Impaired Awareness in People with Severe Alcohol-Related Cognitive Deficits Including Korskoff’s Syndrome: A Network Analysis
Abstract
1. Introduction
- I.
- How does impaired awareness relate to cognitive, behavioral, physical, and social functioning and to QoL in people with KS or other alcohol-related cognitive disorders living in Dutch LTCFs?
- II.
- What other variables play a central role in the network?
2. Materials and Methods
2.1. Participants and Setting
2.2. Measurements
2.2.1. Sociodemographic and Clinical Characteristics
2.2.2. Awareness
2.2.3. Neuropsychiatric Symptoms
2.2.4. Cognitive, Physical, and Social Functioning
2.2.5. Cognitive Functioning
2.2.6. Physical Functioning
2.2.7. Social Functioning
2.2.8. Quality of Life
2.2.9. Network and Statistical Analysis
3. Results
3.1. Demographic and Clinical Characteristics
3.2. Network Analysis
3.3. Associations with PCRS
3.4. Associations with AES
4. Discussion
4.1. Awareness and Apathy
4.2. Apathy
4.3. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Arts, N.J.; Walvoort, S.J.; Kessels, R.P. Korsakoff’s syndrome: A critical review. Neuropsychiatr. Dis. Treat. 2017, 13, 2875–2890. [Google Scholar] [CrossRef] [PubMed]
- Blansjaar, B.A.; Horjus, M.C.; Nijhuis, H.G.J. Prevalence of the Korsakoff syndrome in The Hague, The Netherlands. Acta Psychiatr. Scand. 1987, 75, 604–607. [Google Scholar] [CrossRef] [PubMed]
- Kopelman, M.D. The Korsakoff syndrome. Br. J. Psychiatry. 1995, 166, 154–173. [Google Scholar] [CrossRef] [PubMed]
- Kopelman, M.D. What is the Korsakoff syndrome?—A paper in tribute to Prof Alwyn Lishman. Cogn. Neuropsychiatry 2022, 27, 296–313. [Google Scholar] [CrossRef]
- Pitel, A.L.; Eustache, F.; Beaunieux, H. Component processes of memory in alcoholism: Pattern of compromise and neural substrates. Handb. Clin. Neurol. 2014, 125, 211–225. [Google Scholar]
- Brion, M.; Pitel, A.-L.; Beaunieux, H.; Maurage, P. Revisiting the continuum hypothesis: Toward an in-depth exploration of executive functions in korsakoff syndrome. Front. Hum. Neurosci. 2014, 8, 498. [Google Scholar] [CrossRef]
- Gerridzen, I.J.; Hertogh, C.; Depla, M.F.; Veenhuizen, R.B.; Verschuur, E.M.L.; Joling, K.J. Neuropsychiatric Symptoms in People with Korsakoff Syndrome and Other Alcohol-Related Cognitive Disorders Living in Specialized Long-Term Care Facilities: Prevalence, Severity, and Associated Caregiver Distress. J. Am. Med. Dir. Assoc. 2018, 19, 240–247. [Google Scholar] [CrossRef]
