Clinical Outcome and Prognosis of a Nosocomial Outbreak of COVID-19
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Hospital Quarantine against COVID-19
2.3. Variables
2.4. Statistical Analyses
2.5. Ethics Statement
3. Results
3.1. Clinical Characteristics
3.2. Hospital Course and Outcomes
3.3. Factors Associated with Mortality
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Qu, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. Clinical Characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Thomas, A.; Suresh, M. Assessment of COVID-19 prevention and protection measures in hospitals. Clean. Eng. Technol. 2022, 7, 100440. [Google Scholar] [CrossRef]
- Jang, W.; Kim, B.; Kim, E.S.; Song, K.-H.; Moon, S.M.; Lee, M.J.; Park, J.Y.; Kim, J.-Y.; Shin, M.J.; Lee, H.; et al. Are the Current Guidelines Sufficient to Establish Infection Control Strategies for COVID-19 Related Issues in Hospitals? J. Korean Med. Sci. 2021, 36, e343. [Google Scholar] [CrossRef]
- Shi, H.J.; Lee, J.B.; Kang, H.J.; Choi, M.K.; Eom, J.S. Importance of Wearing Facial Masks to Prevent Nosocomial Transmissions of COVID-19. Korean J. Health Infect. Control. Prev. 2021, 26, 101–107. [Google Scholar] [CrossRef]
- Biernat, M.M.; Zińczuk, A.; Biernat, P.; Bogucka-Fedorczuk, A.; Kwiatkowski, J.; Kalicińska, E.; Marciniak, D.; Simon, K.; Wróbel, T. Nosocomial outbreak of SARS-CoV-2 infection in a haematological unit—High mortality rate in infected patients with haematologic malignancies. J. Clin. Virol. 2020, 130, 104574. [Google Scholar] [CrossRef]
- Carter, B.; Collins, J.T.; Barlow-Pay, F.; Rickard, F.; Bruce, E.; Verduri, A.; Quinn, T.J.; Mitchell, E.; Price, A.; Vilches-Moraga, A.; et al. Nosocomial COVID-19 infection: Examining the risk of mortality. The COPE-Nosocomial Study (COVID in Older PEople). J. Hosp. Infect. 2020, 106, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Luong-Nguyen, M.; Hermand, H.; Abdalla, S.; Cabrit, N.; Hobeika, C.; Brouquet, A.; Goéré, D.; Sauvanet, A. Nosocomial infection with SARS-CoV-2 within Departments of Digestive Surgery. J. Visc. Surg. 2020, 157, S13–S18. [Google Scholar] [CrossRef]
- Zhou, Q.; Gao, Y.; Wang, X.; Liu, R.; Du, P.; Wang, X.; Zhang, X.; Lu, S.; Wang, Z.; Shi, Q.; et al. Nosocomial infections among patients with COVID-19, SARS and MERS: A rapid review and meta-analysis. Ann. Transl. Med. 2020, 8, 629. [Google Scholar] [CrossRef]
- Borges, V.; Isidro, J.; Macedo, F.; Neves, J.; Silva, L.; Paiva, M.; Barata, J.; Catarino, J.; Ciobanu, L.; Duarte, S.; et al. Nosocomial Outbreak of SARS-CoV-2 in a “Non-COVID-19” Hospital Ward: Virus Genome Sequencing as a Key Tool to Understand Cryptic Transmission. Viruses 2021, 13, 604. [Google Scholar] [CrossRef]
