Auditory Domain Sensitivity and Neuroplasticity-Based Targeted Cognitive Training in Autism Spectrum Disorder
Abstract
1. Introduction
2. Methods
- (a)
- Auditory Sweeps (called “Sound Sweeps” in the platform) target auditory processing speed (units = msec). Two successive frequency-modulated tone sweeps were presented, and participants indicated whether the frequency increased or decreased within each tone;
- (b)
- Sound Discrimination (“Fine Tuning”) targets auditory perception and processing speed (units = msec). Participants indicated which one of two confusable syllables was presented;
- (c)
- Syllable Ordering (“Syllable Stacks”) targets auditory memory (units = # items). Participants reported the order of presented syllables in a serial memory span task;
- (d)
- Auditory Spatial Match (“Memory Grid”) targets auditory memory (units = # items). Participants matched identical cards representing syllables.
Data Analyses
3. Results
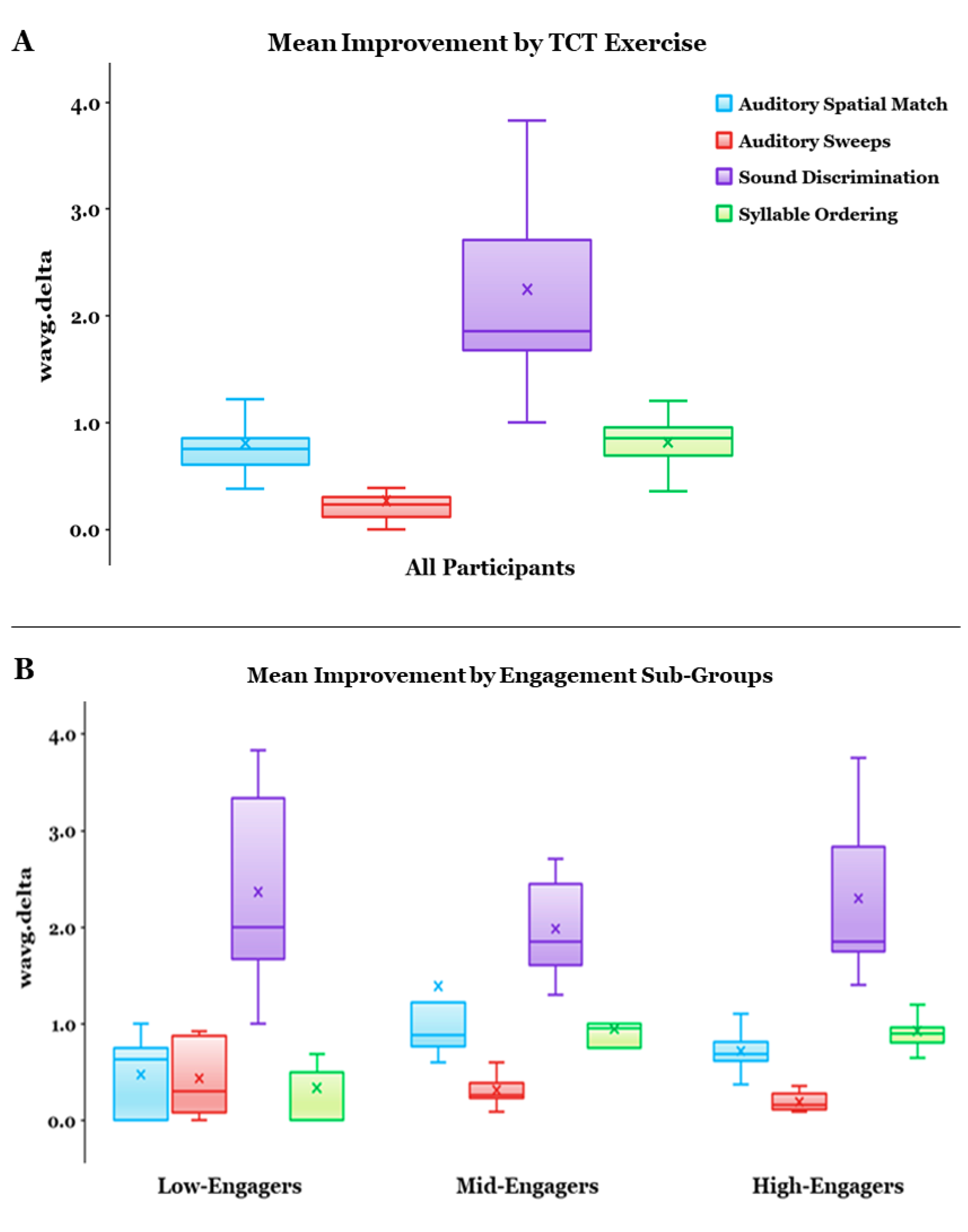
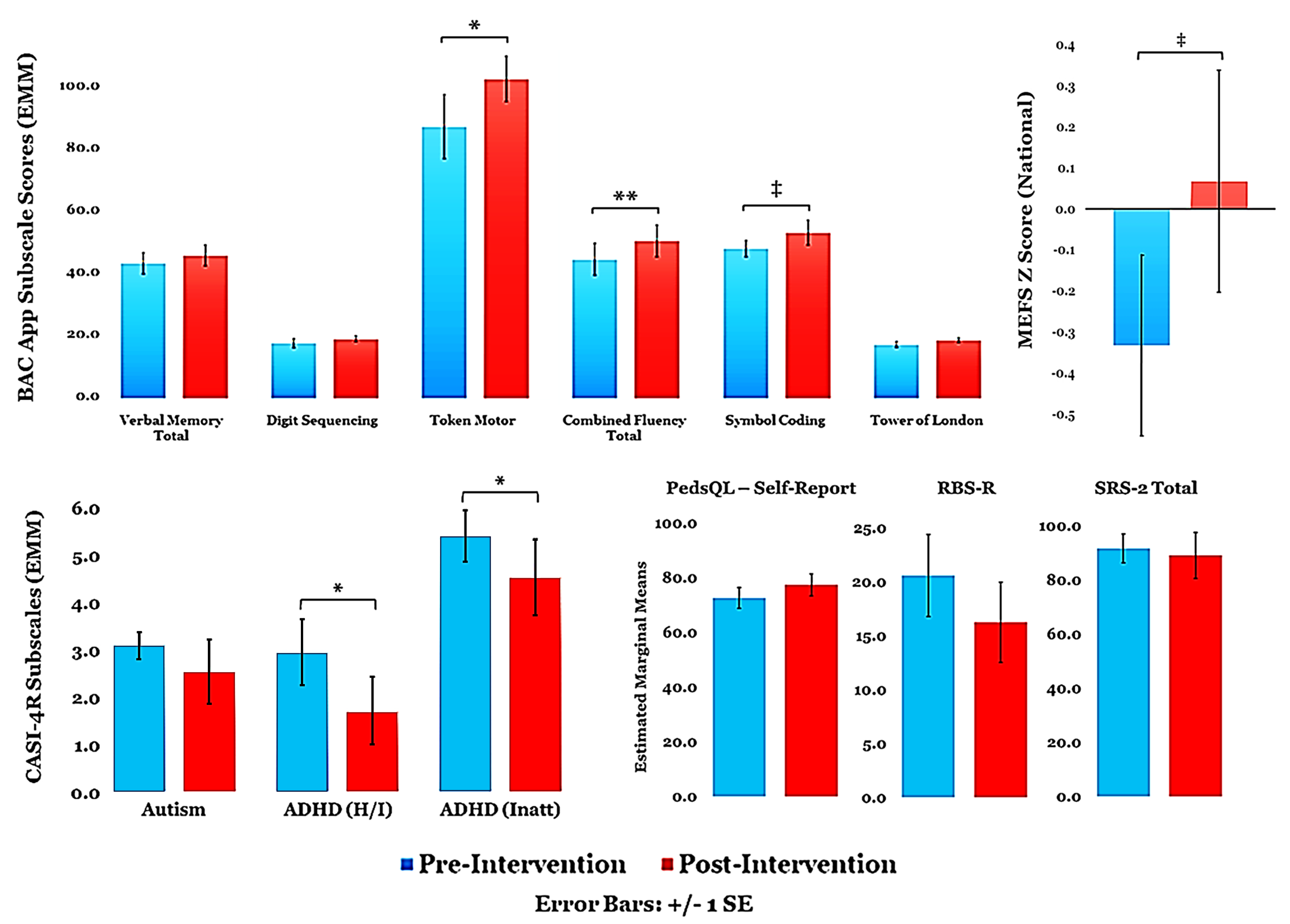
3.1. Overall Effect of Auditory-Based TCT
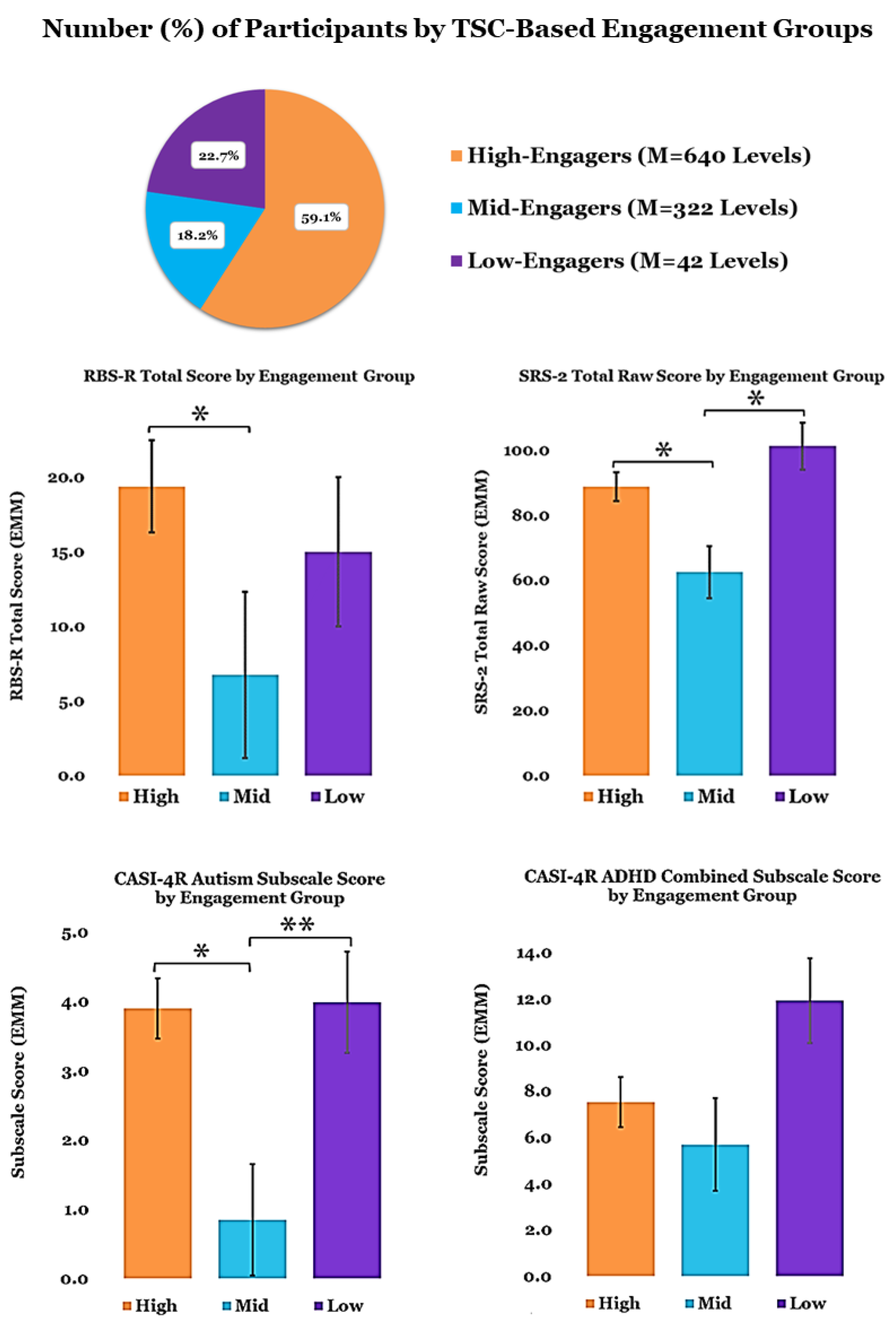
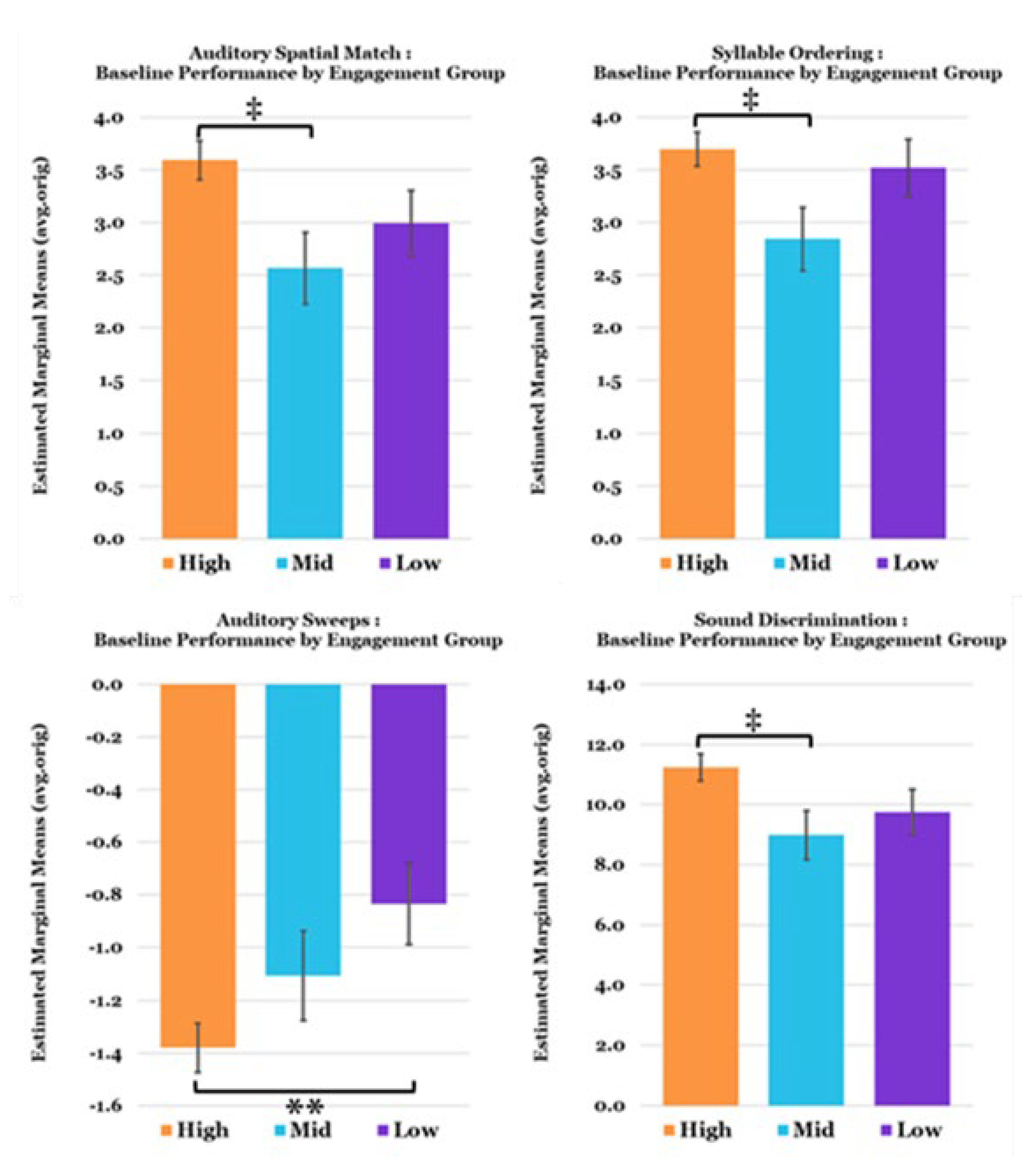
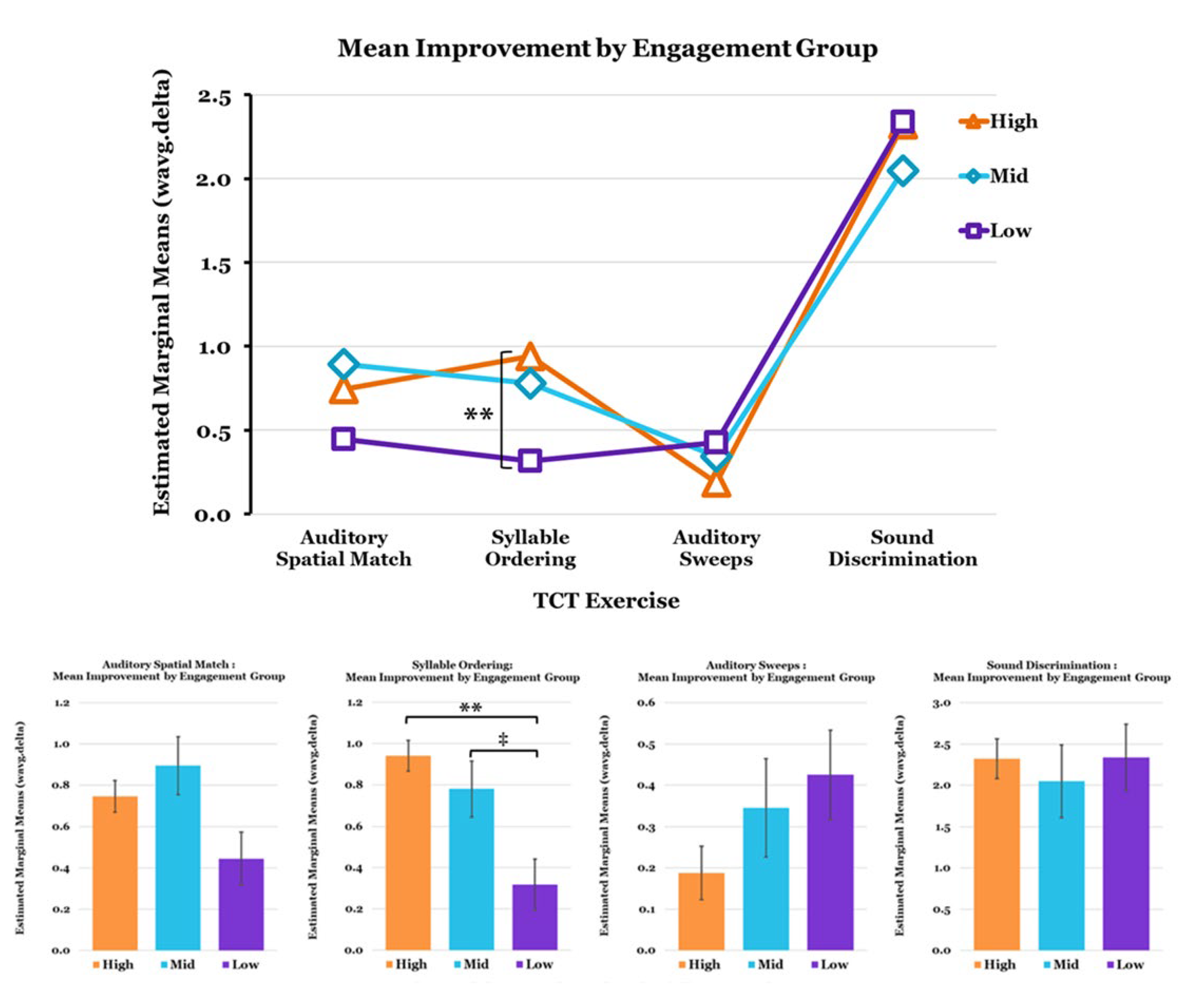
3.2. TCT Performance
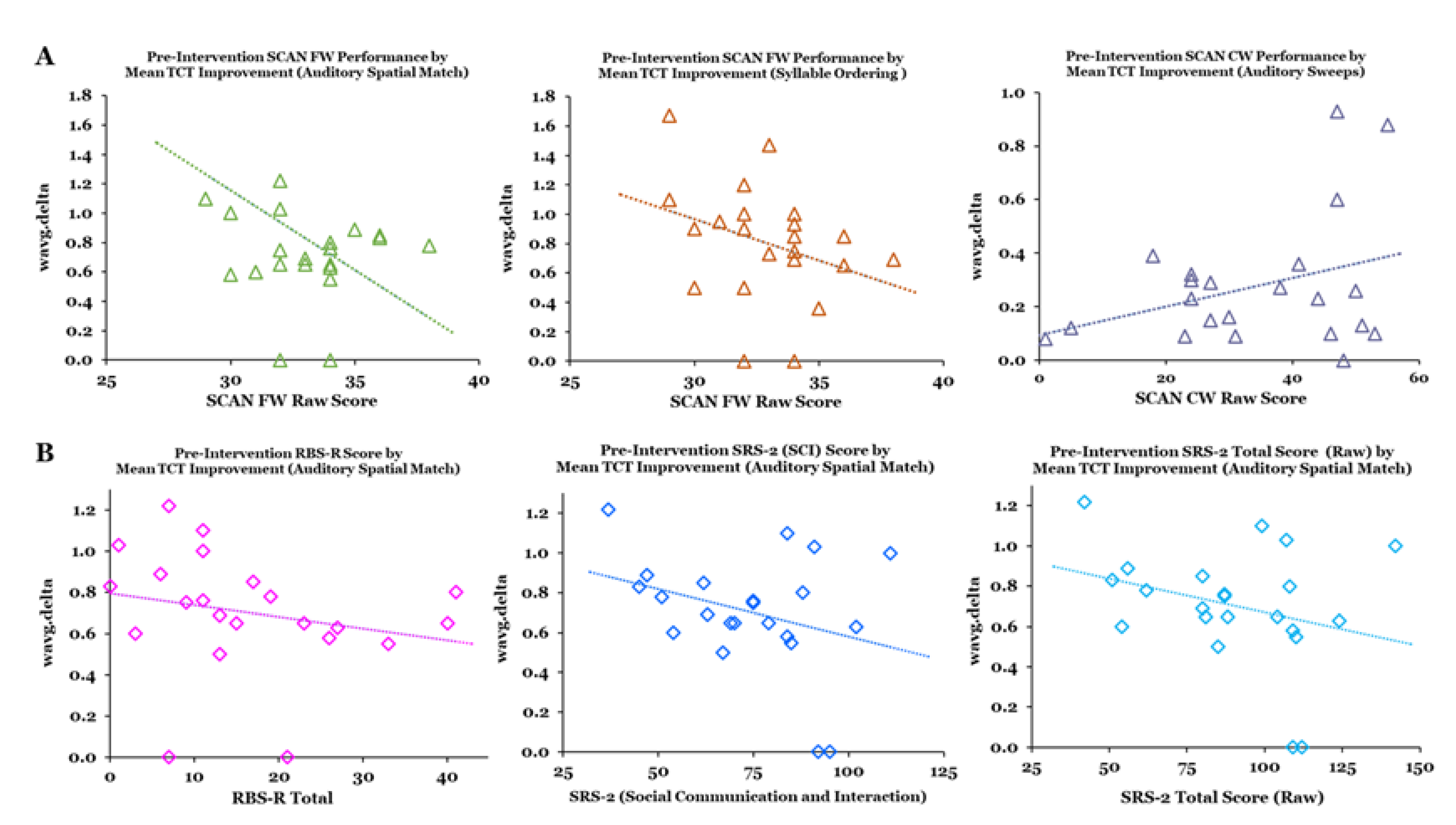
3.3. Relationships between Metrics and Predictors of TCT Performance
4. Discussion
Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Kanner, L. Autistic disturbances of affective contact. Acta Paedopsychiatr. 1968, 35, 100–136. [Google Scholar]
- Grapel, J.N.; Cicchetti, D.V.; Volkmar, F.R. Sensory features as diagnostic criteria for autism: Sensory features in autism. Yale J. Biol. Med. 2015, 88, 69–71. [Google Scholar]
- Thye, M.D.; Bednarz, H.M.; Herringshaw, A.J.; Sartin, E.B.; Kana, R.K. The impact of atypical sensory processing on social impairments in autism spectrum disorder. Dev. Cogn. Neurosci. 2018, 29, 151–167. [Google Scholar] [CrossRef]
- Robertson, C.E.; Baron-Cohen, S. Sensory perception in autism. Nat. Rev. Neurosci. 2017, 18, 671–684. [Google Scholar] [CrossRef]
- McCormick, C.; Hepburn, S.; Young, G.S.; Rogers, S.J. Sensory symptoms in children with autism spectrum disorder, other developmental disorders and typical development: A longitudinal study. Autism 2016, 20, 572–579. [Google Scholar] [CrossRef]
