Peri-Operative Risk Factors Associated with Post-Operative Cognitive Dysfunction (POCD): An Umbrella Review of Meta-Analyses of Observational Studies
Abstract
1. Introduction
2. Materials and Methods
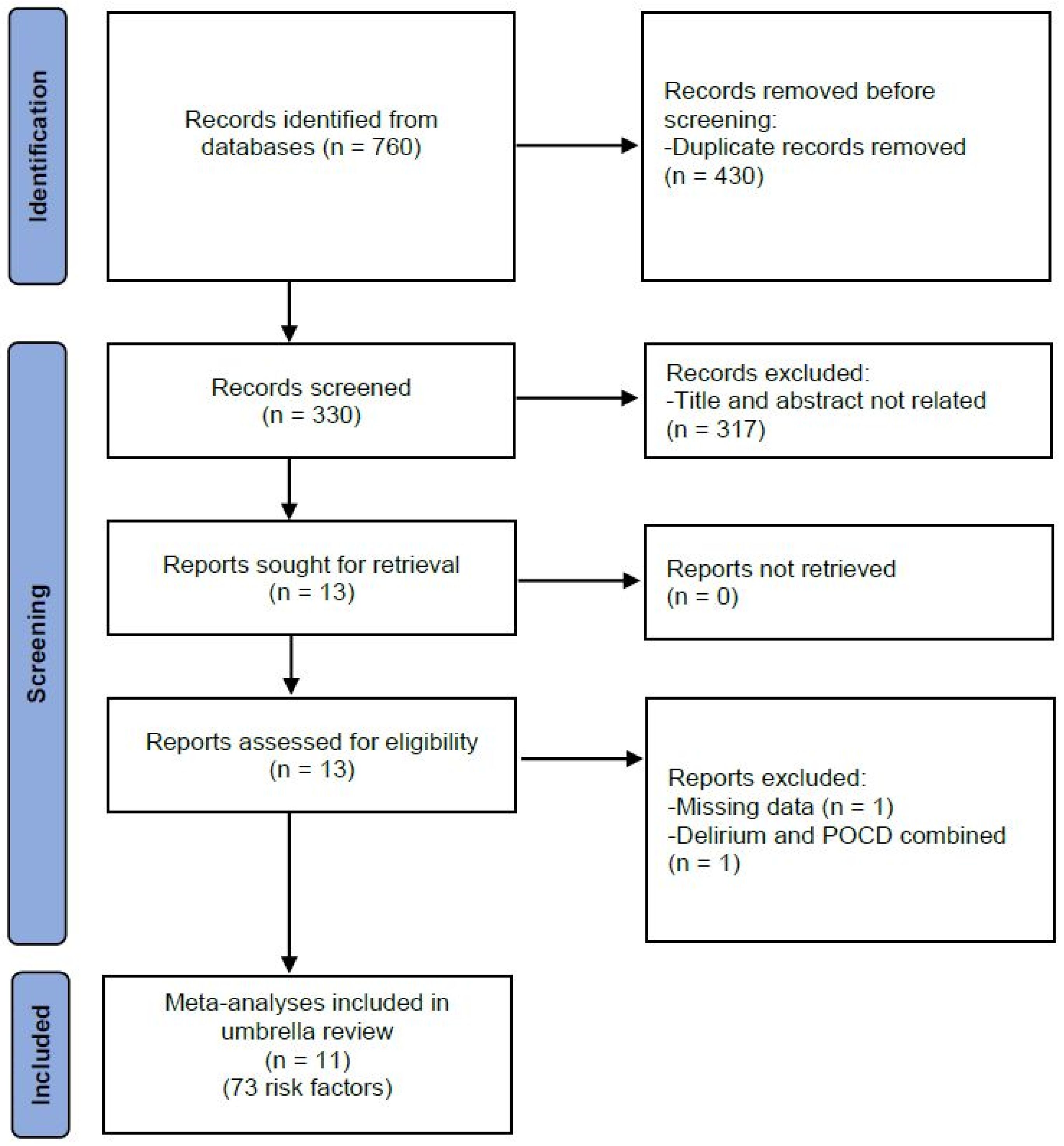
2.1. Literature Search and Selection Criteria
2.2. Data Extraction
2.3. Data Analysis
2.4. Quality Assessment of the Meta-Analyses and Evidence Grading
- Convincing (class I); where the number of cases is >1000, statistically significant using a p value of <1 × 10−6, I2 < 50%, 95% prediction interval excludes the null, the largest included individual study has a statistically significant effect (p ≤ 0.05), no small-study effects and no excess significance bias.
- Highly suggestive (class II); where the number of cases is >1000, statistically significant using a p value of <1 × 10−6, the largest included individual study has a statistically significant effect (p ≤ 0.05) and class I criteria not met.
- Suggestive (class III); where the number of cases is >1000, p value of <1 × 10−3 and Class I—II criteria not met.
- Weak (class IV); statistically significant (p ≤ 0.05) and class I—III criteria not met.
- No evidence (class V); no statistical significance (p > 0.05).