- Boshier, F.A.T.; Venturini, C.; Stirrup, O.; Guerra-Assunção, J.A.; Alcolea-Medina, A.; Becket, A.H.; Byott, M.; Charalampous, T.; Filipe, A.D.S.; Frampton, D.; et al. The Alpha variant was not associated with excess nosocomial SARS-CoV-2 infection in a multi-centre UK hospital study. J. Infect. 2021, 83, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Paltansing, S.; Sikkema, R.S.; de Man, S.J.; Koopmans, M.; Munnink, B.O.; de Man, P. Transmission of SARS-CoV-2 among healthcare workers and patients in a teaching hospital in The Netherlands confirmed by whole-genome sequencing. J. Hosp. Infect. 2021, 110, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Ponsford, M.J.; Jefferies, R.; Davies, C.; Farewell, D.; Humphreys, I.R.; Jolles, S.; Fairbairn, S.; Lewis, K.; Menzies, D.; Benjamin, A.; et al. Burden of nosocomial COVID-19 in Wales: Results from a multicentre retrospective observational study of 2508 hospitalised adults. Thorax 2021, 76, 1246–1249. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Xiao, J.; Deng, A.; Zhang, Y.; Zhuang, Y.; Hu, T.; Li, J.; Tu, H.; Li, B.; Zhou, Y.; et al. Transmission Dynamics of an Outbreak of the COVID-19 Delta Variant B.1.617.2—Guangdong Province, China, May–June 2021. China CDC Wkly. 2021, 3, 584–586. [Google Scholar] [CrossRef]
- Itoh, N.; Akazawa, N.; Ishikane, M.; Kawabata, T.; Kawamura, D.; Chikusa, T.; Kodama, E.N.; Ohmagari, N. Lessons learned from an outbreak of COVID-19 in the head and neck surgery ward of a Japanese cancer center during the sixth wave by Omicron. J. Infect. Chemother. 2022, 28, 1610–1615. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Lee, J.; Park, H.; Lim, Y.-J.; Kim, E.O.; Park, M.-S.; Kim, S.-H. Nosocomial Outbreak by Delta Variant from a Fully Vaccinated Patient. J. Korean Med. Sci. 2022, 37, e133. [Google Scholar] [CrossRef] [PubMed]
- Sohn, Y.J.; Shin, P.J.; Oh, W.S.; Kim, E.; Kim, Y.; Kim, Y.K. Clinical Characteristics of Patients Who Contracted the SARS-CoV-2 Omicron Variant from an Outbreak in a Single Hospital. Yonsei Med. J. 2022, 63, 790–793. [Google Scholar] [CrossRef]
- Hawkins, L.P.A.; Pallett, S.J.C.; Mazzella, A.; Anton-Vazquez, V.; Rosas, L.; Jawad, S.M.; Shakespeare, D.; Breathnach, A. Transmission dynamics and associated mortality of nosocomial COVID-19 throughout 2021: A retrospective study at a large teaching hospital in London. J. Hosp. Infect. 2023, 133, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.-C.; Chan, V.W.-M.; Yuen, L.L.-H.; AuYeung, C.H.-Y.; Leung, J.O.-Y.; Li, C.-K.; Kwok, M.O.-T.; So, S.Y.-C.; Chen, J.H.-K.; Chiu, K.H.-Y.; et al. Infection of healthcare workers despite a high vaccination rate during the fifth wave of COVID-19 due to Omicron variant in Hong Kong. Infect. Prev. Pr. 2023, 5, e124. [Google Scholar] [CrossRef]
- Jia, Z.; Gong, W. Will Mutations in the Spike Protein of SARS-CoV-2 Lead to the Failure of COVID-19 Vaccines? J. Korean Med. Sci. 2021, 36, e124. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Thambiraja, T.S.; Karuppanan, K.; Subramaniam, G. Omicron and Delta variant of SARS-CoV-2: A comparative computational study of spike protein. J. Med. Virol. 2022, 94, 1641–1649. [Google Scholar] [CrossRef]
- Jang, Y.; Kim, I.J.; Moon, S.-S.; Kim, S.B.; Lee, J. Correlation Between Third Dose of COVID-19 Vaccines and Regional Case Fatality Rates During the Omicron Wave in Korea. J. Korean Med. Sci. 2022, 37, e347. [Google Scholar] [CrossRef] [PubMed]
- Statistics Korea. COVID-19 Dashboard. Available online: http://kosis.kr/covid/covid_index.do (accessed on 1 December 2022).
