Glycemic Control in Critically Ill COVID-19 Patients: Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Literature Search
2.3. Eligibility Criteria
2.4. Data Extraction
2.5. Statistical Analysis
3. Results
3.1. Study Selection
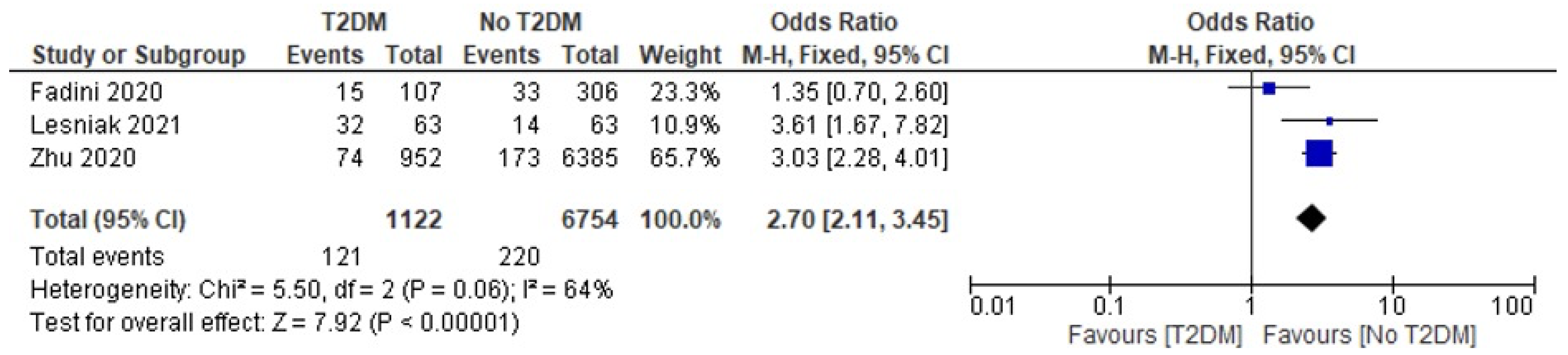
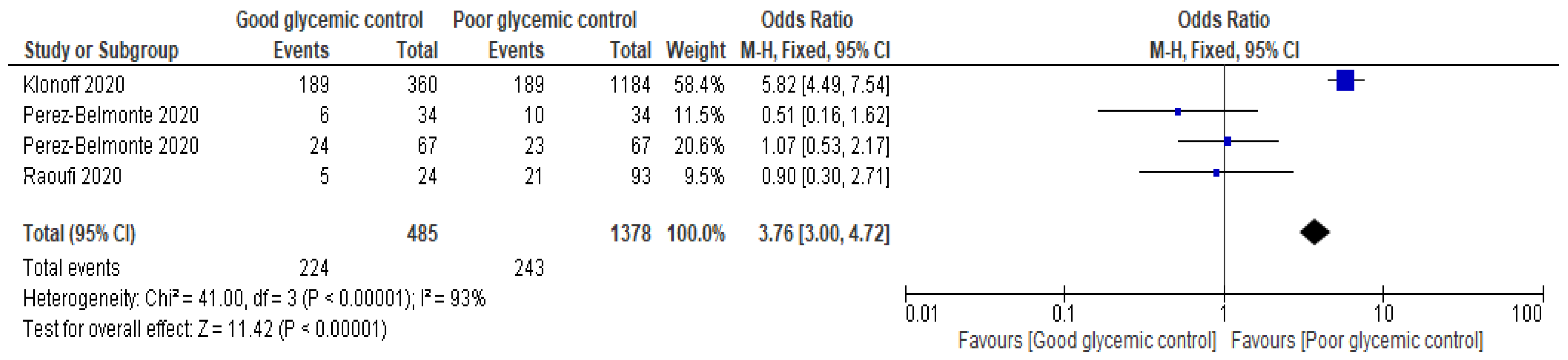
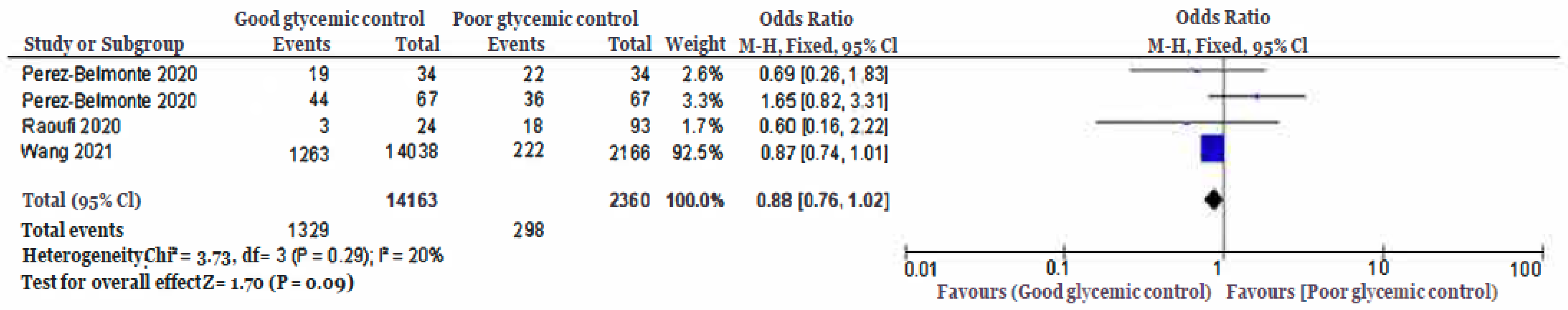
3.2. Meta-Analysis Results
- Death
- Death
3.3. Complications
4. Discussion
4.1. Glucose Control in Hospital
4.2. Association between Glycemic Control and COVID-19 Outcomes
4.3. Diabetes-Associated Complications
4.4. Interpretation
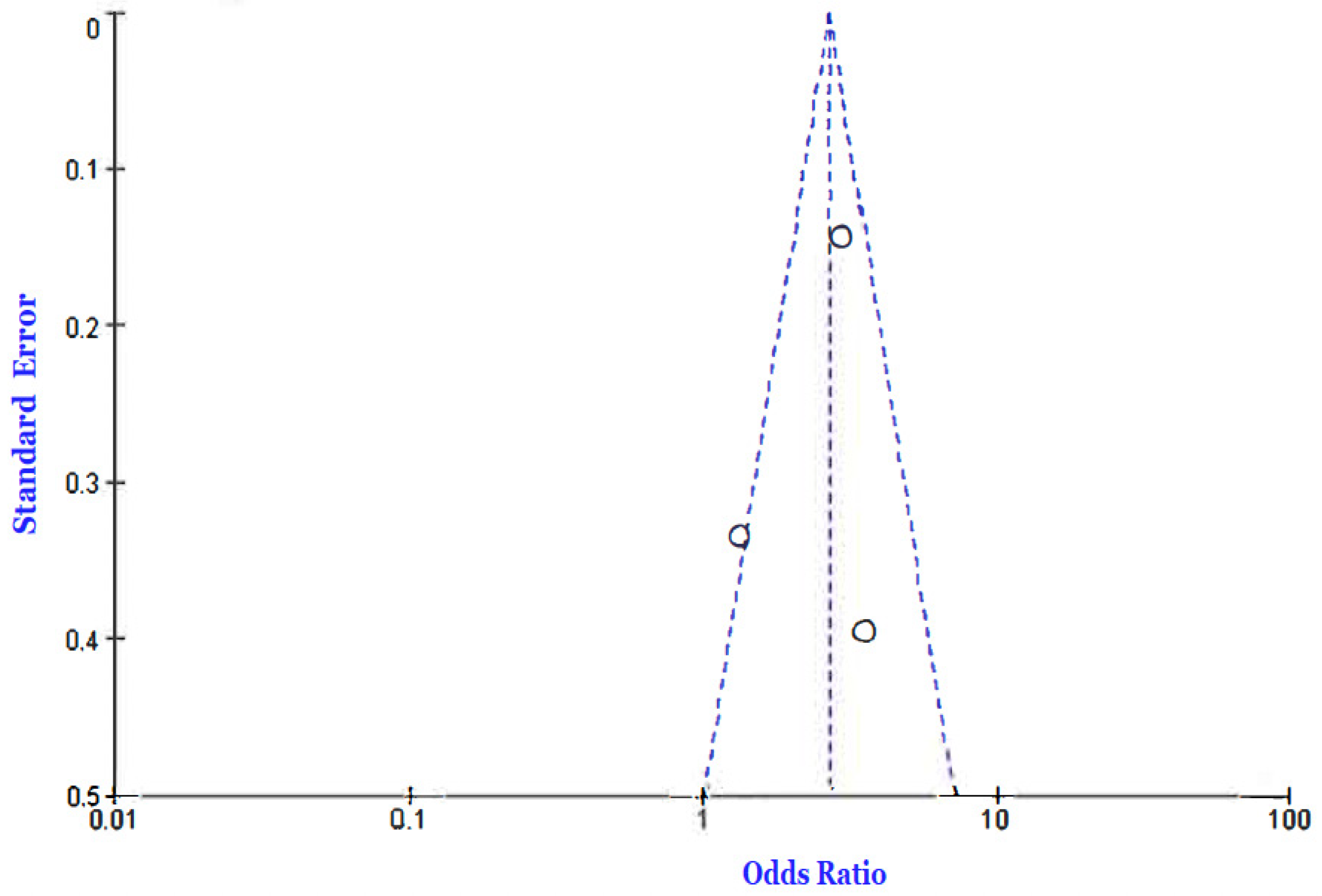
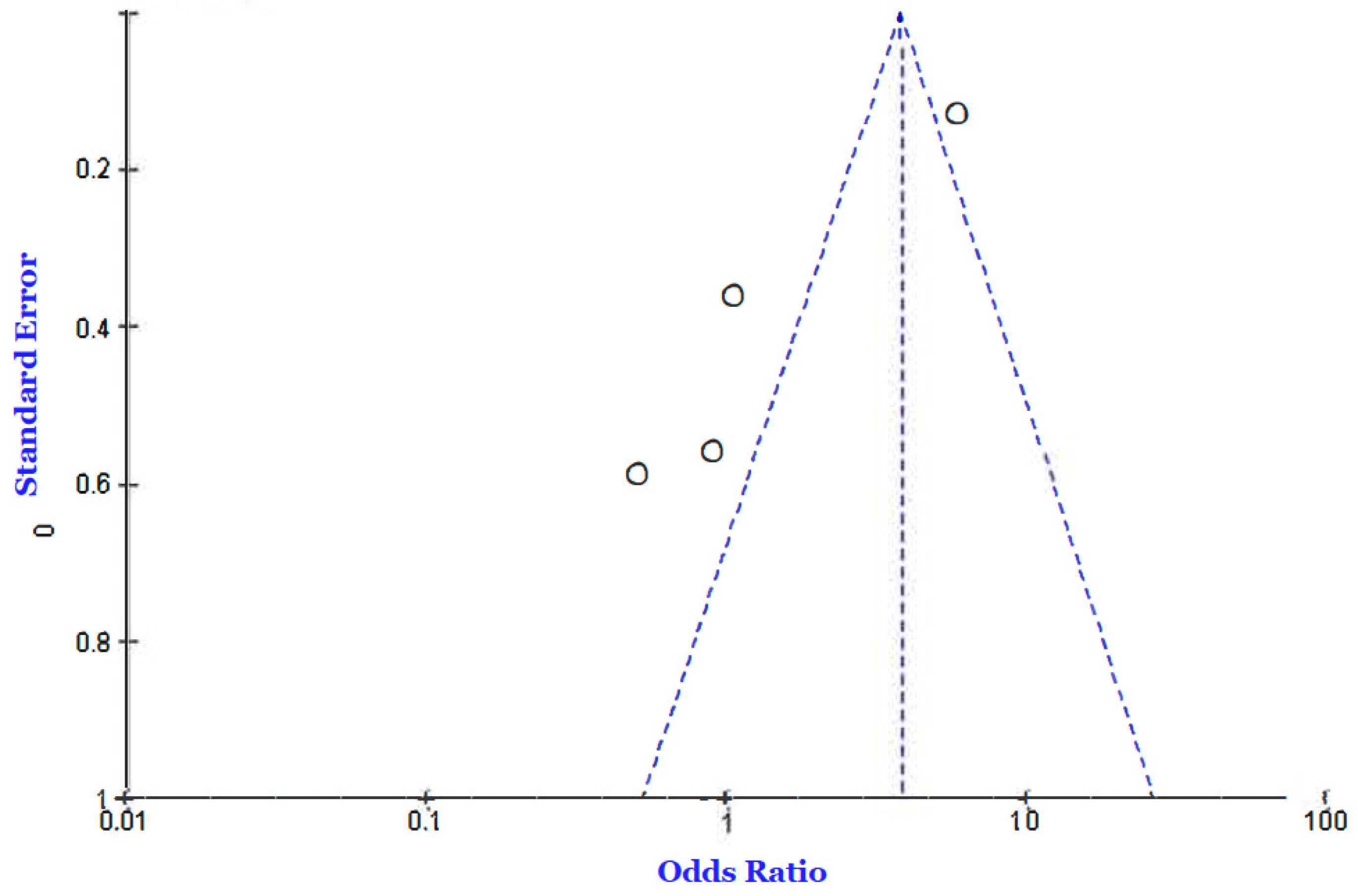
4.5. Strengths and Limitations of the Study
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hartmann, B.; Verket, M.; Balfanz, P.; Hartmann, N.U.; Jacobsen, M.; Brandts, J.; Dreher, M.; Kossack, N.; Häckl, D.; Marx, N.; et al. Glycaemic variability is associated with all-cause mortality in COVID-19 patients with ARDS, a retrospective subcohort study. Sci. Rep. 2022, 12, 9862. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Mathew, J.; Davis, G.M.; Shephardson, A.; Levine, A.; Louard, R.; Urrutia, A.; Perez-Guzman, C.; Umpierrez, G.E.; Peng, L.; et al. Continuous Glucose Monitoring in the Intensive Care Unit During the COVID-19 Pandemic. Diabetes Care 2021, 44, 847–849. [Google Scholar] [CrossRef] [PubMed]
- Klonoff, D.C.; Messler, J.C.; Umpierrez, G.E.; Peng, L.; Booth, R.; Crowe, J.; Garrett, V.; McFarland, R.; Pasquel, F.J. Association Between Achieving Inpatient Glycemic Control and Clinical Outcomes in Hospitalized Patients With COVID-19: A Multicenter, Retrospective Hospital-Based Analysis. Diabetes Care 2021, 44, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Fadini, G.P.; Morieri, M.L.; Boscari, F.; Fioretto, P.; Maran, A.; Busetto, L.; Bonora, B.M.; Selmin, E.; Arcidiacono, G.; Pinelli, S.; et al. Newly-diagnosed diabetes and admission hyperglycemia predict COVID-19 severity by aggravating respiratory deterioration. Diabetes Res. Clin. Pract. 2020, 168, 108374. [Google Scholar] [CrossRef]
