Diagnostic Accuracy of Endoscopic Ultrasonography in Selecting Patients for Endoscopic Submucosal Dissection for Early Gastrointestinal Neoplasms
Abstract
1. Introduction
2. Methods
3. EUS Technique
4. Data Analysis
- -
- True-positive lesions with submucosal invasion;
- -
- True negative lesions without submucosal invasion;
- -
- False negative lesions with submucosal invasion (understaged by EUS);
- -
- False positive lesions without submucosal invasion (overstaged by EUS).
5. Results
5.1. Patients Demographics and Tumor Characteristics and Locations
5.2. Operative Procedures and Pathological Findings
5.3. Overall Diagnostic Accuracy
6. Discussion
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Han, Y.; Sun, S.; Guo, J.; Ge, N.; Wang, S.; Liu, X.; Wang, G.; Hu, J.; Wang, S. Is endoscopic ultrasonography useful for endoscopic submucosal dissection? Endosc. Ultrasound 2016, 5, 284–290. [Google Scholar] [CrossRef]
- Coda, S.; Lee, S.Y.; Gotoda, T. Endoscopic mucosal resection and endoscopic submucosal dissection as treatments for early gastrointestinal cancers in Western countries. Gut Liver 2007, 1, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Stein, H.J.; Feith, M.; Bruecher, B.L.; Naehrig, J.; Sarbia, M.; Siewert, J.R. Early esophageal cancer pattern of lymphatic spread and prognostic factors for long-term survival after surgical resection. Ann. Surg. 2005, 242, 566–575. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Hofstetter, W.; Rashid, A.; Swisher, S.G.; Correa, A.M.; Ajani, J.A.; Hamilton, S.R.; Wu, T.T. Significance of the depth of tumor invasion and lymph node metastasis in superficially invasive (T1) esophageal adenocarcinoma. Am. J. Surg. Pathol. 2005, 29, 1079–1085. [Google Scholar] [CrossRef] [PubMed]
