Effect of Photobiomodulation Combined with Physiotherapy on Functional Performance in Children with Myelomeningo-Cele-Randomized, Blind, Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
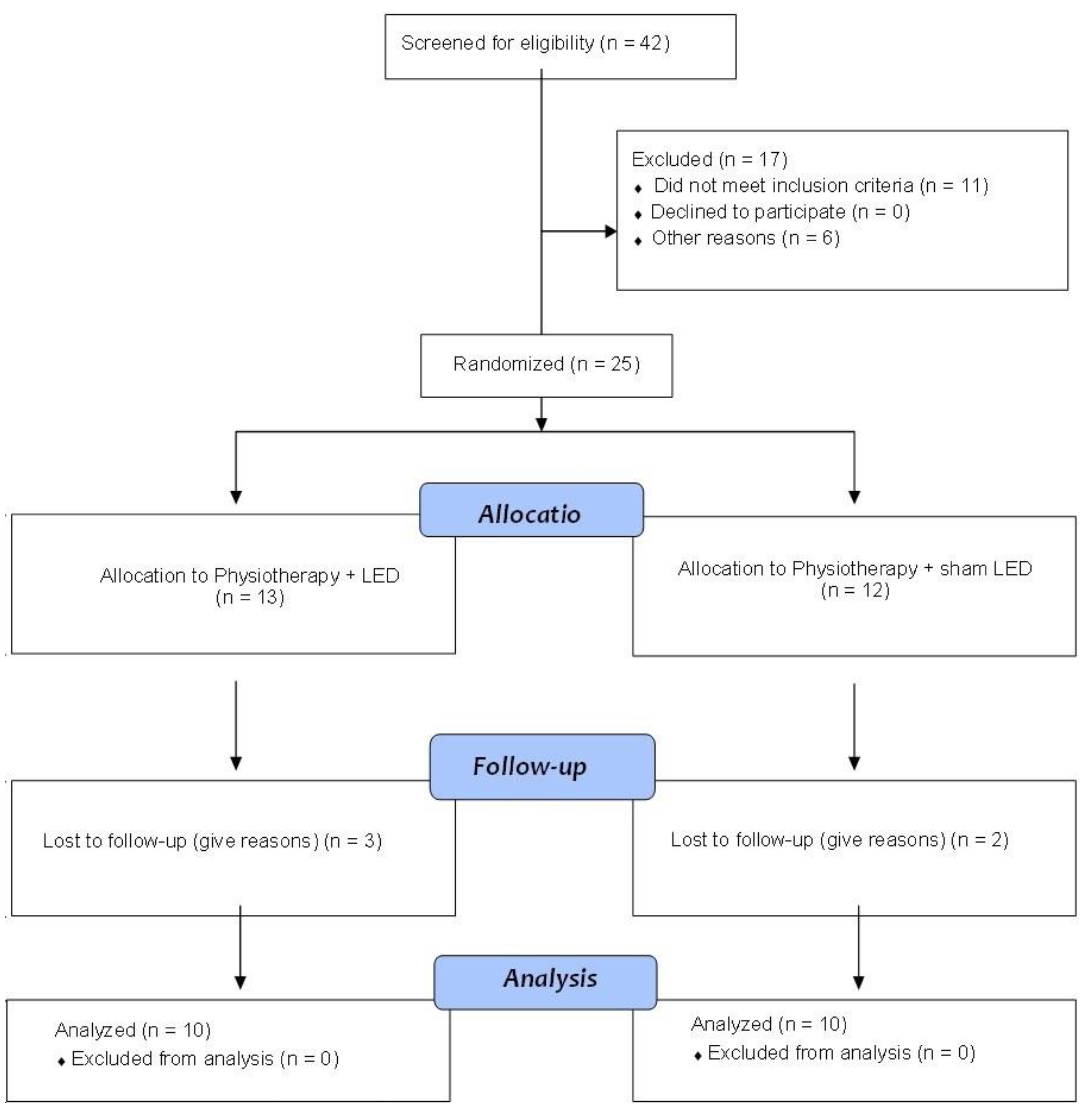
2.1. Participants
2.2. Randomization
2.3. Experimental Design
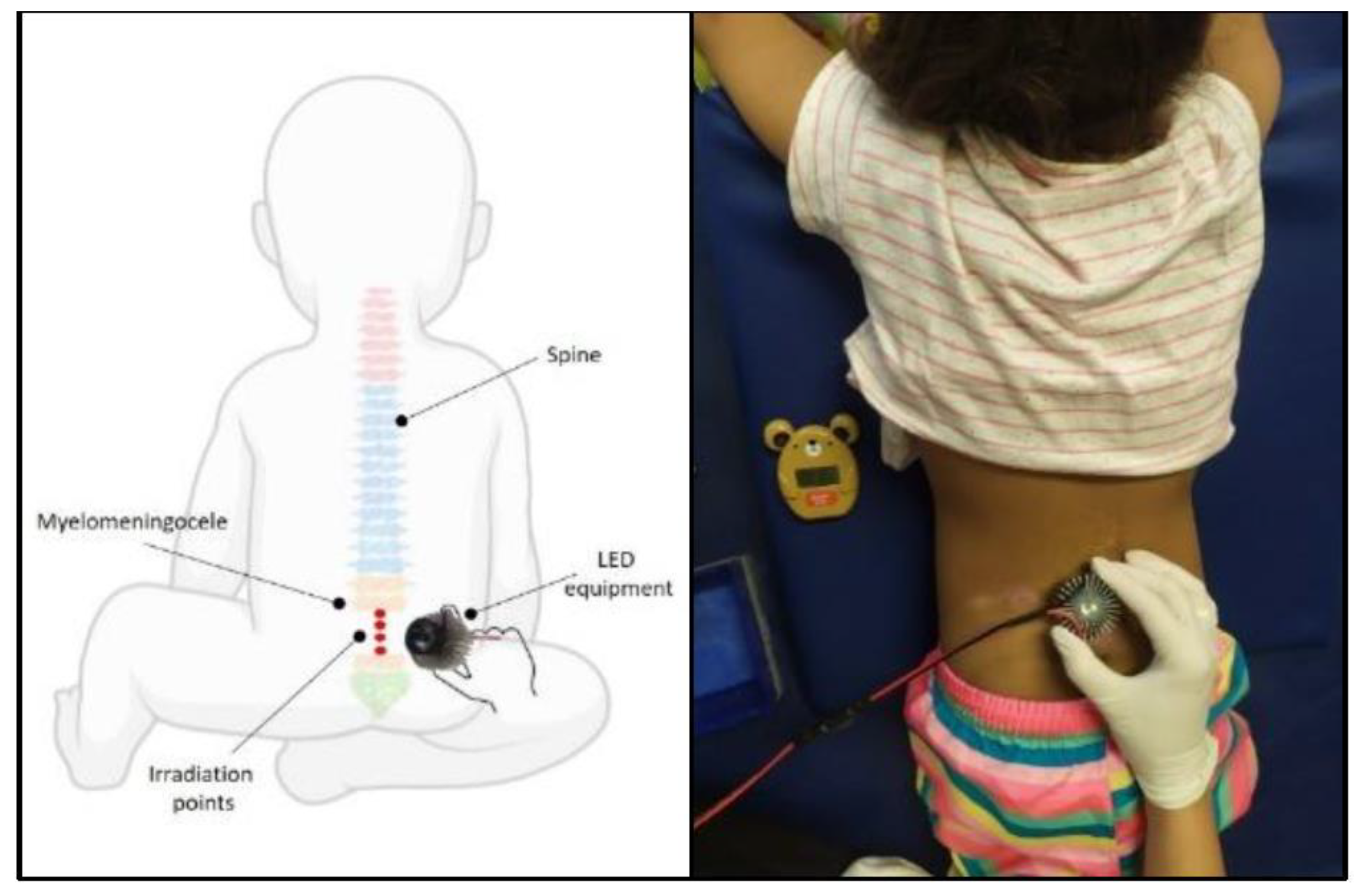
2.4. Intervention
Neurofunctional Physiotherapy
- Hanging positions;
- Squatting to pick up objects from the floor;
- Going up and down steps;
- Sitting down and standing up from a chair;
- Changing positions from kneeling to semi-kneeling and standing;
- Walking with or without assistance around obstacles, and stable and unstable surfaces.
