Increased Risk for Pulmonary Embolism among Patients with Ankylosing Spondylitis—Results from a Large Database Analysis
Abstract
1. Introduction
2. Methods
2.1. Settings
2.2. Sample and Design
2.3. Study Variables
2.4. Statistical Analysis
3. Results
3.1. Study Population
3.2. Medication Use
3.3. Prevalence of PE
3.4. Incident PE Events
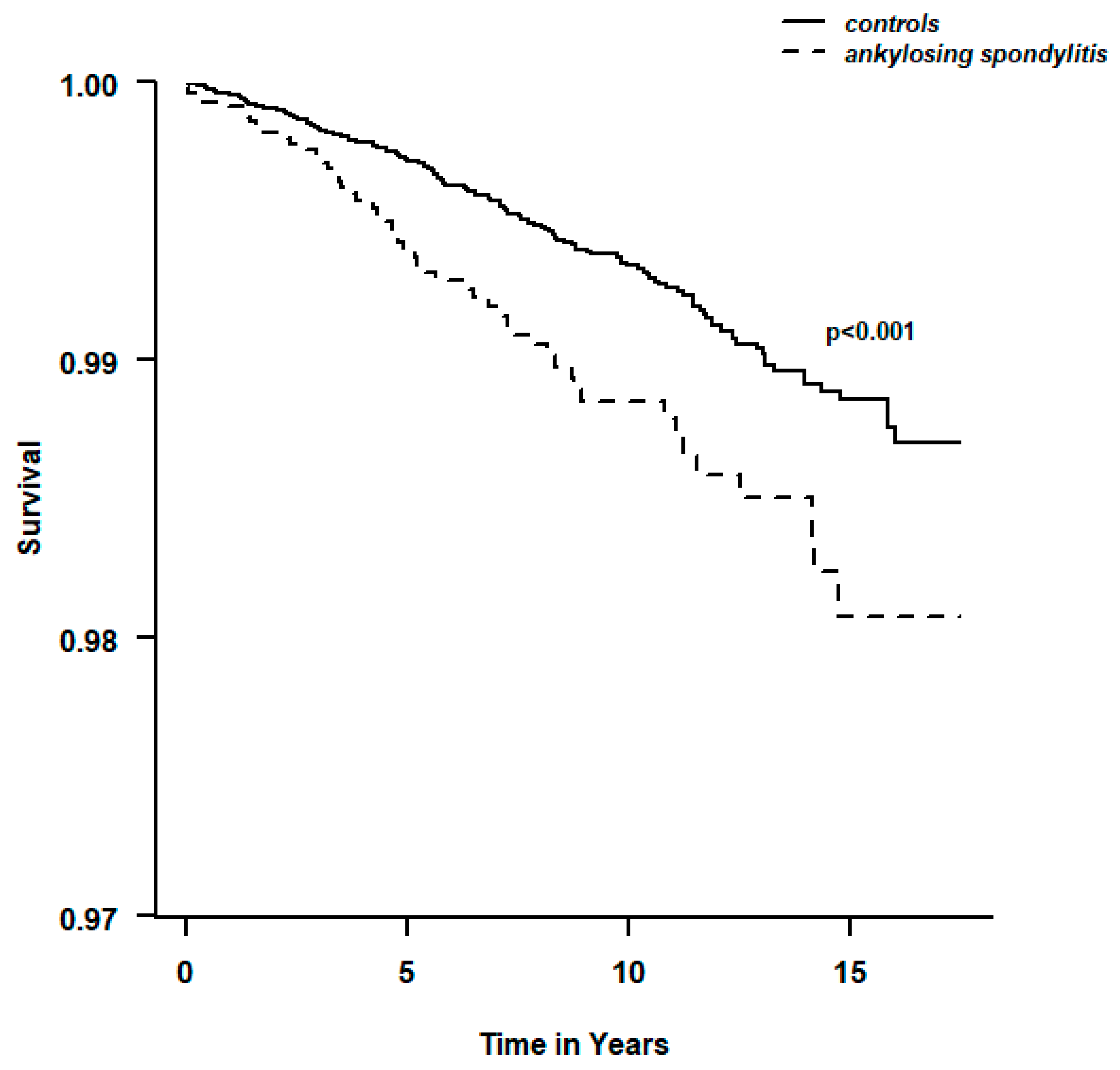
3.5. Risk of PE
3.6. Subgroup Analysis
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhu, W.; He, X.; Cheng, K.; Zhang, L.; Chen, D.; Wang, X.; Qiu, G.; Cao, X.; Weng, X. Ankylosing Spondylitis: Etiology, Pathogenesis, and Treatments. Bone Res. 2019, 7, 22. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, F.; Helliwell, P. Ankylosing Spondylitis: A Review. EMJ 2017, 2, 134–139. [Google Scholar] [CrossRef]
- Proft, F.; Poddubnyy, D. Ankylosing Spondylitis and Axial Spondyloarthritis: Recent Insights and Impact of New Classification Criteria. Ther. Adv. Musculoskelet. 2018, 10, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Rusman, T.; Van Bentum, R.E.; Van Der Horst-Bruinsma, I.E. Sex and Gender Differences in Axial Spondyloarthritis: Myths and Truths. Rheumatology 2020, 59 (Suppl. 4), iv38–iv46. [Google Scholar] [CrossRef]
- López-Medina, C.; Dougados, M.; Ruyssen-Witrand, A.; Moltó, A. Evaluation of Concomitant Peripheral Arthritis in Patients with Recent Onset Axial Spondyloarthritis: 5-Year Results from the DESIR Cohort. Arthritis Res. Ther. 2019, 21, 139. [Google Scholar] [CrossRef] [PubMed]
- Taurog, J.D.; Chhabra, A.; Colbert, R.A. Ankylosing Spondylitis and Axial Spondyloarthritis. N. Engl. J. Med. 2016, 374, 2563–2574. [Google Scholar] [CrossRef]
- Van Der Horst-Bruinsma, I.E.; Nurmohamed, M.T.; Landewé, R.B.M. Comorbidities in Patients with Spondyloarthritis. Rheum. Dis. Clin. N. Am. 2012, 38, 523–538. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Wendling, D.; Botteman, M.F.; Carter, J.A.; Rao, S.; Cifaldi, M. Clinical and Economic Burden of Extra-Articular Manifestations in Ankylosing Spondylitis Patients Treated with Anti-Tumor Necrosis Factor Agents. J. Med. Econ. 2012, 15, 1054–1063. [Google Scholar] [CrossRef]
- Kahn, S.R.; De Wit, K. Pulmonary Embolism. N. Engl. J. Med. 2022, 387, 45–57. [Google Scholar] [CrossRef]