- Gerridzen, I.J.; Moerman-van den Brink, W.G.; Depla, M.F.; Verschuur, E.M.; Veenhuizen, R.B.; van der Wouden, J.C.; Hertogh, C.M.; Joling, K.J. Prevalence and severity of behavioural symptoms in patients with Korsakoff syndrome and other alcohol-related cognitive disorders: A systematic review. Int. J. Geriatr. Psychiatry 2017, 32, 256–273. [Google Scholar] [CrossRef]
- Chen, L.-F.; Lin, C.-E. Wernicke’s Encephalopathy Mimicking an Acute Psychotic Disorder. J. Neuropsychiatry Clin. Neurosci. 2012, 24, E15–E16. [Google Scholar] [CrossRef]
- Ahmed, S.; Akadiri, T.V.; Ata, S.; Ayub, S. An Unusual Presentation of Catatonia in Non-alcoholic Wernicke Encephalopathy. Cureus 2021, 13, e12537. [Google Scholar] [CrossRef]
- van Dam, M.J.; van Meijel, B.; Postma, A.; Oudman, E. Health problems and care needs in patients with Korsakoff’s syndrome: A systematic review. J. Psychiatr. Ment. Health Nurs. 2020, 27, 460–481. [Google Scholar] [PubMed]
- Oudman, E.; van Dam, M.; Postma, A. Social and emotional loneliness in Korsakoff’s syndrome. Cogn. Neuropsychiatry 2018, 23, 307–320. [Google Scholar] [CrossRef] [PubMed]
- Drost, R.; Postma, A.; Oudman, E. Cognitive and affective theory of mind in Korsakoff’s syndrome. Acta Neuropsychiatr. 2018, 31, 128–134. [Google Scholar] [CrossRef]
- Oudman, E.; Zwart, E. Quality of life of patients with Korsakoff’s syndrome and patients with dementia: A cross-sectional study. J. Am. Med. Dir. Assoc. 2012, 13, 778–781. [Google Scholar] [CrossRef] [PubMed]
- Egger, J.I.; Wester, A.J.; De Mey, H.R.; Derksen, J.J. Korsakoff’s syndrome on the MMPI-2. Acta Neuropsychiatr. 2002, 14, 231–236. [Google Scholar] [CrossRef]
- Mograbi, D.C.; Morris, R.G. Anosognosia. Cortex 2018, 103, 385–386. [Google Scholar] [CrossRef]
- Sunderaraman, P.; Cosentino, S. Integrating the Constructs of Anosognosia and Metacognition: A Review of Recent Findings in Dementia. Curr. Neurol. Neurosci. Rep. 2017, 17, 27. [Google Scholar] [CrossRef]
- Gerridzen, I.J.; Joling, K.J.; Depla, M.F.; Veenhuizen, R.B.; Verschuur, E.M.L.; Twisk, J.W.R.; Hertogh, C. Awareness and its relationships with neuropsychiatric symptoms in people with Korsakoff syndrome or other alcohol-related cognitive disorders living in specialized nursing homes. Int. J. Geriatr. Psychiatry 2019, 34, 836–845. [Google Scholar] [CrossRef]
- Sitek, E.J.; Thompson, J.C.; Craufurd, D.; Snowden, J.S. Unawareness of deficits in Huntington’s disease. J. Huntingtons Dis. 2014, 3, 125–135. [Google Scholar] [CrossRef]
- Pennington, C.; Duncan, G.; Ritchie, C. Altered awareness of cognitive and neuropsychiatric symptoms in Parkinson’s disease and Dementia with Lewy Bodies: A systematic review. Int. J. Geriatr. Psychiatry 2021, 36, 15–30. [Google Scholar] [CrossRef]