- James, P.; Schafer, E.; Wolfe, J.; Matthews, L.; Browning, S.; Oleson, J.; Sorensen, E.; Rance, G.; Shiels, L.; Dunn, A. Increased rate of listening difficulties in autistic children. J. Commun. Disord. 2022, 99, 106252. [Google Scholar] [CrossRef]
- Robertson, A.E.; Simmons, D.R. The relationship between sensory sensitivity and autistic traits in the general population. J. Autism Dev. Disord. 2013, 43, 775–784. [Google Scholar] [CrossRef]
- Cascio, C.J.; Woynaroski, T.; Baranek, G.T.; Wallace, M.T. Toward an interdisciplinary approach to understanding sensory function in autism spectrum disorder. Autism Res. 2016, 9, 920–925. [Google Scholar] [CrossRef]
- Matsuba, E.S.M.; Prieve, B.A.; Cary, E.; Pacheco, D.; Madrid, A.; McKernan, E.; Kaplan-Kahn, E.; Russo, N. A Preliminary Study Characterizing Subcortical and Cortical Auditory Processing and Their Relation to Autistic Traits and Sensory Features. J. Autism Dev. Disord. 2022; Online ahead of print. [Google Scholar] [CrossRef]
- Kojovic, N.; Ben Hadid, L.; Franchini, M.; Schaer, M. Sensory Processing Issues and Their Association with Social Difficulties in Children with Autism Spectrum Disorders. J. Clin. Med. 2019, 8, 1508. [Google Scholar] [CrossRef]
- O’Connor, K. Auditory processing in autism spectrum disorder: A review. Neurosci. Biobehav. Rev. 2012, 36, 836–854. [Google Scholar] [CrossRef]
- Maenner, M.J.; Shaw, K.A.; Bakian, A.V.; Bilder, D.A.; Durkin, M.S.; Esler, A.; Furnier, S.M.; Hallas, L.; Hall-Lande, J.; Hudson, A.; et al. Prevalence and Characteristics of Autism Spectrum Disorder Among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2018. MMWR. Surveill. Summ. 2021, 70, 1–16. [Google Scholar] [CrossRef]
- Cakir, J.; Frye, R.E.; Walker, S.J. The lifetime social cost of autism: 1990–2029. Res. Autism Spectr. Disord. 2020, 72, 101502. [Google Scholar] [CrossRef]
- Masi, A.; DeMayo, M.M.; Glozier, N.; Guastella, A.J. An Overview of Autism Spectrum Disorder, Heterogeneity and Treatment Options. Neurosci. Bul.l 2017, 33, 183–193. [Google Scholar] [CrossRef]
