Estimating the Prevalence of Cardiac Amyloidosis in Old Patients with Heart Failure—Barriers and Opportunities for Improvement: The PREVAMIC Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Variables and Data Collection
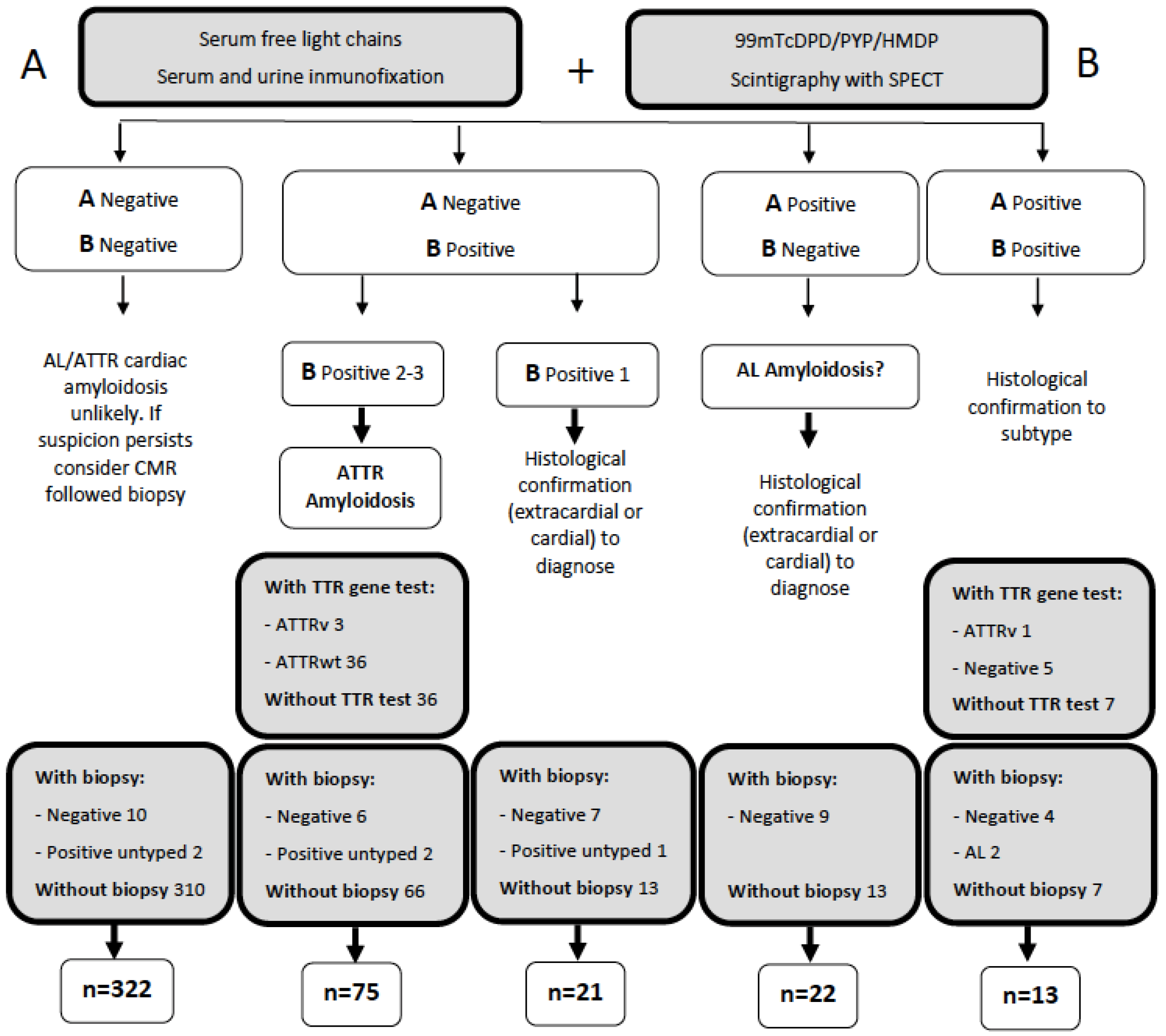
2.3. Diagnosis of CA
2.4. Ethical Aspects
2.5. Statistical Analysis
3. Results
3.1. Prevalence of CA in the Heart Failure Cohort
3.2. Characteristics of the Patients with and without CA
4. Discussion
4.1. Prevalence of Cardiac Amyloidosis. Influence of Age, Sex and Left Ventricular Ejection Fraction
4.2. Barriers and Opportunities for Improvement
4.3. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
PREVAMIC Study Researchers
References
- Conrad, N.; Judge, A.; Tran, J.; Mohseni, H.; Hedgecott, D.; Crespillo, A.P.; Allison, M.; Hemingway, H.; Cleland, J.G.; McMurray, J.J.V.; et al. Temporal trends and patterns in heart failure incidence: A population-based study of 4 million individuals. Lancet 2018, 391, 572–580. [Google Scholar] [CrossRef]
- Sayago-Silva, I.; García-López, F.; Segovia-Cubero, J. Epidemiology of heart failure in Spain over last 20 years. Rev. Esp. 2013, 66, 649–656. [Google Scholar] [CrossRef]
- González-López, E.; López-Sainz, A.; García-Pavía, P. Diagnosis and treatment of transthyretin cardiac amyloidosis. Prog. Hope. Rev. Esp. Cardiol. 2017, 70, 991–1004. [Google Scholar] [CrossRef]
- Benson, M.D.; Buxbaum, J.N.; Eisenberg, D.S.; Merlini, G.; Saraiva, M.J.M.; Sekijima, Y.; Sipe, J.D.; Westermark, P. Amyloid nomenclature 2018: Recommendations by the International Society of Amyloidosis (ISA) nomenclature committee. Amyloid 2018, 25, 215–219. [Google Scholar] [CrossRef]
- Garcia-Pavia, P.; Rapezzi, C.; Adler, Y.; Arad, M.; Basso, C.; Brucato, A.; Burazor, I.; Caforio, A.L.P.; Damy, T.; Eriksson, U.; et al. Diagnosis and treatment of cardiac amyloidosis: A position statement of the ESC Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 2021, 42, 1554–1568. [Google Scholar] [CrossRef]
- Gillmore, J.D.; Maurer, M.S.; Falk, R.H.; Merlini, G.; Damy, T.; Dispenzieri, A.; Wechalekar, A.D.; Berk, J.L.; Quarta, C.C.; Grogan, M.; et al. Nonbiopsy Diagnosis of Cardiac Transthyretin Amyloidosis. Circulation 2016, 133, 2404–2412. [Google Scholar] [CrossRef]
- Wechalekar, A.D.; Gillmore, J.D.; Hawkins, P.N. Systemic amyloidosis. Lancet 2016, 387, 2641–2654. [Google Scholar] [CrossRef]