- Pugliese, G.; Vitale, M.; Resi, V.; Orsi, E. Is diabetes mellitus a risk factor for COronaVIrus Disease 19 (COVID-19)? Acta Diabetol. 2020, 57, 1275–1285. [Google Scholar] [CrossRef]
- Fernández, E.; Cortazar, A.; Bellido, V. Impact of COVID-19 lockdown on glycemic control in patients with type 1 diabetes. Diabetes Res. Clin. Pract. 2020, 166, 108348. [Google Scholar] [CrossRef]
- Zhang, Y.; Cui, Y.; Shen, M.; Zhang, J.; Liu, B.; Dai, M.; Chen, L.; Han, D.; Fan, Y.; Zeng, Y.; et al. Association of diabetes mellitus with disease severity and prognosis in COVID-19: A retrospective cohort study. Diabetes Res. Clin. Pract. 2020, 165, 108227. [Google Scholar] [CrossRef]
- Singh, A.K.; Gupta, R.; Ghosh, A.; Misra, A. Diabetes in COVID-19: Prevalence, pathophysiology, prognosis and practical considerations. Diabetes Metab. Syndr. 2020, 14, 303–310. [Google Scholar] [CrossRef]
- Yang, Y.; Cai, Z.; Zhang, J. Insulin Treatment May Increase Adverse Outcomes in Patients With COVID-19 and Diabetes: A Systematic Review and Meta-Analysis. Front. Endocrinol. 2021, 12, 696087. [Google Scholar] [CrossRef]
- Feldman, E.L.; Savelieff, M.G.; Hayek, S.S.; Pennathur, S.; Kretzler, M.; Pop-Busui, R. COVID-19 and Diabetes: A Collision and Collusion of Two Diseases. Diabetes 2020, 69, 2549–2565. [Google Scholar] [CrossRef]
- Guo, W.; Li, M.; Dong, Y.; Zhou, H.; Zhang, Z.; Tian, C.; Qin, R.; Wang, H.; Shen, Y.; Du, K.; et al. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab. Res. Rev. 2020, 36, e3319. [Google Scholar] [CrossRef]
- McGurnaghan, S.J.; Weir, A.; Bishop, J.; Kennedy, S.; Blackbourn LA, K.; McAllister, D.A.; Hutchinson, S.; Caparrotta, T.M.; Mellor, J.; Jeyam, A.; et al. Risks of and risk factors for COVID-19 disease in people with diabetes: A cohort study of the total population of Scotland. Lancet Diabetes Endocrinol. 2021, 9, 82–93. [Google Scholar] [CrossRef]