- Turetz, M.; Sideris, A.; Friedman, O.; Triphathi, N.; Horowitz, J. Epidemiology, Pathophysiology, and Natural History of Pulmonary Embolism. Semin. Interv. Radiol. 2018, 35, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Zöller, B.; Li, X.; Sundquist, J.; Sundquist, K. Risk of Pulmonary Embolism in Patients with Autoimmune Disorders: A Nationwide Follow-up Study from Sweden. Lancet 2012, 379, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Gendelman, O.; Feifel, A.J.; Tsur, A.M.; Comanhester, D.; Cohen, A.D.; Amital, H. Increased Risk of Venous Thromboembolism among Patients with Familial Mediterranean Fever. J. Thromb. Thrombolysis 2022, 54, 669–674. [Google Scholar] [CrossRef]
- Ungprasert, P.; Srivali, N.; Kittanamongkolchai, W. Ankylosing Spondylitis and Risk of Venous Thromboembolism: A Systematic Review and Meta-Analysis. Lung India 2016, 33, 642. [Google Scholar] [CrossRef]
- Ben-Shabat, N.; Shabat, A.; Watad, A.; Kridin, K.; Bragazzi, N.L.; McGonagle, D.; Comaneshter, D.; Cohen, A.D.; Amital, H. Mortality in Ankylosing Spondylitis According to Treatment: A Nationwide Retrospective Cohort Study of 5,900 Patients from Israel. Arthritis Care Res. 2022, 74, 1614–1622. [Google Scholar] [CrossRef] [PubMed]
- Ramagopalan, S.V.; Wotton, C.J.; Handel, A.E.; Yeates, D.; Goldacre, M.J. Risk of Venous Thromboembolism in People Admitted to Hospital with Selected Immune-Mediated Diseases: Record-Linkage Study. BMC Med. 2011, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Johannesdottir, S.A.; Schmidt, M.; Horváth-Puhó, E.; Sørensen, H.T. Autoimmune Skin and Connective Tissue Diseases and Risk of Venous Thromboembolism: A Population-based Case-control Study. J. Thromb. Haemost. 2012, 10, 815–821. [Google Scholar] [CrossRef] [PubMed]
- Aviña-Zubieta, J.A.; Chan, J.; De Vera, M.; Sayre, E.C.; Choi, H.; Esdaile, J. Risk of Venous Thromboembolism in Ankylosing Spondylitis: A General Population-Based Study. Ann. Rheum. Dis. 2019, 78, 480–485. [Google Scholar] [CrossRef]
- Bai, Y.; Yue, Q.-M.; Sun, H.; Guo, S.-D.; Wang, Z.-Z.; Zhong, P.; Wei, X.-Y.; Sun, L.; Liu, Y.; Shi, X.-B.; et al. Prevalence and Sex- and Age-Related Risk of Pulmonary Embolism in in-Hospital Patients with Atrial Fibrillation: A Multicenter Retrospective Study from China. Ann. Transl. Med. 2020, 8, 1558. [Google Scholar] [CrossRef]
- Stein, P.D.; Huang, H.; Afzal, A.; Noor, H.A. Incidence of Acute Pulmonary Embolism in a General Hospital. Chest 1999, 116, 909–913. [Google Scholar] [CrossRef]
- Mader, R. Atypical Clinical Presentation of Ankylosing Spondylitis. Semin. Arthritis Rheum. 1999, 29, 191–196. [Google Scholar] [CrossRef]
