Post-Surgical Imaging Assessment in Rectal Cancer: Normal Findings and Complications
Abstract
1. Introduction
2. Surgical Options
2.1. Radical Approach
2.2. Local Excision
2.3. Watch and Wait Strategy
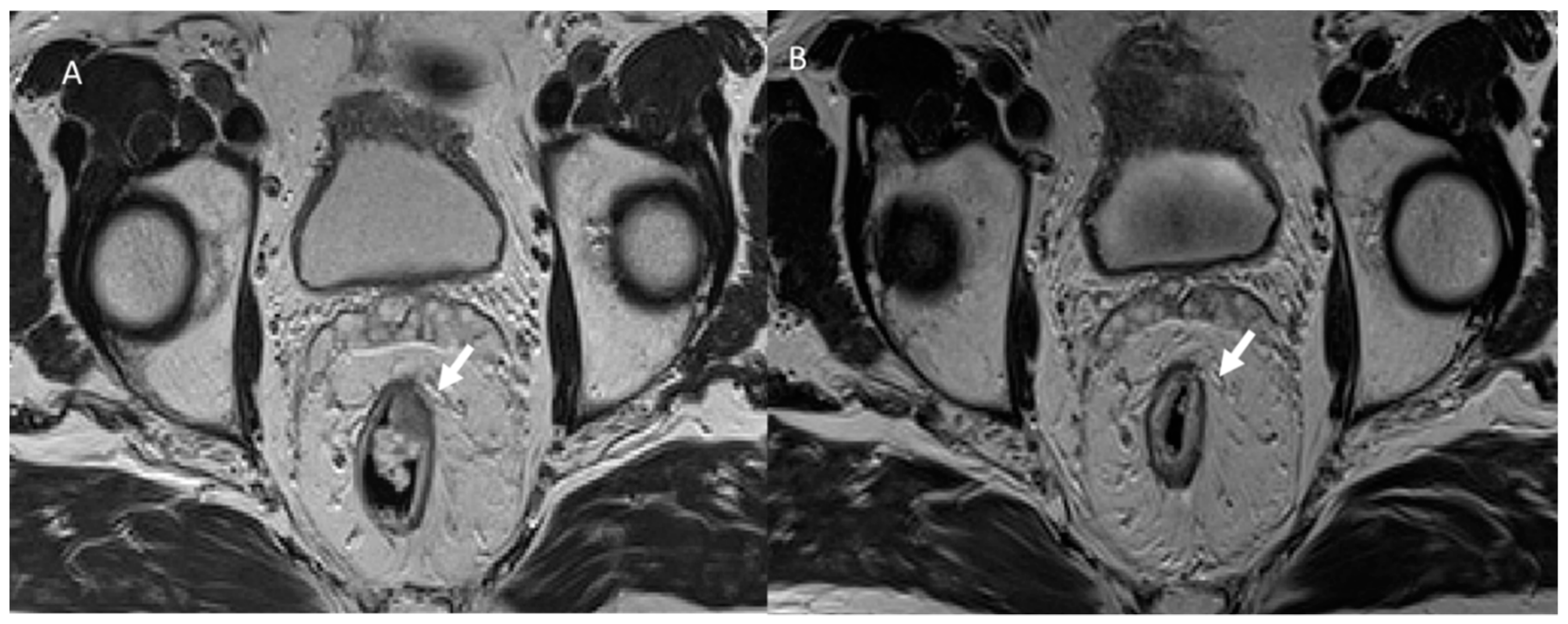
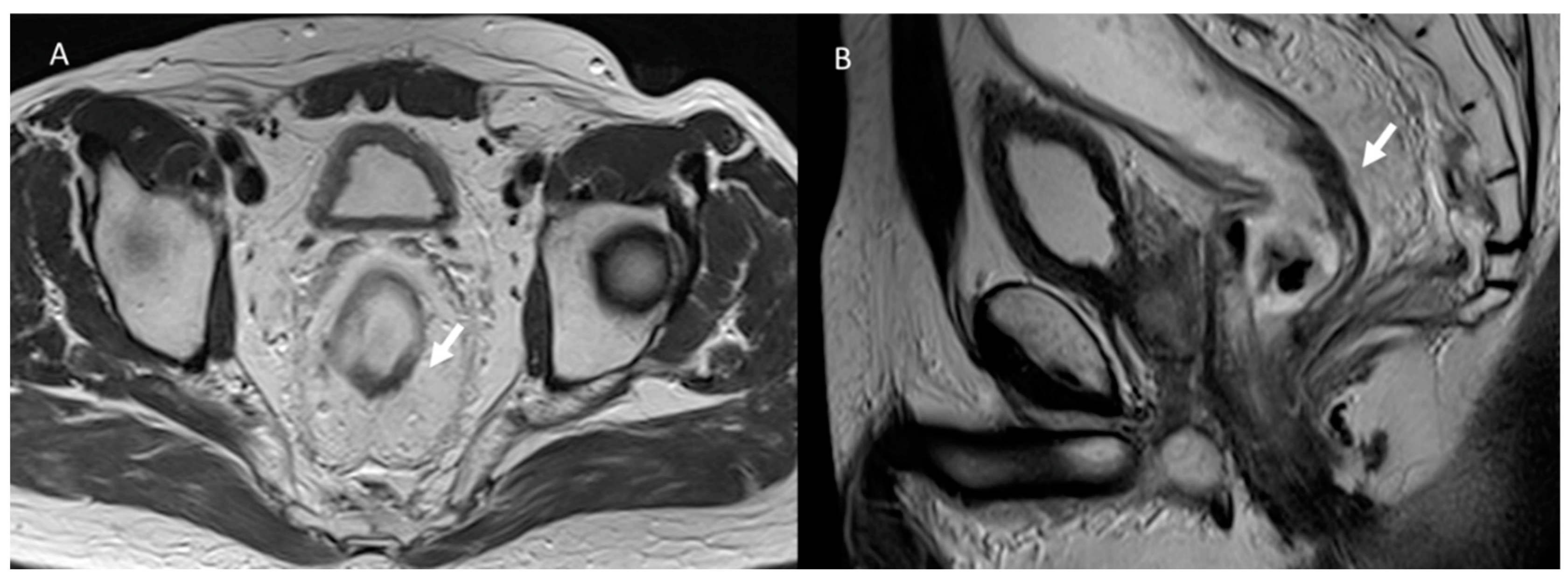
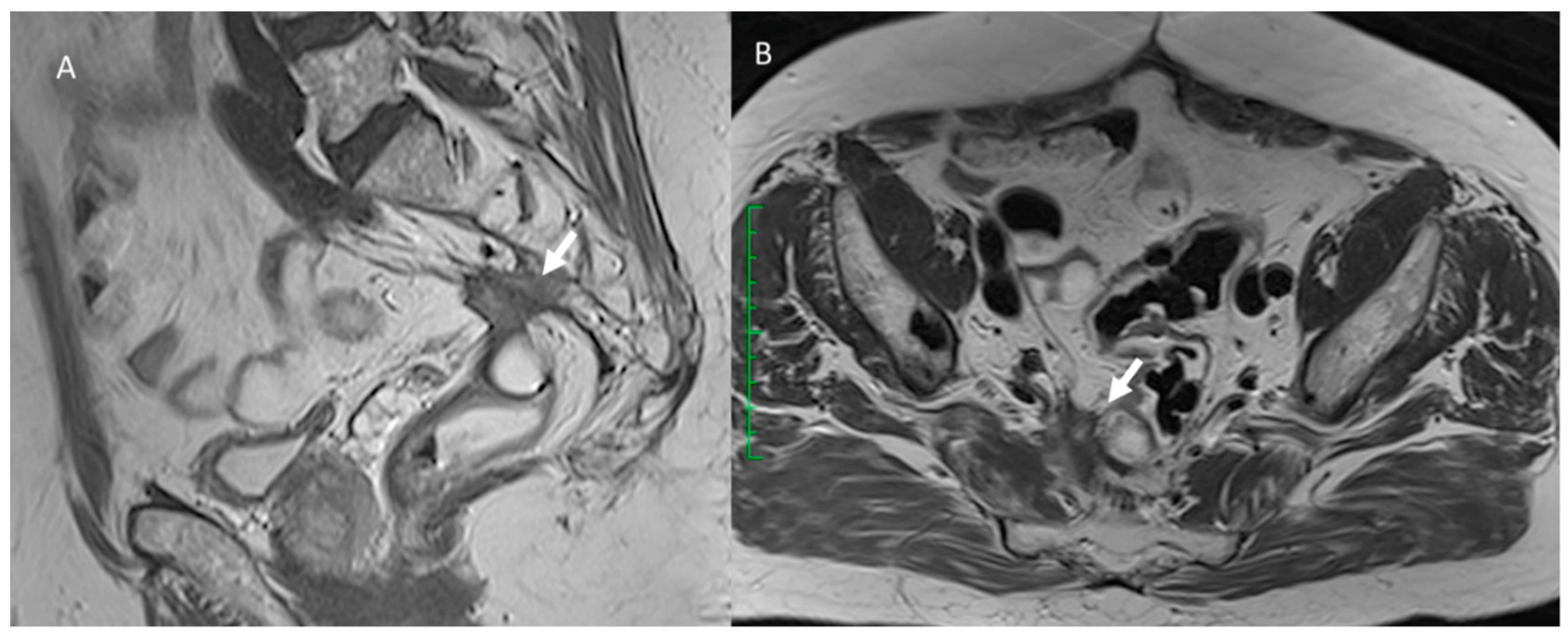
3. Common Postoperative Complications
3.1. Anastomotic Leak
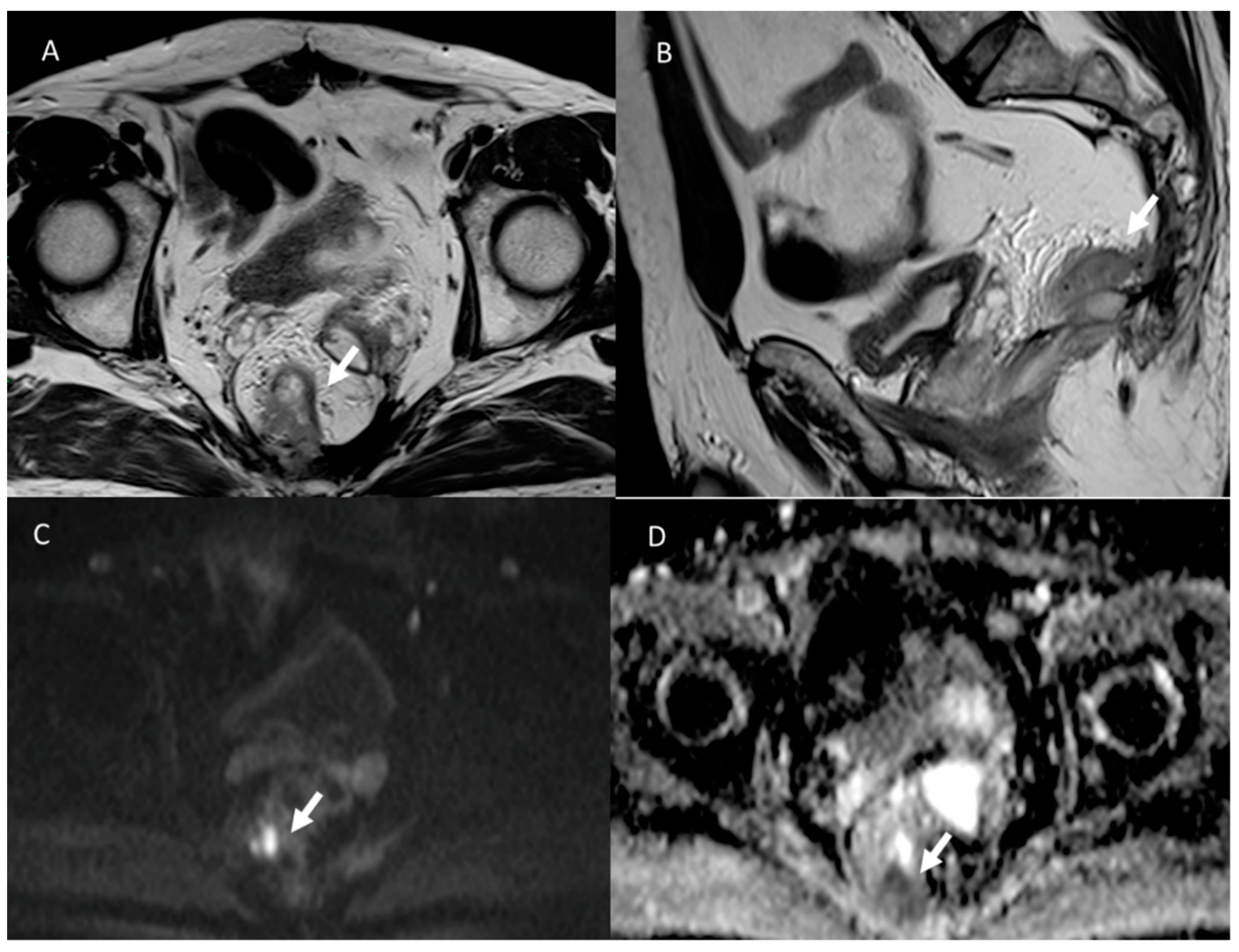
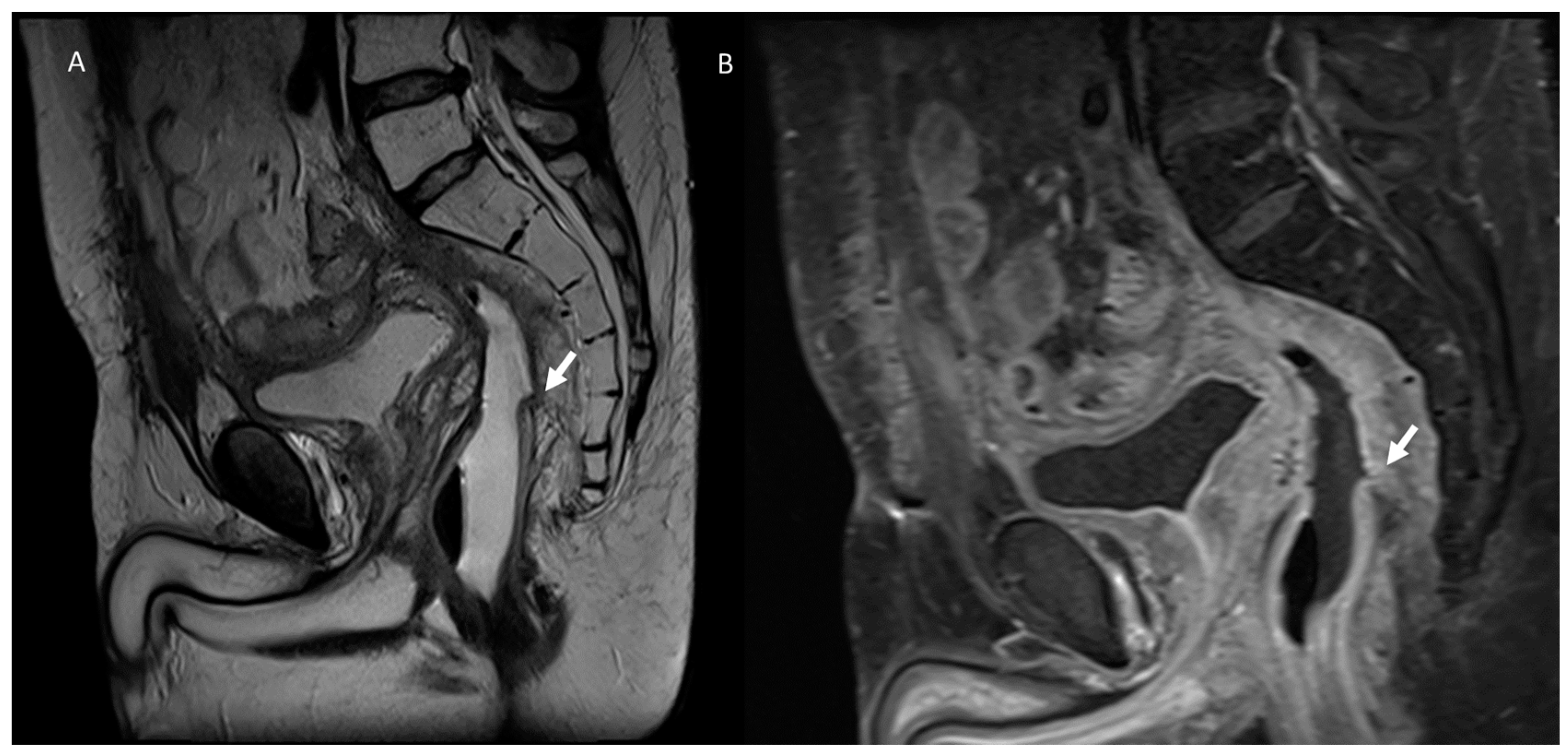
3.2. Fistula
3.3. Bleeding
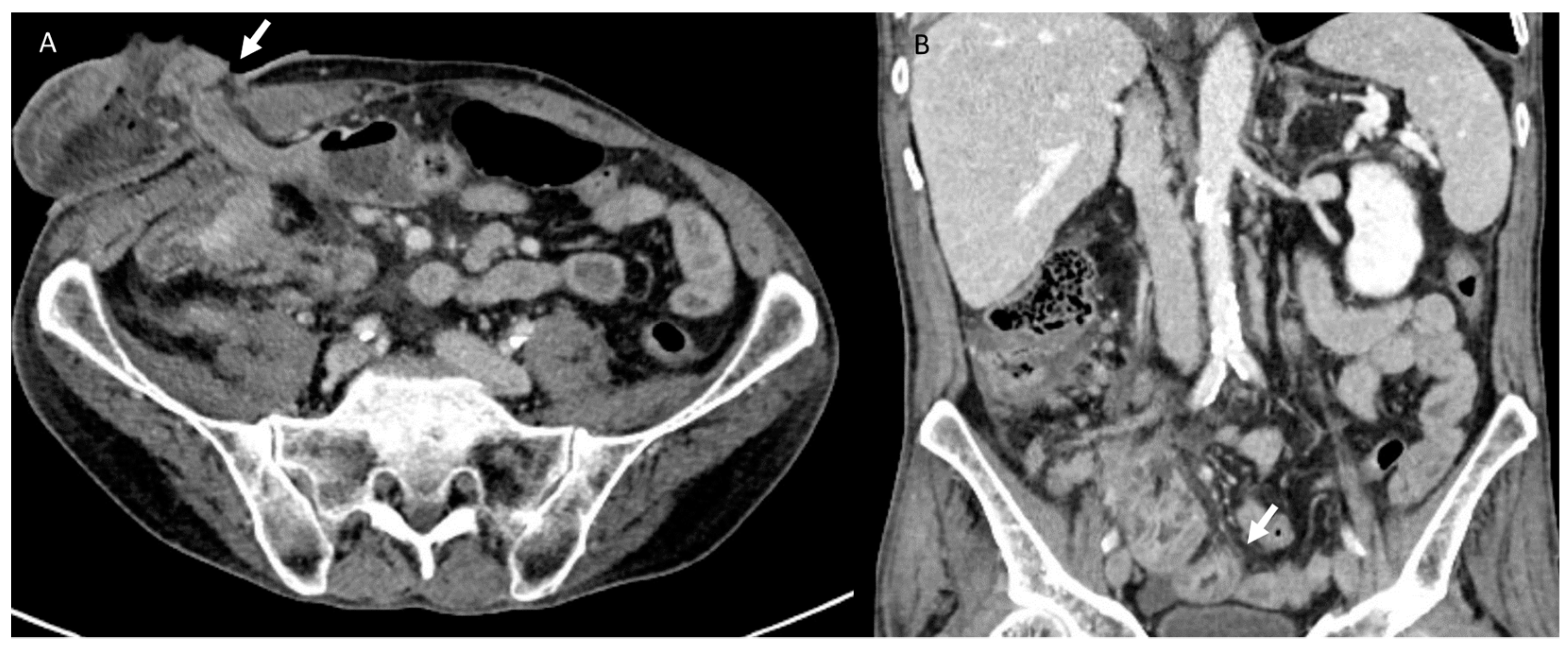
3.4. Urological Injury
3.5. Wound Infections
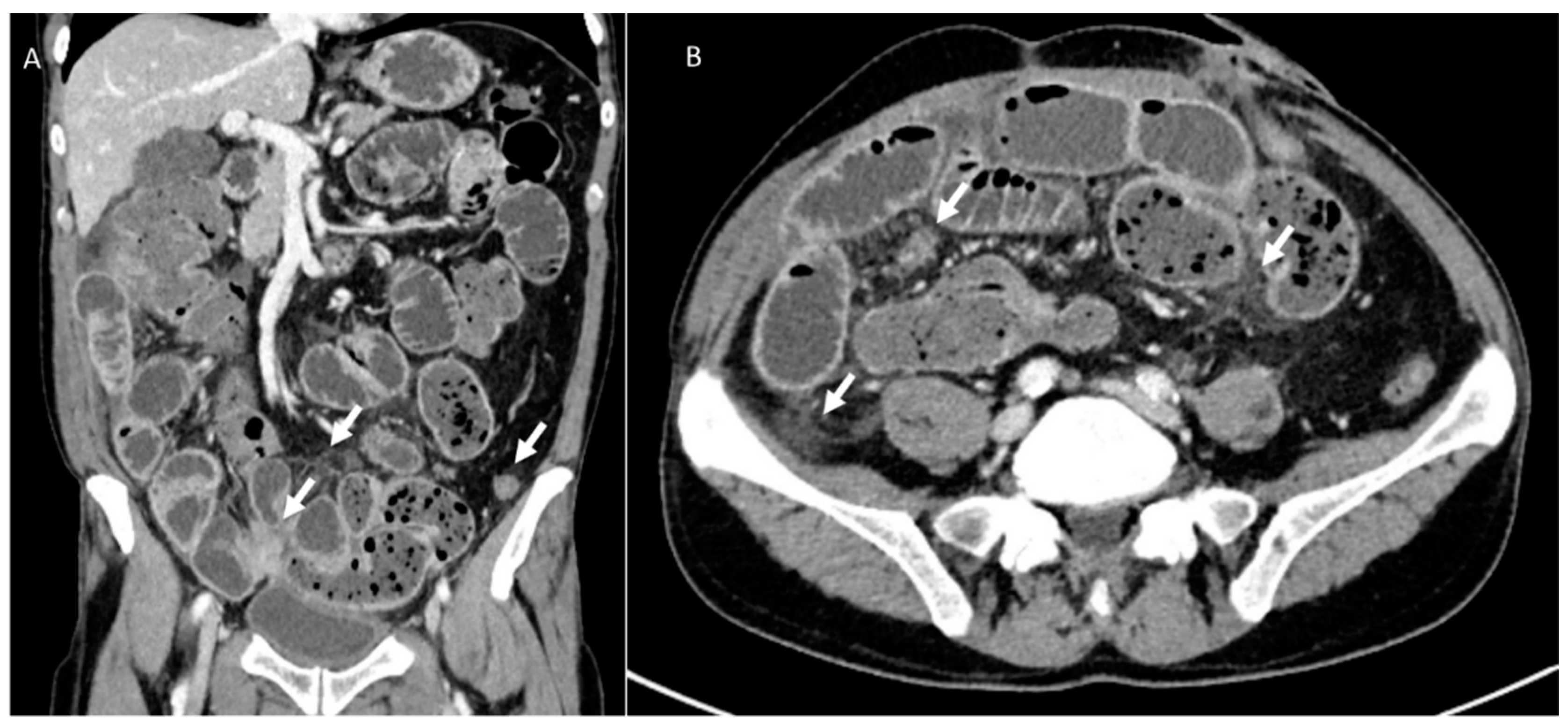
3.6. Hernia
3.7. Local Recurrence
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Pallan, A.; Dedelaite, M.; Mirajkar, N.; Newman, P.A.; Plowright, J.; Ashraf, S. Postoperative complications of colorectal cancer. Clin. Radiol. 2021, 76, 896–907. [Google Scholar] [CrossRef] [PubMed]
- Gamboa, A.C.; Lee, R.M.; Turgeon, M.K.; Varlamos, C.; Regenbogen, S.E.; Hrebinko, K.A.; Holder-Murray, J.; Wiseman, J.T.; Ejaz, A.; Feng, M.P.; et al. Impact of Postoperative Complications on Oncologic Outcomes after Rectal Cancer Surgery: An Analysis of the US Rectal Cancer Consortium. Ann. Surg. Oncol. 2021, 28, 1712–1721. [Google Scholar] [CrossRef]
- Merlotti, A.; Bruni, A.; Borghetti, P.; Ramella, S.; Scotti, V.; Trovò, M.; Chiari, R.; Lohr, F.; Ricardi, U.; Bria, E.; et al. Sequential chemo-hypofractionated RT versus concurrent standard CRT for locally advanced NSCLC: GRADE recommendation by the Italian Association of Radiotherapy and Clinical Oncology (AIRO). Radiol. Med. 2021, 126, 1117–1128. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Kim, Y.S.; Choi, J. Dosimetric analysis of the effects of a temporary tissue expander on the radiotherapy technique. Radiol. Med. 2021, 126, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Falcinelli, L.; Mendichi, M.; Chierchini, S.; Tenti, M.V.; Bellavita, R.; Saldi, S.; Ingrosso, G.; Reggioli, V.; Bini, V.; Aristei, C. Pulmonary function in stereotactic body radiotherapy with helical tomotherapy for primary and metastatic lung lesions. Radiol. Med. 2021, 126, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Arslan, A.; Aktas, E.; Sengul, B.; Tekin, B. Dosimetric evaluation of left ventricle and left anterior descending artery in left breast radiotherapy. Radiol. Med. 2021, 126, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Barra, S.; Guarnieri, A.; di Monale E Bastia, M.B.; Marcenaro, M.; Tornari, E.; Belgioia, L.; Magrini, S.M.; Ricardi, U.; Corvò, R. Short fractionation radiotherapy for early prostate cancer in the time of COVID-19: Long-term excellent outcomes from a multicenter Italian trial suggest a larger adoption in clinical practice. Radiol. Med. 2021, 126, 142–146. [Google Scholar] [CrossRef]
- Lancellotta, V.; Del Regno, L.; Di Stefani, A.; Fionda, B.; Marazzi, F.; Rossi, E.; Balducci, M.; Pampena, R.; Morganti, A.G.; Peris, K.; et al. The role of stereotactic radiotherapy in addition to immunotherapy in the management of melanoma brain metastases: Results of a systematic review. Radiol. Med. 2022, 127, 773–783. [Google Scholar] [CrossRef] [PubMed]
- Tamburini, E.; Tassinari, D.; Ramundo, M.; De Stefano, A.; Viola, M.G.; Romano, C.; Elia, M.T.; Zanaletti, N.; Rudnas, B.; Casadei-Gardini, A.; et al. Adjuvant chemotherapy after neoadjuvant chemo-radiotherapy and surgery in locally advanced rectal cancer. A systematic review of literature with a meta-analysis of randomized clinical trials. Crit. Rev. Oncol. Hematol. 2022, 172, 103627. [Google Scholar] [CrossRef]