- Japan Esophageal Society. Japanese Classification of Esophageal Cancer, 11th Edition: Part II and III. Esophagus 2017, 14, 37–65. [Google Scholar] [CrossRef]
- Participants in the Paris Workshop. The Paris endoscopic classification of superficial neoplastic lesions: Esophagus, stomach, and colon: November 30 to December 1, 2002. Gastrointest. Endosc. 2003, 58 (Suppl. 6), S3–S43. [Google Scholar] [CrossRef]
- Amin, M.B.; Edge, S.B.; Greene, F.L. AJCC Cancer Staging Manual; Springer: New York, NY, USA, 2017; Volume 1024. [Google Scholar]
- Ide, H.; Nakamura, T.; Hayashi, K.; Endo, T.; Kobayashi, A.; Eguchi, R.; Hanyu, F. Esophageal squamous cell carcinoma: Pathology and prognosis. World. J. Surg. 1994, 18, 321–330. [Google Scholar] [CrossRef]
- Kodama, M.; Kakegawa, T. Treatment of superficial cancer of the esophagus: A summary of responses to a questionnaire on superficial cancer of the esophagus in Japan. Surgery 1998, 132, 432–439. [Google Scholar] [CrossRef]
- Takubo, K.; Aida, J.; Sawabe, M.; Kurosomi, M.; Arima, M.; Fujishiro, M.; Arai, T. Early sqamous cell carcinoma of the esophagus: The Japanese viewpoint. Histopathology 2007, 51, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Yanai, H.; Harada, T.; Okamoto, T.; Hirano, A.; Takeo, N.; Yoshida, T.; Okita, K.; Kawano, T. Prognostic value and interobserver agreement of endoscopic ultrasonography for superficial squamous cell carcinoma of the esophagus: A prospective study. Int. J. Gastrointest. Cancer 2003, 34, 1–8. [Google Scholar] [CrossRef]