- Sardu, C.; D’Onofrio, N.; Balestrieri, M.L.; Barbieri, M.; Rizzo, M.R.; Messina, V.; Maggi, P.; Coppola, N.; Paolisso, G.; Marfella, R. Outcomes in Patients with Hyperglycemia Affected by COVID-19: Can We Do More on Glycemic Control? Diabetes Care 2020, 43, 1408–1415. [Google Scholar] [CrossRef]
- Vasbinder, A.; Anderson, E.; Shadid, H.; Berlin, H.; Pan, M.; Azam, T.U.; Khaleel, I.; Padalia, K.; Meloche, C.; O’Hayer, P.; et al. Inflammation, Hyperglycemia, and Adverse Outcomes in Individuals With Diabetes Mellitus Hospitalized for COVID-19. Diabetes Care 2022, 45, 692–700. [Google Scholar] [CrossRef]
- Takahashi, Y.; Matsuura, H.; Domi, H.; Yamamura, H. A continuous intravenous insulin infusion protocol to manage high-dose methylprednisolone-induced hyperglycemia in patients with severe COVID-19. Clin. Diabetes Endocrinol. 2022, 8, 4. [Google Scholar] [CrossRef]
- Halpern, B.; Mancini, M.C. Controlled obesity status: A rarely used concept, but with particular importance in the COVID-19 pandemic and beyond. J. Endocrinol. Investig. 2021, 44, 877–880. [Google Scholar] [CrossRef]
- Boeder, S.; Kobayashi, E.; Ramesh, G.; Serences, B.; Kulasa, K.; Majithia, A.R. Accuracy and Glycemic Efficacy of Continuous Glucose Monitors in Critically Ill COVID-19 Patients: A Retrospective Study. J. Diabetes Sci. Technol. 2022, 19322968221113865. [Google Scholar] [CrossRef]
- Zhou, J.; Tan, J. Diabetes patients with COVID-19 need better blood glucose management in Wuhan, China. Metabolism 2020, 107, 154216. [Google Scholar] [CrossRef]
- Wu, X.; Luo, S.; Zheng, X.; Ding, Y.; Wang, S.; Ling, P.; Yue, T.; Xu, W.; Yan, J.; Weng, J. Glycemic control in children and teenagers with type 1 diabetes around lockdown for COVID-19: A continuous glucose monitoring-based observational study. J. Diabetes Investig. 2021, 12, 1708–1717. [Google Scholar] [CrossRef]