- Orfei, M.; Caltagirone, C.; Spalletta, G. The Evaluation of Anosognosia in Stroke Patients. Cerebrovasc. Dis. 2009, 27, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Mograbi, D.C.; Huntley, J.; Critchley, H. Self-awareness in Dementia: A Taxonomy of Processes, Overview of Findings, and Integrative Framework. Curr. Neurol. Neurosci. Rep. 2021, 21, 69. [Google Scholar] [CrossRef] [PubMed]
- Aalten, P.; Van Valen, E.; Clare, L.; Kenny, G.; Verhey, F. Awareness in dementia: A review of clinical correlates. Aging Ment. Health 2005, 9, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Starkstein, S.E.; Jorge, R.; Mizrahi, R.; Robinson, R.G. A diagnostic formulation for anosognosia in Alzheimer’s disease. J. Neurol. Neurosurg. Psychiatry 2006, 77, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Clare, L.; Marková, I.S.; Roth, I.; Morris, R.G. Awareness in Alzheimer’s disease and associated dementias: Theoretical framework and clinical implications. Aging Ment. Health 2011, 15, 936–944. [Google Scholar] [CrossRef]
- Turró-Garriga, O.; Garre-Olmo, J.; Vilalta-Franch, J.; Conde-Sala, J.L.; de Gracia Blanco, M.; López-Pousa, S. Burden associated with the presence of anosognosia in Alzheimer’s disease. Int. J. Geriatr. Psychiatry 2013, 28, 291–297. [Google Scholar] [CrossRef]
- Starkstein, S.E.; Jorge, R.; Mizrahi, R.; Adrian, J.; Robinson, R.G. Insight and danger in Alzheimer’s disease. Eur. J. Neurol. 2007, 14, 455–460. [Google Scholar] [CrossRef]
- Borgatti, S.P.; Mehra, A.; Brass, D.J.; Labianca, G. Network analysis in the social sciences. Science 2009, 323, 892–895. [Google Scholar] [CrossRef]
- Ma’ayan, A. Introduction to Network Analysis in Systems Biology. Sci. Signal. 2011, 4, tr5. [Google Scholar] [CrossRef]
- Hevey, D. Network analysis: A brief overview and tutorial. Health Psychol. Behav. Med. 2018, 6, 301–328. [Google Scholar] [CrossRef]
- Borsboom, D. A network theory of mental disorders. World Psychiatry 2017, 16, 5–13. [Google Scholar] [CrossRef]
- Borsboom, D.; Cramer, A.O. Network Analysis: An Integrative Approach to the Structure of Psychopathology. Annu. Rev. Clin. Psychol. 2013, 9, 91–121. [Google Scholar] [CrossRef]
- Epskamp, S.; Borsboom, D.; Fried, E.I. Estimating psychological networks and their accuracy: A tutorial paper. Behav. Res. Methods 2018, 50, 195–212. [Google Scholar] [CrossRef] [PubMed]
- Schumacher-Kuiper, M.; Loon, A.; Peeters, C.; Ekkel, M.; Hertogh, C.; Veenhuizen, R. Is There a Relation Between Caregiver Burden and Cognitive Dysfunction in Huntington’s Disease? J. Psychosoc. Rehabil. Ment. Health 2021, 8, 61–71. [Google Scholar] [CrossRef]