- Cellini, F.; Di Franco, R.; Manfrida, S.; Borzillo, V.; Maranzano, E.; Pergolizzi, S.; Morganti, A.G.; Fusco, V.; Deodato, F.; Santarelli, M.; et al. Palliative radiotherapy indications during the COVID-19 pandemic and in future complex logistic settings: The NORMALITY model. Radiol. Med. 2021, 126, 1619–1656. [Google Scholar] [CrossRef] [PubMed]
- Rega, D.; Granata, V.; Romano, C.; D’Angelo, V.; Pace, U.; Fusco, R.; Cervone, C.; Ravo, V.; Tatangelo, F.; Avallone, A.; et al. Watch and Wait Approach for Rectal Cancer Following Neoadjuvant Treatment: The Experience of a High Volume Cancer Center. Diagnostics 2021, 11, 1507. [Google Scholar] [CrossRef]
- Grosek, J.; Košir, J.A.; Novak, J.; Omejc, M.; Tomažič, A.; Norčič, G. Validation of The Slovenian Version of the Low Anterior Resection Syndrome Score for Rectal Cancer Patients after Surgery. Slov. J. Public Health 2019, 58, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Salvestrini, V.; Becherini, C.; Desideri, I.; Caprara, L.; Mariotti, M.; Banini, M.; Pierossi, N.; Scotti, V.; Livi, L.; Bonomo, P. The impact of patient preference in the treatment algorithm for recurrent/metastatic head and neck squamous cell carcinoma. Radiol. Med. 2022, 127, 866–871. [Google Scholar] [CrossRef] [PubMed]
- Lancellotta, V.; Fanetti, G.; Monari, F.; Mangoni, M.; Mazzarotto, R.; Tagliaferri, L.; Gobitti, C.; Lodi Rizzini, E.; Talomo, S.; Turturici, I.; et al. Stereotactic radiotherapy (SRT) for differentiated thyroid cancer (DTC) oligometastases: An AIRO (Italian association of radiotherapy and clinical oncology) systematic review. Radiol. Med. 2022, 127, 681–689. [Google Scholar] [CrossRef]
- Francolini, G.; Jereczek-Fossa, B.A.; Di Cataldo, V.; Simontacchi, G.; Marvaso, G.; Gandini, S.; Corso, F.; Ciccone, L.P.; Zerella, M.A.; Gentile, P.; et al. Stereotactic or conventional radiotherapy for macroscopic prostate bed recurrence: A propensity score analysis. Radiol. Med. 2022, 127, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Parisi, S.; Ferini, G.; Cacciola, A.; Lillo, S.; Tamburella, C.; Santacaterina, A.; Bottari, A.; Brogna, A.; Ferrantelli, G.; Pontoriero, A.; et al. A non-surgical COMBO-therapy approach for locally advanced unresectable pancreatic adenocarcinoma: Preliminary results of a prospective study. Radiol. Med. 2022, 127, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Young, D.O.; Kumar, A.S. Local Excision of Rectal Cancer. Surg. Clin. North Am. 2017, 97, 573–585. [Google Scholar] [CrossRef]
- Granata, V.; Grassi, R.; Fusco, R.; Izzo, F.; Brunese, L.; Delrio, P.; Avallone, A.; Pecori, B.; Petrillo, A. Current status on response to treatment in locally advanced rectal cancer: What the radiologist should know. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 12050–12062. [Google Scholar] [CrossRef] [PubMed]
- Scialpi, M.; Moschini, T.O.; De Filippis, G. PET/contrast-enhanced CT in oncology: “to do, or not to do, that is the question”. Radiol. Med. 2022, 127, 925–927. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Fusco, R.; de Lutio di Castelguidone, E.; Avallone, A.; Palaia, R.; Delrio, P.; Tatangelo, F.; Botti, G.; Grassi, R.; Izzo, F.; et al. Diagnostic performance of gadoxetic acid-enhanced liver MRI versus multidetector CT in the assessment of colorectal liver metastases compared to hepatic resection. BMC Gastroenterol. 2019, 19, 129. [Google Scholar] [CrossRef]
- Masci, G.M.; Ciccarelli, F.; Mattei, F.I.; Grasso, D.; Accarpio, F.; Catalano, C.; Laghi, A.; Sammartino, P.; Iafrate, F. Role of CT texture analysis for predicting peritoneal metastases in patients with gastric cancer. Radiol. Med. 2022, 127, 251–258. [Google Scholar] [CrossRef]