3. Results
3.1. Study Characteristics
3.2. Study Results
3.3. Credibility Assessment
3.4. Sensitivity Analysis
3.5. Quality Assessment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tasbihgou, S.R.; Absalom, A.R. Postoperative neurocognitive disorders. Korean J. Anesthesiol. 2021, 74, 15–22. [Google Scholar] [CrossRef]
- Evered, L.; Silbert, B.; Knopman, D.S.; Scott, D.A.; DeKosky, S.T.; Rasmussen, L.S.; Oh, E.S.; Crosby, G.; Berger, M.; Eckenhoff, R. Recommendations for the nomenclature of cognitive change associated with anaesthesia and surgery—2018. Anesthesiology 2018, 129, 872–879. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Text Revision (DSM-IV-TR®); American Psychiatric Association: Washington, DC, USA, 2010. [Google Scholar]
- Skvarc, D.R.; Berk, M.; Byrne, L.K.; Dean, O.M.; Dodd, S.; Lewis, M.; Marriott, A.; Moore, E.M.; Morris, G.; Page, R.S. Post-operative cognitive dysfunction: An exploration of the inflammatory hypothesis and novel therapies. Neurosci. Biobehav. Rev. 2018, 84, 116–133. [Google Scholar] [CrossRef] [PubMed]
- Eckenhoff, R.G.; Maze, M.; Xie, Z.; Culley, D.J.; Goodlin, S.J.; Zuo, Z.; Wei, H.; Whittington, R.A.; Terrando, N.; Orser, B.A. Perioperative neurocognitive disorder: State of the preclinical science. Anesthesiology 2020, 132, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Fu, H.; Wang, T. Neuroinflammation in perioperative neurocognitive disorders: From bench to the bedside. CNS Neurosci. Ther. 2022, 28, 484–496. [Google Scholar] [CrossRef] [PubMed]
- Rundshagen, I. Postoperative cognitive dysfunction. Dtsch. Ärzteblatt Int. 2014, 111, 119. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.-H.; Ji, M.-H.; Gao, D.-P.; Li, W.-Y.; Yang, J.-J. Association between perioperative blood transfusion and early postoperative cognitive dysfunction in aged patients following total hip replacement surgery. Upsala J. Med. Sci. 2014, 119, 262–267. [Google Scholar] [CrossRef]
- Newman, M.F.; Kirchner, J.L.; Phillips-Bute, B.; Gaver, V.; Grocott, H.; Jones, R.H.; Mark, D.B.; Reves, J.G.; Blumenthal, J.A. Longitudinal assessment of neurocognitive function after coronary-artery bypass surgery. N. Engl. J. Med. 2001, 344, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, Y. Postoperative cognitive dysfunction after cardiac surgery. Anaesth. Pain Intensive Care 2016, 20, S146–S149. [Google Scholar]
- Reis, H.J.; Teixeira, A.L.; Kalman, J.; Bogáts, G.; Babik, B.; Janka, Z.; Teixeira, M.M.; Palotás, A. Different inflammatory biomarker patterns in the cerebro-spinal fluid following heart surgery and major non-cardiac operations. Curr. Drug Metab. 2007, 8, 639–642. [Google Scholar] [CrossRef]
- Hood, R.; Budd, A.; Sorond, F.; Hogue, C. Peri—Operative neurological complications. Anaesthesia 2018, 73, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Evered, L.A.; Silbert, B.S.; Scott, D.A.; Maruff, P.; Ames, D. Prevalence of dementia 7.5 years after coronary artery bypass graft surgery. Anesthesiology 2016, 125, 62–71. [Google Scholar] [CrossRef]
- Ntalouka, M.; Arnaoutoglou, E.; Tzimas, P. Postoperative cognitive disorders: An update. Hippokratia 2018, 22, 147. [Google Scholar] [PubMed]
- Terrando, N.; Eriksson, L.I.; Kyu Ryu, J.; Yang, T.; Monaco, C.; Feldmann, M.; Jonsson Fagerlund, M.; Charo, I.F.; Akassoglou, K.; Maze, M. Resolving postoperative neuroinflammation and cognitive decline. Ann. Neurol. 2011, 70, 986–995. [Google Scholar] [CrossRef] [PubMed]
- Vutskits, L.; Xie, Z. Lasting impact of general anaesthesia on the brain: Mechanisms and relevance. Nat. Rev. Neurosci. 2016, 17, 705–717. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Chen, Y.; Zhang, P.; Chen, G.; Zhou, Y.; Yu, X. The potential mechanism of postoperative cognitive dysfunction in older people. Exp. Gerontol. 2020, 130, 110791. [Google Scholar] [CrossRef]
- Belrose, J.C.; Noppens, R.R. Anesthesiology and cognitive impairment: A narrative review of current clinical literature. BMC Anesthesiol. 2019, 19, 241. [Google Scholar] [CrossRef]
- Safavynia, S.A.; Goldstein, P.A. The role of neuroinflammation in postoperative cognitive dysfunction: Moving from hypothesis to treatment. Front. Psychiatry 2019, 9, 752. [Google Scholar] [CrossRef]
- Wang, W.; Wang, Y.; Wu, H.; Lei, L.; Xu, S.; Shen, X.; Guo, X.; Shen, R.; Xia, X.; Liu, Y. Postoperative cognitive dysfunction: Current developments in mechanism and prevention. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2014, 20, 1908. [Google Scholar] [CrossRef]
- Needham, M.; Webb, C.; Bryden, D. Postoperative cognitive dysfunction and dementia: What we need to know and do. BJA Br. J. Anaesth. 2017, 119, i115–i125. [Google Scholar] [CrossRef]
