Intervortex Venous Anastomosis in the Macula in Central Serous Chorioretinopathy Imaged by En Face Optical Coherence Tomography
Abstract
1. Introduction
2. Materials and Methods
2.1. OCT and OCTA Examination
2.2. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chhablani, J.; Cohen, F.B.; Aymard, P.; Beydoun, T.; Bousquet, E.; Daruich-Matet, A.; Matet, A.; Zhao, M.; Cheung, C.M.G.; Freund, K.B.; et al. Multimodal Imaging-Based Central Serous Chorioretinopathy Classification. Ophthalmol. Retin. 2020, 4, 1043–1046. [Google Scholar] [CrossRef] [PubMed]
- Kaye, R.; Chandra, S.; Sheth, J.; Boon, C.J.F.; Sivaprasad, S.; Lotery, A. Central serous chorioretinopathy: An update on risk factors, pathophysiology and imaging modalities. Prog. Retin. Eye Res. 2020, 79, 100865. [Google Scholar] [CrossRef] [PubMed]
- Spaide, R.F.; Rochepeau, C.; Kodjikian, L.; Garcia, M.A.; Coulon, C.; Burillon, C.; Denis, P.; Delaunay, B.; Mathis, T.; Matet, A.; et al. Choriocapillaris Flow Features Follow a Power Law Distribution: Implications for Characterization and Mechanisms of Disease Progression. Graefe’s Arch. Clin. Exp. Ophthalmol. 2019, 257, 905–912. [Google Scholar] [CrossRef]
- Daruich, A.; Matet, A.; Dirani, A.; Bousquet, E.; Zhao, M.; Farman, N.; Jaisser, F.; Behar-Cohen, F. Central serous chorioretinopathy: Recent findings and new physiopathology hypothesis. Prog. Retin. Eye Res. 2015, 48, 82–118. [Google Scholar] [CrossRef]
- Cheung, C.M.G.; Lee, W.K.; Koizumi, H.; Dansingani, K.; Lai, T.Y.Y.; Freund, K.B. Pachychoroid disease. Eye 2019, 33, 14–33. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.; Ho, M.; Lai, F.H.P.; Ng, D.S.C.; Iu, L.P.L.; Iu, L.P.L.; Chen, L.J.; Chen, L.J.; Mak, A.C.Y.; Mak, A.C.Y.; et al. Analysis of choriocapillaris perfusion and choroidal layer changes in patients with chronic central serous chorioretinopathy randomised to micropulse laser or photodynamic therapy. Br. J. Ophthalmol. 2020, 33, 14–33. [Google Scholar] [CrossRef]
- Teussink, M.M.; Breukink, M.B.; Van Grinsven, M.J.J.P.; Hoyng, C.B.; Jeroen Klevering, B.; Boon, C.J.F.; De Jong, E.K.; Theelen, T. Oct angiography compared to fluorescein and indocyanine green angiography in chronic central serous chorioretinopathy. Investig. Ophthalmol. Vis. Sci. 2015, 56, 5229–5237. [Google Scholar] [CrossRef]