- Boyle, K.M.; Sagar, P.M.; Chalmers, A.G.; Sebag-Montefiore, D.; Cairns, A.; Eardley, I. Surgery for locally recurrent rectal cancer. Dis. Colon Rectum 2005, 48, 929–937. [Google Scholar] [CrossRef]
- Granata, V.; Caruso, D.; Grassi, R.; Cappabianca, S.; Reginelli, A.; Rizzati, R.; Masselli, G.; Golfieri, R.; Rengo, M.; Regge, D.; et al. Structured Reporting of Rectal Cancer Staging and Restaging: A Consensus Proposal. Cancers 2021, 13, 2135. [Google Scholar] [CrossRef]
- Sinaei, M.; Swallow, C.; Milot, L.; Moghaddam, P.A.; Smith, A.; Atri, M. Patterns and signal intensity characteristics of pelvic recurrence of rectal cancer at MR imaging. Radiographics 2013, 33, E171–E187. [Google Scholar] [CrossRef] [PubMed]
- Keller, D.S.; Berho, M.; Perez, R.O.; Wexner, S.D.; Chand, M. The multidisciplinary management of rectal cancer. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 414–429. [Google Scholar] [CrossRef]
- Glynne-Jones, R.; Wyrwicz, L.; Tiret, E.; Brown, G.; Rödel, C.; Cervantes, A.; Arnold, D. Rectal cancer: ESMO Guidelines Committee. Rectal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2017, 28 (Suppl. 4), iv22–iv40, Erratum in Ann. Oncol. 2018, 29 (Suppl. S4), iv263. [Google Scholar] [CrossRef]
- Wei, R.; Crook, C.; Bamford, R. Abdominoperineal Resection. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Monson, J.R.; Weiser, M.R.; Buie, W.D.; Chang, G.J.; Rafferty, J.F.; Buie, W.D.; Rafferty, J. Standards Practice Task Force of the American Society of Colon and Rectal Surgeons. Practice parameters for the management of rectal cancer (revised). Dis. Colon Rectum 2013, 56, 535–550. [Google Scholar] [CrossRef]
- You, Y.N.; Hardiman, K.M.; Bafford, A.; Poylin, V.; Francone, T.D.; Davis, K.; Paquette, I.M.; Steele, S.R.; Feingold, D.L.; On Behalf of the Clinical Practice Guidelines Committee of the American Society of Colon and Rectal Surgeons. The American Society of Colon and Rectal Surgeons Clinical Practice Guidelines for the Management of Rectal Cancer. Dis. Colon Rectum 2020, 63, 1191–1222. [Google Scholar] [CrossRef]
- Weinstein, S.; Osei-Bonsu, S.; Aslam, R.; Yee, J. Multidetector CT of the postoperative colon: Review of normal appearances and common complications. Radiographics 2013, 33, 515–532. [Google Scholar] [CrossRef]
- Emile, S.H.; de Lacy, F.B.; Keller, D.S.; Martin-Perez, B.; Alrawi, S.; Lacy, A.M.; Chand, M. Evolution of transanal total mesorectal excision for rectal cancer: From top to bottom. World J. Gastrointest. Surg. 2018, 10, 28–39. [Google Scholar] [CrossRef]
- Penna, M.; Hompes, R.; Arnold, S.; Wynn, G.; Austin, R.; Warusavitarne, J.; Moran, B.; Hanna, G.B.; Mortensen, N.J.; Tekkis, P.P.; et al. Transanal Total Mesorectal Excision: International Registry Results of the First 720 Cases. Ann. Surg. 2017, 266, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, N. Management of Rectal Cancer. Surg. Clin. North Am. 2020, 100, 615–628. [Google Scholar] [CrossRef]
- Avallone, A.; Pecori, B.; Bianco, F.; Aloj, L.; Tatangelo, F.; Romano, C.; Granata, V.; Marone, P.; Leone, A.; Botti, G.; et al. Critical role of bevacizumab scheduling in combination with pre-surgical chemo-radiotherapy in MRI-defined high-risk locally advanced rectal cancer: Results of the BRANCH trial. Oncotarget 2015, 6, 30394–30407. [Google Scholar] [CrossRef]
- Avallone, A.; Piccirillo, M.C.; Nasti, G.; Rosati, G.; Carlomagno, C.; Di Gennaro, E.; Romano, C.; Tatangelo, F.; Granata, V.; Cassata, A.; et al. Effect of Bevacizumab in Combination With Standard Oxaliplatin-Based Regimens in Patients With Metastatic Colorectal Cancer: A Randomized Clinical Trial. JAMA Netw. Open 2021, 4, e2118475. [Google Scholar] [CrossRef] [PubMed]