- Subramaniam, K.; Luks, T.L.; Fisher, M.; Simpson, G.V.; Nagarajan, S.; Vinogradov, S. Computerized cognitive training restores neural activity within the reality monitoring network in schizophrenia. Neuron 2012, 73, 842–853. [Google Scholar] [CrossRef]
- Fisher, M.; Herman, A.; Stephens, D.B.; Vinogradov, S. Neuroscience-informed computer-assisted cognitive training in schizophrenia. Ann. N. Y. Acad. Sci. 2016, 1366, 90–114. [Google Scholar] [CrossRef]
- Fisher, M.; Holland, C.; Merzenich, M.M.; Vinogradov, S. Using neuroplasticity-based auditory training to improve verbal memory in schizophrenia. Am. J. Psychiatry 2009, 166, 805–811. [Google Scholar] [CrossRef]
- Mishra, J.; Merzenich, M.M.; Sagar, R. Accessible online neuroplasticity-targeted training for children with ADHD. Child Adolesc. Psychiatry Ment. Health 2013, 7, 38. [Google Scholar] [CrossRef]
- Mishra, J.; Sagar, R.; Joseph, A.A.; Gazzaley, A.; Merzenich, M.M. Training sensory signal-to-noise resolution in children with ADHD in a global mental health setting. Transl. Psychiatry 2016, 6, e781. [Google Scholar] [CrossRef]
- Johnston, M.V.; Ishida, A.; Ishida, W.N.; Matsushita, H.B.; Nishimura, A.; Tsuji, M. Plasticity and injury in the developing brain. Brain Dev. 2009, 31, 1–10. [Google Scholar] [CrossRef]
- Huttenlocher, P.R. Morphometric study of human cerebral cortex development. Neuropsychologia 1990, 28, 517–527. [Google Scholar] [CrossRef]
- Ismail, F.Y.; Fatemi, A.; Johnston, M.V. Cerebral plasticity: Windows of opportunity in the developing brain. Eur. J. Paediatr. Neurol. 2017, 21, 23–48. [Google Scholar] [CrossRef]
- Gogtay, N.; Giedd, J.N.; Lusk, L.; Hayashi, K.M.; Greenstein, D.; Vaituzis, A.C.; Nugent, T.F., 3rd; Herman, D.H.; Clasen, L.S.; Toga, A.W.; et al. Dynamic mapping of human cortical development during childhood through early adulthood. Proc. Natl. Acad. Sci. USA 2004, 101, 8174–8179. [Google Scholar] [CrossRef]
- Crone, E.A.; Dahl, R.E. Understanding adolescence as a period of social-affective engagement and goal flexibility. Nat. Rev. Neurosci. 2012, 13, 636–650. [Google Scholar] [CrossRef]
- Kadosh, K.C.; Linden, D.E.J.; Lau, J.Y.F. Plasticity during childhood and adolescence: Innovative approaches to investigating neurocognitive development. Dev. Sci. 2013, 16, 574–583. [Google Scholar] [CrossRef]