2.5. Photobiomodulation Protocol
2.6. Assessments
2.6.1. Functional Mobility and Caregiver Assistance
2.6.2. Electrical Muscle Activity
2.7. Sample Calculation
2.8. Statistical Analysis
3. Results
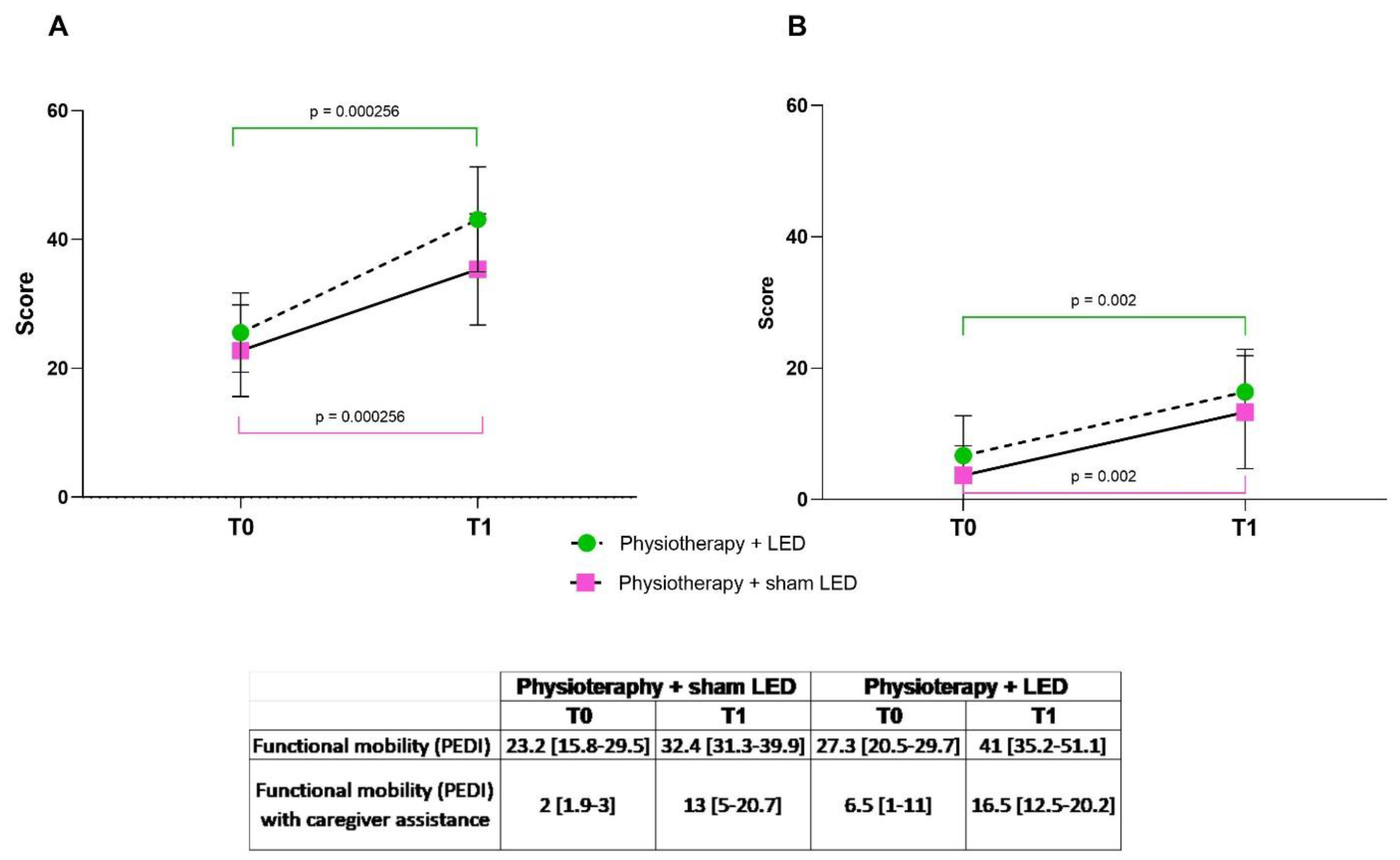
3.1. Functional Mobility and Caregiver Assistance
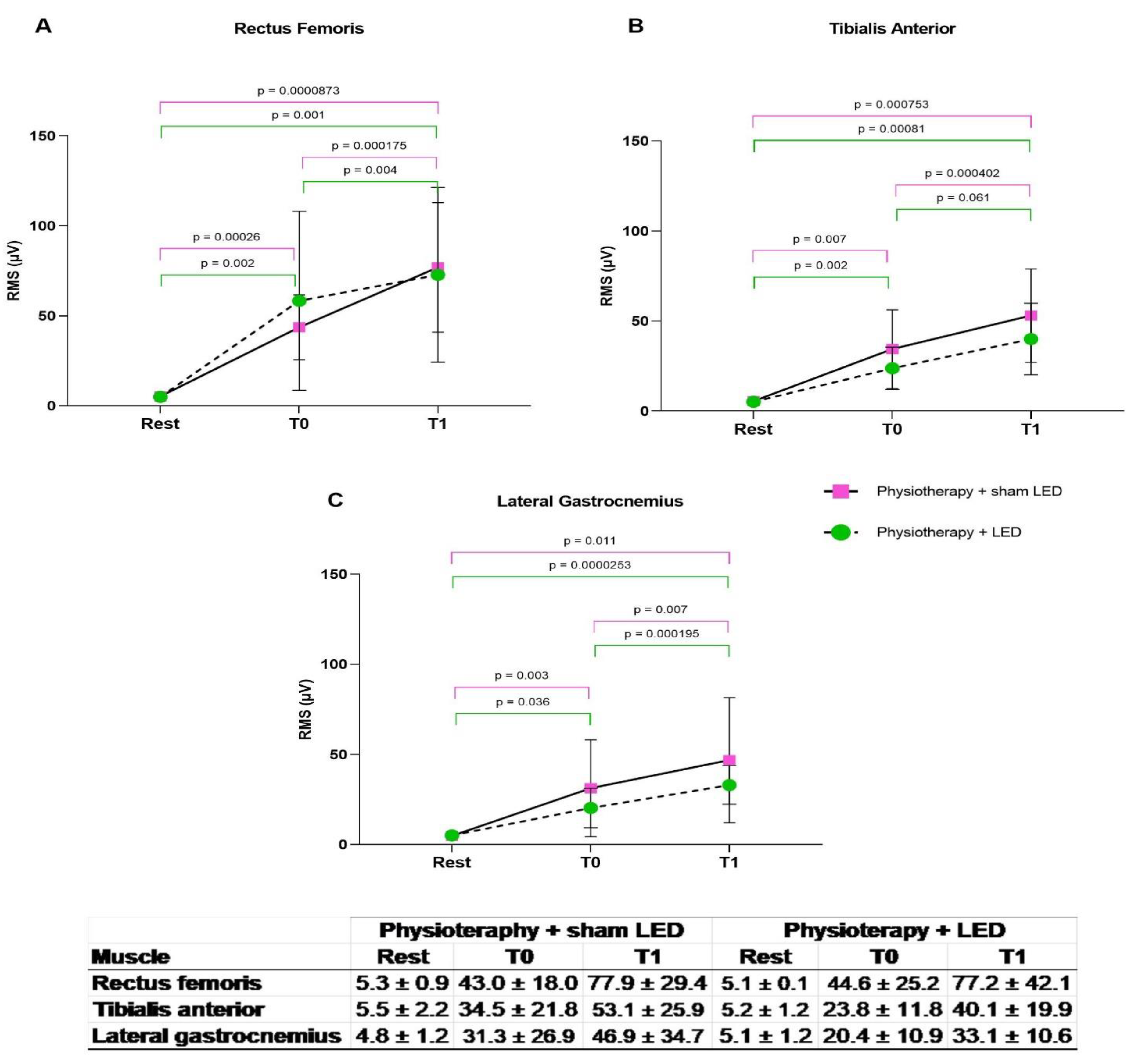
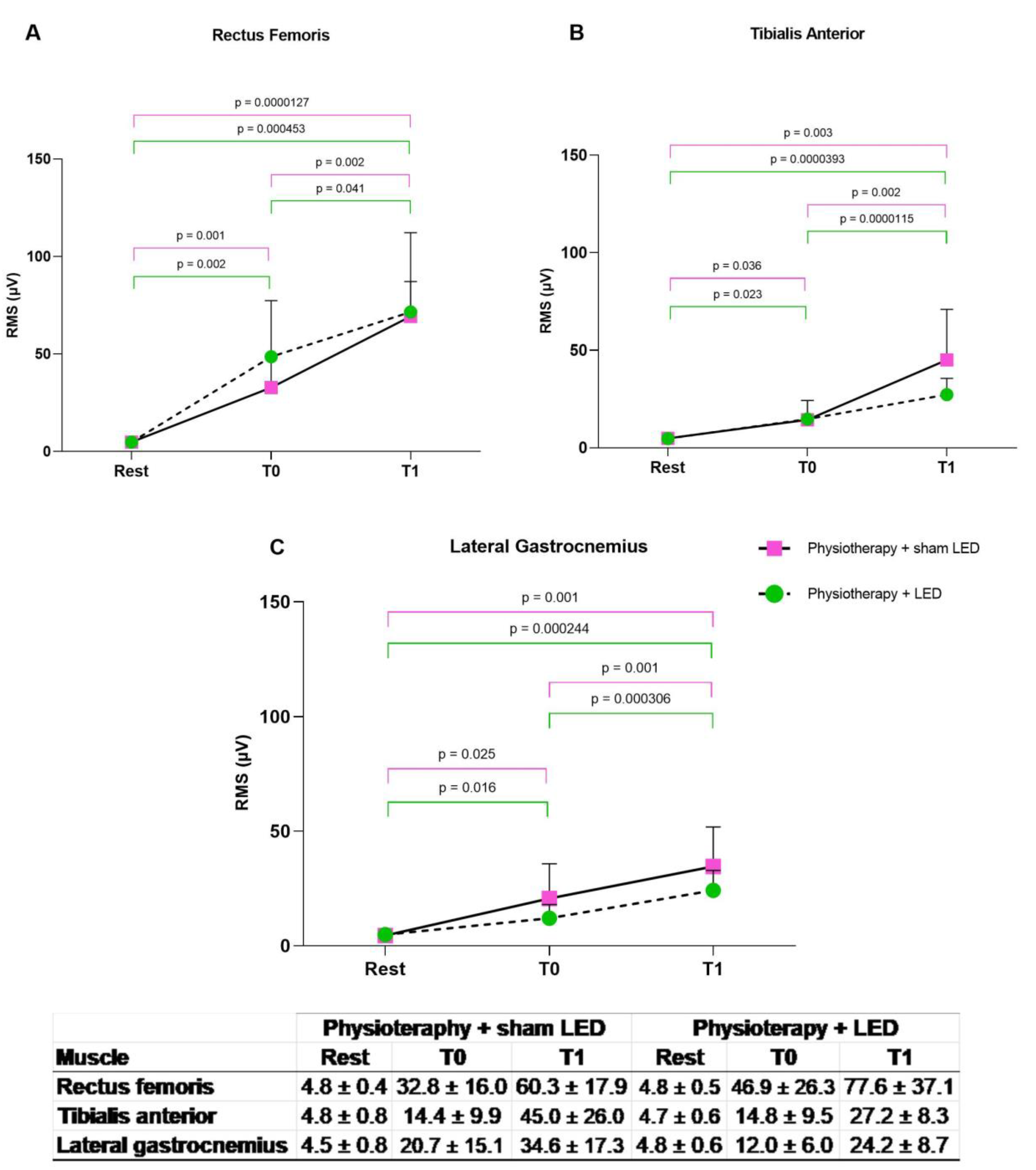
3.2. Electrical Muscle Activity
3.3. Compromised Lower Limbs
More Compromised Lower Limbs
3.4. Less Compromised Lower Limbs
4. Discussion
Improvements in Electrical
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Copp, A.J.; Adzick, N.S.; Chitty, L.S.; Fletcher, J.M.; Holmbeck, G.N.; Shaw, G.M. Spina bifida. Nat. Rev. Dis. Prim. 2015, 1, 1–45. [Google Scholar] [CrossRef] [PubMed]
- Kural, C.; Solmaz, I.; Tehli, O.; Temiz, C.; Kutlay, M.; Daneyemez, M.K.; Izci, Y. Evaluation and Management of Lumbosacral Myelomeningoceles in Children. Eurasian J. Med. 2015, 47, 174–178. [Google Scholar] [CrossRef] [PubMed]
- Oria, M.; Figueira, R.L.; Scorletti, F.; Sbragia, L.; Owens, K.; Li, Z.; Pathak, B.; Corona, M.U.; Marotta, M.; Encinas, J.L.; et al. CD200-CD200R imbalance correlates with microglia and pro- inflammatory activation in rat spinal cords exposed to amniotic fluid in retinoic acid-induced spina bifida. Sci. Rep. 2018, 8, 10638. [Google Scholar] [CrossRef] [PubMed]
- Marotta, M.; Fernández-Martín, A.; Oria, M.; Fontecha, C.G.; Giné, C.; Martínez-Ibáñez, V.; Carreras, E.; Belfort, M.A.; Pelizzo, G.; Peiró, J.L. Isolation, characterization, and differentiation of multipotent neural progenitor cells from human cerebrospinal fluid in fetal cystic myelomeningocele. Stem. Cell. Res. 2017, 22, 33–42. [Google Scholar] [CrossRef]
- Danzer, E.; Joyeux, L.; Flake, A.W.; Deprest, J. Fetal surgical intervention for myelomeningocele: Lessons learned, outcomes, and future implications. Dev. Med. Child. Neurol. 2020, 62, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Sansom, J.K.; Teulier, C.; Smith, B.A.; Moerchen, V.; Muraszko, K.U.B. Muscle Activation Patterns in Infants with Myelomeningocele Stepping on a Treadmill. Pediatr. Phys. Ther. 2014, 25, 278–289. [Google Scholar] [CrossRef]