- Clancy, C.; Burke, J.P.; Albert, M.R.; O’Connell, P.R.; Winter, D.C. Transanal endoscopic microsurgery versus standard transanal excision for the removal of rectal neoplasms: A systematic review and meta-analysis. Dis. Colon Rectum 2015, 58, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Arezzo, A.; Passera, R.; Saito, Y.; Sakamoto, T.; Kobayashi, N.; Sakamoto, N.; Yoshida, N.; Naito, Y.; Fujishiro, M.; Niimi, K.; et al. Systematic review and meta-analysis of endoscopic submucosal dissection versus transanal endoscopic microsurgery for large noninvasive rectal lesions. Surg. Endosc. 2014, 28, 427–438. [Google Scholar] [CrossRef]
- Bedrikovetski, S.; Fitzsimmons, T.; Perry, J.; Vather, R.; Carruthers, S.; Selva-Nayagam, S.; Thomas, M.L.; Moore, J.W.; Sammour, T. Personalized total neoadjuvant therapy (pTNT) for advanced rectal cancer with tailored treatment sequencing based on clinical stage at presentation. ANZ J. Surg. 2022. ahead of print. [Google Scholar] [CrossRef]
- Habr-Gama, A.; Perez, R.O.; Nadalin, W.; Sabbaga, J.; Ribeiro, U., Jr.; Silva e Sousa, A.H., Jr.; Campos, F.G.; Kiss, D.R.; Gama-Rodrigues, J. Operative versus nonoperative treatment for stage 0 distal rectal cancer following chemoradiation therapy: Long-term results. Ann. Surg. 2004, 240, 711–717. [Google Scholar] [CrossRef]
- Habr-Gama, A.; Perez, R.O.; Wynn, G.; Marks, J.; Kessler, H.; Gama-Rodrigues, J. Complete clinical response after neoadjuvant chemoradiation therapy for distal rectal cancer: Characterization of clinical and endoscopic findings for standardization. Dis. Colon Rectum 2010, 53, 1692–1698. [Google Scholar] [CrossRef]
- Habr-Gama, A.; São Julião, G.P.; Vailati, B.B.; Sabbaga, J.; Aguilar, P.B.; Fernandez, L.M.; Araújo, S.E.A.; Perez, R.O. Organ Preservation in cT2N0 Rectal Cancer After Neoadjuvant Chemoradiation Therapy: The Impact of Radiation Therapy Dose-escalation and Consolidation Chemotherapy. Ann. Surg. 2019, 269, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Chadi, S.A.; Malcomson, L.; Ensor, J.; Riley, R.D.; Vaccaro, C.A.; Rossi, G.L.; Daniels, I.R.; Smart, N.J.; Osborne, M.E.; Beets, G.L.; et al. Factors affecting local regrowth after watch and wait for patients with a clinical complete response following chemoradiotherapy in rectal cancer (InterCoRe consortium): An individual participant data meta-analysis. Lancet Gastroenterol. Hepatol. 2018, 3, 825–836. [Google Scholar] [CrossRef]
- Dossa, F.; Chesney, T.R.; Acuna, S.A.; Baxter, N.N. A watch-and-wait approach for locally advanced rectal cancer after a clinical complete response following neoadjuvant chemoradiation: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2017, 2, 501–513. [Google Scholar] [CrossRef] [PubMed]
- van der Valk, M.J.M.; Hilling, D.E.; Bastiaannet, E.; Meershoek-Klein Kranenbarg, E.; Beets, G.L.; Figueiredo, N.L.; Habr-Gama, A.; Perez, R.O.; Renehan, A.G.; IWWD Consortium. Long-term outcomes of clinical complete responders after neoadjuvant treatment for rectal cancer in the International Watch & Wait Database (IWWD): An international multicentre registry study. Lancet 2018, 391, 2537–2545. [Google Scholar] [CrossRef] [PubMed]
- Ganeshan, D.; Nougaret, S.; Korngold, E.; Rauch, G.M.; Moreno, C.C. Locally recurrent rectal cancer: What the radiologist should know. Abdom. Radiol. 2019, 44, 3709–3725. [Google Scholar] [CrossRef]
- Cutolo, C.; Dell’Aversana, F.; Fusco, R.; Grazzini, G.; Chiti, G.; Simonetti, I.; Bruno, F.; Palumbo, P.; Pierpaoli, L.; Valeri, T.; et al. Combined Hepatocellular-Cholangiocarcinoma: What the Multidisciplinary Team Should Know. Diagnostics 2022, 12, 890. [Google Scholar] [CrossRef]