- González-López, E.; Gallego-Delgado, M.; Guzzo-Merello, G.; de Haro-Del Moral, F.J.; Cobo-Marcos, M.; Robles, C.; Bornstein, B.; Salas, C.; Lara-Pezzi, E.; Alonso-Pulpon, L.; et al. Wild-type transthyretin amyloidosis as a cause of heart failure with preserved ejection fraction. Eur. Heart J. 2015, 3, 2585–2594. [Google Scholar] [CrossRef]
- Castaño, A.; Narotsky, D.L.; Hamid, N.; Khalique, O.K.; Morgenstern, R.; DeLuca, A.; Rubin, J.; Chiuzan, C.; Nazif, T.; Vahl, T.; et al. Unveiling transthyretin cardiac amyloidosis and its predictors among elderly patients with severe aortic stenosis undergoing transcatheter aortic valve replacement. Eur. Heart J. 2017, 38, 2879–2887. [Google Scholar] [CrossRef]
- Scully, P.R.; Treibel, T.A.; Fontana, M.; Lloyd, G.; Mullen, M.; Pugliese, F.; Hartman, N.; Hawkins, P.N.; Menezes, L.J.; Moon, J.C. Prevalence of Cardiac Amyloidosis in Patients Referred for Transcatheter Aortic Valve Replacement. J. Am. Coll. Cardiol. 2018, 7, 463–464. [Google Scholar] [CrossRef]
- Aimo, A.; Merlo, M.; Porcari, A.; Gergiopoulos, G.; Pagura, L.; Vergaro, G.; Sinagra, G.; Emdin, S.; Rapezzi, C. Redefining the epidemiology of cardiac amyloidosis. A systematic review and meta-analysis of screening studies. Eur. J. Heart Fail. 2022, 24, 2342–2351. [Google Scholar] [CrossRef]
- Salamanca-Bautista, P.; Ruiz-Hueso, R.; González-Franco, A.; Casado-Cerrada, J.; Formiga, F.; Llàcer-Iborra, P.; Amores-Arriaga, B.; Conde-Martel, A.; Manzano-Espinosa, L.; Aramburu-Bodas, O. Prevalence of cardiac amyloidosis in Spanish patients with heart failure: The PREVAMIC study design. Span. J. Med. 2022, 2, 46–50. [Google Scholar] [CrossRef]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.F.; Coats, A.J.S.; Falk, V.; Gonzalez-Juanate, J.R.; a Harjo, V.P.; Jankowsk, E.A.; et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. J. Heart Fail. 2016, 18, 891–897. [Google Scholar] [CrossRef]
- Mueller, C.; McDonald, K.; de Boer, R.A.; Maisel, A.; Cleland, J.G.; Kozhuharov, N.; Coats, A.J.; Metra, M.; Mebazaa, A.; Ruschitzka, F.; et al. Heart Failure Association of the European Society of Cardiology practical guidance on the use of natriuretic peptide concentrations. Eur. J. Heart Fail. 2019, 21, 715–731. [Google Scholar] [CrossRef]