- Schoenmakers, M.A.G.C.; Uiterwaal, C.S.P.M.; Gulmans, V.A.M.; Gooskens, R.H.J.M.; Helders, P.J.M. Determinants of functional independence and quality of life in children with spina bifida. Clin. Rehabil. 2005, 19, 677–685. [Google Scholar] [CrossRef]
- Steinhart, S.; Kornitzer, E.; Baron, A.B.; Wever, C.; Shoshan, L.; Katz-Leurer, M. Independence in self-care activities in children with myelomeningocele: Exploring factors based on the International Classification of Function model. Disabil. Rehabil. 2018, 40, 62–68. [Google Scholar] [CrossRef]
- Lee, D.K.; Sansom, J.K. Early Treadmill Practice in Infants Born with Myelomeningocele: A Pilot Study. Pediatr. Phys. Ther. 2019, 31, 68–75. [Google Scholar] [CrossRef]
- Luft, A.R. Rehabilitation and Plasticity. Front. Neurol. Neurosci. 2013, 32, 88–94. [Google Scholar]
- Harris, S.R.; Winstein, C.J. The Past, Present, and Future of Neurorehabilitation: From, NUSTEP Through IV STEP. Pediatr Phys Ther. 2017, 29, S2–S9. [Google Scholar] [CrossRef] [PubMed]
- Krakauer, J.W.; Hadjiosif, A.M.; Xu, J.; Wong, A.L.; Haith, A.M. Motor learning. Compr. Physiol. 2019, 9, 613–663. [Google Scholar] [PubMed]
- Krishnan, C.; Washabaugh, E.P.; Reid, C.E.; Althoen, M.M.; Ranganathan, R. Learning new gait patterns: Age-related differences in skill acquisition and interlimb transfer. Exp. Gerontol. 2018, 111, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Freitas, L.F.D.; Hamblin, M.R. Proposed Mechanisms of Photobiomodulation or Low-Level Light Therapy. IEEE J. Sel. Top. Quantum Electron. 2016, 22, 7000417. [Google Scholar] [CrossRef]
- Hashmi, J.T.; Huang, Y.Y.; Osmani, B.Z.; Sharma, S.K.; Naeser, M.A.; Hamblin, M.R. Role of low-level laser therapy in neurorehabilitation. PMR 2010, 2, S292–S305. [Google Scholar] [CrossRef]
- Heiskanen, V.; Hamblin, M.R. Photobiomodulation: Lasers vs Light Emitting Diodes? Photochem. Photobiol. Sci. 2018, 17, 1003–1017. [Google Scholar] [CrossRef]
- Calderhead, R.G. Review article the photobiological basics behind light-emitting diode (Led) phototherapy. Laser Ther. 2007, 16, 97–108. [Google Scholar] [CrossRef]