- Gotoda, T.; Yanagisawa, A.; Sasako, M.; Ono, H.; Nakanishi, Y.; Shimoda, T.; Kato, Y. Incindence of lymph node metastasis from early gastric cancer: Estimation with a large number of cases at two large centers. Gastric Cancer 2000, 3, 219–225. [Google Scholar] [CrossRef]
- Gotoda, T. Endoscopic resection of early gastric cancer. Gastric Cancer 2007, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Uraoka, T.; Parra-Blanco, A.; Yahagi, N. Colorectal endoscopic submucosal dissection: Is it suitable in western countries? J. Gastroenterol. Hepatol. 2013, 28, 406–414. [Google Scholar] [CrossRef] [PubMed]
- Fujimori, T.; Fuji, S.; Saito, N.; Sugihara, K. Pathological diagnosis of early colorectal carcinoma and its clinical implications. Digestion 2009, 79 (Suppl. 1), 40–51. [Google Scholar] [CrossRef]
- Tsuzuki, T.; Okada, H.; Kawahara, Y.; Nasu, J.; Takenaka, R.; Inoue, M.; Kawano, S.; Kita, M.; Hori, K.; Yamamoto, K. Usefulness and problems of endoscopic ultrasonography in prediction of the depth of tumor invasion in early gastric cancer. Acta Med. Okayama 2011, 65, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.W. The Role of Endosonography in the Staging of Gastrointestinal Cancers. Clin. Endosc. 2015, 48, 297–301. [Google Scholar] [CrossRef]
- Trindade, A.J.; Berzin, T.M. Clinical controversies in endoscopic ultrasound. Gastroenterol. Rep. 2013, 1, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Xu, T.; Zhang, Q.; Liu, J.; Ding, Z.; Hou, X. The New American Joint Committee on Cancer T staging system for stomach: Increased complexity without clear improvement in predictive accuracy for endoscopic ultrasound. BMC Gastroenterol. 2021, 21, 255. [Google Scholar] [CrossRef]