- Iacono, P.; Tedeschi, M.; Boccassini, B.; Chiaravalloti, A.; Varano, M.; Parravano, M. Chronic Central Serous Chorioretinopathy: Early and Late Morphological and Functional Changes After Verteporfin Photodynamic Therapy. Retina 2019, 39, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Su, Y.; Li, L.; Qi, H.; Zheng, H.; Chen, C. Effect of photodynamic therapy on optical coherence tomography angiography in eyes with chronic central serous chorioretinopathy. Ophthalmologica 2017, 237, 167–172. [Google Scholar] [CrossRef]
- Matet, A.; Daruich, A.; Hardy, S.; Behar-Cohen, F. Patterns of Choriocapillaris Flow Signal Voids in Central Serous Chorioretinopathy. Retina 2019, 39, 2178–2188. [Google Scholar] [CrossRef]
- Spaide, R.F.; Gemmy Cheung, C.M.; Matsumoto, H.; Kishi, S.; Boon, C.J.F.; van Dijk, E.H.C.; Mauget-Faysse, M.; Behar-Cohen, F.; Hartnett, M.E.; Sivaprasad, S.; et al. Venous overload choroidopathy: A hypothetical framework for central serous chorioretinopathy and allied disorders. Prog. Retin. Eye Res. 2022, 86, 100973. [Google Scholar] [CrossRef] [PubMed]
- Battista, M.; Borrelli, E.; Parravano, M.; Gelormini, F.; Tedeschi, M.; De Geronimo, D.; Sacconi, R.; Querques, L.; Bandello, F.; Querques, G. OCTA characterisation of microvascular retinal alterations in patients with central serous chorioretinopathy. Br. J. Ophthalmol. 2020, 104, 1453–1457. [Google Scholar] [CrossRef] [PubMed]
- Rochepeau, C.; Kodjikian, L.; Garcia, M.A.; Coulon, C.; Burillon, C.; Denis, P.; Delaunay, B.; Mathis, T. Optical Coherence Tomography Angiography Quantitative Assessment of Choriocapillaris Blood Flow in Central Serous Chorioretinopathy. Am. J. Ophthalmol. 2018, 194, 26–34. [Google Scholar] [CrossRef]
- Dansingani, K.K.; Balaratnasingam, C.; Naysan, J.; Freund, K.B. En Face Imaging of Pachychoroid Spectrum Disorders with Swept-Source Optical Coherence Tomography. Retina 2016, 36, 499–516. [Google Scholar] [CrossRef]