- Li, Y.-K.; Peng, S.; Li, L.-Q.; Wang, Q.; Ping, W.; Zhang, N.; Fu, X.-N. Clinical and Transmission Characteristics of COVID-19—A Retrospective Study of 25 Cases from a Single Thoracic Surgery Department. Curr. Med. Sci. 2020, 40, 295–300. [Google Scholar] [CrossRef]
- Leducq, V.; Couturier, J.; Granger, B.; Jolivet, S.; Morand-Joubert, L.; Robert, J.; Denis, M.; Salauze, B.; Goldstein, V.; Zafilaza, K.; et al. Investigation of healthcare-associated COVID-19 in a large French hospital group by whole-genome sequencing. Microbiol. Res. 2022, 263, 127133. [Google Scholar] [CrossRef]
- Klompas, M.; Pandolfi, M.C.; Nisar, A.B.; Baker, M.A.; Rhee, C. Association of Omicron vs Wild-type SARS-CoV-2 Variants with Hospital-Onset SARS-CoV-2 Infections in a US Regional Hospital System. JAMA 2022, 328, 296–298. [Google Scholar] [CrossRef] [PubMed]
- Eckart, A.; Hauser, S.I.; Kutz, A.; Haubitz, S.; Hausfater, P.; Amin, D.; Amin, A.; Huber, A.; Mueller, B.; Schuetz, P. Combination of the National Early Warning Score (NEWS) and inflammatory biomarkers for early risk stratification in emergency department patients: Results of a multinational, observational study. BMJ Open 2019, 9, e024636. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.Y. COVID-19 in South Korea. Postgrad Med. J. 2020, 96, 399–402. [Google Scholar] [CrossRef]
- Nyberg, T.; Ferguson, N.M.; Nash, S.G.; Webster, H.H.; Flaxman, S.; Andrews, N.; Hinsley, W.; Bernal, J.L.; Kall, M.; Bhatt, S.; et al. Comparative analysis of the risks of hospitalisation and death associated with SARS-CoV-2 omicron (B.1.1.529) and delta (B.1.617.2) variants in England: A cohort study. Lancet 2022, 399, 1303–1312. [Google Scholar] [CrossRef]