- Gopnik, A.; O’Grady, S.; Lucas, C.G.; Griffiths, T.L.; Wente, A.; Bridgers, S.; Aboody, R.; Fung, H.; Dahl, R.E. Changes in cognitive flexibility and hypothesis search across human life history from childhood to adolescence to adulthood. Proc. Natl. Acad. Sci. USA 2017, 114, 7892–7899. [Google Scholar] [CrossRef]
- Selemon, L.D. A role for synaptic plasticity in the adolescent development of executive function. Transl. Psychiatry 2013, 3, e238. [Google Scholar] [CrossRef]
- Fuhrmann, D.; Knoll, L.J.; Blakemore, S.J. Adolescence as a Sensitive Period of Brain Development. Trends Cogn. Sci. 2015, 19, 558–566. [Google Scholar] [CrossRef]
- Nelson, E.E.; Lau, J.Y.F.; Jarcho, J.M. Growing pains and pleasures: How emotional learning guides development. Trends Cogn. Sci. 2014, 18, 99–108. [Google Scholar] [CrossRef]
- Nelson, E.E.; Leibenluft, E.; McClure, E.B.; Pine, D.S. The social re-orientation of adolescence: A neuroscience perspective on the process and its relation to psychopathology. Psychol. Med. 2005, 35, 163–174. [Google Scholar] [CrossRef]
- Vetter, N.C.; Leipold, K.; Kliegel, M.; Phillips, L.H.; Altgassen, M. Ongoing development of social cognition in adolescence. Child Neuropsychology 2013, 19, 615–629. [Google Scholar] [CrossRef]
- Blakemore, S.J. Development of the social brain in adolescence. J. R. Soc. Med. 2012, 105, 111–116. [Google Scholar] [CrossRef]
- Suzuki, K.; Takagai, S.; Tsujii, M.; Ito, H.; Nishimura, T.; Tsuchiya, K.J. Sensory processing in children with autism spectrum disorder and the mental health of primary caregivers. Brain Dev. 2019, 41, 341–351. [Google Scholar] [CrossRef]
- Howlin, P.; Goode, S.; Hutton, J.; Rutter, M. Adult outcome for children with autism. J. Child Psychol. Psychiatry 2004, 45, 212–229. [Google Scholar] [CrossRef]
- Nicholas, D.B.; Hodgetts, S.; Zwaigenbaum, L.; Smith, L.E.; Shattuck, P.; Parr, J.R.; Conlon, O.; Germani, T.; Mitchell, W.; Sacrey, L.; et al. Research needs and priorities for transition and employment in autism: Considerations reflected in a “Special Interest Group” at the International Meeting for Autism Research. Autism Res. 2017, 10, 15–24. [Google Scholar] [CrossRef]