- Perugini, E.; Guidalotti, P.L.; Salvi, F.; Cooke, R.M.T.; Pettinato, C.; Riva, L.; Leone, O.; Farsad, M.; Ciliberti, P.; Bacchi-Reggiani, L.; et al. Noinvasive etiologic diagnosis of cardiaca amyloidosis using 99mTC-3,3-diphosphono-1,2-propanodicarboxylic acid scintigraphy. J. Am. Coll. Cardiol. 2005, 46, 1076–1084. [Google Scholar] [CrossRef]
- Mohammed, S.F.; Mirzoyev, S.A.; Edwards, W.D.; Dogan, A.; Grogan, D.R.; Dunlay, S.M.; Roger, V.L.; Gertz, M.A.; Dispenzieri, A.; Zeldenrust, S.R.; et al. Left ventricular amyloid deposition in patients with heart failure and preserved ejection fraction. JACC Heart Fail. 2014, 2, 113–122. [Google Scholar] [CrossRef]
- Lindmark, K.; Pilebro, B.; Sundström, T.; Lindqvist, P. Prevalence of wild type transthyretin cardiac amyloidosis in a heart failure clinic. ESC Heart Fail. 2021, 8, 745–749. [Google Scholar] [CrossRef]
- Hahn, V.S.; Yanek, L.R.; Vaishnav, J.; Ying, W.; Vaidya, D.; Lee, Y.Z.J.; Riley, S.J.; Subramanya, V.; Brown, E.E.; Hopkins, C.D.; et al. Endomyocardial Biopsy Characterization of Heart Failure With Preserved Ejection Fraction and Prevalence of Cardiac Amyloidosis. JACC Heart Fail. 2020, 8, 712–724. [Google Scholar] [CrossRef]
- Devesa, A.; Blasco, A.C.; Lázaro, A.M.P.; Askari, E.; Lapeña, G.; Talavera, S.G.; Urquía, M.T.; Olleros, C.R.; Tuñón, J.; Ibáñez, B.; et al. Prevalence of transthyretin amyloidosis in patients with heart failure and no left ventricular hypertrophy. ESC Heart Fail. 2021, 8, 2856–2865. [Google Scholar] [CrossRef]
- AbouEzzeddine, O.F.; Davies, D.R.; Scott, C.G.; Fayyaz, A.U.; Askew, J.W.; McKie, P.M.; Noseworthy, P.A.; Johnson, G.B.; Dunlay, S.M.; Borlaug, B.A.; et al. Prevalence of Transthyretin Amyloid Cardiomyopathy in Heart Failure With Preserved Ejection Fraction. JAMA Cardiol. 2021, 6, 1267–1274. [Google Scholar] [CrossRef]
- Sainz, A.L.; Moral, F.J.D.H.-D.; Dominguez, F.; Restrepo-Cordoba, A.; Amor-Salamanca, A.; Hernandez-Hernandez, A.; Ruiz-Guerrero, L.; Krsnik, I.; Cobo-Marcos, M.; Castro, V.; et al. Prevalence of cardiac amyloidosis among elderly patients with systolic heart failure or conduction disorders. Amyloid 2019, 26, 156–163. [Google Scholar] [CrossRef]
- Uusitalo, V.; Suomalainen, O.; Loimaala, A.; Mätzke, S.; Heliö, T. Prognostic Value of 99mTc-HMDP Scintigraphy in Elderly Patients With Chronic Heart Failure. Heart Lung Circ. 2022, 31, 629–637. [Google Scholar] [CrossRef]