- Kim, B.-G.; Kang, M.; Lim, J.; Lee, J.; Kang, D.; Kim, M.; Kim, J.; Park, H.; Min, K.H.; Cho, J.; et al. Comprehensive risk assessment for hospital-acquired pneumonia: Sociodemographic, clinical, and hospital environmental factors associated with the incidence of hospital-acquired pneumonia. BMC Pulm. Med. 2022, 22, 21. [Google Scholar] [CrossRef]
- Kim, S.H.; Choi, H.S.; Jin, E.S.; Choi, H.; Lee, H.; Lee, S.-H.; Lee, C.Y.; Lee, M.G.; Kim, Y. Predicting severe outcomes using national early warning score (NEWS) in patients identified by a rapid response system: A retrospective cohort study. Sci. Rep. 2021, 11, 1–8. [Google Scholar] [CrossRef]
- Jia, H.; Li, L.; Li, W.; Hou, T.; Ma, H.; Yang, Y.; Wu, A.; Liu, Y.; Wen, J.; Yang, H.; et al. Impact of Healthcare-Associated Infections on Length of Stay: A Study in 68 Hospitals in China. BioMed. Res. Int. 2019, 2019, 2590563. [Google Scholar] [CrossRef]
- Kwon, S.L.; Oh, J. COVID-19 vaccination program in South Korea: A long journey toward a new normal. Health Policy Technol. 2022, 11, 100601. [Google Scholar] [CrossRef]
- Lee, C.M.; Lee, E.; Park, W.B.; Choe, P.G.; Song, K.-H.; Kim, E.S.; Park, S.-W. Breakthrough COVID-19 Infection during the Delta Variant Dominant Period: Individualized Care Based on Vaccination Status Is Needed. J. Korean Med. Sci. 2022, 37. [Google Scholar] [CrossRef]
- Nomura, S.; Eguchi, A.; Yoneoka, D.; Kawashima, T.; Tanoue, Y.; Murakami, M.; Sakamoto, H.; Maruyama-Sakurai, K.; Gilmour, S.; Shi, S.; et al. Reasons for being unsure or unwilling regarding intention to take COVID-19 vaccine among Japanese people: A large cross-sectional national survey. Lancet Reg. Health West. Pac. 2021, 14, 100223. [Google Scholar] [CrossRef] [PubMed]
- Meister, T.; Pisarev, H.; Kolde, R.; Kalda, R.; Suija, K.; Milani, L.; Karo-Astover, L.; Piirsoo, M.; Uusküla, A. Clinical characteristics and risk factors for COVID-19 infection and disease severity: A nationwide observational study in Estonia. PLoS ONE 2022, 17, e0270192. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Syed-Ahmed, M.; Narayanan, M. Immune Dysfunction and Risk of Infection in Chronic Kidney Disease. Adv. Chronic Kidney Dis. 2019, 26, 8–15. [Google Scholar] [CrossRef]