- Shiihara, H.; Sonoda, S.; Terasaki, H.; Kakiuchi, N.; Yamashita, T.; Uchino, E.; Murao, F.; Sano, H.; Mitamura, Y.; Sakamoto, T. Quantitative analyses of diameter and running pattern of choroidal vessels in central serous chorioretinopathy by en face images. Sci. Rep. 2020, 10, 9591. [Google Scholar] [CrossRef]
- Spaide, R.F.; Ngo, W.K.; Barbazetto, I.; Sorenson, J.A. Sausaging and Bulbosities of the Choroidal Veins in Central Serous Chorioretinopathy. Retina 2022, 42, 1638–1644. [Google Scholar] [CrossRef] [PubMed]
- Kishi, S.; Matsumoto, H. A new insight into pachychoroid diseases: Remodeling of choroidal vasculature. Graefe’s Arch. Clin. Exp. Ophthalmol. = Albr. von Graefes Arch. fur Klin. und Exp. Ophthalmol. 2022, 42, 3405–3417. [Google Scholar] [CrossRef]
- Matsumoto, H.; Hoshino, J.; Mukai, R.; Nakamura, K.; Kikuchi, Y.; Kishi, S.; Akiyama, H. Vortex Vein Anastomosis at the Watershed in Pachychoroid Spectrum Diseases. Ophthalmol. Retin. 2020, 4, 938–945. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Hoshino, J.; Mukai, R.; Nakamura, K.; Kishi, S.; Akiyama, H. Pulsation of anastomotic vortex veins in pachychoroid spectrum diseases. Sci. Rep. 2021, 11, 14942. [Google Scholar] [CrossRef]
- Spaide, R.F.; Ledesma-Gil, G.; Gemmy Cheung, C.M. Intervortex Venous Anastomosis in Pachychoroid-Related Disorders. Retina Publish Ah. 2020, 11, 14942. [Google Scholar] [CrossRef]
- Moreno-Morillo, F.J.; Fernández-Vigo, J.I.; Burgos-Blasco, B.; Llorente-La Orden, C.; Vidal-Villegas, B.; Santos-Bueso, E. Optical coherence tomography angiography of choroidal nodules in neurofibromatosis type-1: A case series. Eur. J. Ophthalmol. 2022, 32, NP91–NP94. [Google Scholar] [CrossRef] [PubMed]
- Ramtohul, P.; Cabral, D.; Oh, D.; Galhoz, D.; Freund, K.B. En face Ultrawidefield OCT of the Vortex Vein System in Central Serous Chorioretinopathy. Ophthalmol. Retin. 2022. [Google Scholar] [CrossRef]
- Terao, N.; Imanaga, N.; Wakugawa, S.; Sawaguchi, S.; Tamashiro, T.; Yamauchi, Y.; Koizumi, H. Short Axial Length Is Related to Asymmetric Vortex Veins in Central Serous Chorioretinopathy. Ophthalmol. Sci. 2021, 1, 100071. [Google Scholar] [CrossRef] [PubMed]