- Grynszpan, O.; Perbal, S.; Pelissolo, A.; Fossati, P.; Jouvent, R.; Dubal, S.; Perez-Diaz, F. Efficacy and specificity of computer-assisted cognitive remediation in schizophrenia: A meta-analytical study. Psychol. Med. 2011, 41, 163–173. [Google Scholar] [CrossRef]
- Vinogradov, S.; Fisher, M.; de Villers-Sidani, E. Cognitive training for impaired neural systems in neuropsychiatric illness. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2012, 37, 43–76. [Google Scholar] [CrossRef]
- Peijnenborgh, J.C.; Hurks, P.M.; Aldenkamp, A.P.; Vles, J.S.; Hendriksen, J.G. Efficacy of working memory training in children and adolescents with learning disabilities: A review study and meta-analysis. Neuropsychol. Rehabil. 2016, 26, 645–672. [Google Scholar] [CrossRef]
- Spencer-Smith, M.; Klingberg, T. Benefits of a working memory training program for inattention in daily life: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0119522. [Google Scholar] [CrossRef]
- Melby-Lervag, M.; Hulme, C. Is working memory training effective? A meta-analytic review. Dev. Psychol. 2013, 49, 270–291. [Google Scholar] [CrossRef]
- Holmes, J.; Gathercole, S.E.; Dunning, D.L. Adaptive training leads to sustained enhancement of poor working memory in children. Dev. Sci. 2009, 12, F9–F15. [Google Scholar] [CrossRef]
- Klingberg, T.; Fernell, E.; Olesen, P.J.; Johnson, M.; Gustafsson, P.; Dahlstrom, K.; Gillberg, C.G.; Forssberg, H.; Westerberg, H. Computerized training of working memory in children with ADHD--a randomized, controlled trial. J. Am. Acad. Child Adolesc. Psychiatry 2005, 44, 177–186. [Google Scholar] [CrossRef]
- Klingberg, T. Training and plasticity of working memory. Trends Cogn. Sci. 2010, 14, 317–324. [Google Scholar] [CrossRef]
- Dahlin, E.; Backman, L.; Neely, A.S.; Nyberg, L. Training of the executive component of working memory: Subcortical areas mediate transfer effects. Restor. Neurol. Neurosci. 2009, 27, 405–419. [Google Scholar] [CrossRef]
- Adcock, R.A.; Dale, C.; Fisher, M.; Aldebot, S.; Genevsky, A.; Simpson, G.V.; Nagarajan, S.; Vinogradov, S. When top-down meets bottom-up: Auditory training enhances verbal memory in schizophrenia. Schizophr. Bull. 2009, 35, 1132–1141. [Google Scholar] [CrossRef]