- Rockett, R.J.; Arnott, A.; Lam, C.; Sadsad, R.; Timms, V.; Gray, K.-A.; Eden, J.-S.; Chang, S.; Gall, M.; Draper, J.; et al. Revealing COVID-19 transmission in Australia by SARS-CoV-2 genome sequencing and agent-based modeling. Nat. Med. 2020, 26, 1398–1404. [Google Scholar] [CrossRef] [PubMed]
- Blackstone, J.; Stirrup, O.; Mapp, F.; Panca, M.; Copas, A.; Flowers, P.; Hockey, L.; Price, J.; Partridge, D.; Peters, C.; et al. Protocol for the COG-UK hospital-onset COVID-19 infection (HOCI) multicentre interventional clinical study: Evaluating the efficacy of rapid genome sequencing of SARS-CoV-2 in limiting the spread of COVID-19 in UK NHS hospitals. BMJ Open 2022, 12, e052514. [Google Scholar] [CrossRef]
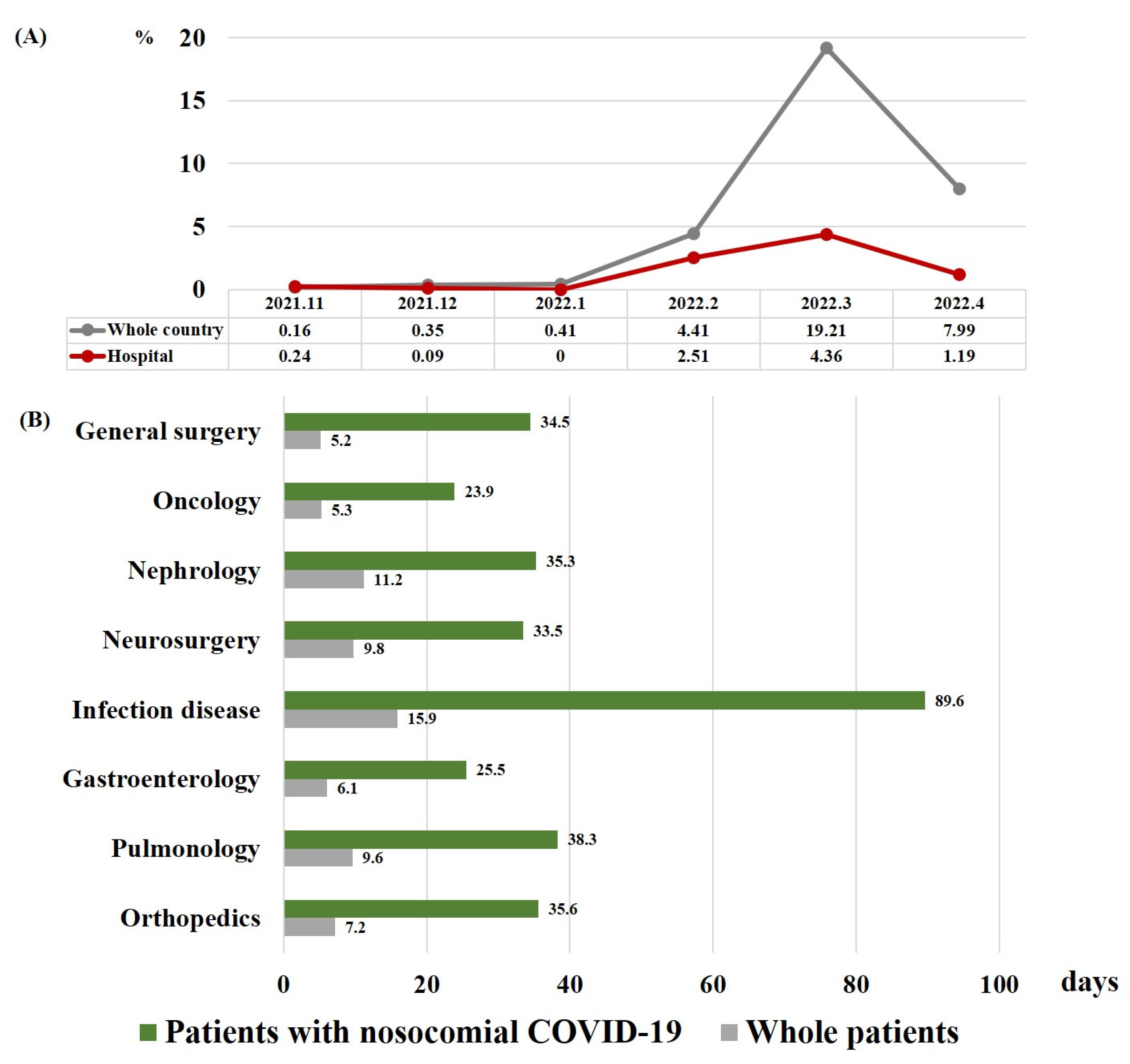
| Variable | All Patients (n = 167) | Survivor (n = 153) | Non-Survivor (n = 14) | p-Value |
|---|---|---|---|---|
| Sex, male | 105 (63%) | 97 (63%) | 8 (57%) | 0.774 |
| Age, year | 69 (57–81) | 68 (57–79) | 78 (66–89) | 0.047 |
| BMI, kg/m2 | 23.4 (20.8–26.2) | 23.4 (20.1–26.2) | 21.9 (18.6–24.5) | 0.126 |
| Co-morbidity | ||||
| Hypertension | 84 (50%) | 76 (50%) | 8 (57%) | 0.781 |
| Diabetes | 53 (32%) | 47 (31%) | 6 (43%) | 0.376 |
| Cancer | 42 (25%) | 36 (24%) | 6 (43%) | 0.119 |
| Chronic kidney disease | 29 (17%) | 24 (16%) | 5 (36%) | 0.071 |
| History of CVA | 24 (14%) | 20 (13%) | 4 (28.6%) | 0.121 |
| Chronic airway disease | 13 (7.8%) | 10 (6.5%) | 3 (21%) | 0.081 |
| Liver cirrhosis | 11 (6.6%) | 9 (5.9%) | 2 (14.3%) | 0.232 |
| Congestive heart failure | 10 (6.0%) | 10 (6.5%) | 0 | 1.000 |
| Department | ||||