- Gerridzen, I.J.; Goossensen, M.A. Patients with Korsakoff syndrome in nursing homes: Characteristics, comorbidity, and use of psychotropic drugs. Int. Psychogeriatr. 2014, 26, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Prigatano, G.P.; Fordyce, D.J. Neuropsychological Rehabilitation after Brain Injury; Johns Hopkins University Press: Baltimore, MA, USA, 1986. [Google Scholar]
- Kaufer, D.I.; Cummings, J.L.; Ketchel, P.; Smith, V.; MacMillan, A.; Shelley, T.; Lopez, O.L.; DeKosky, S.T. Validation of the NPI-Q, a brief clinical form of the Neuropsychiatric Inventory. J. Neuropsychiatry Clin. Neurosci. 2000, 12, 233–239. [Google Scholar] [CrossRef]
- Zuidema, S.; Smalbrugge, M.; Bil, W.; Geelen, R.; Kok, R.; Luijendijk, H.; van der Stelt, I.; van Strien, A.; Vink, M.; Vreeken, H. Multidisciplinary Guideline Problem Behaviour in Dementia; Verenso: Utrecht, The Netherlands, 2018. [Google Scholar]
- Lueken, U.; Seidl, U.; Völker, L.; Schweiger, E.; Kruse, A.; Schröder, J. Development of a short version of the Apathy Evaluation Scale specifically adapted for demented nursing home residents. Am. J. Geriatr. Psychiatry 2007, 15, 376–385. [Google Scholar] [CrossRef]
- Leontjevas, R.; Evers-Stephan, A.; Smalbrugge, M.; Pot, A.M.; Thewissen, V.; Gerritsen, D.L.; Koopmans, R.T. A Comparative Validation of the Abbreviated Apathy Evaluation Scale (AES-10) With the Neuropsychiatric Inventory Apathy Subscale Against Diagnostic Criteria of Apathy. J. Am. Med. Dir. Assoc. 2012, 13, 308.e1–308.e6. [Google Scholar] [CrossRef] [PubMed]
- Hirdes, J.P.; Ljunggren, G.; Morris, J.N.; Frijters, D.H.; Finne Soveri, H.; Gray, L.; Björkgren, M.; Gilgen, R. Reliability of the interRAI suite of assessment instruments: A 12-country study of an integrated health information system. BMC Health Serv. Res. 2008, 8, 277. [Google Scholar] [CrossRef]
- Morris, J.N.; Fries, B.E.; Mehr, D.R.; Hawes, C.; Phillips, C.; Mor, V.; Lipsitz, L.A. MDS Cognitive Performance Scale. J. Gerontol. 1994, 49, M174–M182. [Google Scholar] [CrossRef]
- Morris, J.N.; Fries, B.E.; Morris, S.A. Scaling ADLs within the MDS. J. Gerontol. A Biol. Sci. Med. Sci. 1999, 54, M546–M553. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.Y.; Kim, H. The Revised Index for Social Engagement in Long-Term Care Facilities: A Psychometric Study. J. Nurs. Res. 2017, 25, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Priebe, S.; Huxley, P.; Knight, S.; Evans, S. Application and results of the Manchester Short Assessment of Quality of Life (MANSA). Int. J. Soc. Psychiatry 1999, 45, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Azur, M.J.; Stuart, E.A.; Frangakis, C.; Leaf, P.J. Multiple imputation by chained equations: What is it and how does it work? Int. J. Methods Psychiatr. Res. 2011, 20, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Peeters, C.F.; Bilgrau, A.E.; van Wieringen, W.N. rags2ridges: A One-Stop-Shop for Graphical Modeling of High-Dimensional Precision Matrices. arXiv 2020, arXiv:2010.05619. [Google Scholar] [CrossRef]
- Schäfer, J.; Strimmer, K. A Shrinkage Approach to Large-Scale Covariance Matrix Estimation and Implications for Functional Genomics. Stat. Appl. Genet. Mol. Biol. 2005, 4, 32. [Google Scholar] [CrossRef]
- Fruchterman, T.M.J.; Reingold, E.M. Graph drawing by force-directed placement. Softw. Pract. Exp. 1991, 21, 1129–1164. [Google Scholar] [CrossRef]
- Alexander, C.M.; Martyr, A.; Gamble, L.D.; Savage, S.A.; Quinn, C.; Morris, R.G.; Clare, L. Does awareness of condition help people with mild-to-moderate dementia to live well? BMC Geriatr. 2021, 21, 1–15. [Google Scholar] [CrossRef]
- Conde-Sala, J.L.; Reñé-Ramírez, R.; Turró-Garriga, O.; Gascón-Bayarri, J.; Juncadella-Puig, M.; Moreno-Cordón, L.; Viñas-Diez, V.; Garre-Olmo, J. Clinical differences in patients with Alzheimer’s disease according to the presence or absence of anosognosia: Implications for perceived quality of life. J. Alzheimers Dis. 2013, 33, 1105–1116. [Google Scholar] [CrossRef]
- de Groot, A.M.; Ekkel, M.R.; Smalbrugge, M.; Hertogh, C.M.P.M.; Veenhuizen, R.B. Impaired Awareness of Functional Deficits in People with Huntington’s Disease Living in Specialized Long Term Care Facilities; European Geriatric Medicine Society: London, UK, 2022. [Google Scholar]
- Derouesné, C.; Thibault, S.; Lagha-Pierucci, S.; Baudouin-Madec, V.; Ancri, D.; Lacomblez, L. Decreased awareness of cognitive deficits in patients with mild dementia of the Alzheimer type. Int. J. Geriatr. Psychiatry 1999, 14, 1019–1030. [Google Scholar] [CrossRef]
- Starkstein, S.E. Anosognosia in Alzheimer’s disease: Diagnosis, frequency, mechanism and clinical correlates. Cortex 2014, 61, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Belfort, T.; Simões, P.; de Sousa, M.F.B.; Santos, R.L.; Barbeito, I.; Torres, B.; Dourado, M.C.N. The Relationship Between Social Cognition and Awareness in Alzheimer Disease. J. Geriatr. Psychiatry Neurol. 2018, 31, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Blansjaar, B.A.; Takens, H.; Zwinderman, A.H. The course of alcohol amnestic disorder: A three-year follow-up study of clinical signs and social disabilities. Acta Psychiatr. Scand. 1992, 86, 240–246. [Google Scholar] [CrossRef]