- Schlemper, R.J.; Riddell, R.H.; Kato, Y.; Borchard, F.; Cooper, H.S.; Dawsey, S.M.; Dixon, M.F.; Fenoglio-Preiser, C.M.; Fléjou, J.F.; Geboes, K.; et al. The Vienna classification of gastrointestinal epithelial neoplasia. Gut 2000, 47, 251–255. [Google Scholar] [CrossRef]
- Ishihara, R.; Arima, M.; Iizuka, T.; Oyama, T.; Katada, C.; Kato, M.; Goda, K.; Goto, O.; Tanaka, K.; Yano, T.; et al. Japan Gastroenterological Endoscopy Society Guidelines Committee of ESD/EMR for Esophageal Cancer. Endoscopic submucosal dissection/endoscopic mucosal resection guidelines for esophageal cancer. Dig. Endosc. 2020, 32, 452–493. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.H.; Hyung, W.J.; Cho, G.S.; Kim, M.C.; Han, S.U.; Kim, W.; Ryu, S.W.; Lee, H.J.; Song, K.Y. Morbidity and mortality of laparoscopic gastrectomy versus open gastrectomy for gastric cancer: An interim report—A phase III multicenter, prospective, randomized Trial (KLASS Trial). Ann. Surg. 2010, 251, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.I.; Kim, Y.A.; Kim, C.G.; Ryu, K.W.; Kim, Y.W.; Sim, J.A.; Yun, Y.H.; Choi, I.J. Serial intermediate-term quality of life comparison after endoscopic submucosal dissection versus surgery in early gastric cancer patients. Surg. Endosc. 2018, 32, 2114–2122. [Google Scholar] [CrossRef] [PubMed]
- Tae, C.H.; Shim, K.N.; Kim, B.W.; Kim, J.H.; Hong, S.J.; Baik, G.H.; Song, H.J.; Kim, Y.S.; Jang, S.H.; Jung, H.K. Comparison of subjective quality of life after endoscopic submucosal resection or surgery for early gastric cancer. Sci. Rep. 2020, 10, 6680. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, N.; Saito, Y.; Uraoka, T.; Matsuda, T.; Suzuki, H.; Fujii, T. Treatment strategy for laterally spreading tumors in Japan: Before and after the introduction of endoscopic submucosal dissection. J. Gastroenterol. Hepatol. 2009, 24, 1387–1392. [Google Scholar] [CrossRef]