| Orthopedics | 32 (19%) | 32 (21%) | 0 | 0.074 |
| Pulmonology | 28 (17%) | 23 (15%) | 5 (36%) | 0.062 |
| Gastroenterology | 23 (14%) | 20 (13%) | 3 (21%) | 0.414 |
| Infection disease | 18 (11%) | 17 (11%) | 1 (7%) | 1.000 |
| Neurosurgery | 15 (9.0%) | 15 (9.2%) | 0 | 0.618 |
| Nephrology | 15 (9.0%) | 13 (8.5%) | 2 (14.3%) | 0.365 |
| Oncology | 12 (7.2%) | 10 (6.5%) | 2 (14.3%) | 0.265 |
| General surgery | 6 (3.6%) | 6 (3.9%) | 0 | 1.000 |
| Cardiology | 5 (3.0%) | 5 (3.3%) | 0 | 1.000 |
| Neurology | 4 (2.3%) | 4 (2.6%) | 0 | 1.000 |
| Others | 9 (5.4%) | 8 (5.2%) | 1 (7.1%) | 0.554 |
| Diagnosis | ||||
| Cancer | 31 (19%) | 28 (19%) | 3 (21%) | 0.726 |
| Fracture | 18 (11%) | 17 (11%) | 1 (7%) | 1.000 |
| CVA | 18 (11%) | 18 (12%) | 0 | 0.368 |
| Pneumonia | 17 (10%) | 13 (8.5%) | 4 (29%) | 0.039 |
| Abdomen infection | 16 (9.6%) | 16 (11%) | 0 | 0.366 |
| Osteomyelitis or arthritis | 16 (9.6%) | 14 (9.2%) | 2 (14%) | 0.627 |
| Spine fracture and stenosis | 11 (6.6%) | 11 (7.2%) | 0 | 0.602 |
| Urinary tract infection | 10 (6.0%) | 9 (5.9%) | 1 (7.1%) | 0.594 |
| Acute or chronic renal failure | 9 (5.4%) | 9 (5.9%) | 0 | 1.000 |
| Others | 21 (12%) | 18 (11.8%) | 3 (21.4%) | 0.389 |
| Vaccination | ||||
| None | 37 (22%) | 28 (18.3%) | 9 (64.3%) | <0.001 |
| First | 7 (4.2%) | 7 (4.6%) | 0 | 1.000 |
| Tozinameran | 5 (71.4%) | |||
| Covishield | 2 (28.6%) | |||
| Second | 36 (21.6%) | 36 (23.5%) | 0 | 0.042 |
| Tozinameran | 20 (55.6%) | |||
| Covishield | 10 (27.8%) | |||
| Elasomeran | 6 (16.7%) | |||
| Third | 87 (52.1%) | 82 (53.6%) | 5 (35.7%) | 0.266 |
| Tozinameran | 74 (85.1%) | 71 (86.6%) | 3 (60%) | |
| Elasomeran | 13 (14.9%) | 11 (13.4%) | 2 (40%) | |
| Type of hospital room | ||||
| Single person room | 14 (8.4%) | 11 (7.2%) | 3 (21.4%) | 0.098 |
| Three-person room | 9 (5.4%) | 9 (5.9%) | 0 | 1.000 |