- McDermott, F.D.; Heeney, A.; Kelly, M.E.; Steele, R.J.; Carlson, G.L.; Winter, D.C. Systematic review of preoperative, intraoperative and postoperative risk factors for colorectal anastomotic leaks. Br. J. Surg. 2015, 102, 462–479. [Google Scholar] [CrossRef]
- Hyman, N.; Manchester, T.L.; Osler, T.; Burns, B.; Cataldo, P.A. Anastomotic leaks after intestinal anastomosis: It’s later than you think. Ann. Surg. 2007, 245, 254–258. [Google Scholar] [CrossRef]
- van Helsdingen, C.P.; Jongen, A.C.; de Jonge, W.J.; Bouvy, N.D.; Derikx, J.P. Consensus on the definition of colorectal anastomotic leakage: A modified Delphi study. World J. Gastroenterol. 2020, 26, 3293–3303. [Google Scholar] [CrossRef] [PubMed]
- Rahbari, N.N.; Weitz, J.; Hohenberger, W.; Heald, R.J.; Moran, B.; Ulrich, A.; Holm, T.; Wong, W.D.; Tiret, E.; Moriya, Y.; et al. Definition and grading of anastomotic leakage following anterior resection of the rectum: A proposal by the International Study Group of Rectal Cancer. Surgery 2010, 147, 339–351. [Google Scholar] [CrossRef]
- Sparreboom, C.L.; van Groningen, J.T.; Lingsma, H.F.; Wouters, M.W.J.M.; Menon, A.G.; Kleinrensink, G.J.; Jeekel, J.; Lange, J.F.; Dutch ColoRectal Audit Group. Different Risk Factors for Early and Late Colorectal Anastomotic Leakage in a Nationwide Audit. Dis. Colon Rectum 2018, 61, 1258–1266. [Google Scholar] [CrossRef]
- Zhan, T.C.; Zhang, D.K.; Gu, J.; Li, M. Surgical complications after different therapeutic approaches for locally advanced rectal cancer. World J. Gastrointest. Oncol. 2019, 11, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Bertelsen, C.A.; Andreasen, A.H.; Jørgensen, T.; Harling, H.; Danish Colorectal Cancer Group. Anastomotic leakage after anterior resection for rectal cancer: Risk factors. Colorectal Dis. 2010, 12, 37–43. [Google Scholar] [CrossRef]
- Samji, K.B.; Kielar, A.Z.; Connolly, M.; Fasih, N.; Doherty, G.; Chung, A.; Hache, E. Anastomotic Leaks After Small- and Large-Bowel Surgery: Diagnostic Performance of CT and the Importance of Intraluminal Contrast Administration. AJR Am. J. Roentgenol. 2018, 210, 1259–1265. [Google Scholar] [CrossRef]
- Blumetti, J.; Abcarian, H. Management of low colorectal anastomotic leak: Preserving the anastomosis. World J. Gastrointest. Surg. 2015, 7, 378–383. [Google Scholar] [CrossRef]
- Li, N.; Wakim, J.; Koethe, Y.; Huber, T.; Schenning, R.; Gade, T.P.; Hunt, S.J.; Park, B.J. Multicenter assessment of augmented reality registration methods for image-guided interventions. Radiol. Med. 2022, 127, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Doeksen, A.; Tanis, P.J.; Wüst, A.F.; Vrouenraets, B.C.; van Lanschot, J.J.; van Tets, W.F. Radiological evaluation of colorectal anastomoses. Int. J. Colorectal Dis. 2008, 23, 863–868. [Google Scholar] [CrossRef] [PubMed]
- Pozzessere, C.; Boudiaf, M.; Cirigliano, A.; Dohan, A.; Mazzei, M.A.; Barat, M.; Volterrani, L.; Soyer, P. MR-enterography: Role in the assessment of suspected anastomotic recurrence of Crohn disease after ileocolic resection. Radiol. Med. 2022, 127, 238–250. [Google Scholar] [CrossRef] [PubMed]
- VanBuren, W.M.; Lightner, A.L.; Kim, S.T.; Sheedy, S.P.; Woolever, M.C.; Menias, C.O.; Fletcher, J.G. Imaging and Surgical Management of Anorectal Vaginal Fistulas. Radiographics 2018, 38, 1385–1401. [Google Scholar] [CrossRef]
- Hori, M.; Oto, A.; Orrin, S.; Suzuki, K.; Baron, R.L. Diffusion-weighted MRI: A new tool for the diagnosis of fistula in ano. J. Magn. Reson. Imaging 2009, 30, 1021–1026. [Google Scholar] [CrossRef] [PubMed]
- Tonolini, M. Elucidating vaginal fistulas on CT and MRI. Insights Imaging 2019, 10, 123. [Google Scholar] [CrossRef] [PubMed]