- Bramley, P.; McArthur, K.; Blayney, A.; McCullagh, I. Risk factors for postoperative delirium: An umbrella review of systematic reviews. Int. J. Surg. 2021, 93, 106063. [Google Scholar] [CrossRef] [PubMed]
- Rengel, K.F.; Pandharipande, P.P.; Hughes, C.G. Postoperative delirium. La Presse Médicale 2018, 47, e53–e64. [Google Scholar] [CrossRef] [PubMed]
- Aromataris, E.; Fernandez, R.; Godfrey, C.M.; Holly, C.; Khalil, H.; Tungpunkom, P. Summarizing systematic reviews: Methodological development, conduct and reporting of an umbrella review approach. JBI Evid. Implement. 2015, 13, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Int. J. Surg. 2021, 88, 105906. [Google Scholar] [CrossRef] [PubMed]
- Marx, W.; Veronese, N.; Kelly, J.T.; Smith, L.; Hockey, M.; Collins, S.; Trakman, G.L.; Hoare, E.; Teasdale, S.B.; Wade, A. The dietary inflammatory index and human health: An umbrella review of meta-analyses of observational studies. Adv. Nutr. 2021, 12, 1681–1690. [Google Scholar] [CrossRef] [PubMed]
- Radua, J.; Ramella-Cravaro, V.; Ioannidis, J.P.; Reichenberg, A.; Phiphopthatsanee, N.; Amir, T.; Yenn Thoo, H.; Oliver, D.; Davies, C.; Morgan, C. What causes psychosis? An umbrella review of risk and protective factors. World Psychiatry 2018, 17, 49–66. [Google Scholar] [CrossRef] [PubMed]
- Greaves, D.; Psaltis, P.J.; Davis, D.H.J.; Ross, T.J.; Ghezzi, E.S.; Lampit, A.; Smith, A.E.; Keage, H.A.D. Risk Factors for Delirium and Cognitive Decline following Coronary Artery Bypass Grafting Surgery: A Systematic Review and Meta-Analysis. J. Am. Heart Assoc. 2020, 9, e017275. [Google Scholar] [CrossRef]
- Monk, T.G.; Weldon, B.C.; Garvan, C.W.; Dede, D.E.; Van Der Aa, M.T.; Heilman, K.M.; Gravenstein, J.S. Predictors of cognitive dysfunction after major noncardiac surgery. J. Am. Soc. Anesthesiol. 2008, 108, 18–30. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G. Quantifying heterogeneity in a meta—Analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef]
- Ioannidis, J.P.; Trikalinos, T.A. An exploratory test for an excess of significant findings. Clin. Trials 2007, 4, 245–253. [Google Scholar] [CrossRef]
- Hovens, I.B.; van Leeuwen, B.L.; Mariani, M.A.; Kraneveld, A.D.; Schoemaker, R.G. Postoperative cognitive dysfunction and neuroinflammation; cardiac surgery and abdominal surgery are not the same. Brain Behav. Immun. 2016, 54, 178–193. [Google Scholar] [CrossRef] [PubMed]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017, 358, j4008. [Google Scholar] [CrossRef]
- Aceto, P.; Lai, C.; de Crescenzo, F.; Crea, M.A.; Di Franco, V.; Pellicano, G.R.; Perilli, V.; Lai, S.; Papanice, D.; Sollazzi, L. Cognitive decline after carotid endarterectomy: Systematic review and meta-analysis. Eur. J. Anaesthesiol. 2020, 37, 1066–1074. [Google Scholar] [CrossRef] [PubMed]
- Feinkohl, I.; Winterer, G.; Pischon, T. Obesity and post-operative cognitive dysfunction: A systematic review and meta-analysis. Diabetes/Metab. Res. Rev. 2016, 32, 643–651. [Google Scholar] [CrossRef]
- Feinkohl, I.; Winterer, G.; Pischon, T. Diabetes is associated with risk of postoperative cognitive dysfunction: A meta-analysis. Diabetes/Metab. Res. Rev. 2017, 33, e2884. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Wang, K.; Gu, T.; Du, B.; Song, J. Association between APOE epsilon 4 allele and postoperative cognitive dysfunction: A meta-analysis. Int. J. Neurosci. 2014, 124, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Feinkohl, I.; Winterer, G.; Spies, C.D.; Pischon, T. Cognitive Reserve and the Risk of Postoperative Cognitive Dysfunction. Dtsch. Arztebl. Int. 2017, 114, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Feinkohl, I.; Winterer, G.; Pischon, T. Hypertension and risk of post-operative cognitive dysfunction (POCD): A systematic review and meta-analysis. Clin. Pract. Epidemiol. Ment. Health 2017, 13, 27–42. [Google Scholar] [CrossRef]
- Feinkohl, I.; Winterer, G.; Pischon, T. Associations of dyslipidaemia and lipid-lowering treatment with risk of postoperative cognitive dysfunction: A systematic review and meta-analysis. J. Epidemiol. Community Health 2018, 72, 499–506. [Google Scholar] [CrossRef]
- Ghezzi, E.S.; Ross, T.J.; Davis, D.; Psaltis, P.J.; Loetscher, T.; Keage, H.A.D. Meta-Analysis of Prevalence and Risk Factors for Cognitive Decline and Improvement after Transcatheter Aortic Valve Implantation. Am. J. Cardiol. 2020, 127, 105–112. [Google Scholar] [CrossRef]
- Liu, X.; Yu, Y.; Zhu, S. Inflammatory markers in postoperativedelirium (POD) and cognitive dysfunction (POCD): A meta-analysis of observational studies. PLoS ONE 2018, 13, e0195659. [Google Scholar] [CrossRef] [PubMed]
- Fu, C.; Lin, J.; Gong, G.; Zhong, W.; Chen, H.; Luo, X. Inflammatory markers in postoperative cognitive dysfunction for patients undergoing total hip arthroplasty: A meta-analysis. Aging Clin. Exp. Res. 2022, 34, 277–288. [Google Scholar] [CrossRef]