- Aalten, P.; van Valen, E.; de Vugt, M.E.; Lousberg, R.; Jolles, J.; Verhey, F.R. Awareness and behavioral problems in dementia patients: A prospective study. Int. Psychogeriatrics 2006, 18, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Salmon, E.; Perani, D.; Herholz, K.; Marique, P.; Kalbe, E.; Holthoff, V.; Delbeuck, X.; Beuthien-Baumann, B.; Pelati, O.; Lespagnard, S.; et al. Neural correlates of anosognosia for cognitive impairment in Alzheimer’s disease. Hum. Brain Mapp 2006, 27, 588–597. [Google Scholar] [CrossRef]
- Jacus, J.P. Awareness, apathy, and depression in Alzheimer’s disease and mild cognitive impairment. Brain Behav. 2017, 7, e00661. [Google Scholar] [CrossRef]
- Robert, P.; Clairet, S.; Benoit, M.; Koutaich, J.; Bertogliati, C.; Tible, O.; Caci, H.; Borg, M.; Brocker, P.; Bedoucha, P. The apathy inventory: Assessment of apathy and awareness in Alzheimer’s disease, Parkinson’s disease and mild cognitive impairment. Int. J. Geriatr. Psychiatry 2002, 17, 1099–1105. [Google Scholar] [CrossRef]
- Starkstein, S.E.; Brockman, S.; Bruce, D.; Petracca, G. Anosognosia is a significant predictor of apathy in Alzheimer’s disease. J. Neuropsychiatry Clin. Neurosci. 2010, 22, 378–383. [Google Scholar] [CrossRef]
- Oscar-Berman, M. Function and dysfunction of prefrontal brain circuitry in alcoholic Korsakoff’s syndrome. Neuropsychol. Rev. 2012, 22, 154–169. [Google Scholar] [CrossRef]
- Kessels, R.P.; Moerman-van den Brink, W.; Rensen, Y.; van Aken, L.; Walvoort, S.J.; Egger, J.I. The Relation between Behavioral, Emotional and Cognitive Apathy and Everyday Executive Dysfunction in Alcoholic Korsakoff’s Syndrome. Arch. Clin. Psychiatry 2021, 48, 178–181. [Google Scholar]
- Richard, E.; Schmand, B.; Eikelenboom, P.; Yang, S.C.; Ligthart, S.A.; Moll van Charante, E.P.; van Gool, W.A. Symptoms of apathy are associated with progression from mild cognitive impairment to Alzheimer’s disease in non-depressed subjects. Dement. Geriatr. Cogn. Disord. 2012, 33, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.W.; Grossman, H.T.; Sano, M. Why Do They Just Sit? Apathy as a Core Symptom of Alzheimer Disease. Am. J. Geriatr. Psychiatry 2019, 27, 395–405. [Google Scholar] [CrossRef] [PubMed]
- van Dorst, M.E.G.; Rensen, Y.C.M.; Husain, M.; Kessels, R.P.C. Behavioral, Emotional and Social Apathy in Alcohol-Related Cognitive Disorders. J. Clin. Med. 2021, 10, 2447. [Google Scholar] [CrossRef] [PubMed]