- Bruno, M.; Casta, A.; Burton, A.; Grodin, J.L. Transthyretin amyloid cardiomyopathy in women: Frequency, characteristics, and diagnostic challenges. Heart Fail. Rev. 2021, 26, 35–45. [Google Scholar] [CrossRef]
- Kroi, F.; Fischer, N.; Gezin, A.; Hashim, M.; Rozembaum, M.H. Estimating the gender distribution of patients with wild-type transthyretin amyloid cardiomyopathy: A systematic review and meta-analysis. Cardiol. Ther. 2021, 10, 41–55. [Google Scholar] [CrossRef]
- Lauppe, R.E.; Hansen, J.L.; Gerdesköld, C.; Rozenbaum, M.H.; Strand, A.M.; Vakevainen, M.; Kuusisto, J.; Gude, E.; Gustafsson, F.; Smith, J.G. Nationwide prevalence and characteristics of transthyretin amyloid cardiomyopathy in Sweden. Open Heart. 2021, 8, e001755. [Google Scholar] [CrossRef]
- Asif, T.; Araujo, T.; Singh, V.; Malhotra, S. High prevalence of heart failure with reduced ejection fraction in patients with transthyretin cardiac amyloidosis. J. Nucl. Cardiol. 2020, 27, 1044–1046. [Google Scholar] [CrossRef]
- Barge-Caballero, G.; Barge-Caballero, E.; López-Pérez, M.; Bilbao-Quesada, R.; González-Babarro, E.; Gómez-Otero, I.; López-López, A.; Gutiérrez-Feijoo, M.; Varela-Román, A.; González-Juanatey, C.; et al. Cardiac amyloidosis: Description of a series of 143 cases. Med. Clin. 2022, 159, 207–213. [Google Scholar] [CrossRef]
- Rowczenio, D.M.; Noor, J.; Gillmore, J.D.; Lachmann, H.J.; Whelan, C.; Hawkins, P.N.; Obici, L.; Westermark, P.; Grateau, G.; Wechalekar, A.D. Online Registry for mutations in Hereditary Amyloidosis including nomenclature recommendations. Hum. Mutat. 2014, 35, E2403–E2412. [Google Scholar] [CrossRef]
- Choi, Y.-J.; Kim, D.; Rhee, T.-M.; Lee, H.-J.; Park, J.-B.; Lee, S.-P.; Chang, S.-A.; Kim, Y.-J.; Jeon, E.-S.; Oh, J.K.; et al. Left atrial reservoir strain as a novel predictor of new-onset atrial fibrillation in light-chain-type cardiac amyloidosis. Eur. Heart J. Cardiovasc. Imaging, 2023; online ahead of print. [Google Scholar] [CrossRef]
- Bandera, F.; Martone, R.; Chacko, L.; Ganesananthan, S.; Gilbertson, J.A.; Ponticos, M.; Lane, T.; Martinez-Naharro, A.; Whelan, C.; Quarta, C.; et al. Clinical importance of left atrial infiltration in cardiac transthyretin amyloidosis. JACC Cardiovasc. Imaging 2022, 15, 17–29. [Google Scholar] [CrossRef]
- Davies, D.R.; Redfield, M.M.; Scott, C.G.; Minamisawa, M.; Grogan, M.; Dispenzieri, A.; Chareonthaitawee, P.; Shah, A.M.; Shah, S.J.; Wehbe, R.M.; et al. A simple score to identify increased risk of transthyretin amyloid cardiomyopathy in heart failure with preserved ejection fraction. JAMA Cardiol. 2022, 7, 1036–1044. [Google Scholar] [CrossRef]