- Ha, R.K.; Han, K.S.; Park, S.S.; Sohn, D.K.; Hong, C.W.; Kim, B.C.; Kim, B. Efficacy and Safety of Endoscopic Clipping for Acute Anastomotic Bleeding After Colorectal Surgery. Ann. Coloproctol. 2022, 38, 262–265. [Google Scholar] [CrossRef] [PubMed]
- Iacobellis, F.; Brillantino, A.; Di Serafino, M.; Dell’Aversano Orabona, G.; Grassi, R.; Cappabianca, S.; Scaglione, M.; Romano, L. Economic and clinical benefits of immediate total-body CT in the diagnostic approach to polytraumatized patients: A descriptive analysis through a literature review. Radiol. Med. 2022, 127, 637–644. [Google Scholar] [CrossRef]
- Casal Núñez, J.E.; Vigorita, V.; Ruano Poblador, A.; Gay Fernández, A.M.; Toscano Novella, M.Á.; Cáceres Alvarado, N.; Pérez Dominguez, L. Presacral venous bleeding during mobilization in rectal cancer. World J. Gastroenterol. 2017, 23, 1712–1719. [Google Scholar] [CrossRef]
- Halabi, W.J.; Jafari, M.D.; Nguyen, V.Q.; Carmichael, J.C.; Mills, S.; Pigazzi, A.; Stamos, M.J. Ureteral injuries in colorectal surgery: An analysis of trends, outcomes, and risk factors over a 10-year period in the United States. Dis. Colon Rectum 2014, 57, 179–186. [Google Scholar] [CrossRef]
- Esparaz, A.M.; Pearl, J.A.; Herts, B.R.; LeBlanc, J.; Kapoor, B. Iatrogenic urinary tract injuries: Etiology, diagnosis, and management. Semin. Interv. Radiol. 2015, 32, 195–208. [Google Scholar] [CrossRef]
- Abboudi, H.; Ahmed, K.; Royle, J.; Khan, M.S.; Dasgupta, P.; N’Dow, J. Ureteric injury: A challenging condition to diagnose and manage. Nat. Rev. Urol. 2013, 10, 108–115. [Google Scholar] [CrossRef]
- Potenta, S.E.; D’Agostino, R.; Sternberg, K.M.; Tatsumi, K.; Perusse, K. CT Urography for Evaluation of the Ureter. Radiographics 2015, 35, 709–726. [Google Scholar] [CrossRef]
- Ascenti, G.; Cicero, G.; Bertelli, E.; Papa, M.; Gentili, F.; Ciccone, V.; Manetta, R.; Gandolfo, N.; Cardone, G.; Miele, V. CT-urography: A nationwide survey by the Italian Board of Urogenital Radiology. Radiol. Med. 2022, 127, 577–588. [Google Scholar] [CrossRef]
- Tamura, K.; Matsuda, K.; Yokoyama, S.; Iwamoto, H.; Mizumoto, Y.; Murakami, D.; Nakamura, Y.; Yamaue, H. Defunctioning loop ileostomy for rectal anastomoses: Predictors of stoma outlet obstruction. Int. J. Colorectal Dis. 2019, 34, 1141–1145. [Google Scholar] [CrossRef]
- Malik, T.; Lee, M.J.; Harikrishnan, A.B. The incidence of stoma related morbidity—A systematic review of randomised controlled trials. Ann. R. Coll. Surg. Engl. 2018, 100, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Shabbir, J.; Britton, D.C. Stoma complications: A literature overview. Colorectal Dis. 2010, 12, 958–964. [Google Scholar] [CrossRef] [PubMed]
- Toh, J.W.; Lim, R.; Keshava, A.; Rickard, M.J. The risk of internal hernia or volvulus after laparoscopic colorectal surgery: A systematic review. Colorectal Dis. 2016, 18, 1133–1141. [Google Scholar] [CrossRef] [PubMed]
- De Muzio, F.; Cutolo, C.; Granata, V.; Fusco, R.; Ravo, L.; Maggialetti, N.; Brunese, M.C.; Grassi, R.; Grassi, F.; Bruno, F.; et al. CT study protocol optimization in acute non-traumatic abdominal settings. Eur. Rev. Med. Pharmacol. Sci. 2022, 26, 860–878. [Google Scholar] [CrossRef]
- Millet, I.; Boutot, D.; Faget, C.; Pages-Bouic, E.; Molinari, N.; Zins, M.; Taourel, P. Assessment of Strangulation in Adhesive Small Bowel Obstruction on the Basis of Combined CT Findings: Implications for Clinical Care. Radiology 2017, 285, 798–808. [Google Scholar] [CrossRef] [PubMed]
- Rajebhosale, R.; Miah, M.; Currie, F.; Thomas, P. Closed loop obstruction and adhesive intestinal obstruction in perineal hernia. BMJ Case Rep. 2020, 13, e238112. [Google Scholar] [CrossRef] [PubMed]