- Dickinson, D.; Tenhula, W.; Morris, S.; Brown, C.; Peer, J.; Spencer, K.; Li, L.; Gold, J.M.; Bellack, A.S. A randomized, controlled trial of computer-assisted cognitive remediation for schizophrenia. Am. J. Psychiatry 2010, 167, 170–180. [Google Scholar] [CrossRef]
- Fisher, N.; Happe, F. A training study of theory of mind and executive function in children with autistic spectrum disorders. J. Autism Dev. Disord. 2005, 35, 757–771. [Google Scholar] [CrossRef]
- Kenworthy, L.; Yerys, B.E.; Anthony, L.G.; Wallace, G.L. Understanding executive control in autism spectrum disorders in the lab and in the real world. Neuropsychol. Rev. 2008, 18, 320–338. [Google Scholar] [CrossRef]
- Allen, M.L.; Hartley, C.; Cain, K. iPads and the Use of “Apps” by Children with Autism Spectrum Disorder: Do They Promote Learning? Front. Psychol. 2016, 7, 1305. [Google Scholar] [CrossRef]
- Esposito, M.; Sloan, J.; Tancredi, A.; Gerardi, G.; Postiglione, P.; Fotia, F.; Napoli, E.; Mazzone, L.; Valeri, G.; Vicari, S. Using Tablet Applications for Children With Autism to Increase Their Cognitive and Social Skills. J. Spec. Educ. Technol. 2017, 32, 199–209. [Google Scholar] [CrossRef]
- Tseng, A.; Biagianti, B.; Francis, S.M.; Conelea, C.A.; Jacob, S. Social Cognitive Interventions for Adolescents with Autism Spectrum Disorders: A Systematic Review. J. Affect. Disord. 2020, 274, 199–204. [Google Scholar] [CrossRef]
- Fisher, M.; Loewy, R.; Carter, C.; Lee, A.; Ragland, J.D.; Niendam, T.; Schlosser, D.; Pham, L.; Miskovich, T.; Vinogradov, S. Neuroplasticity-based auditory training via laptop computer improves cognition in young individuals with recent onset schizophrenia. Schizophr. Bull. 2015, 41, 250–258. [Google Scholar] [CrossRef]
- Nahum, M.; Fisher, M.; Loewy, R.; Poelke, G.; Ventura, J.; Nuechterlein, K.H.; Hooker, C.I.; Green, M.F.; Merzenich, M.; Vinogradov, S. A novel, online social cognitive training program for young adults with schizophrenia: A pilot study. Schizophr. Research. Cogn. 2014, 1, e11–e19. [Google Scholar] [CrossRef]
- Varni, J.W.; Seid, M.; Rode, C.A. The PedsQL: Measurement model for the pediatric quality of life inventory. Med. Care 1999, 37, 126–139. [Google Scholar] [CrossRef]
- Constantino, J.N.; Gruber, C.P. Social Responsiveness Scale: SRS-2, 2nd ed.; Western Psychological Services: Torrane, CA, USA, 2012; p. 106. [Google Scholar]