- Monk, T.G.; Price, C.C. Postoperative cognitive disorders. Curr. Opin. Crit. Care 2011, 17. [Google Scholar] [CrossRef] [PubMed]
- Mortasawi, A.; Arnrich, B.; Walter, J.; Frerichs, I.; Rosendahl, U.; Ennker, J. Impact of age on the results of coronary artery bypass grafting. Asian Cardiovasc. Thorac. Ann. 2004, 12, 324–329. [Google Scholar] [CrossRef]
- Zhang, W.-Y.; Wu, W.-L.; Gu, J.-J.; Sun, Y.; Ye, X.-F.; Qiu, W.-J.; Su, C.-Q.; Zhang, S.-Q.; Ye, W.-Q. Risk factors for postoperative delirium in patients after coronary artery bypass grafting: A prospective cohort study. J. Crit. Care 2015, 30, 606–612. [Google Scholar] [CrossRef]
- Scott, D.A.; Evered, L.; Maruff, P.; MacIsaac, A.; Maher, S.; Silbert, B.S. Cognitive function before and after left heart catheterization. J. Am. Heart Assoc. 2018, 7, e008004. [Google Scholar] [CrossRef] [PubMed]
- Robinson, T.N.; Wu, D.S.; Pointer, L.F.; Dunn, C.L.; Moss, M. Preoperative cognitive dysfunction is related to adverse postoperative outcomes in the elderly. J. Am. Coll. Surg. 2012, 215, 12–17. [Google Scholar] [CrossRef]
- Clegg, A.; Young, J.; Iliffe, S.; Rikkert, M.O.; Rockwood, K. Frailty in elderly people. Lancet 2013, 381, 752–762. [Google Scholar] [CrossRef]
- Olotu, C. Postoperative neurocognitive disorders. Curr. Opin. Anesthesiol. 2020, 33, 101–108. [Google Scholar] [CrossRef]
- McCrimmon, R.J.; Ryan, C.M.; Frier, B.M. Diabetes and cognitive dysfunction. Lancet 2012, 379, 2291–2299. [Google Scholar] [CrossRef]
- Vagelatos, N.T.; Eslick, G.D. Type 2 diabetes as a risk factor for Alzheimer’s disease: The confounders, interactions, and neuropathology associated with this relationship. Epidemiol. Rev. 2013, 35, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Brandt, M.; Harder, K.; Walluscheck, K.P.; Fraund, S.; Böning, A.; Cremer, J. Coronary artery bypass surgery in diabetic patients. J. Card. Surg. 2004, 19, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Puskas, F.; Grocott, H.P.; White, W.D.; Mathew, J.P.; Newman, M.F.; Bar-Yosef, S. Intraoperative hyperglycemia and cognitive decline after CABG. Ann. Thorac. Surg. 2007, 84, 1467–1473. [Google Scholar] [CrossRef]
- Desborough, J. The stress response to trauma and surgery. Br. J. Anaesth. 2000, 85, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Moller, J.T.; Cluitmans, P.; Rasmussen, L.S.; Houx, P.; Rasmussen, H.; Canet, J.; Rabbitt, P.; Jolles, J.; Larsen, K.; Hanning, C. Long-term postoperative cognitive dysfunction in the elderly: ISPOCD1 study. Lancet 1998, 351, 857–861. [Google Scholar] [CrossRef]
- Rasmussen, L.; O’brien, J.; Silverstein, J.; Johnson, T.; Siersma, V.; Canet, J.; Jolles, J.; Hanning, C.; Kuipers, H.; Abildstrom, H. Is peri-Operative cortisol secretion related to post-operative cognitive dysfunction? Acta Anaesthesiol. Scand. 2005, 49, 1225–1231. [Google Scholar] [CrossRef]
- Travica, N.; Aslam, H.; O’Neil, A.; Lane, M.M.; Berk, M.; Gamage, E.; Walder, K.; Liu, Z.S.; Segasby, T.; Marx, W. Brain Derived Neurotrophic Factor in Perioperative Neurocognitive Disorders: Current Evidence and Future Directions. Neurobiol. Learn. Mem. 2022, 193, 107656. [Google Scholar] [CrossRef]
- Bekker, A.Y.; Weeks, E.J. Cognitive function after anaesthesia in the elderly. Best Pract. Res. Clin. Anaesthesiol. 2003, 17, 259–272. [Google Scholar] [CrossRef]
- Silbert, B.; Evered, L.; Scott, D.A.; McMahon, S.; Choong, P.; Ames, D.; Maruff, P.; Jamrozik, K. Preexisting cognitive impairment is associated with postoperative cognitive dysfunction after hip joint replacement surgery. Anesthesiology 2015, 122, 1224–1234. [Google Scholar] [CrossRef]
- Nadelson, M.; Sanders, R.; Avidan, M. Perioperative cognitive trajectory in adults. Br. J. Anaesth. 2014, 112, 440–451. [Google Scholar] [CrossRef]
- van Sinderen, K.; Schwarte, L.A.; Schober, P. Diagnostic criteria of postoperative cognitive dysfunction: A focused systematic review. Anesthesiol. Res. Pract. 2020, 2020, 7384394. [Google Scholar] [CrossRef] [PubMed]
| Outcome | Study Design | Level of Comparison | n_Study | Participants, n | n_Cases | Type of Effect Size Metric | Effect Size (95% CI) | 95% CI Prediction Intervals | p Value | I2 | Largest Study Effect Size (95% CI) | PUBLICATION Bias | Small-Study Effect or Excess Significance Bias | Evidence Class |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pre-operative factors | ||||||||||||||
| AF (transcatheter aortic valve Implantation) [41] | Prospective | Yes vs. no | 3 | 410 | 58 | Odds ratio | 0.25 (−0.1, 0.59) | −1.98, 2.47 | 1.57 × 10−1 | 0% | 0.72 (−0.25, 1.68) | No | Neither | NS |
| Age (transcatheter aortic valve implantation) [41] | Prospective | Continuous | 2 | 340 | 35 | Mean difference | 0.07 (−0.07, 0.2) | NE | 3.34 × 10−1 | 31% | 0.15 (−0.03, 0.34) | NE | Neither | NS |