- Kapoor, R.; Timsina, L.R.; Gupta, N.; Kaur, H.; Vidger, A.J.; Pollander, A.M.; Jacobi, J.; Khare, S.; Rahman, O. Maintaining Blood Glucose Levels in Range (70-150 mg/dL) is Difficult in COVID-19 Compared to Non-COVID-19 ICU Patients-A Retrospective Analysis. J. Clin. Med. 2020, 9, 3635. [Google Scholar] [CrossRef]
- Gianchandani, R.; Esfandiari, N.H.; Ang, L.; Iyengar, J.; Knotts, S.; Choksi, P.; Pop-Busui, R. Managing Hyperglycemia in the COVID-19 Inflammatory Storm. Diabetes 2020, 69, 2048–2053. [Google Scholar] [CrossRef] [PubMed]
- Christoforidis, A.; Kavoura, E.; Nemtsa, A.; Pappa, K.; Dimitriadou, M. Coronavirus lockdown effect on type 1 diabetes management omicronn children wearing insulin pump equipped with continuous glucose monitoring system. Diabetes Res. Clin. Pract. 2020, 166, 108307. [Google Scholar] [CrossRef] [PubMed]
- Scott, E.S.; Jenkins, A.; Fulcher, G. Challenges of diabetes management during the COVID-19 pandemic. Med. J. Aust. 2020, 213, 56–57.e1. [Google Scholar] [CrossRef] [PubMed]
- Holman, N.; Knighton, P.; Kar, P.; O’Keefe, J.; Curley, M.; Weaver, A.; Barron, E.; Bakhai, C.; Khunti, K.; Wareham, N.J.; et al. Risk factors for COVID-19-related mortality in people with type 1 and type 2 diabetes in England: A population-based cohort study. Lancet Diabetes Endocrinol. 2020, 8, 823–833. [Google Scholar] [CrossRef]
- Lesniak, C.; Ong, R.; Akula, M.S.; Douedi, S.; Akoluk, A.; Soomro, R.; Copca-Alvarez, A.; Purewal, T.; Patel, I.; Upadhyaya, V.; et al. Inpatient glycemic control and outcome of COVID-19 patients: A retrospective cohort. SAGE Open Med. 2021, 9, 20503121211039105. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V. A Cochrane Handbook for Systematic Reviews of Interventions. In Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2022. [Google Scholar]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Mishra, Y.; Prashar, M.; Sharma, D.; Akash Kumar, V.P.; Tilak, T.V.S.V.G.K. Diabetes, COVID 19 and mucormycosis: Clinical spectrum and outcome in a tertiary care medical center in Western India. Diabetes Metab. Syndr. 2021, 15, 102196. [Google Scholar] [CrossRef]