- da Silva, F.C.; Gomes, A.O.; da Costa Palácio, P.R.; Politti, F.; de Fátima Teixeira da Silva, D.; Mesquita-Ferrari, R.A.; Fernandes, K.P.S.; Bussadori, S.K. Photobiomodulation improves motor response in patients with spinal cord injury submitted to electromyographic evaluation: Randoized clinical trial. Lasers Med Sci. 2018, 33, 883–890. [Google Scholar] [CrossRef]
- da Silva, F.C.; Silva, T.; Gomes, A.O.; Costa Palácio, P.R.d.; Andreo, L.; Gonçalves, M.L.L.; Silva, D.F.T.; Horliana, A.C.R.T.; Motta, L.J.; Mesquita-Ferrari, R.A.; et al. Sensory and motor responses after photobiomodulation associated with physiotherapy in patients with incomplete spinal cord injury: Clinical, randomized trial. Lasers Med. Sci. 2020, 35, 1751–1758. [Google Scholar] [CrossRef]
- Kleim, J.A.; Jones, T.A. Principles of experience-dependent neural plasticity: Implications for rehabilitation after brain damage. J. Speech Lang. Hear. Res. 2008, 51, S225–S239. [Google Scholar] [CrossRef]
- Blundell, S.W.; Shepherd, R.B.; Dean, C.M.; Adams, R.D.; Cahill, B.M. Functional strength training in cerebral palsy: A pilot study of a group circuit training class for children aged 4–8 years. Clin. Rehabil. 2003, 17, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Shemy, S.A.E. Trunk endurance and gait changes after core stability training in children with hemiplegic cerebral palsy: A randomized controlled trial. J. Back Musculoskelet. Rehabil. 2018, 31, 1159. [Google Scholar] [CrossRef] [PubMed]
- Aizawa, C.Y.P.; Morales, M.P.; Lundberg, C.; Moura, M.C.D.S.; Pinto, F.C.G.; Voos, M.C.; Hasue, R.H. Conventional physical therapy and physical therapy based on reflex stimulation showed similar results in children with myelomeningocele. Arq. De Neuro-Psiquiatria. 2017, 75, 160–166. [Google Scholar] [CrossRef]