| Age (coronary artery bypass) [28] | Prospective | Continuous | 22 | 2881 | 1397 | Mean difference | 0.27 (0.14, 0.41) | −0.38, 0.93 | 9.47 × 10−5 | 92% | 1.49 (1.3, 1.68) | No | Neither | III |
| Age (carotid endarterectomy) [34] | Prospective | Continuous | 10 | 884 | 122 | Mean difference | 0.1 (0.03, 0.17) | 0.02, 0.18 | 3.19 × 10−3 | 0% | 0.24 (0, 0.49) | No | Neither | IV |
| APOE4 (cardiac) [37] | Prospective | Yes vs. no | 3 | 247 | 54 | Odds ratio | 0.26 (−0.13, 0.65) | −2.27, 2.79 | 1.85 × 10−1 | 0% | 0.38 (−0.21, 0.98) | No | Neither | NS |
| APOE4 (non-cardiac) [37] | Prospective | Yes vs. no | 3 | 2928 | 829 | Odds ratio | 0.36 (−0.03, 0.75) | −3.89, 4.61 | 7.16 × 10−2 | 77% | 0.68 (−0.92, 2.28) | No | Neither | NS |
| Arrhythmia (coronary artery bypass) [28] | Prospective | Yes vs. no | 7 | 945 | 400 | Odds ratio | 0.12 (−0.15, 0.4) | −0.23, 0.48 | 3.75 × 10−1 | 0% | 1.32 (0.19, 2.45) | No | Neither | NS |
| BMI (coronary artery bypass) [28] | Prospective | Continuous | 5 | 910 | 471 | Mean difference | 0.14 (−0.01, 0.28) | −0.36, 0.63 | 6.36 × 10−2 | 75% | 0.32 (0.23, 0.42) | No | Neither | NS |
| BMI (transcatheter aortic valve implantation) [41] | Prospective | Continuous | 2 | 340 | 35 | Mean difference | 0.04 (−0.06, 0.15) | NE | 4.22 × 10−1 | 0% | 0.07 (−0.12, 0.26) | NE | No excess significance | NS |
| Cognition: all tests (coronary artery bypass) [28] | Prospective | Continuous | 3 | 155 | 82 | Mean difference | 0.2 (0.04, 0.36) | −0.82, 1.22 | 1.26 × 10−2 | 0% | 0.3 (0.05, 0.56) | No | Neither | IV |
| Cognition: MMSE (coronary artery bypass) [28] | Prospective | Continuous | 2 | 120 | 68 | Mean difference | 0.23 (0.05, 0.41) | NE | 1.19 × 10−2 | 0% | 0.31 (0.06, 0.56) | NE | No excess significance | IV |
| Cognitive impairment (coronary artery bypass) [28] | Prospective | Yes vs. no | 4 | 749 | 214 | Odds ratio | 0.06 (−0.19, 0.3) | −0.48, 0.6 | 6.50 × 10−1 | 0% | 0.25 (−0.21, 0.71) | No | Neither | NS |
| Cognitive impairment (transcatheter aortic valve implantation) [41] | Prospective | Yes vs. no | 2 | 340 | 35 | Odds ratio | −0.13 (−0.73, 0.47) | NE | 6.72 × 10−1 | 8% | −0.01 (−0.54, 0.52) | NE | No excess significance * | NS |
| Contralateral stenonsis (carotid endarterectomy) [34] | Prospective | Yes vs. no | 3 | 252 | 92 | Odds ratio | 0.32 (−0.18, 0.83) | −2.96, 3.61 | 2.12 × 10−1 | 0% | 0.35 (−0.86, 1.56) | No | Neither | NS |
| C-reactive protein [42] | Prospective | Continuous | 10 | 594 | 211 | Mean difference | 0.21 (0.08, 0.33) | −0.16, 0.58 | 1.14 × 10−3 | 56% | 0.64 (0.33, 0.94) | No | Neither | NS |
| C-reactive protein (hip arthroplasty) [43] | Prospective | Continuous | 8 | 744 | 208 | Mean difference | 0.23 (0.11, 0.35) | −0.11, 0.56 | 2.01 × 10−4 | 55% | 0.5 (0.27, 0.73) | No | Either | IV |
| Depression (coronary artery bypass) [28] | Prospective | Yes vs. no | 2 | 330 | 188 | Odds ratio | 0.68 (0.06, 1.3) | NE | 3.12 × 10−2 | 62% | 1.01 (0.43, 1.58) | NE | Neither | IV |
| Depression: all test (coronary artery bypass) [28] | Prospective | Yes vs. no | 4 | 426 | 127 | Mean difference | 0.28 (−0.1, 0.67) | −1.53, 2.1 | 1.48 × 10−1 | 92% | 0.82 (0.61, 1.02) | No | Neither | NS |
| Diabetes [36] | Prospective | Yes vs. no | 13 | 2554 | 815 | Odds ratio | 0.16 (0.01, 0.32) | −0.33, 0.65 | 4.13 × 10−2 | 71% | 2.17 (0.36, 3.98) | No | No small-study effect * | IV |
| Diabetes (transcatheter aortic valve implantation) [41] | Prospective | Yes vs. no | 2 | 340 | 35 | Odds ratio | −0.15 (−0.64, 0.33) | NE | 5.34 × 10−1 | 0% | −0.09 (−0.62, 0.44) | NE | No excess significance * | NS |
| Diabetes (carotid endarterectomy) [34] | Prospective | Yes vs. no | 12 | 1888 | 493 | Odds ratio | 0.12 (−0.06, 0.3) | −0.22, 0.46 | 1.83 × 10−1 | 15% | 0.9 (0.34, 1.46) | No | Neither | NS |
| Diabetes (coronary artery bypass) [28] | Prospective | Yes vs. no | 17 | 2968 | 1273 | Odds ratio | 0.2 (0.1, 0.3) | 0.06, 0.34 | 4.63 × 10−5 | 5% | 0.57 (0.08, 1.06) | No | Neither | III |
| Dyslipidemia (carotid endarterectomy) [34] | Prospective | Yes vs. no | 8 | 766 | 374 | Odds ratio | 0.03 (−0.22, 0.28) | −0.29, 0.34 | 8.38 × 10−1 | 0% | 0.53 (−0.66, 1.72) | No | Neither | NS |
| Dyslipidemia (coronary artery bypass) [28] | Prospective | Yes vs. no | 6 | 840 | 227 | Odds ratio | 0.08 (−0.17, 0.33) | −0.54, 0.71 | 5.13 × 10−1 | 36% | 0.86 (0.24, 1.47) | No | Neither | NS |
| Education [38] | Prospective | Continuous | 8 | 2535 | 680 | Odds ratio | −0.06 (−0.09, −0.03) | −0.14, 0.01 | 3.74 × 10−5 | 44% | −0.01 (−0.06, 0.03) | Yes | Small-study effects | IV |
| Education (coronary artery bypass) [28] | Prospective | Continuous | 6 | 538 | 243 | Mean difference | 0.14 (0.05, 0.22) | 0.02, 0.26 | 1.67 × 10−3 | 0% | 0.3 (−0.04, 0.63) | No | Neither | IV |