- Ungprasert, P.; Srivali, N.; Wijarnpreecha, K.; Charoenpong, P.; Knight, E.L. Non-Steroidal Anti-Inflammatory Drugs and Risk of Venous Thromboembolism: A Systematic Review and Meta-Analysis. Rheumatology 2015, 54, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Molander, V.; Bower, H.; Frisell, T.; Delcoigne, B.; Di Giuseppe, D.; Askling, J. Venous Thromboembolism with JAK Inhibitors and Other Immune-Modulatory Drugs: A Swedish Comparative Safety Study among Patients with Rheumatoid Arthritis. Ann. Rheum. Dis. 2023, 82, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Verma, I.; Krishan, P.; Syngle, A. Predictors of Atherosclerosis in Ankylosing Spondylitis. Rheumatol. Ther. 2015, 2, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Toussirot, E. The Risk of Cardiovascular Diseases in Axial Spondyloarthritis. Current Insights. Front. Med. 2021, 8, 782150. [Google Scholar] [CrossRef]
- Bĕlohlávek, J.; Dytrych, V.; Linhart, A. Pulmonary Embolism, Part I: Epidemiology, Risk Factors and Risk Stratification, Pathophysiology, Clinical Presentation, Diagnosis and Nonthrombotic Pulmonary Embolism. Exp. Clin. Cardiol. 2013, 18, 129–138. [Google Scholar] [PubMed]
| Characteristics | Patients with AS (n = 5825) | Controls (n = 28,356) | p-Value |
|---|---|---|---|
| Age, mean ± SD; median | 50.0 ± 16; 49.3 | 49.8 ± 16; 49.1 | NS |
| Men, n (%) | 3701 (63.5) | 17,975 (63.4.) | NS |
| Arab ethnicity, n (%) | 999 (17.2) | 4914, 17.3) | NS |
| Smoking history, n (%) | 2032 (34.9) | 9379 (33.1) | <0.01 |
| Recruitment periods, n (%) | NS | ||
| 2002–2007 | 1754 (30.1) | 8576 (30.2) | |
| 2008–2013 | 2103 (36.1) | 10,212 (36.0) | |
| 2014–2018 | 1968 (33.8) | 9568 (33.7) | |
| Socioeconomic status a, n (%) | NS | ||
| Low | 787 (14.4) | 4082 (15.4) | |
| Medium | 3837 (70.3) | 18,570 (69.9) | |
| High | 834 (17.6) | 3907 (14.7) | |
| Body Mass Index b n (%) | <0.001 | ||
| <18.5 kg/m2 | 97 (2.3) | 469 (2.5) | |
| 18.5–24.9 kg/m2 | 1413 (33.9) | 7029 (37.4) | |
| 25–29.9 kg/m2 | 1428 (34.2) | 6788 (36.1) | |
| ≥30 kg/m2 | 1235 (29.6) | 4499 (23.9) | |
| AS Treatment, n (%) | |||
| Use of any TNFi | 1760 (30.2) | ||
| Use of any DMARD | 1687 (29.0) | ||
| Use of any NSAID | 5648 (97.0) | ||
| TNFi only | 737 (12.7) | - | |
| DMARDs only | 664 (11.4) | - | |
| Sulfasalazine only | 372 (6.4) | ||
| Methotrexate only | 153 (2.6) | ||
| Sulfasalazine and Methotrexate | 550 (9.4) | ||
| NSAIDs only | 3249 (55.8) | - | |
| DMARDs and TNFi | 1023 (17.6) | - | |
| TNFi and Methotrexate | 664 (11.4) | ||
| TNFi and Sulfasalazine | 770 (13.2) | ||
| TNFi, Methotrexate, and Sulfasalazine | 411 (7.1) | ||