- Huda, A.; Castaño, A.; Niyogi, A.; Schumacher, J.; Stewart, M.; Bruno, M.; Hu, M.; Ahmad, F.S.; Deo, R.C.; Shah, S.J. A machine learning model for identifying patients at risk for wild-type transthyretin amyloid cardiomyopathy. Nat. Commun. 2021, 12, 2725. [Google Scholar] [CrossRef]
- Sammani, A.; Jansen, M.; de Vries, N.M.; de Jonge, N.; Baas, A.F.; Riele, A.S.J.M.T.; Asselbergs, F.W.; Oerlemans, M.I.F.J. Automatic identification of patients with unexplained left ventricular hypertrophy in electronic health record data to improve targeted treatment and family screening. Front. Cardiovasc. Med. 2022, 9, 768847. [Google Scholar] [CrossRef]
- Brons, M.; Muller, S.A.; Rutten, F.H.; van der Meer, M.G.; Vrancken, A.F.J.E.; Minnema, M.C.; Baas, A.F.; Asselbergs, F.W.; Oerlemans, M.I.F.J. Evaluation of the cardiac amyloidosis clinical pathway implementation: A real-world experience. Eur. Heart J. Open 2022, 2, oeac011. [Google Scholar] [CrossRef]
- Kittleson, M.M.; Ruberg, F.L.; Ambardekar, A.V.; Brannagan, T.H.; Cheng, R.K.; Clarke, J.O.; Dember, L.M.; Frantz, J.G.; Hershberger, R.E.; Maurer, M.S.; et al. 2023 ACC Expert Consensus Decision Pathway on comprehensive multidisciplinary care for the patient with cardiac amyloidosis: A report of the American College of Cardiology Solutions set oversight committee. J. Am. Coll. Cardiol. 2023; Online ahead of print. [Google Scholar] [CrossRef]
- Sperry, B.W.; Khoury, J.A.; Raza, S.; Rosenthal, J.L. Comprehensive approach to cardiac amyloidosis care: Considerations in starting an amyloidosis program. Heart Fail Rev. 2022, 27, 1559–1565. [Google Scholar] [CrossRef]

| Cardiac Amyloidosis n = 91 (20.1%) | No Cardiac Amyloidosis n = 372 (79.9%) | All n = 453 (100%) | p Value | |
|---|---|---|---|---|
| Demography/physical examination Age, median (IQR), years Women, n (%) BMI, median (IQR) Kg/m2 SBP, median (IQR), mmHg Outpatients | 88 [85–91] 36 (39.9) 27.1 [25.0–32.7] | 83 [78–87] 193 (53.5) 30.1 [26.6–33.9] | 85 [79–88] 229 (50.6) 29.8 [26.1–32.7] | <0.001 0.019 0.002 |
| 126 [109–140] 50 (54.9) | 129 [116–142] 263 (70.1) | 128 [115–142] 313 (69.0) | 0.442 0.003 | |