| Euroscore (coronary artery bypass) [28] | Prospective | Continuous | 4 | 582 | 233 | Mean difference | 0.23 (0.14, 0.31) | 0, 0.45 | 2.09 × 10−7 | 10% | 0.3 (0.16, 0.45) | No | Neither | IV |
| Hypercholestrolemia [40] | Prospective | Yes vs. no | 12 | 1538 | 436 | Odds ratio | −0.04 (−0.12, 0.04) | −0.13, 0.05 | 3.47 × 10−1 | 0% | 0.86 (0.24, 1.47) | No | Neither | NS |
| Hypertension [39] | Prospective | Yes vs. no | 24 | 4247 | 1385 | Odds ratio | 0.03 (−0.03, 0.1) | −0.15, 0.22 | 3.28 × 10−1 | 35% | 0.92 (0.02, 1.83) | No | No small-study effect * | NS |
| Hypertension (carotid endarterectomy) [34] | Prospective | Yes vs. no | 12 | 1887 | 1266 | Odds ratio | −0.01 (−0.15, 0.13) | −0.17, 0.15 | 9.00 × 10−1 | 0% | 0.61(−1.01, 2.23) | No | Neither | NS |
| Hypertension (transcatheter aortic valve implantation) [41] | Prospective | Yes vs. no | 2 | 340 | 35 | Odds ratio | 0.15 (−1.01, 1.32) | NE | 7.99 × 10−1 | 28% | 0.59 (−0.54, 1.72) | NE | No excess significance * | NS |
| Hypertension (coronary artery bypass) [28] | Prospective | Yes vs. no | 15 | 2115 | 871 | Odds ratio | 0.36 (0.21, 0.51) | −0.03, 0.75 | 4.91 × 10−6 | 34% | 1 (0.6, 1.41) | No | Neither | IV |
| Interleukin 1β (hip arthroplasty) [43] | Prospective | Continuous | 5 | 247 | 90 | Mean difference | 0.19 (0.04, 0.34) | −0.17, 0.55 | 1.14 × 10−2 | 26% | 0.39 (0.09, 0.68) | No | Neither | IV |
| Interleukin 6 [42] | Prospective | Continuous | 16 | 986 | 373 | Mean Difference | 0.15 (0.08, 0.22) | 0, 0.29 | 2.43 × 10−5 | 17% | 0.6 (0.34, 0.86) | No | Neither | IV |
| Interleukin 6 (hip arthroplasty) [43] | Prospective | Continuous | 6 | 699 | 182 | Mean difference | 0.1 (0.03, 0.17) | 0, 0.21 | 7.78 × 10−3 | 0% | 0.24(−0.08, 0.57) | No | Neither | IV |
| Kidney injury (coronary artery bypass) [28] | Prospective | Yes vs. no | 4 | 749 | 275 | Odds ratio | 0.11 (−0.51, 0.72) | −2.26, 2.47 | 7.35 × 10−1 | 54% | 0.79 (0.01, 1.57) | No | Excess significance bias | NS |
| LVEF% (coronary artery bypass) [28] | Prospective | Continuous | 9 | 1225 | 623 | Mean Difference | 0.14 (0.04, 0.24) | −0.15, 0.43 | 4.72 × 10−3 | 62% | 0.64 (0.32, 0.97) | Yes | Small-study effects | IV |
| Male sex (transcatheter aortic valve implantation) [41] | Prospective | Male vs. female | 2 | 340 | 35 | Odds ratio | 0.13 (−0.26, 0.53) | NE | 5.12 × 10−1 | 0% | 0.27 (−0.7, 1.23) | NE | No excess significance * | NS |
| Male sex (carotid endarterectomy) [34] | Prospective | Male vs. female | 12 | 1888 | 1397 | Odds ratio | −0.05 (−0.15, 0.05) | −0.16, 0.06 | 3.28 × 10−1 | 0% | 0.88 (−0.32, 2.07) | No | Neither | NS |
| Male sex (coronary artery bypass) [28] | Prospective | Male vs. female | 18 | 2403 | 1069 | Odds ratio | 0.01 (−0.11, 0.14) | −0.12, 0.15 | 8.25 × 10−1 | 0% | 0.65 (−1.12, 2.43) | No | Neither | NS |
| Obesity [35] | Prospective | Yes vs. no | 3 | 696 | 164 | Odds ratio | 0.33 (−0.16, 0.81) | −5.03, 5.68 | 1.85 × 10−1 | 78% | 1.76 (0.19, 3.33) | No | Excess significance | NS |
| One kg higher body weight (cardiac) [35] | Prospective | Yes vs. no | 2 | 293 | 80 | Odds ratio | −0.11 (−0.51, 0.28) | NE | 5.67 × 10−1 | 49% | 0 (−0.06, 0.06) | NE | No excess significance * | NS |
| Peripheral vascular disease (coronary artery bypass) [28] | Prospective | Yes vs. no | 4 | 856 | 329 | Odds ratio | 0.04 (−0.49, 0.57) | −2, 2.09 | 8.69 × 10−1 | 57% | 0.81 (0.09, 1.53) | No | Excess significance bias | NS |
| Pre-operative symptoms (carotid endarterectomy) [34] | Prospective | Yes vs. no | 8 | 1196 | 596 | Odds ratio | 0.17 (−0.01, 0.35) | −0.06, 0.39 | 6.72 × 10−2 | 0% | 0.85 (0.02, 1.69) | No | Neither | IV |
| Previous MI <90 days (coronary artery bypass) [28] | Prospective | Yes vs. no | 3 | 418 | 152 | Odds ratio | 0.06 (−0.29, 0.42) | −2.26, 2.39 | 7.24 × 10−1 | 0% | 0.27 (−0.35, 0.88) | No | Neither | NS |
| Previous MI history (coronary artery bypass) [28] | Prospective | Yes vs. no | 7 | 1011 | 455 | Odds ratio | 0.08 (−0.11, 0.27) | −0.33, 0.49 | 3.93 × 10−1 | 25% | 0.33 (−0.12, 0.79) | No | Neither | NS |
| Previous stroke, TIA, CVA (coronary artery bypass) [28] | Prospective | Yes vs. no | 5 | 745 | 287 | Odds ratio | 0.49 (0.21, 0.77) | 0.04, 0.94 | 5.82 × 10−4 | 0% | 0.86 (0.35, 1.38) | No | Neither | IV |
| s100b [42] | Prospective, case-control | Continuous | 5 | 232 | 108 | Mean Difference | 0.27 (0.02, 0.53) | −0.62, 1.17 | 3.57 × 10−2 | 74% | 1.13 (0.69, 1.56) | No | Neither | IV |
| S100b (hip arthroplasty) [43] | Prospective | Continuous | 3 | 245 | 58 | Mean difference | 0.23 (0.1, 0.36) | −0.71, 1.17 | 5.96 × 10−4 | 5% | 0.31 (−0.03, 0.64) | No | Neither | IV |
| Smoking current/history (coronary artery bypass) [28] | Prospective | Yes vs. no | 9 | 1560 | 713 | Odds ratio | 0.02 (−0.25, 0.28) | −0.78, 0.82 | 8.94 × 10−1 | 68% | 1.49 (−0.28, 3.27) | No | Neither | NS |