- Lam, K.S.; Aman, M.G. The Repetitive Behavior Scale-Revised: Independent validation in individuals with autism spectrum disorders. J. Autism Dev. Disord. 2007, 37, 855–866. [Google Scholar] [CrossRef]
- Gadow, K.D.; Sprafkin, J. Child and Adolescent Symptom Inventory-4R; Checkmate Plus: Stony Brook, NY, USA, 2005. [Google Scholar]
- Atkins, A.S.; Tseng, T.; Vaughan, A.; Twamley, E.W.; Harvey, P.; Patterson, T.; Narasimhan, M.; Keefe, R.S. Validation of the tablet-administered Brief Assessment of Cognition (BAC App). Schizophr. Res. 2017, 181, 100–106. [Google Scholar] [CrossRef]
- White, R.E.; Carlson, S.M. What would Batman do? Self-distancing improves executive function in young children. Dev. Sci. 2016, 19, 419–426. [Google Scholar] [CrossRef]
- Keith, R.W. Development and standardization of SCAN-A: Test of auditory processing disorders in adolescents and adults. J. Am. Acad. Audio.l 1995, 6, 286–292. [Google Scholar]
- Dale, C.L.; Brown, E.G.; Fisher, M.; Herman, A.B.; Dowling, A.F.; Hinkley, L.B.; Subramaniam, K.; Nagarajan, S.S.; Vinogradov, S. Auditory Cortical Plasticity Drives Training-Induced Cognitive Changes in Schizophrenia. Schizophr. Bull. 2016, 42, 220–228. [Google Scholar] [CrossRef]
- Biagianti, B.; Fisher, M.; Loewy, R.; Brandrett, B.; Ordorica, C.; LaCross, K.; Schermitzler, B.; McDonald, M.; Ramsay, I.; Vinogradov, S. Specificity and Durability of Changes in Auditory Processing Efficiency After Targeted Cognitive Training in Individuals With Recent-Onset Psychosis. Front. Psychiatry 2020, 11, 857. [Google Scholar] [CrossRef]
| Participants (N = 25) | |
|---|---|
| Age (years) | |
| Mean | 17.4 |
| SD | 5.0 |
| Sex (%) | |
| Male | 84.0 |
| Female | 16.0 |
| Race (%) | |
| American Indian/Alaska Native | 4.0 |
| Asian | 4.0 |
| Black/African American | 8.0 |
| White/Caucasian | 84.0 |
| Other | 4.0 |
| Neurodevelopmental Diagnoses (%) | |
| ADHD, ASD | 40.0 |
| ASD | 44.0 |
| ADHD, ASD, OCD | 12.0 |
| ASD, OCD | 4.0 |
| M | SD | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| avg.orig | |||||||||||
| 1. Auditory Spatial Match | 3.27 | 0.76 | r | ||||||||
| p | |||||||||||
| 2. Syllable Ordering | 3.50 | 0.65 | r | 0.659 ** | |||||||