- Flores, M.B.; Manella, K.J.; Ardolino, E.M. Relationship between Movement Quality, Functional Ambulation Status, and Spatiotemporal Gait Parameters in Children with Myelomeningocele. Phys. Occup. Ther. Pediatr. 2020, 40, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Neves, A.; Visicatto, L.P.; Oliveira, A.B.d.; Adriana, N.; Ferreira, C. Effects of Kinesio taping in rectus femoris activity and sit-to-stand movement in children with unilateral cerebral palsy: Placebo-controlled, repeated-measure design. Disabil. Rehabil. 2019, 41, 2049–2059. [Google Scholar]
- Hermens, H.J. Development of recommendations for, SEMG sensors and sensor placement procedures. J. Electromyogr. Kinesiol. 2000, 10, 361–374. [Google Scholar] [CrossRef] [PubMed]
- Novak, I.; Morgan, C.; Fahey, M.; Finch-Edmondson, M.; Galea, C.; Hines, A.; Langdon, K.; Mc Namara, M.; Paton, M.C.; Popat, H.; et al. State of the Evidence Traffic Lights 2019: Systematic Review of Interventions for Preventing and Treating Children with Cerebral Palsy. Curr. Neurol. Neurosci. Rep. 2020, 20, 2–21. [Google Scholar] [CrossRef] [PubMed]
- Pantall, A.; Teulier, C.U.B.D. Changes in muscle activation patterns in response to enhanced sensory input during treadmill stepping in infants born with myelomeningocele. Hum. Mov. Sci. 2012, 31, 1670–1687. [Google Scholar] [CrossRef]
- Khan, F.; Amatya, B.; Galea, M.P.; Gonzenbach, R. Neurorehabilitation: Applied neuroplasticity. J. Neurol. 2017, 264, 603–615. [Google Scholar] [CrossRef]
- Mapaisansin, P.; Suriyaamarit, D.; Boonyong, S. The development of sit-to-stand in typically developing children aged 4 to 12 years: Movement time, trunk and lower extremity joint angles, and joint moments. Gait Posture 2020, 76, 14–21. [Google Scholar] [CrossRef]
- Bobbert, M.F.; Kistemaker, D.A.; Vaz, M.A.; Ackermann, M. Searching for strategies to reduce the mechanical demands of the sit-to-stand task with a muscle-actuated optimal control model. Clin. Biomech. 2016, 37, 83–90. [Google Scholar] [CrossRef] [PubMed]
- McDonald, C.M.; Jaffe, K.M.; Mosca, V.S.; Shurtleff, D.B. Ambulatory Outcome of Children with Myelomeningocele: Effect of Lower-Extremity Muscle Strength. Dev. Med. Child Neurol. 1991, 33, 482–490. [Google Scholar] [CrossRef] [PubMed]