| Statin use [40] | Prospective | Yes vs. no | 8 | 1804 | 445 | Odds ratio | −0.15 (−0.31, 0) | −0.54, 0.24 | 5.76 × 10−2 | 41% | 0.17 (−0.14, 0.48) | No | Neither | IV |
| Statin (carotid endarterectomy) [34] | Prospective | Yes vs. no | 3 | 1279 | 741 | Odds ratio | −0.31 (−0.49, −0.14) | −1.77, 1.14 | 4.31 × 10−4 | 21% | −0.18 (−0.56, 0.2) | No | Neither | IV |
| Stroke/TIA (transcatheter aortic valve implantation) [41] | Prospective | Yes vs. no | 2 | 340 | 35 | Odds ratio | 0.17 (−0.41, 0.74) | NE | 5.70 × 10−1 | 0% | 0.56 (−0.42, 1.54) | NE | No excess significance * | NS |
| Tumour necrosis factor alpha (hip arthroplasty) [43] | Prospective | Continuous | 5 | 412 | 127 | Mean difference | 0.17 (0.08, 0.27) | 0.02, 0.33 | 4.65 × 10−4 | 0% | 0.26 (0.03, 0.49) | NE | Neither | IV |
| Intraoperative | ||||||||||||||
| Aortic cross-clamping time (coronary artery bypass) [28] | Prospective | Continuous | 7 | 608 | 275 | Mean Difference | 0.13 (0.05, 0.21) | 0, 0.26 | 2.46 × 10−3 | 6% | 0.52 (0.19, 0.85) | No | Neither | IV |
| CPB time (coronary artery bypass) [28] | Prospective | Continuous | 13 | 1829 | 942 | Mean Difference | 0.1 (0.06, 0.15) | 0.05, 0.16 | 8.88 × 10−8 | 0% | 0.49 (0.11, 0.86) | Yes | Small-study effects | IV |
| Cross-clamping duration (carotid endarterectomy) [34] | Prospective | Continuous | 10 | 892 | 130 | Mean Difference | 0.1 (0.02, 0.19) | −0.09, 0.29 | 1.38 × 10−2 | 29% | 0.48 (0.14, 0.82) | Yes | Small-study effects | IV |
| Cerebral protection device (Transcatheter Aortic Valve Implantation) [41] | Prospective | Yes vs. no | 2 | 127 | 47 | Odds ratio | 0 (−0.41, 0.4) | NE | 9.91 × 10−1 | 0% | 0.18 (−0.37, 0.74) | NE | No excess significance * | NS |
| Hyperperfusion (carotid endarterectomy) [34] | Prospective | Yes vs. no | 5 | 417 | 52 | Odds ratio | 1.97 (1.55, 2.39) | 1.29, 2.65 | 4.18 × 10−20 | 0% | 2.91 (1.06, 4.75) | No | Neither | IV |
| Intubation time (coronary artery bypass) [28] | Prospective | Continuous | 6 | 1193 | 589 | Mean Difference | 0.41 (−0.29, 1.1) | −2.19, 3 | 2.5 × 10−1 | 99% | 1.7 (1.61, 1.8) | No | Neither | NS |
| Number of grafts (coronary artery bypass) [28] | Prospective | Continuous | 7 | 1113 | 536 | Mean Difference | 0.07 (0.01, 0.12) | −0.01, 0.14 | 2.96 × 10−2 | 0% | 0.26 (−0.03, 0.54) | Yes | Small-study effects | IV |
| Selective shunting placement (carotid endarterectomy) [34] | Prospective | Yes vs. no | 2 | 220 | 9 | Odds ratio | 0.7 (−0.07, 1.46) | NE | 7.62 × 10−2 | 0% | 0.76 (−0.43, 1.95) | NE | No excess significance * | NS |
| Surgery duration (coronary artery bypass) [28] | Prospective | Continuous | 6 | 727 | 261 | Mean Difference | 0.13 (0.06, 0.21) | 0.03, 0.24 | 3.174 × 10−4 | 0% | 0.24 (0.05, 0.43) | No | Neither | IV |
| Total microemboli (coronary artery bypass) [28] | Prospective | Continuous | 4 | 791 | 434 | Mean Difference | 0.09 (0.02, 0.15) | −0.07, 0.24 | 1.68 × 10−2 | 0% | 0.18 (−0.1, 0.47) | No | Neither | IV |
| Post-operative | ||||||||||||||
| Arrhythmia (coronary artery bypass) [28] | Prospective | Yes vs. no | 6 | 1045 | 487 | Odds ratio | 0.19 (0.01, 0.36) | −0.07, 0.44 | 4.22 × 10−2 | 0% | 0.65 (−0.02, 1.32) | No | Neither | IV |
| C-reactive protein (hip arthroplasty) [43] | Prospective | Continuous | 2 | 74 | 25 | Mean difference | 0.83 (−0.31, 1.96) | NE | 1.53 × 10−1 | 96% | 1.41 (1.1, 1.72) | No | Neither | NS |
| Delirium (coronary artery bypass) [28] | Prospective | Yes vs. no | 3 | 355 | 148 | Odds ratio | 1 (0.46, 1.54) | −2.92, 4.92 | 2.54 × 10−4 | 6% | 2.02 (0.39, 3.65) | No | Excess significance bias | IV |
| LOS in ICU (coronary artery bypass) [28] | Prospective | Continuous | 7 | 1059 | 547 | Mean Difference | 0.43 (−0.13, 1) | −1.64, 2.5 | 1.31 × 10−1 | 99% | 1.49 (1.4, 1.58) | No | Neither | NS |
| Interleukin 6 (hip arthroplasty) [43] | Prospective | Continuous | 3 | 131 | 60 | Mean difference | 0.52 (0.32, 0.72) | −1.18, 2.22 | 3.41 × 10−1 | 24% | 0.63 (0.31, 0.95) | No | Neither | NS |
| Interleukin 1β (hip arthroplasty) [43] | Prospective | Continuous | 3 | 131 | 60 | Mean difference | 0.12 (−0.1, 0.34) | −2.01, 2.25 | 2.99 × 10−1 | 39% | 0.37 (0.04, 0.71) | No | Neither | NS |
| Stroke (transcatheter aortic valve implantation) [41] | Prospective | Yes vs. no | 3 | 325 | 47 | Odds ratio | −0.35 (−1.24, 0.55) | −7.8, 7.1 | 4.48 × 10−1 | 22% | 0.3 (−1.51, 2.1) | No | Neither | NS |
| S100b (hip arthroplasty) [43] | Prospective | Continuous | 3 | 131 | 60 | Mean difference | 0.51 (0.3,0.72) | −1.38, 2.39 | 1.85 × 10−1 | 31% | 0.62 (0.3, 0.9) | No | Neither | NS |
| Tumour necrosis factor alpha (hip arthroplasty) [43] | Prospective | Continuous | 2 | 97 | 42 | Mean difference | 0.21 (0.01, 0.41) | NE | 3.97 × 10−2 | 0% | 0.3 (−0.02, 0.62) | NE | No excess significance * | IV |