- Hosoda, Y.; Yoshikawa, M.; Miyake, M.; Tabara, Y.; Ahn, J.; Woo, S.J.; Honda, S.; Sakurada, Y.; Shiragami, C.; Nakanishi, H.; et al. CFH and VIPR2 as susceptibility loci in choroidal thickness and pachychoroid disease central serous chorioretinopathy. Proc. Natl. Acad. Sci. USA 2018, 115, 6261–6266. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Mukai, R.; Saito, K.; Hoshino, J.; Kishi, S.; Akiyama, H. Vortex vein congestion in the monkey eye: A possible animal model of pachychoroid. PLoS ONE 2022, 17, e0274137. [Google Scholar] [CrossRef]
- Shiihara, H.; Sonoda, S.; Terasaki, H.; Kakiuchi, N.; Shinohara, Y.; Tomita, M.; Sakamoto, T. Automated segmentation of en face choroidal images obtained by optical coherent tomography by machine learning. Jpn. J. Ophthalmol. 2018, 62, 643–651. [Google Scholar] [CrossRef]
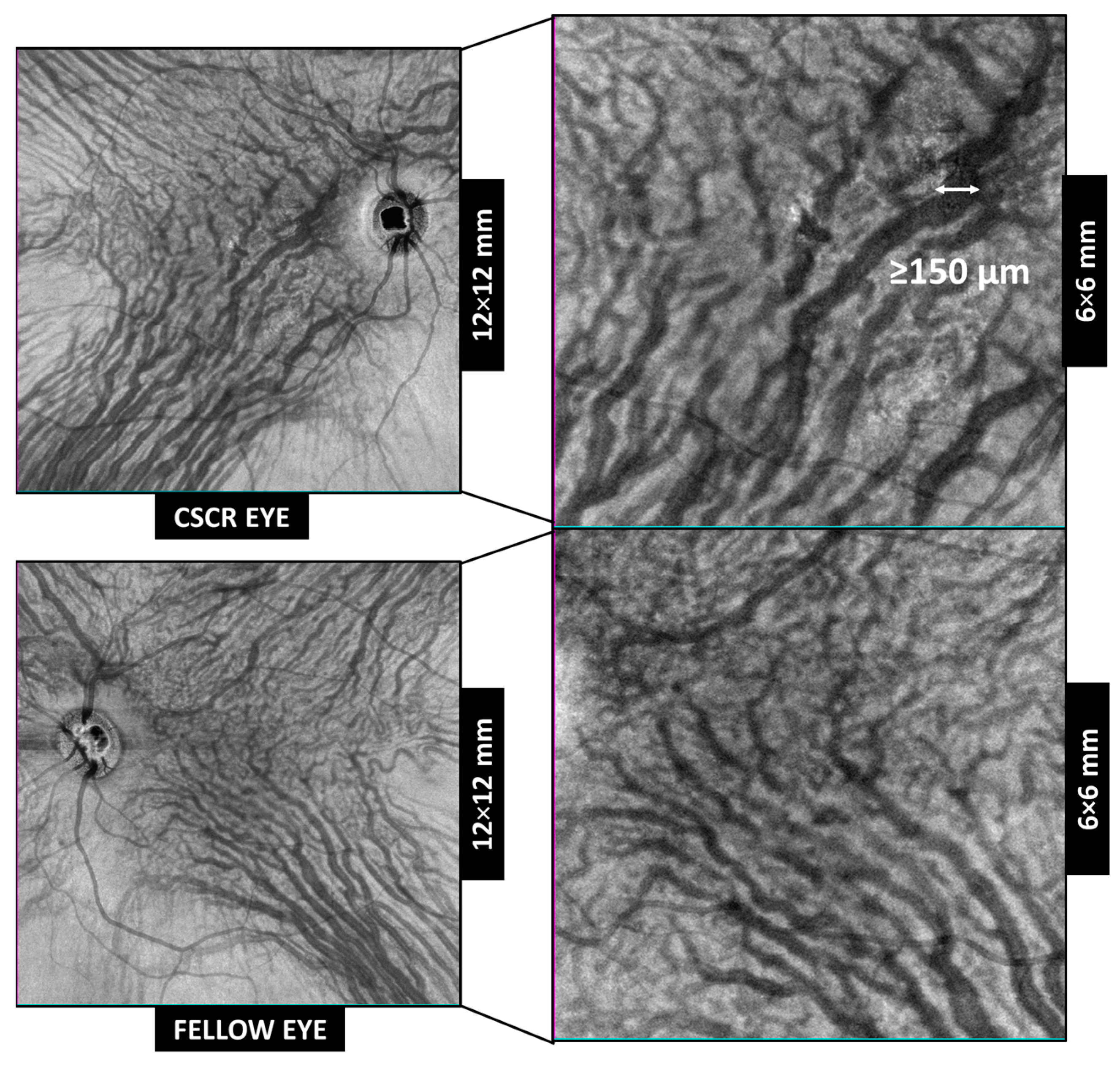

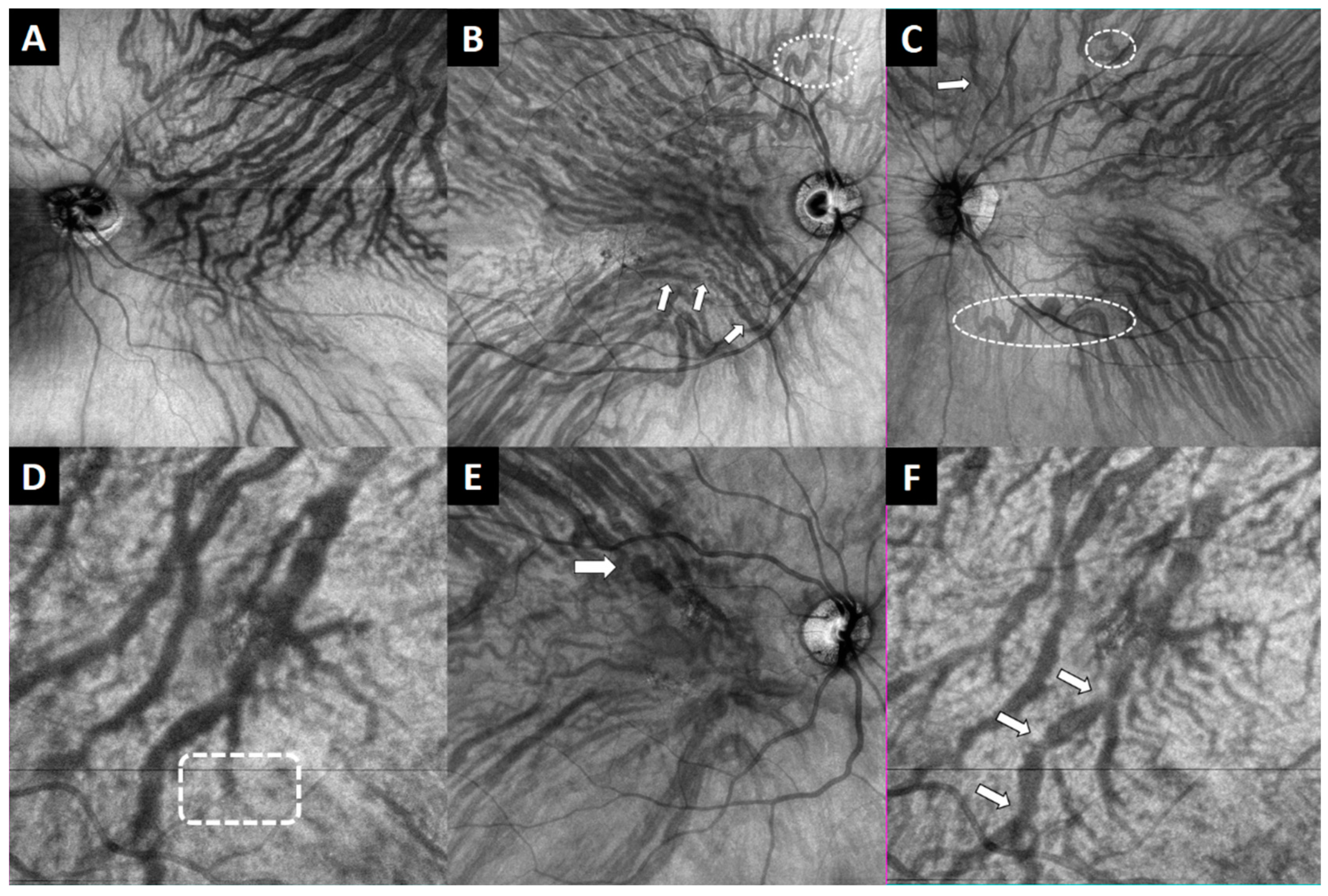

| Group | % Patients with Anastomosis | Nº Macular Anastomosis |
|---|---|---|
| 1. Chronic unilateral CSCR (persistent SRF) (N = 135) | 79.2% (107/135) | 2.9 ± 1.8 (0 to 8) |
| 2. Fellow unaffected eye (N = 135) | 51.8% (70/135) | 2.1 ± 1.7 (0 to 8) |
| 3. Controls (N = 110 eyes, 57 patients) | 58.2% (64/110) | 1.5 ± 1.6 (0 to 6) |
| p-value ANOVA | <0.01 * | <0.01 * |
| p-value (paired comparison) | 1 vs. 2: <0.01 * | 1 vs. 2: <0.01 * |
| 1 vs. 3: <0.01 * | 1 vs. 3: <0.01 * | |
| 2 vs. 3: 0.068 | 2 vs. 3: <0.01 * |
| Parameter | Group Studied (n = 77 Eyes) | Kappa Value |
|---|---|---|
| Qualitative assessment: presence or absence of anastomoses | CSCR eyes | 0.847 |
| Control eyes | 0.860 | |
| Quantitative assessment: number of anastomoses | CSCR eyes | 0.922 |
| Control eyes | 0.848 |
| Group | Asymmetries (%) | Abrupt Choroidal Vessels (%) | Sausaging (%) | BULBOSITIES (%) | Corkscrew Vessels (%) |
|---|---|---|---|---|---|
| 1. Chronic unilateral CSCR (persistent SRF) (N = 135) | 17% (23/135) 11 superior and 12 inferior predominance | 7.4% (10/135) | 23.7% (32/135) | 22.9% (31/135) | 15.5% (21/135) |
| 2. Fellow unaffected eye (N = 135) | 8.9% (12/135) 9 superior and 3 inferior predominance | 2.9% (4/135) | 7.4% (10/135) | 14.8% (20/135) | 6.6% (9/135) |