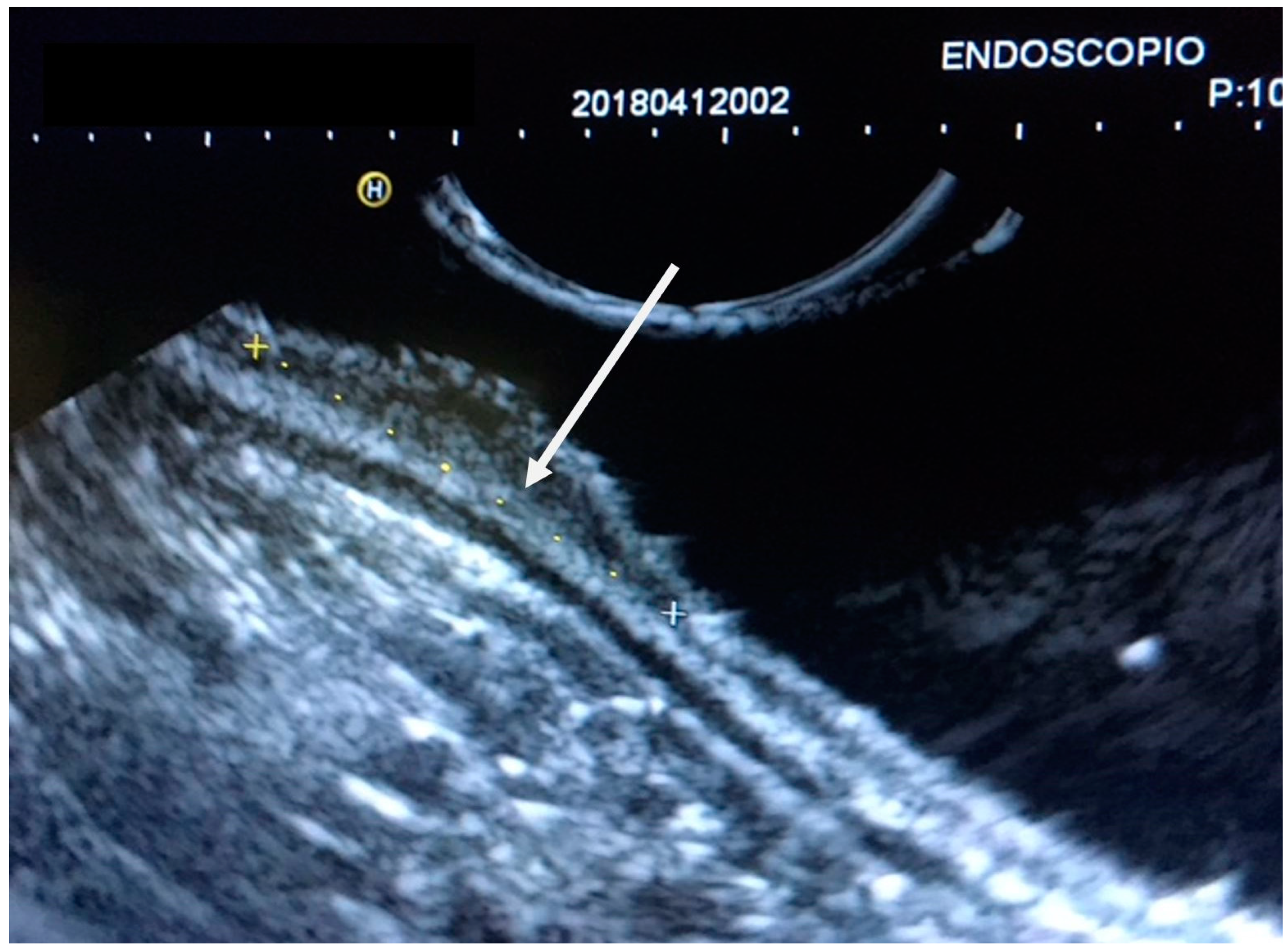
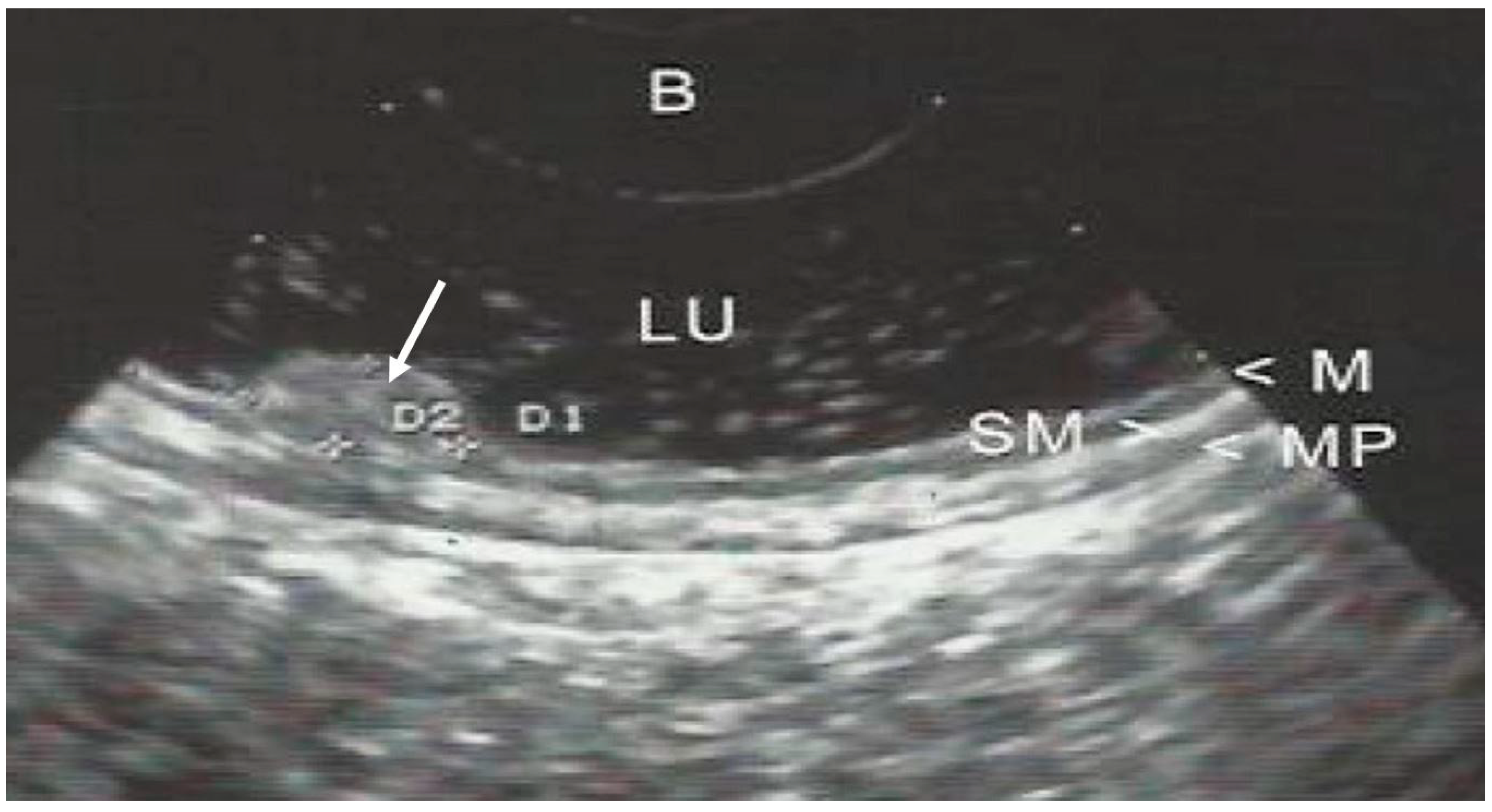
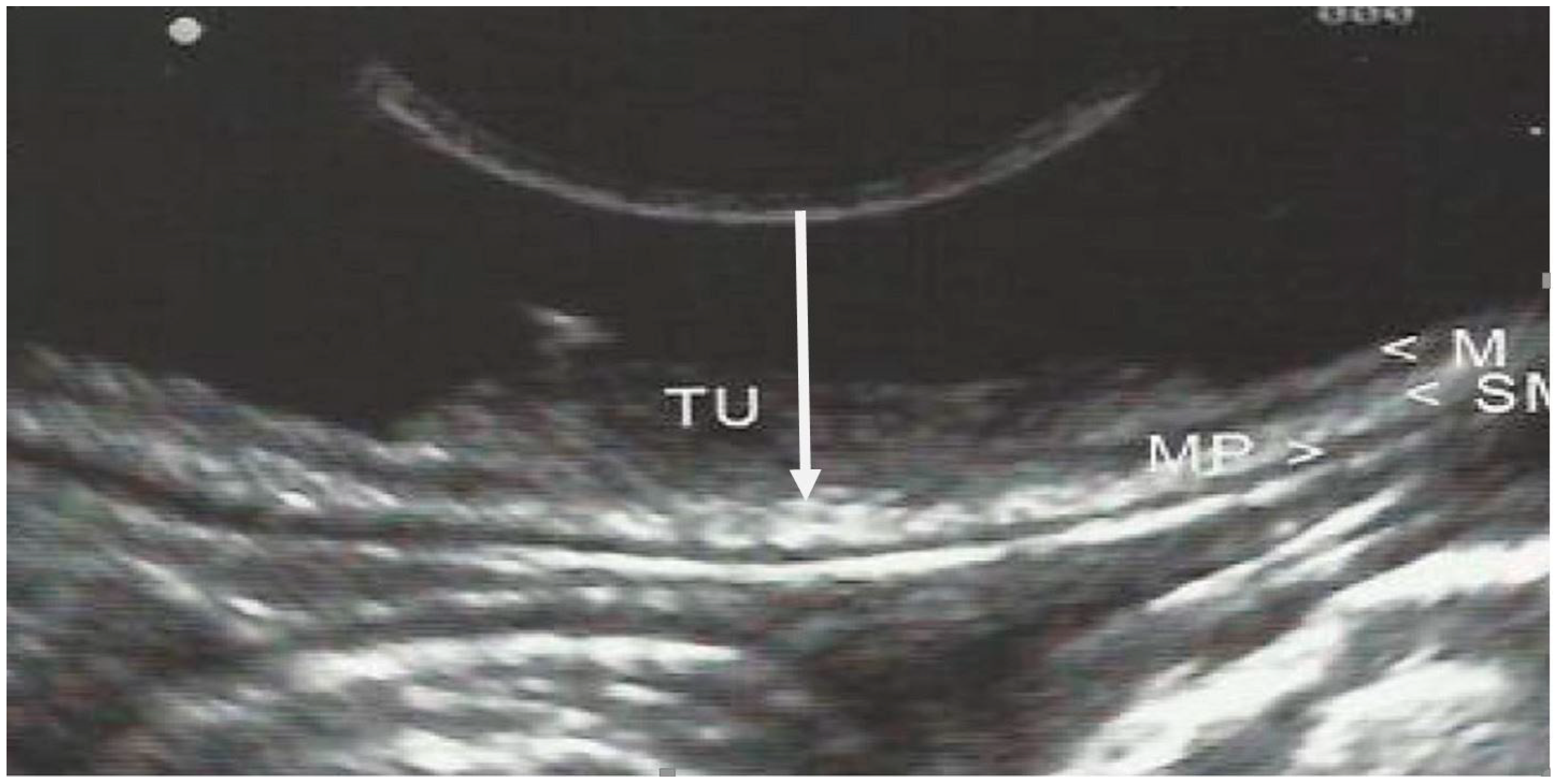
| Variable | N (%) |
|---|---|
| Noninvasive low-grade neoplasm | 81 (27.64%) |
| Noninvasive high-grade neoplasm | 90 (30.71%) |
| Invasive Adenocarcinoma | 114 (38.90%) |
| Carcinoid tumor | 8 (2.73%) |
| Variable | N (%) | |
|---|---|---|
| Gender | Male | 176 (60) |
| Female | 117 (40) | |
| Tumor Location | ||
| Esophagus | Upper third | 8 (2.7) |
| Middle third | 15 (5.1) | |
| Lower third | 18 (2.7) | |
| Stomach | Fundus | 12 (4.1) |
| Corpus | 41 (13.9) | |
| Antrum | 53 (18.1) | |
| Gastro-jejunal anastomosis | 4 (1.4) | |
| Duodenum | 13 (4.4) | |
| Sigmoid colon | 8 (6.1) | |
| Rectum | 121 (41.3) | |
| EUS layer invasion | Mucosa | 155 (52.9) |
| Submucosa | 32 (10.9) | |
| Proper muscle | 32 (10.9) | |
| Subserosa | 74 (25.3) | |
| Variable | N (%) | |
|---|---|---|
| Operative procedure | ESD (endoscopic submucosal dissection) | 175 (59.7) |
| Esophagectomy | 21 (7.2) | |
| Total gastrectomy | 10 (3.4) | |
| Distal subtotal gastrectomy | 21 (7.2) | |
| Lower anterior resection | 66 (22.5) | |
| Histological tumor depth | Mucosa | 136 (46.4) |
| Submucosa | 49 (16.7) | |
| Proper muscle | 34 (11.6) | |
| Subserosa | 64 (21.8) | |
| Serosa exposure | 10 (3.4) |
| Tumor Location | N | Accuracy Correctly Diagnosed | Incorrectly Diagnosed | Understaged | Overstaged |
|---|---|---|---|---|---|
| Esophagus | 41 | 39 (95.1) | 2 (4.9) | 2 (4.9) | - |
| Stomach | 106 | 88 (83) | 18 (16.9) | 15 (14.1) | 3 (2.8) |