- Salehpour, F.; Mahmoudi, J.; Kamari, F.; Sadigh-Eteghad, S.; Rasta, S.H.; Hamblin, M.R. Brain Photobiomodulation Therapy: A Narrative Review. Mol. Neurobiol. 2018, 55, 6601–6636. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Dmitriev, A.E.; Cardoso, M.J.; Viers-costello, A.G.; Borke, R.C.; Streeter, J.; Anders, J.J. 810 nm Wavelength Light: An Effective Therapy for Transected or Contused Rat Spinal Cord. Lasers Surg. Med. 2009, 41, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Paula, A.A.; Nicolau, R.A.; Lima, M.D.O.; Angel, M.; Salgado, C.; Cogo, J.C. Low-intensity laser therapy effect on the recovery of traumatic spinal cord injury. Lasers Med. Sci. 2014, 9, 1849–1859. [Google Scholar] [CrossRef]
- Veronez, S.; Assis, L.; Campo, P.D.; Oliveira, F.D.; Castro, G.d.; Renno, A.C.M.; Medalha, C.C. Effects of different fluences of low-level laser therapy in an experimental model of spinal cord injury in rats. Lasers Med. Sci. 2016, 32, 343–349. [Google Scholar] [CrossRef]
- Zein, R.; Selting, W.; Hamblin, M.R. Review of light parameters and photobiomodulation efficacy: Dive into complexity. J. Biomed. Opt. 2018, 23, 120901. [Google Scholar] [CrossRef]

| Parameter | |
|---|---|
| Center wavelength [nm] | 850 |
| Spectral bandwidth (FWHM) [nm] | 20 |
| Operating mode | Continuous wave |
| Average radiant power [mW] | 500 |
| Polarization | Random |
| Aperture diameter [cm] | 1.9 |
| Irradiance at aperture [mW/cm2] | 176 |
| Beam profile | Multimode |
| Beam spot size at target [cm2] | 2.84 |
| Irradiance at target [mW/cm2] | 176 |
| Exposure duration per point [s] | 50 |
| Radiant exposure [J/cm2] | 9 |
| Radiant energy per point [J] | 25 |
| Number of points irradiated | 4 |
| Radiant energy per session [J] | 100 |
| Area irradiated per session [cm2] | 11.34 |
| Application technique | Contact |
| Number of treatment sessions | 24 |
| Frequency of treatment sessions | 2× per week |
| Total radiant energy [J] | 2400 |
| Características | Physiotherapy + LED (n = 10) | Physiotherapy + Sham LED (n = 10) |
|---|---|---|
| Age | 3.8 ± 1.6 | 3.2 ± 1.6 |
| Sex (girls/boys) | 9/1 | 10/0 |
| Lesion level n (%) | ||
| Upper lumbar | 0 | 0 |
| Lower lumbar | 10 (100%) | 10 (100%) |
| Sacral | 0 | 0 |