- Raoufi, M.; Khalili, S.; Mansouri, M.; Mahdavi, A.; Khalili, N. Well-controlled vs poorly-controlled diabetes in patients with COVID-19: Are there any differences in outcomes and imaging findings? Diabetes Res. Clin. Pract. 2020, 166, 108286. [Google Scholar] [CrossRef]
- Pérez-Belmonte, L.M.; Torres-Peña, J.D.; López-Carmona, M.D.; Ayala-Gutiérrez, M.M.; Fuentes-Jiménez, F.; Huerta, L.J.; Muñoz, J.A.; Rubio-Rivas, M.; Madrazo, M.; Garcia, M.G.; et al. Mortality and other adverse outcomes in patients with type 2 diabetes mellitus admitted for COVID-19 in association with glucose-lowering drugs: A nationwide cohort study. BMC Med. 2020, 18, 359. [Google Scholar] [CrossRef]
- Wang, B.; Glicksberg, B.S.; Nadkarni, G.N.; Vashishth, D. Evaluation and management of COVID-19-related severity in people with type 2 diabetes. BMJ Open Diabetes Res. Care 2021, 9, e002299. [Google Scholar] [CrossRef]
- Bhatti, J.M.; Raza, S.A.; Shahid, M.O.; Akhtar, A.; Ahmed, T.; Das, B. Association between glycemic control and the outcome in hospitalized patients with COVID-19. Endocrine 2022, 77, 213–220. [Google Scholar] [CrossRef]
- Zhu, L.; She, Z.; Cheng, X.; Qin, J.; Zhang, X.; Cai, J.; Lei, F.; Wang, H.; Xie, J.; Wang, W.; et al. Association of Blood Glucose Control and Outcomes in Patients with COVID-19 and Pre-existing Type 2 Diabetes. Cell Metab. 2020, 31, 1068–1077.e3. [Google Scholar] [CrossRef]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical Characteristics of 138 Hospitalized Patients with 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA 2020, 323, 1061–1069. [Google Scholar] [CrossRef]
- Foppa, L.; Alessi, J.; Nemetz, B.; de Matos, R.; Telo, G.H.; Schaan, B.D. Quality of care in patients with type 1 diabetes during the COVID-19 pandemic: A cohort study from Southern Brazil. Diabetol. Metab. Syndr. 2022, 14, 75. [Google Scholar] [CrossRef]