| PE before AS/Index date | 24 (0.4) | 60 (0.2) | <0.01 |
| Mean time between AS/Index to PE diagnosis, years | 5.7 ± 4 | 6.7 ± 8 | NS |
| Outcomes | Variables | AS (n = 5825) | Controls (n = 28,355) |
|---|---|---|---|
| Pulmonary Embolism | Events, n (%) | 52 (0.9) | 152 (0.5) |
| Follow-up time, median (IQR) | 7.35 (3.4–11.4) | 7.50 (3.5–11.7) | |
| Cumulative patient’s years | 44,784 | 222,059 | |
| Incidence rate per 10,000 person-years, (95%CI) | 11.6 (8.7–15.2) | 6.8 (5.8–8.0) | |
| Unadjusted HR (95%CI) | 1.70 (1.24 to 2.33) | reference | |
| Multivariate HR (95%CI) | 1.59 (1.04 to 2.42) | reference |
| Subgroup | AS n (%) | Controls n (%) | HR (95% CI) | p-Value |
|---|---|---|---|---|
| Age | ||||
| ≤49 years | 9 (0.3) | 16 (0.1) | 2.79 (1.23 to 6.31) | 0.014 |
| >49 years | 43 (1.5) | 136 (1.0) | 1.58 (1.12 to 2.23) | 0.009 |
| Sex | ||||
| Men | 31 (0.8) | 88 (0.5) | 1.74 (1.16 to 2.63) | 0.008 |
| Women | 21 (1.0) | 64 (0.6) | 1.65 (1.01 to 2.70) | 0.047 |
| AS Treatment | ||||
| TNFi | 5 (0.4) | 17 (0.3) | 1.44 (0.53 to 3.91) | 0.472 |
| DMARDs only | 6 (0.7) | 22 (0.5) | 1.37 (0.55 to 3.38) | 0.494 |
| NSAIDs only | 41 (1.2) | 114 (0.7) | 1.79 (1.25 to 2.55) | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gendelman, O.; Simon, N.; Ben-Shabat, N.; Patt, Y.S.; McGonagle, D.; Cohen, A.D.; Amital, H.; Watad, A. Increased Risk for Pulmonary Embolism among Patients with Ankylosing Spondylitis—Results from a Large Database Analysis. J. Clin. Med. 2024, 13, 2790. https://doi.org/10.3390/jcm13102790
Gendelman O, Simon N, Ben-Shabat N, Patt YS, McGonagle D, Cohen AD, Amital H, Watad A. Increased Risk for Pulmonary Embolism among Patients with Ankylosing Spondylitis—Results from a Large Database Analysis. Journal of Clinical Medicine. 2024; 13(10):2790. https://doi.org/10.3390/jcm13102790
Chicago/Turabian StyleGendelman, Omer, Neta Simon, Niv Ben-Shabat, Yonatan Shneor Patt, Dennis McGonagle, Arnon Dov Cohen, Howard Amital, and Abdulla Watad. 2024. "Increased Risk for Pulmonary Embolism among Patients with Ankylosing Spondylitis—Results from a Large Database Analysis" Journal of Clinical Medicine 13, no. 10: 2790. https://doi.org/10.3390/jcm13102790
APA StyleGendelman, O., Simon, N., Ben-Shabat, N., Patt, Y. S., McGonagle, D., Cohen, A. D., Amital, H., & Watad, A. (2024). Increased Risk for Pulmonary Embolism among Patients with Ankylosing Spondylitis—Results from a Large Database Analysis. Journal of Clinical Medicine, 13(10), 2790. https://doi.org/10.3390/jcm13102790