| Heart disease/Devices Coronary artery disease, n (%) Moderate/severe valve disease, n (%) Previous HF, n (%) Pacemaker, n (%) | 27 (29.7) 42 (39.6) 77 (84.6) 19 (20.9) | 112 (30.9) 189 (48.0) 321 (88.7) 53 (14.7) | 139 (30.7) 231 (46.4) 398 (87.9) 72 (15.9) | 0.411 0.253 0.397 0.125 |
| Other comorbidities Diabetes mellitus, n (%) Hypertension, n (%) Dyslipidemia, n (%) Cerebrovascular disease, n (%) COPD, n (%) | 27 (29.7) 78 (85.7) 50 (54.9) 11 (12.1) 9 (9.9) | 151 (41.7) 321 (88.7) 211 (58.3) 69 (19.1) 69 (19.1) | 178 (39.3) 399 (88.1) 261 (57.6) 80 (17.7) 78 (17.2) | 0.137 0.732 0.810 0.295 0.103 |
| Red flags Low BP, previous hypertension, n (%) History of syncope, n (%) History of back pain, n (%) History of anaemia, n (%) Intolerance of ACEI/ARBs, n (%) Intolerance of beta-blockers, n (%) Intolerance of CCBs, n (%) Intolerance of digoxin, n (%) Carpal tunnel syndrome, n (%) Spinal stenosis, n (%) Biceps tendon ruptura, n (%) Periorbital purpura, n (%) Macroglosia, n (%) MGUS, n (%) Peripheral neuropathy, n (%) Autonomic dysfuncion, n (%) Nephrotic syndrome, n (%) | 22 (24.2) 7 (7.7) 31 (34.1) 41 (45.1) 8 (8.8) 15 (16.5) 2 (2.2) 4 (4.4) 21 (23.1) 8 (8.8) 3 (3.3) 1 (1.1) 2 (2.2) 11 (12.1) 11 (12.1) 2 (2.2) 0 (0) | 48 (13.3) 36 (9.9) 119 (32.9) 170 (47.0) 12 (3.3) 23(6.4) 5 (1.4) 8 (2.2) 17 (4.7) 19 (5.2) 1 (0.3) 1 (0.3) 1 (0.3) 16 (4.4) 20 (5.5) 8 (2.2) 6 (1.7) | 70 (15.5) 43 (9.5) 150 (33.1) 211 (46.6) 20 (4.4) 38 (8.4) 7 (1.5) 12 (2.6) 38 (8.4) 27 (6.0) 4 (0.9) 2 (0.4) 3 (0.7) 27 (6.0) 31 (6.8) 10 (2.2) 6 (1.3) | 0.033 0.564 0.077 0.811 0.014 0.009 0.116 0.578 <0.001 0.001 0.012 0.772 0.221 0.047 0.068 0.980 0.478 |
| Signs and symptoms Dyspnoea, n (%) Fatigue, n (%) Muscular weakness, n (%) Eye symptoms, n (%) Dry cough, n (%) Angina, n (%) Palpitations, n (%) Weightloss, n (%) Diarrhea, n (%) Constipation, n (%) Paresthesia, n (%) Delusionsm n (%) | 67 (73.6) 69 (75.8) 44 (48.4) 15 (16.5) 14 (15.4) 10 (11) 13 (14.3) 17 (18.7) 9 (9.9) 17 (18.7) 12 (13.2) 8 (8.8) | 260 (71.8) 196 (54.1) 108 (29.3) 68 (18.8) 55 (15.2) 30 (8.3) 80 (22.1) 36 (9.9) 21 (5.8) 82 (22.7) 27 (7.5) 16 (4.4) | 327 (72.2) 265 (58.5) 152 (33.6) 83 (18.3) 69 (15.2) 40 (8.8) 93 (20.5) 53 (11.7) 30 (6.6) 99 (21.9) 39 88.6) 24 (5.3) | 0.573 0.001 0.002 0.866 0.992 0.487 0.248 0.067 0.363 0.715 0.214 0.227 |
| Functional assessment Previous NYHA class III-IV, n (%) Barthel Index, median (IQR), points Pfeiffer Questionnaire, errors | 27 (31.0) 90 [70–100] 1 [0–2] | 79 (22.8) 90 [80–100] 1 [0–2] | 106 (24.4) 90 [75–100] 1 [0–2] | 0.108 0.237 0.575 |