| Four-person room | 46 (28%) | 42 (28%) | 4 (29%) | 1.000 |
| Five-person room | 8 (4.8%) | 6 (3.9%) | 2 (14.3%) | 0.137 |
| Six-person room | 87 (52%) | 82 (53.6%) | 5 (35.7%) | 0.266 |
| Intensive care unit | 3 (1.8%) | 3 (2.0%) | 0 | 1.000 |
| Variable | All patients (n = 167) | Survivor (n = 153) | Non-Survivor (n = 14) | p-Value |
|---|---|---|---|---|
| Duration, days | ||||
| From admission to COVID-19 infection | 11 (7–23) | 10 (7–23) | 12 (8–34) | 0.344 |
| Hospital days | 24 (15–42) | 24 (15–43) | 27 (14–43) | 0.995 |
| Symptoms | ||||
| Fever | 47 (28%) | 40 (26%) | 7 (50%) | 0.068 |
| Cough | 28 (17%) | 25 (16%) | 3 (21%) | 0.707 |
| Dyspnea | 9 (11%) | 14 (9.2%) | 5 (36%) | 0.012 |
| Asymptomatic state | 86 (52%) | 83 (54%) | 3 (21%) | 0.024 |
| Status before COVID-19 | ||||
| Ventilator | 4 (2.4%) | 3 (2.0%) | 1 (7.1%) | 0.298 |
| Antibiotics | 101 (61%) | 90 (59%) | 11 (79%) | 0.168 |
| Steroid | 11 (6.6%) | 8 (5.2%) | 3 (21%) | 0.052 |
| Status after COVID-19 | ||||
| Pneumonia | 27 (16%) | 17 (11%) | 10 (71%) | <0.001 |
| Sepsis | 11 (6.6%) | 2 (1.3%) | 9 (64%) | <0.001 |
| Ventilator | 10 (6.0%) | 4 (2.6%) | 6 (43%) | <0.001 |
| Change of antibiotics | 40 (24%) | 29 (19%) | 11 (79%) | <0.001 |
| NEWS | 2 (1–4) | 2 (1–3) | 4 (3–6) | <0.001 |
| Medication for COVID-19 | ||||
| Remdesivir | 145 (87%) | 131 (86%) | 14 (100%) | 0.219 |
| Paxlovid | 3 (1.8%) | 3 (2.0%) | 0 | 1.000 |
| Characteristics | Unadjusted HR | 95% CI | p-Value | Adjusted HR | 95% CI | p-Value |
|---|---|---|---|---|---|---|
| Age ≥ 70 years | 1.335 | 0.463–3.847 | 0.593 | 4.909 | 0.853–28.240 | 0.075 |
| Male | 0.775 | 0.269–2.235 | 0.638 | 0.943 | 0.264–3.375 | 0.928 |
| BMI ≤ 23.4 kg/m2 | 1.368 | 0.475–3.943 | 0.562 | |||
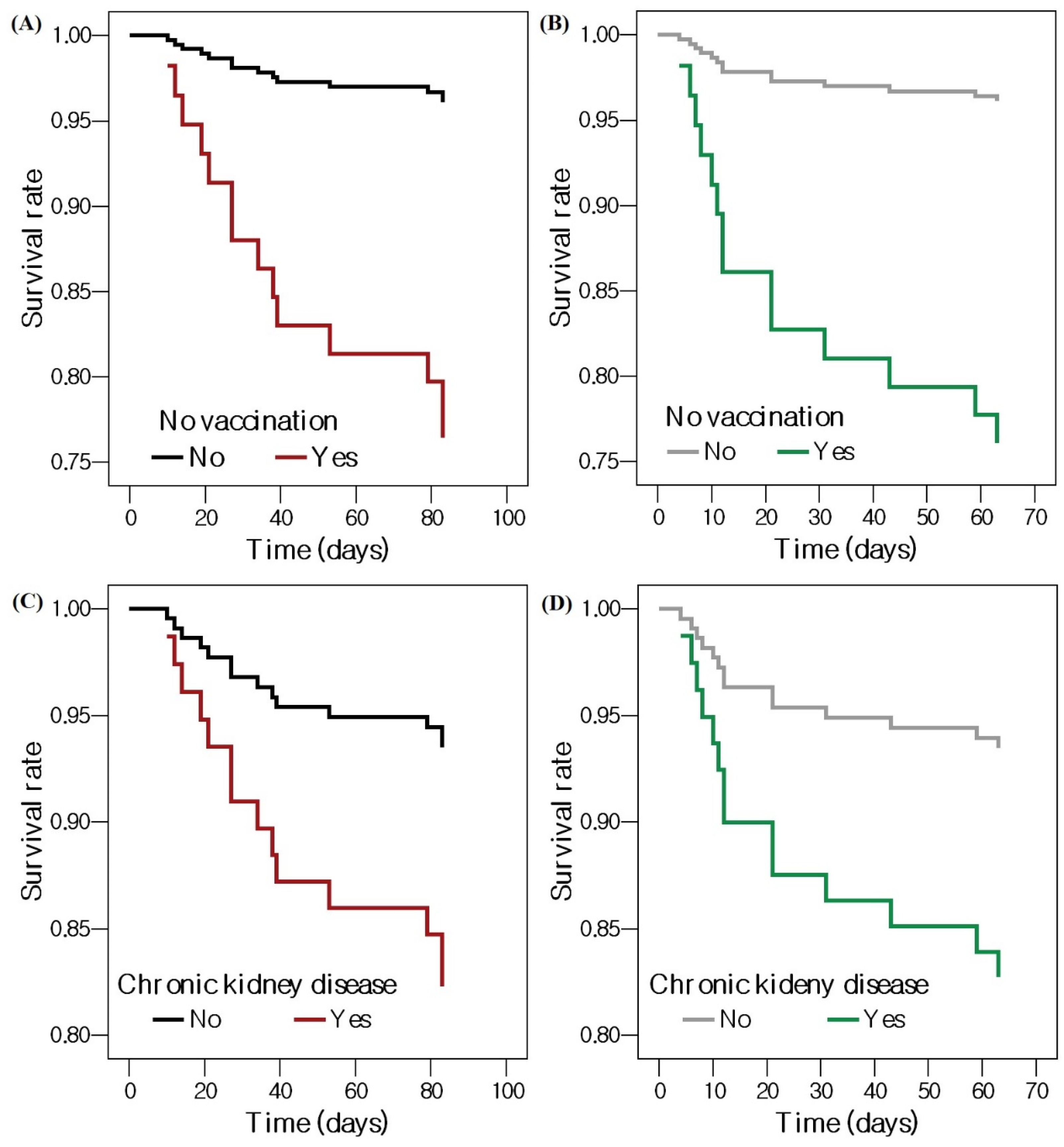
| No vaccination | 6.747 | 2.260–20.145 | 0.001 | 5.944 | 1.626–21.733 | 0.007 |