- Oey, M.J.; Brouwer, V.; Buijs, M.J.; Wijnia, J.W.; Postma, A.; Oudman, E. Unraveling Apathy in Korsakoff Syndrome Patients Receiving Long-Term Care with and without Cerebrovascular Comorbidity. Alcohol. Clin. Exp. Res. 2021, 45, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Alexander, C.M.; Martyr, A.; Savage, S.A.; Morris, R.G.; Clare, L. Measuring Awareness in People with Dementia: Results of a Systematic Scoping Review. J. Geriatr. Psychiatry Neurol. 2021, 34, 335–348. [Google Scholar] [CrossRef] [PubMed]
- de Ruijter, N.S.; Schoonbrood, A.M.G.; van Twillert, B.; Hoff, E.I. Anosognosia in dementia: A review of current assessment instruments. Alzheimers Dement. 2020, 12, e12079. [Google Scholar] [CrossRef] [PubMed]
- Villalobos, D.; Bilbao, Á.; Espejo, A.; García-Pacios, J. Efficacy of an intervention programme for rehabilitation of awareness of deficit after acquired brain injury: A pilot study. Brain Inj. 2018, 32, 158–166. [Google Scholar] [CrossRef]
| Node Name | Variable | Mean [SD] | Count [%] | Missing [%] |
|---|---|---|---|---|
| Sociodemographic and clinical characteristics | ||||
| Age | Age | 63.2 [7.9] | 0 [0] | |
| Sex | Gender (male) | 166 [77.2] | 0 [0] | |
| LoS | Length of stay in in LCTF (years) | 6.7 [5.6] | 0 [0] | |
| Edu | Education (category) | 31 [14.4] | ||
| Elementary/lower | 124 [67.4] | |||
| Secondary | 43 [23.4] | |||
| Higher/university | 17 [9.2] | |||
| Single | Single, divorced, or widow(er) | 188 [91.7] | 10 [4.7] | |
| Use of ≥1 psychotropic drugs | 137 [63.7] | 0 [0] | ||
| AP. | Antipsychotic drugs | 103 [47.9] | ||
| AD. | Antidepressant drugs | 79 [36.7] | ||
| BZ. | Benzodiazepines | 66 [30.7] | ||
| Questionnaires (↑: higher is better, ↓: lower is better) | ||||
| PCRS | Awareness discrepancy score (−120–120) ↓ | 40.0 [20.0] | 0 [0] | |
| AES | Apathy Evaluation Scale (10–40) ↓ | 25.3 [6.0] | 2 [0.9] | |
| NPI | Neuropsychiatric Symptoms (severity score) ↓ | |||
| NPI-Psy | Psychosis subscale | 0 [0] | ||
| Present | 145 [67.4%] | |||
| Severity: 2 subscales (0–6) | 0.86 [1.5] | |||
| NPI-Ag | Agitation | 0 [0] | ||
| Present | 175 [81.4%] | |||
| Severity: 3 subscales (0–9) | 3.2 [2.6] | |||
| NPI-Anx | Anxiety subscale | 0 [0] | ||
| Present | 54 [25.1%] | |||
| Severity 1 subscale (0–3) | 0.52 [1.0] | |||
| NPI-Ap | Apathy subscale | 0 [0] | ||
| Present | 98 [45.6%] | |||
| Severity: 1 subscale (0–3) | 0.81 [1.0] | |||
| NPI-Dep | Depression subscale | 0 [0] | ||
| Present | 91 [42.3%] | |||
| Severity: 1 subscale (0–3) | 0.82 [1.1] | |||