| Duodenum | 13 | 13 (100) | - | - | - |
| Gastro-jejunal anastomosis | 4 | 3 (75) | 1 (25) | 1 (25) | - |
| Sigmoid colon | 8 | 6 (75) | 2 (25) | 2 (25) | - |
| Rectum | 121 | 93 (76.9) | 28 (23.1) | 18 (14.9) | 10 (8.3) |
| Total | 293 | 242 (82.6) | 51 (17.4) | 38 (12.9) | 13 (4.4) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gambitta, P.; Fontana, P.; Fanetti, I.; Veglia, G.; Vertemati, M.; Armellino, A.; Aseni, P. Diagnostic Accuracy of Endoscopic Ultrasonography in Selecting Patients for Endoscopic Submucosal Dissection for Early Gastrointestinal Neoplasms. J. Clin. Med. 2023, 12, 2505. https://doi.org/10.3390/jcm12072505
Gambitta P, Fontana P, Fanetti I, Veglia G, Vertemati M, Armellino A, Aseni P. Diagnostic Accuracy of Endoscopic Ultrasonography in Selecting Patients for Endoscopic Submucosal Dissection for Early Gastrointestinal Neoplasms. Journal of Clinical Medicine. 2023; 12(7):2505. https://doi.org/10.3390/jcm12072505
Chicago/Turabian StyleGambitta, Pietro, Paola Fontana, Ilaria Fanetti, Giulia Veglia, Maurizio Vertemati, Antonio Armellino, and Paolo Aseni. 2023. "Diagnostic Accuracy of Endoscopic Ultrasonography in Selecting Patients for Endoscopic Submucosal Dissection for Early Gastrointestinal Neoplasms" Journal of Clinical Medicine 12, no. 7: 2505. https://doi.org/10.3390/jcm12072505
APA StyleGambitta, P., Fontana, P., Fanetti, I., Veglia, G., Vertemati, M., Armellino, A., & Aseni, P. (2023). Diagnostic Accuracy of Endoscopic Ultrasonography in Selecting Patients for Endoscopic Submucosal Dissection for Early Gastrointestinal Neoplasms. Journal of Clinical Medicine, 12(7), 2505. https://doi.org/10.3390/jcm12072505