| 3. Controls (N = 110 eyes, 57 patients) | 1.8% (2/110) 1 superior and 1 inferior predominance | 1% (1/110) | 8.1% (9/110) | 7.3% (8/110) | 4.5% (5/110) |
| p-value ANOVA | <0.001 * | 0.028 | 0.073 | 0.105 | 0.03 |
| p-value (paired comparison) | 1 vs 2: <0.001 * | 1 vs 2: <0.001 * | 1 vs 2: 0.206 | 1 vs 2: 0.195 | 1 vs 2: 0.015 * |
| 1 vs 3: <0.001 * | 1 vs 3: <0.001 * | 1 vs 3: 0.311 | 1 vs 3: 0.129 | 1 vs 3: 0.293 | |
| 2 vs 3: <0.001 * | 2 vs 3: 0.864 | 2 vs 3: 0.434 | 2 vs 3: 0.591 | 2 vs 3: 0.547 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Vigo, J.I.; Rego-Lorca, D.; Moreno-Morillo, F.J.; Burgos-Blasco, B.; Valverde-Megías, A.; Méndez-Hernández, C.; López-Guajardo, L.; Donate-López, J. Intervortex Venous Anastomosis in the Macula in Central Serous Chorioretinopathy Imaged by En Face Optical Coherence Tomography. J. Clin. Med. 2023, 12, 2088. https://doi.org/10.3390/jcm12062088
Fernández-Vigo JI, Rego-Lorca D, Moreno-Morillo FJ, Burgos-Blasco B, Valverde-Megías A, Méndez-Hernández C, López-Guajardo L, Donate-López J. Intervortex Venous Anastomosis in the Macula in Central Serous Chorioretinopathy Imaged by En Face Optical Coherence Tomography. Journal of Clinical Medicine. 2023; 12(6):2088. https://doi.org/10.3390/jcm12062088
Chicago/Turabian StyleFernández-Vigo, José Ignacio, Daniela Rego-Lorca, Francisco Javier Moreno-Morillo, Bárbara Burgos-Blasco, Alicia Valverde-Megías, Carmen Méndez-Hernández, Lorenzo López-Guajardo, and Juan Donate-López. 2023. "Intervortex Venous Anastomosis in the Macula in Central Serous Chorioretinopathy Imaged by En Face Optical Coherence Tomography" Journal of Clinical Medicine 12, no. 6: 2088. https://doi.org/10.3390/jcm12062088
APA StyleFernández-Vigo, J. I., Rego-Lorca, D., Moreno-Morillo, F. J., Burgos-Blasco, B., Valverde-Megías, A., Méndez-Hernández, C., López-Guajardo, L., & Donate-López, J. (2023). Intervortex Venous Anastomosis in the Macula in Central Serous Chorioretinopathy Imaged by En Face Optical Coherence Tomography. Journal of Clinical Medicine, 12(6), 2088. https://doi.org/10.3390/jcm12062088