- Molinelli, V.; Angeretti, M.G.; Duka, E.; Tarallo, N.; Bracchi, E.; Novario, R.; Fugazzola, C. Role of MRI and added value of diffusion-weighted and gadolinium-enhanced MRI for the diagnosis of local recurrence from rectal cancer. Abdom. Radiol. 2018, 43, 2903–2912. [Google Scholar] [CrossRef]
- Horvat, N.; Carlos Tavares Rocha, C.; Clemente Oliveira, B.; Petkovska, I.; Gollub, M.J. MRI of Rectal Cancer: Tumor Staging, Imaging Techniques, and Management. Radiographics 2019, 39, 367–387. [Google Scholar] [CrossRef] [PubMed]
- Renfro, L.A.; Grothey, A.; Kerr, D.; Haller, D.G.; André, T.; Van Cutsem, E.; Saltz, L.; Labianca, R.; Loprinzi, C.L.; Adjuvant Colon Cancer Endpoints (ACCENT) Group; et al. Survival following early-stage colon cancer: An ACCENT-based comparison of patients versus a matched international general population. Ann. Oncol. 2015, 26, 950–958. [Google Scholar] [CrossRef] [PubMed]
- Deandrea, S.; Sardanelli, F.; Calabrese, M.; Ferré, F.; Vainieri, M.; Sestini, E.; Caumo, F.; Saguatti, G.; Bucchi, L.; Cataliotti, L. Provision of follow-up care for women with a history of breast cancer following the 2016 position paper by the Italian Group for Mammographic Screening and the Italian College of Breast Radiologists by SIRM: A survey of Senonetwork Italian breast centres. Radiol. Med. 2022, 127, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Rokan, Z.; Simillis, C.; Kontovounisios, C.; Moran, B.; Tekkis, P.; Brown, G. Locally Recurrent Rectal Cancer According to a Standardized MRI Classification System: A Systematic Review of the Literature. J. Clin. Med. 2022, 11, 3511. [Google Scholar] [CrossRef] [PubMed]
- Masaki, T.; Matsuoka, H.; Kishiki, T.; Kojima, K.; Tonari, A.; Aso, N.; Beniya, A.; Iioka, A.; Wakamatsu, T.; Sunami, E. Site-specific risk factors for local recurrence after rectal cancer surgery. Surg. Oncol. 2021, 37, 101540. [Google Scholar] [CrossRef] [PubMed]
- Moore, H.G.; Shoup, M.; Riedel, E.; Minsky, B.D.; Alektiar, K.M.; Ercolani, M.; Paty, P.B.; Wong, W.D.; Guillem, J.G. Colorectal Cancer Pelvic Recurrences: Determinants of Resectability. Dis. Colon Rectum 2004, 47, 1599–1606. [Google Scholar] [CrossRef] [PubMed]
- Bruno, F.; Marrelli, A.; Tommasino, E.; Martinese, G.; Gagliardi, A.; Pertici, L.; Pagliei, V.; Palumbo, P.; Arrigoni, F.; Di Cesare, E.; et al. Advanced MRI imaging of nerve roots in lumbar radiculopathy due to discoradicular conflict: DWI, DTI, and T2 mapping with clinical and neurophysiological correlations. Radiol. Med. 2022. ahead of print. [Google Scholar] [CrossRef]
- Borgheresi, A.; De Muzio, F.; Agostini, A.; Ottaviani, L.; Bruno, A.; Granata, V.; Fusco, R.; Danti, G.; Flammia, F.; Grassi, R.; et al. Lymph Nodes Evaluation in Rectal Cancer: Where Do We Stand and Future Perspective. J. Clin. Med. 2022, 11, 2599. [Google Scholar] [CrossRef]
- Lee, S.L.; Shin, Y.R.; Kim, K. The added value of pelvic surveillance by MRI during postoperative follow-up of rectal cancer, with a focus on abbreviated MRI. Eur. Radiol. 2020, 30, 3113–3124. [Google Scholar] [CrossRef]
- Lambregts, D.M.; van Heeswijk, M.M.; Delli Pizzi, A.; van Elderen, S.G.; Andrade, L.; Peters, N.H.; Kint, P.A.; Osinga-de Jong, M.; Bipat, S.; Beets-Tan, R.G.; et al. Diffusion-weighted MRI to assess response to chemoradiotherapy in rectal cancer: Main interpretation pitfalls and their use for teaching. Eur. Radiol. 2017, 27, 4445–4454. [Google Scholar] [CrossRef]
- Renzulli, M.; Brandi, N.; Argalia, G.; Brocchi, S.; Farolfi, A.; Fanti, S.; Golfieri, R. Morphological, dynamic and functional characteristics of liver pseudolesions and benign lesions. Radiol. Med. 2022, 127, 129–144. [Google Scholar] [CrossRef]