| RISE | Social participation (0–6) ↑ | 4.27 [1.8] | 0 [0] | |
| CPS | Cognitive impairment (0–6) ↓ | 2.55 [1.6] | 0 [0] | |
| ADL | Activities of daily living (0–6) ↓ | 1.09 [1.2] | 0 [0] | |
| IADL | Instrumental activities of daily living (0–48) ↓ | 39.1 [8.3] | 0 [0] | |
| MANSA | Quality of life (12–84) ↑ | 61.0 [9.6] | 3 [1.4] | |
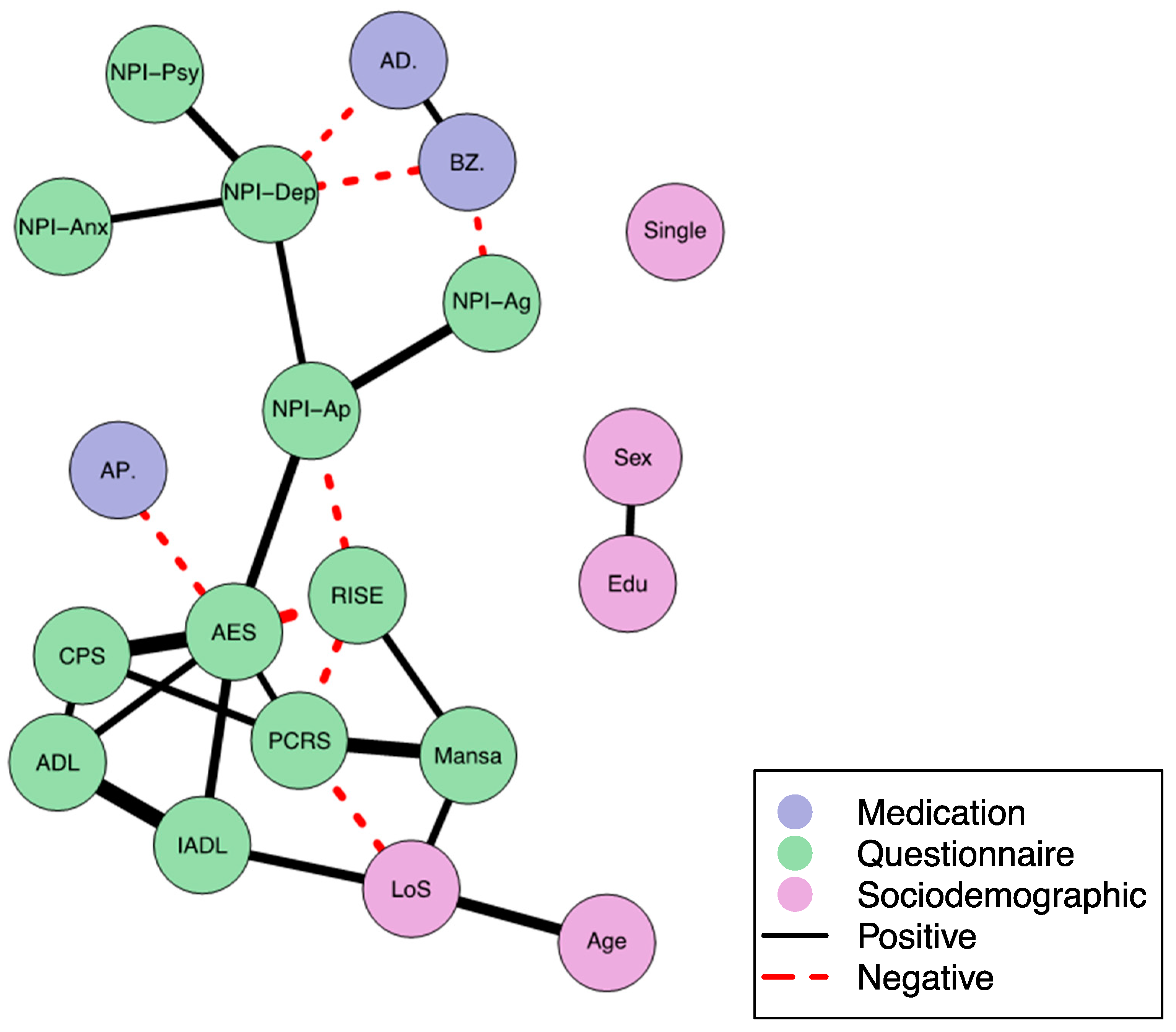
| Centrality Scores | Number of Connections | ||||
|---|---|---|---|---|---|
| Eigenvector | Betweenness | Degree | Positive | Negative | |
| AES | 1 | 74 | 7 | 5 | 2 |
| CPS | 0.679 | 0 | 3 | 3 | 0 |
| ADL | 0.622 | 0 | 3 | 3 | 0 |
| IADL | 0.567 | 0 | 3 | 3 | 0 |
| PCRS | 0.476 | 40 | 5 | 3 | 2 |
| NPI-Ap | 0.451 | 120 | 4 | 3 | 1 |
| NPI-Ag | 0.140 | 0 | 2 | 1 | 1 |
| NPI-Dep | 0.131 | 102 | 5 | 3 | 2 |
| Mansa | 0.099 | 32 | 3 | 3 | 0 |
| LoS | 0.074 | 32 | 4 | 3 | 1 |
| NPI-Psy | 0.032 | 0 | 1 | 1 | 0 |
| NPI-Anx | 0.029 | 0 | 1 | 1 | 0 |
| Age | 0.023 | 0 | 1 | 1 | 0 |
| RISE | 0 | 76 | 4 | 1 | 3 |
| Sex | 0 | 0 | 1 | 1 | 0 |
| BZ. | 0 | 8 | 3 | 1 | 2 |
| AP. | 0 | 0 | 1 | 0 | 1 |
| AD. | 0 | 0 | 2 | 1 | 1 |
| Edu | 0 | 0 | 1 | 1 | 0 |
| Single | 0 | 0 | 0 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fidder, H.; Veenhuizen, R.B.; Gerridzen, I.J.; van Wieringen, W.N.; Smalbrugge, M.; Hertogh, C.M.P.M.; van Loon, A.M. Impaired Awareness in People with Severe Alcohol-Related Cognitive Deficits Including Korskoff’s Syndrome: A Network Analysis. J. Clin. Med. 2023, 12, 3139. https://doi.org/10.3390/jcm12093139
Fidder H, Veenhuizen RB, Gerridzen IJ, van Wieringen WN, Smalbrugge M, Hertogh CMPM, van Loon AM. Impaired Awareness in People with Severe Alcohol-Related Cognitive Deficits Including Korskoff’s Syndrome: A Network Analysis. Journal of Clinical Medicine. 2023; 12(9):3139. https://doi.org/10.3390/jcm12093139
Chicago/Turabian StyleFidder, Hester, Ruth B. Veenhuizen, Ineke J. Gerridzen, Wessel N. van Wieringen, Martin Smalbrugge, Cees M. P. M. Hertogh, and Anouk M. van Loon. 2023. "Impaired Awareness in People with Severe Alcohol-Related Cognitive Deficits Including Korskoff’s Syndrome: A Network Analysis" Journal of Clinical Medicine 12, no. 9: 3139. https://doi.org/10.3390/jcm12093139
APA StyleFidder, H., Veenhuizen, R. B., Gerridzen, I. J., van Wieringen, W. N., Smalbrugge, M., Hertogh, C. M. P. M., & van Loon, A. M. (2023). Impaired Awareness in People with Severe Alcohol-Related Cognitive Deficits Including Korskoff’s Syndrome: A Network Analysis. Journal of Clinical Medicine, 12(9), 3139. https://doi.org/10.3390/jcm12093139






