4% | <0.001 |
| ILD | 1 | 5.6% | 11 | 40.7% | 23 | 51.1% | 0.004 |
| Arthralgia | 8 | 44.4% | 22 | 81.5% | 25 | 55.6% | 0.025 |
| Arthritis | 2 | 11.1% | 15 | 55.6% | 16 | 35.6% | 0.010 |
| ANA-positive | 9 | 50.0% | 14 | 51.9% | 21 | 46.7% | 0.908 |
| Anti Jo1 | 4 | 22.2% | 16 | 59.3% | 13 | 28.9% | 0.013 |
| CK IU/L AS ± SD | 3663.6 ± 3257.8 | 3570.7 ± 5460 | 3198 ± 3996 | 0.336 * | |||
| AST IU/L AS ± SD | 147.2 ± 95.7 | 198 ± 337.1 | 143 ± 181.3 | 0.286 * | |||
| ALT IU/L AS ± SD | 128.2 ± 111.1 | 106.6 ± 122.3 | 96.7 ± 111.2 | 0.169 * | |||
| CRP mg/L AS ± SD | 19.3 ± 15.1 | 24.0 ± 24.2 | 21.6 ± 24.6 | 0.786 * | |||
| Age (years) AS ± SD | 54.8 ± 13.6 | 47.2 ± 15.4 | 47.6 ± 17.0 | 0.223 * | |||
| Authors | N of Patients | Diagnosis | Capillaroscopy Finding |
|---|---|---|---|
| Ganczarczyk et al. [17] | 35 | PM 19 DM 16 | Avascular zones were observed in 94% of patients with DM and 42% of patients with PM. Enlarged capillaries were present in 56% of DM patients and 21% of PM patients. |
| Mugii et al. [10] | 50 | DM | Scleroderma capillaroscopic patterns were found in 74% of patients: early in 24%, active in 46%, and late in 4%. |
| Shenvandan et al. [13] | 150 | DM 81 ADM 25 JDM 25 PM 19 | Among DM patients, 72.8% exhibited a scleroderma capillaroscopic pattern, while 27.16% had nonspecific changes. Among patients with ADM, 76% had a scleroderma capillaroscopic pattern, 16% had nonspecific changes, and 8% had normal findings. In JDM patients, 72% had a scleroderma capillaroscopic pattern and 27% had nonspecific changes. In PM patients, 42% showed a scleroderma capillaroscopic pattern, 47% had nonspecific changes, and 11% had normal findings. |
| Bergman et al. [20] | 11 | DM | Overall, 63% of patients had scleroderma capillaroscopic changes, while 36% had normal findings. |
| Ingegnoli et al. [21] | 8 | JDM | Scleroderma changes were present in 63% of the subjects, with no patients showing normal capillaroscopic findings. |
| Nascif et al. [22] | 13 | JDM | In the active phase, 92% (12/13) of patients had scleroderma capillaroscopic changes. |
| Pavlov Dolijanovic et al. [23] | 26 | PM 19 DM 7 | Among PM patients, 68.42% had normal findings and 31.57% had nonspecific changes. In DM patients, 71.1% had a scleroderma pattern, while 28.9% had nonspecific changes. |
| Manfredi et al. [14] | 52 | DM 29 PM 23 | A scleroderma capillaroscopic pattern was observed in 69% of DM patients, while none of the PM patients had a scleroderma pattern. |
| Shenvandan et al. [18] | 27 | DM | A scleroderma pattern was present in 88.9% of patients, with an early form in 3%, active form in 33%, and late form in 52%. Nonspecific changes were found in 11%. |
| Kubo et al. [15] | 70 | DM 52 PM 18 | A scleroderma capillaroscopic pattern was observed in 65.4% of patients with dermatomyositis (DM) and 27.8% of patients with polymyositis (PM). |
| Our study | 90 | DM 37 PM 35 ADM 13 JDM 5 | DM patients: 81.1% exhibited scleroderma capillaroscopic changes, 16.2% had nonspecific changes, and 2.7% had normal findings. In PM patients: 2.9% had scleroderma capillaroscopic changes, 48.6% had nonspecific changes, and 48.6% had normal findings. For patients with amyopathic dermatomyositis (ADM): 76.9% had scleroderma capillaroscopic changes, while 23.1% had nonspecific changes. In JDM patients: 80% had scleroderma capillaroscopic changes and 20% had nonspecific changes. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bogojevic, M.; Markovic Vlaisavljevic, M.; Medjedovic, R.; Strujic, E.; Pravilovic Lutovac, D.; Pavlov-Dolijanovic, S. Nailfold Capillaroscopy Changes in Patients with Idiopathic Inflammatory Myopathies. J. Clin. Med. 2024, 13, 5550. https://doi.org/10.3390/jcm13185550
Bogojevic M, Markovic Vlaisavljevic M, Medjedovic R, Strujic E, Pravilovic Lutovac D, Pavlov-Dolijanovic S. Nailfold Capillaroscopy Changes in Patients with Idiopathic Inflammatory Myopathies. Journal of Clinical Medicine. 2024; 13(18):5550. https://doi.org/10.3390/jcm13185550
Chicago/Turabian StyleBogojevic, Milan, Milica Markovic Vlaisavljevic, Rifat Medjedovic, Elvira Strujic, Dragana Pravilovic Lutovac, and Slavica Pavlov-Dolijanovic. 2024. "Nailfold Capillaroscopy Changes in Patients with Idiopathic Inflammatory Myopathies" Journal of Clinical Medicine 13, no. 18: 5550. https://doi.org/10.3390/jcm13185550
APA StyleBogojevic, M., Markovic Vlaisavljevic, M., Medjedovic, R., Strujic, E., Pravilovic Lutovac, D., & Pavlov-Dolijanovic, S. (2024). Nailfold Capillaroscopy Changes in Patients with Idiopathic Inflammatory Myopathies. Journal of Clinical Medicine, 13(18), 5550. https://doi.org/10.3390/jcm13185550