- Mehta, P.B.; Kohn, M.A.; Koliwad, S.K.; Rushakoff, R.J. Lack of association between either outpatient or inpatient glycemic control and COVID-19 illness severity or mortality in patients with diabetes. BMJ Open Diabetes Res. Care 2021, 9, e002203. [Google Scholar] [CrossRef]


| Author/Year | Study Design | Country | Participants | Intervention | Results | Conclusion |
|---|---|---|---|---|---|---|
| Mishra et al. (2021) [28] | Cohort | India | 32 | Blood glucose was monitored in CAM patients. | In patients with CAM, 87.5% had Diabetes Mellitus as the most common co-morbidity. | Uncontrolled BG in CAM patients reported high fatality. |
| Lesniak et al. (2021) [25] | Cohort | USA | 292 | Glycemic control among diabetic and non-diabetic COVID-19 patients in-hospital mortality | Significant fatality and hospitalization among COVID-19 inpatients diabetic and non-diabetic. | Study shows link between mortality and BG levels, suggesting the advantages of well-managed BG. |
| Raoufi et al. (2020) [29] | Cohort | Iran | 117 | Two groups of COVID-19 patients based on HbA1c values screened. | No difference in mortality and recovery rate between two groups (p = 0.54 and p = 0.85, respectively). | No significant difference in clinical outcomes and chest CT severity scores between the two groups. |
| Perez-Belmonte et al. (2020) [30] | Cohort | Spain | 2666 | All COVID-19 patients with type 2 diabetes mellitus on hypoglycemic medication in the Spanish Society of Internal Medicine’s registry followed for mortality | Covid patients with home glucose-lowering drugs showed a significant association with in-hospital deaths, need for ICU, mechanical ventilation and increase hospital stay. | In patients with type 2 diabetes mellitus admitted for COVID-19, at-home glucose-lowering drugs showed no significant association with mortality and adverse outcomes. |