| p | 0.001 | ||||||||||
| 3. Auditory Sweeps | −1.21 | 0.42 | r | −0.476 ** | −0.374 ‡ | ||||||
| p | 0.017 | 0.052 | |||||||||
| 4. Sound Discrimination | 10.49 | 1.95 | r | 0.500 ** | 0.700 ** | −0.635 ** | |||||
| p | 0.012 | 0.000 | 0.001 | ||||||||
| wavg.delta | |||||||||||
| 5. Auditory Spatial Match | 0.70 | 0.29 | r | −0.006 | −0.086 | −0.552 ** | 0.023 | ||||
| p | 0.490 | 0.360 | 0.006 | 0.462 | |||||||
| 6. Syllable Ordering | 0.77 | 0.35 | r | 0.171 | 0.094 | −0.667 ** | 0.193 | 0.590 ** | |||
| p | 0.236 | 0.346 | 0.001 | 0.208 | 0.003 | ||||||
| 7. Auditory Sweeps | 0.27 | 0.24 | r | −0.360 ‡ | −0.329 ‡ | 0.890 ** | −0.607 ** | −0.612 ** | −0.566 ** | ||
| p | 0.059 | 0.078 | 0.000 | 0.002 | 0.002 | 0.005 | |||||
| 8. Sound Discrimination | 2.28 | 0.83 | r | −0.340 ‡ | −0.176 | 0.268 | −0.325 ‡ | −0.144 | 0.189 | 0.072 | |
| p | 0.071 | 0.229 | 0.126 | 0.081 | 0.273 | 0.212 | 0.382 | ||||
| num | |||||||||||
| 9. TCT Levels Completed | 446.64 | 255.61 | r | 0.458 * | 0.244 | −0.613 ** | 0.433 * | 0.332 ‡ | 0.727 ** | −0.448 * | 0.037 |
| p | 0.021 | 0.150 | 0.002 | 0.028 | 0.076 | 0.000 | 0.024 | 0.439 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tseng, A.; DuBois, M.; Biagianti, B.; Brumley, C.; Jacob, S. Auditory Domain Sensitivity and Neuroplasticity-Based Targeted Cognitive Training in Autism Spectrum Disorder. J. Clin. Med. 2023, 12, 1635. https://doi.org/10.3390/jcm12041635
Tseng A, DuBois M, Biagianti B, Brumley C, Jacob S. Auditory Domain Sensitivity and Neuroplasticity-Based Targeted Cognitive Training in Autism Spectrum Disorder. Journal of Clinical Medicine. 2023; 12(4):1635. https://doi.org/10.3390/jcm12041635
Chicago/Turabian StyleTseng, Angela, Megan DuBois, Bruno Biagianti, Caroline Brumley, and Suma Jacob. 2023. "Auditory Domain Sensitivity and Neuroplasticity-Based Targeted Cognitive Training in Autism Spectrum Disorder" Journal of Clinical Medicine 12, no. 4: 1635. https://doi.org/10.3390/jcm12041635
APA StyleTseng, A., DuBois, M., Biagianti, B., Brumley, C., & Jacob, S. (2023). Auditory Domain Sensitivity and Neuroplasticity-Based Targeted Cognitive Training in Autism Spectrum Disorder. Journal of Clinical Medicine, 12(4), 1635. https://doi.org/10.3390/jcm12041635