| Outcome | Evidence Class | Study, n | Participants, n | Effect Size (95% CI) | p Value | I2 |
|---|---|---|---|---|---|---|
| Pre-operative factors | ||||||
| Age (coronary artery bypass) | III | 22 | 2881 | 0.27 (0.14, 0.41) | 9.47 × 10−5 | 92% |
| Age (carotid endarterectomy) | IV | 10 | 884 | 0.1 (0.03, 0.17) | 3.19 × 10−3 | 0% |
| Cognition: All tests (coronary artery bypass) | IV | 3 | 155 | 0.2 (0.04, 0.36) | 1.26 × 10−2 | 0% |
| Cognition: MMSE (coronary artery bypass) | IV | 2 | 120 | 0.23 (0.05, 0.41) | 1.19 × 10−2 | 0% |
| C-reactive protein (hip arthroplasty) | IV | 8 | 744 | 0.23 (0.11, 0.35) | 2.01 × 10−4 | 55% |
| Depression (coronary artery bypass) | IV | 2 | 330 | 0.68 (0.06, 1.3) | 3.12 × 10−2 | 62% |
| Diabetes | IV | 13 | 2554 | 0.16 (0.01, 0.32) | 4.13 × 10−2 | 71% |
| Diabetes (coronary artery bypass) | III | 17 | 2968 | 0.2 (0.1, 0.3) | 4.63 × 10−5 | 5% |
| Education | IV | 8 | 2535 | −0.06 (−0.09, −0.03) | 3.74 × 10−5 | 44% |
| Education (coronary artery bypass) | IV | 6 | 538 | 0.14 (0.05, 0.22) | 1.67 × 10−3 | 0% |
| Euroscore (coronary artery bypass) | IV | 4 | 582 | 0.23 (0.14, 0.31) | 2.09 × 10−7 | 10% |
| Hypertension (coronary artery bypass) | IV | 15 | 2115 | 0.36 (0.21, 0.51) | 4.91 × 10−6 | 34% |
| Interleukin 1β (hip arthroplasty) | IV | 5 | 247 | 0.19 (0.04, 0.34) | 1.14 × 10−2 | 26% |
| Interleukin 6 | IV | 16 | 986 | 0.15 (0.08, 0.22) | 2.43 × 10−5 | 17% |
| Interleukin 6 (hip arthroplasty) | IV | 6 | 699 | 0.1 (0.03, 0.17) | 7.78 × 10−3 | 0% |
| LVEF% (coronary artery bypass) | IV | 9 | 1225 | 0.14 (0.04, 0.24) | 4.72 × 10−3 | 62% |
| Previous stroke, TIA, CVA (coronary artery bypass) | IV | 5 | 745 | 0.49 (0.21, 0.77) | 5.82 × 10−4 | 0% |
| S100b | IV | 5 | 232 | 0.27 (0.02, 0.53) | 3.57 × 10−2 | 74% |
| S100b (hip arthroplasty) | IV | 3 | 245 | 0.23 (0.1, 0.36) | 5.96 × 10−4 | 5% |
| Statin (carotid endarterectomy) | IV | 3 | 1279 | −0.31 (−0.49, −0.14) | 4.31 × 10−4 | 21% |
| Tumour necrosis factor alpha (hip arthroplasty) | IV | 5 | 412 | 0.17 (0.08, 0.27) | 4.65 × 10−4 | 0% |
| Intra-operative factors | ||||||
| Aortic cross-clamping time (coronary artery bypass) | IV | 7 | 608 | 0.13 (0.05, 0.21) | 2.46 × 10−3 | 6% |
| CPB time (coronary artery bypass) | IV | 13 | 1829 | 0.1 (0.06, 0.15) | 8.88 × 10−8 | 0% |
| Cross-clamping duration (carotid endarterectomy) | IV | 10 | 893 | 0.1 (0.02, 0.19) | 1.38 × 10−2 | 29% |
| Hyperperfusion (carotid endarterectomy) | IV | 5 | 417 | 1.97 (1.55, 2.39) | 4.18 × 10−20 | 0% |
| Number of grafts (coronary artery bypass) | IV | 7 | 1113 | 0.07 (0.01, 0.12) | 2.96 × 10−2 | 0% |
| Surgery duration (coronary artery bypass) | IV | 6 | 727 | 0.13 (0.06, 0.21) | 3.17 × 10−4 | 0% |
| Total microemboli (coronary artery bypass) | IV | 4 | 791 | 0.09 (0.02, 0.15) | 1.68 × 10−2 | 0% |
| Post-operative factors | ||||||
| Arrhythmia (coronary artery bypass) | IV | 6 | 1045 | 0.19 (0.01, 0.36) | 4.22 × 10−2 | 0% |
| Delirium (coronary artery bypass) | IV | 3 | 355 | 1 (0.46, 1.54) | 2.54 × 10−4 | 6% |
| Tumour necrosis factor alpha (hip arthroplasty) | IV | 2 | 97 | 0.21 (0.01, 0.41) | 3.97 × 10−2 | 0% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Travica, N.; Lotfaliany, M.; Marriott, A.; Safavynia, S.A.; Lane, M.M.; Gray, L.; Veronese, N.; Berk, M.; Skvarc, D.; Aslam, H.; et al. Peri-Operative Risk Factors Associated with Post-Operative Cognitive Dysfunction (POCD): An Umbrella Review of Meta-Analyses of Observational Studies. J. Clin. Med. 2023, 12, 1610. https://doi.org/10.3390/jcm12041610
Travica N, Lotfaliany M, Marriott A, Safavynia SA, Lane MM, Gray L, Veronese N, Berk M, Skvarc D, Aslam H, et al. Peri-Operative Risk Factors Associated with Post-Operative Cognitive Dysfunction (POCD): An Umbrella Review of Meta-Analyses of Observational Studies. Journal of Clinical Medicine. 2023; 12(4):1610. https://doi.org/10.3390/jcm12041610
Chicago/Turabian StyleTravica, Nikolaj, Mojtaba Lotfaliany, Andrew Marriott, Seyed A. Safavynia, Melissa M. Lane, Laura Gray, Nicola Veronese, Michael Berk, David Skvarc, Hajara Aslam, and et al. 2023. "Peri-Operative Risk Factors Associated with Post-Operative Cognitive Dysfunction (POCD): An Umbrella Review of Meta-Analyses of Observational Studies" Journal of Clinical Medicine 12, no. 4: 1610. https://doi.org/10.3390/jcm12041610
APA StyleTravica, N., Lotfaliany, M., Marriott, A., Safavynia, S. A., Lane, M. M., Gray, L., Veronese, N., Berk, M., Skvarc, D., Aslam, H., Gamage, E., Formica, M., Bishop, K., & Marx, W. (2023). Peri-Operative Risk Factors Associated with Post-Operative Cognitive Dysfunction (POCD): An Umbrella Review of Meta-Analyses of Observational Studies. Journal of Clinical Medicine, 12(4), 1610. https://doi.org/10.3390/jcm12041610