| Wang et al. (2021) [31] | Cohort | USA | 16,504 | COVID-19 patients with DM on Insulin, metformin, ACEIs, angiotensin receptor blockers, and corticosteroids. | The HR of longitudinal HbA1c for risk of ICU = 1.12 (per 1% increase, p < 0.001) and 1.48 (comparing group with poor (HbA1c ≥ 9%) and adequate glycemic control (HbA1c 6–9%), p < 0.001). | Combination of metformin, insulin, and corticosteroids prevents COVID-19 patients with T2D developing severe complications. |
| Boeder et al. (2022) [17] | Cohort | USA | 24 | CGM in critically ill COVID-19 patient in emergency department | Individuals’ glycemic control improved when CGM was used (mean difference –30.7 mg/dL). Mean absolute relative difference of 14.8% and 99.5% of CGM. | BG management in COVID-19 patients improved during IV therapy. |
| Klonoff et al. (2020) [3] | Cohort | USA | 1544 | Patients grouped according to achieved mean glucose category in mg/dL. | In non-ICU patients, severe hyperglycemia BG >13.88 mmol on days 2–3 linked with high fatality (HR) 7.17; 95% CI 2.62–19.62) compared with patients with BG <7.77 mmol/L). In patients admitted directly to the ICU. Severe hyperglycemia on admission reported increased mortality (HR 3.14; 95% CI 1.44–6.88). | Hyperglycemia highly linked with poor outcomes in COVID-19 inpatients. |
| Bhatti et al. (2022) [32] | Cohort | Pakistan | 638 | COVID-19 patients with and undiagnosed DM. | Mortality was lowest in Patients with HbA1c of <7% (p < 0.001). Need for ICU admission highest in patients with HbA1c between 7 and 10% (p 0.002). | Effective glycemic control is related to lower risk of fatality in COVID-19 infections. |
| Zhang et al. (2020) [7] | Cohort | China | 52 | Retrospectively hospitalized COVID-19 patients with and without DM reviewed. | COVID-19 patients with diabetes were more likely to develop severe or critical disease conditions with more complications and had higher incidence rates of antibiotic therapy, non-invasive and invasive mechanical ventilation, and death (11.1% vs. 4.1%). | COVID-19 patients with diabetes showed poor clinical outcomes. |