| Laboratory Haemoglobin, median (IQR), mg/dl Creatinine, median (IQR), mg/dl Sodium, median (IQR), mEq/L Potassium, median (IQR), mEq/L Troponin T-hs, median (IQR), ng/L NT-proBNP, median (IQR), pg/mL Ca125, median (IQR), U/mL | 12.8 [11.5–13.7] 1.3 [1.0–1.8] 141 [137–143] 4.4 [4.1–4.9] 81.7 [40.2–132.5] 4398 [2035–8452] 33.1 [12.7–94.4] | 12.4 [11.0–13.8] 1.3 [0.9–1.7] 141 [139–143] 4.3 [4.0–4.8] 32.2 [22.0–58.0] 2420 [1368–4352] 17.7 [10.5–35.7] | 12.4 [11.1–13.8] 1.3 [1.0–1.7] 141 [139–143] 4.4 [4.0–4.8] 38.0 [24.0–72.0] 2651 [1498–5271] 18.5 [11.0–39.5] | 0.206 0.828 0.182 0.194 <0.001 0.001 0.065 |
| Treatment Beta-blockers, n (%) ACEIs, n (%) ARBs, n (%) Aldosterone antagonists, n (%) Sacubitril-valsartan, n (%) Digoxin, n (%) CCBs, n (%) Nitrates, n (%) Loop diuretics, n (%) Thiazide diuretics, n (%) i-SGLT2s, n (%) Anticoagulants, n (%) | 46 (50.5) 23 (25.3) 23 (25.3) 25 (27.2) 6 (6.6) 3 (3.3) 14 (15.4) 7 (7.7) 77 (84.6) 13 (14.3) 1 (1.1) 59 (64.8) | 249 (68.8) 102 (28.2) 112 (30.9) 99 (27.3) 34 (9.4) 29 (8.0) 104 (28.7) 50 (13.8) 328 (90.6) 51 (14.1) 27 (7.5) 234 (62.9) | 295 (65.1) 125 (27.6) 135 (29.8) 124 (27.4) 40 (8.8) 32 (7.1) 118 (26.0) 57 (12.6) 405 (89.4) 64 (14.1) 28 (6.2) 293 (64.7) | 0.004 0.852 0.572 0.756 0.695 0.209 0.034 0.286 0.209 0.948 0.077 0.959 |
| Cardiac Amyloidosis n = 91 (20.1%) | No Cardiac Amyloidosis n = 372 (79.9%) | All n = 453 (100%) | p Value | |
|---|---|---|---|---|
| Electrocardiogram Atrio-ventricular block Atrial fibrillation Low voltage Pseudo-myocardial infarction pattern Right bundle branch block Left bundle branch block Left ventricular hypertrophy | 9 (9.9) 66 (72.5) 28 (30.8) 16 (17.6) 25 (27.5) 19 (20.9) 19 (20.9) | 37 (10.0) 250 (69.1) 59 (16.3) 65 (18.0) 92 (25.4) 98 (27.1) 121 (33.4 | 46 (10.1) 316 (69.8) 87 (19.2) 81 (17.9) 117 (25.8) 117 (25.8) 140 (30.9) | 0.918 0.687 0.004 0.361 0.774 0.458 0.047 |
| Echocardiogram LVEF < 40% LVEF 40–49% LVEF > 50% SWT, median (IQR), mm PW, median (IQR), mm LV mass index (IQR), g/m2 Left atrium (mm) Aortic stenosis Aortic regurgitation Mitral stenosis Mitral regurgitation Tricuspid regurgitation TAPSE, median (IQR), mm Pericardial effusion Speckled myocardium | 8 (9.6) 14 (16.9) 61 (73.5) 17 [14–19] 14 [13–17] 151 [125–187] 47 [43–51] 16 (20.1) 40 (52.6) 2 (2.5) 61 (78.2) 56 (71.8) 18 [16–23] 22 (24.2) 16 (17.6) | 42 (12.1) 37 (10.6) 259 (77.3) 14 [13–15] 13 [12–14] 135 [107–171] 46 [42–50] 75 (22.2) 152 (45.4) 26 (7.7) 265 (78.2) 220 (65.7) 20 [17–25] 37 (10.2) 8 (2.2) | 50 (11.6) 51 (11.8) 330 (76.6) 14 [13–16] 13 [12–14] 138 [112–174] 47 [43–52] 91 (21.9) 192 (46.7) 28 (6.7) 326 (78.2) 276 (66.8) 20 [17–25] 59 (13.0) 24 (5.3) | 0.263 <0.001 <0.001 0.056 0.325 0.030 0.066 0.187 0.157 0.104 0.290 <0.001 <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz-Hueso, R.; Salamanca-Bautista, P.; Quesada-Simón, M.A.; Yun, S.; Conde-Martel, A.; Morales-Rull, J.L.; Suárez-Gil, R.; García-García, J.Á.; Llàcer, P.; Fonseca-Aizpuru, E.M.; et al. Estimating the Prevalence of Cardiac Amyloidosis in Old Patients with Heart Failure—Barriers and Opportunities for Improvement: The PREVAMIC Study. J. Clin. Med. 2023, 12, 2273. https://doi.org/10.3390/jcm12062273
Ruiz-Hueso R, Salamanca-Bautista P, Quesada-Simón MA, Yun S, Conde-Martel A, Morales-Rull JL, Suárez-Gil R, García-García JÁ, Llàcer P, Fonseca-Aizpuru EM, et al. Estimating the Prevalence of Cardiac Amyloidosis in Old Patients with Heart Failure—Barriers and Opportunities for Improvement: The PREVAMIC Study. Journal of Clinical Medicine. 2023; 12(6):2273. https://doi.org/10.3390/jcm12062273
Chicago/Turabian StyleRuiz-Hueso, Rocío, Prado Salamanca-Bautista, Maria Angustias Quesada-Simón, Sergi Yun, Alicia Conde-Martel, José Luis Morales-Rull, Roi Suárez-Gil, José Ángel García-García, Pau Llàcer, Eva María Fonseca-Aizpuru, and et al. 2023. "Estimating the Prevalence of Cardiac Amyloidosis in Old Patients with Heart Failure—Barriers and Opportunities for Improvement: The PREVAMIC Study" Journal of Clinical Medicine 12, no. 6: 2273. https://doi.org/10.3390/jcm12062273
APA StyleRuiz-Hueso, R., Salamanca-Bautista, P., Quesada-Simón, M. A., Yun, S., Conde-Martel, A., Morales-Rull, J. L., Suárez-Gil, R., García-García, J. Á., Llàcer, P., Fonseca-Aizpuru, E. M., Amores-Arriaga, B., Martínez-González, Á., Armengou-Arxe, A., Peña-Somovilla, J. L., López-Reboiro, M. L., & Aramburu-Bodas, Ó., on behalf of the PREVAMIC Investigators Group. (2023). Estimating the Prevalence of Cardiac Amyloidosis in Old Patients with Heart Failure—Barriers and Opportunities for Improvement: The PREVAMIC Study. Journal of Clinical Medicine, 12(6), 2273. https://doi.org/10.3390/jcm12062273