| Ambulator (yes/no) | 0/10 | 0/10 |
| Arnold Chiari malformation (yes/no) | 10/0 | 10/0 |
| Tethered spinal cord (yes/no) | 0/10 | 0/10 |
| Neurogenic bladder (yes/no) | 10/0 | 10/0 |
| Neurogenic intestine (yes/no) | 10/0 | 10/0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, T.; Tobelem, D.d.C.; Malavazzi, T.C.D.S.; Mendonça, J.F.B.d.; Andreo, L.; Chavantes, M.C.; Tempestini Horliana, A.C.R.; Turcio, K.H.L.; Gomes, A.O.; Deana, A.M.; et al. Effect of Photobiomodulation Combined with Physiotherapy on Functional Performance in Children with Myelomeningo-Cele-Randomized, Blind, Clinical Trial. J. Clin. Med. 2023, 12, 2920. https://doi.org/10.3390/jcm12082920
Silva T, Tobelem DdC, Malavazzi TCDS, Mendonça JFBd, Andreo L, Chavantes MC, Tempestini Horliana ACR, Turcio KHL, Gomes AO, Deana AM, et al. Effect of Photobiomodulation Combined with Physiotherapy on Functional Performance in Children with Myelomeningo-Cele-Randomized, Blind, Clinical Trial. Journal of Clinical Medicine. 2023; 12(8):2920. https://doi.org/10.3390/jcm12082920
Chicago/Turabian StyleSilva, Tamiris, Daysi da Cruz Tobelem, Tainá Caroline Dos Santos Malavazzi, Juliana Fernandes Barreto de Mendonça, Lucas Andreo, Maria Cristina Chavantes, Anna Carolina Ratto Tempestini Horliana, Karina Helga Leal Turcio, Andréa Oliver Gomes, Alessandro Melo Deana, and et al. 2023. "Effect of Photobiomodulation Combined with Physiotherapy on Functional Performance in Children with Myelomeningo-Cele-Randomized, Blind, Clinical Trial" Journal of Clinical Medicine 12, no. 8: 2920. https://doi.org/10.3390/jcm12082920
APA StyleSilva, T., Tobelem, D. d. C., Malavazzi, T. C. D. S., Mendonça, J. F. B. d., Andreo, L., Chavantes, M. C., Tempestini Horliana, A. C. R., Turcio, K. H. L., Gomes, A. O., Deana, A. M., Fernandes, K. P. S., Motta, L. J., Mesquita-Ferrari, R. A., Brugnera, A., Nammour, S., & Bussadori, S. K. (2023). Effect of Photobiomodulation Combined with Physiotherapy on Functional Performance in Children with Myelomeningo-Cele-Randomized, Blind, Clinical Trial. Journal of Clinical Medicine, 12(8), 2920. https://doi.org/10.3390/jcm12082920