| Fadini et al. (2020) [4] | Cohort | Italy | 413 | Retrospectively COVID patients with diabetes analyzed for composite of ICU admission or death. | ICU admission/death in 37.4% of patients with diabetes compared to 20.3% in those without diabetes. (RR 1.85; 95% CI 1.33–2.57; p < 0.001). | Newly diagnosed diabetes is key determinant of COVID-19 severity. |
| Zhu et al. (2020) [33] | Cohort | China | 7337 | Retrospectively studying subjects with COVID-19 and diabetes for prognosis | Subjects with DM needed more medical interventions. Higher death rates (7.8% versus 2.7%; (HR = 1.49) and multiple organ injuries than the non-diabetic patients. | DM is a key predictor for COVID-19 infection progression. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chander, S.; Deepak, V.; Kumari, R.; Leys, L.; Wang, H.Y.; Mehta, P.; Sadarat, F. Glycemic Control in Critically Ill COVID-19 Patients: Systematic Review and Meta-Analysis. J. Clin. Med. 2023, 12, 2555. https://doi.org/10.3390/jcm12072555
Chander S, Deepak V, Kumari R, Leys L, Wang HY, Mehta P, Sadarat F. Glycemic Control in Critically Ill COVID-19 Patients: Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2023; 12(7):2555. https://doi.org/10.3390/jcm12072555
Chicago/Turabian StyleChander, Subhash, Vishal Deepak, Roopa Kumari, Lorenzo Leys, Hong Yu Wang, Puja Mehta, and FNU Sadarat. 2023. "Glycemic Control in Critically Ill COVID-19 Patients: Systematic Review and Meta-Analysis" Journal of Clinical Medicine 12, no. 7: 2555. https://doi.org/10.3390/jcm12072555
APA StyleChander, S., Deepak, V., Kumari, R., Leys, L., Wang, H. Y., Mehta, P., & Sadarat, F. (2023). Glycemic Control in Critically Ill COVID-19 Patients: Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 12(7), 2555. https://doi.org/10.3390/jcm12072555