| Comorbidity | ||||||
| Cancer | 2.392 | 0.930–6.896 | 0.106 | 2.713 | 0.687–10.713 | 0.154 |
| Chronic airway disease | 3.409 | 0.950–12.230 | 0.060 | 2.088 | 0.295–14.781 | 0.461 |
| Chronic kidney disease | 2.901 | 0.972–8.658 | 0.056 | 6.963 | 1.182–41.014 | 0.032 |
| History of CVA | 2.495 | 0.782–7.956 | 0.122 | 2.610 | 0.558–12.215 | 0.223 |
| Liver cirrhosis | 2.495 | 0.558–11.150 | 0.231 | 1.017 | 0.112–9.218 | 0.988 |
| Single person room admission | 3.104 | 0.856–11.133 | 0.082 | 3.030 | 0.587–15.642 | 0.186 |
| Pulmonology | 2.861 | 0.959–8.539 | 0.059 | 1.630 | 0.199–13.371 | 0.649 |
| Pneumonia | 3.621 | 1.135–11.547 | 0.030 | 4.908 | 0.669–36.028 | 0.118 |
| NEWS ≥ 2 | 8.538 | 1.117–65.273 | 0.039 | 5.303 | 0.571–49.279 | 0.142 |
| Antibiotics before diagnosis | 2.434 | 0.679–8.724 | 0.172 | 2.354 | 0.405–13.663 | 0.340 |
| Steroid before diagnosis | 3.905 | 1.089–14.003 | 0.037 | 2498 | 0.479–13.018 | 0.277 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.H.; Kim, T.; Choi, H.; Shin, T.R.; Sim, Y.S. Clinical Outcome and Prognosis of a Nosocomial Outbreak of COVID-19. J. Clin. Med. 2023, 12, 2279. https://doi.org/10.3390/jcm12062279
Kim SH, Kim T, Choi H, Shin TR, Sim YS. Clinical Outcome and Prognosis of a Nosocomial Outbreak of COVID-19. Journal of Clinical Medicine. 2023; 12(6):2279. https://doi.org/10.3390/jcm12062279
Chicago/Turabian StyleKim, Sang Hyuk, Taehee Kim, Hayoung Choi, Tae Rim Shin, and Yun Su Sim. 2023. "Clinical Outcome and Prognosis of a Nosocomial Outbreak of COVID-19" Journal of Clinical Medicine 12, no. 6: 2279. https://doi.org/10.3390/jcm12062279
APA StyleKim, S. H., Kim, T., Choi, H., Shin, T. R., & Sim, Y. S. (2023). Clinical Outcome and Prognosis of a Nosocomial Outbreak of COVID-19. Journal of Clinical Medicine, 12(6), 2279. https://doi.org/10.3390/jcm12062279