- Grosu, S.; Schäfer, A.O.; Baumann, T.; Manegold, P.; Langer, M.; Gerstmair, A. Differentiating locally recurrent rectal cancer from scar tissue: Value of diffusion-weighted MRI. Eur. J. Radiol. 2016, 85, 1265–1270. [Google Scholar] [CrossRef]
- Sinha, R.; Rajiah, P.; Ramachandran, I.; Sanders, S.; Murphy, P.D. Diffusion-weighted MR imaging of the gastrointestinal tract: Technique, indications, and imaging findings. Radiographics 2013, 33, 655–676. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; Belli, A.; Danti, G.; Bicci, E.; Cutolo, C.; Petrillo, A.; Izzo, F. Diffusion weighted imaging and diffusion kurtosis imaging in abdominal oncological setting: Why and when. Infect. Agent. Cancer. 2022, 17, 25. [Google Scholar] [CrossRef] [PubMed]
- Hupkens, B.J.P.; Maas, M.; Martens, M.H.; Deserno, W.M.L.L.G.; Leijtens, J.W.A.; Nelemans, P.J.; Bakers, F.C.H.; Lambregts, D.M.J.; Beets, G.L.; Beets-Tan, R.G.H. MRI surveillance for the detection of local recurrence in rectal cancer after transanal endoscopic microsurgery. Eur. Radiol. 2017, 27, 4960–4969. [Google Scholar] [CrossRef] [PubMed]
- Lambregts, D.M.; Lahaye, M.J.; Heijnen, L.A.; Martens, M.H.; Maas, M.; Beets, G.L.; Beets-Tan, R.G. MRI and diffusion-weighted MRI to diagnose a local tumour regrowth during long-term follow-up of rectal cancer patients treated with organ preservation after chemoradiotherapy. Eur. Radiol. 2016, 26, 2118–2125. [Google Scholar] [CrossRef] [PubMed]
- Colosio, A.; Soyer, P.; Rousset, P.; Barbe, C.; Nguyen, F.; Bouché, O.; Hoeffel, C. Value of diffusion-weighted and gadolinium-enhanced MRI for the diagnosis of pelvic recurrence from colorectal cancer. J. Magn. Reson. Imaging 2014, 40, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Fusco, R.; Granata, V.; Sansone, M.; Rega, D.; Delrio, P.; Tatangelo, F.; Romano, C.; Avallone, A.; Pupo, D.; Giordano, M.; et al. Validation of the standardized index of shape tool to analyze DCE-MRI data in the assessment of neo-adjuvant therapy in locally advanced rectal cancer. Radiol. Med. 2021, 126, 1044–1054. [Google Scholar] [CrossRef]
- Orlacchio, A.; Guastoni, C.; Beretta, G.D.; Cosmai, L.; Galluzzo, M.; Gori, S.; Grassedonio, E.; Incorvaia, L.; Marcantoni, C.; Netti, G.S.; et al. SIRM-SIN-AIOM: Appropriateness criteria for evaluation and prevention of renal damage in the patient undergoing contrast medium examinations-consensus statements from Italian College of Radiology (SIRM), Italian College of Nephrology (SIN) and Italian Association of Medical Oncology (AIOM). Radiol. Med. 2022, 127, 534–542. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Ma, X.; Li, S.; Li, Z.; Jia, Y.; Xia, Y.; Wang, M.; Shen, F.; Lu, J. MRI-Based Radiomics of Rectal Cancer: Assessment of the Local Recurrence at the Site of Anastomosis. Acad. Radiol. 2021, 28 (Suppl. S1), S87–S94. [Google Scholar] [CrossRef]
- Gitto, S.; Bologna, M.; Corino, V.D.A.; Emili, I.; Albano, D.; Messina, C.; Armiraglio, E.; Parafioriti, A.; Luzzati, A.; Mainardi, L.; et al. Diffusion-weighted MRI radiomics of spine bone tumors: Feature stability and machine learning-based classification performance. Radiol. Med. 2022, 127, 518–525. [Google Scholar] [CrossRef]
- Zerunian, M.; Pucciarelli, F.; Caruso, D.; Polici, M.; Masci, B.; Guido, G.; De Santis, D.; Polverari, D.; Principessa, D.; Benvenga, A.; et al. Artificial intelligence based image quality enhancement in liver MRI: A quantitative and qualitative evaluation. Radiol. Med. 2022. ahead of print. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; De Muzio, F.; Cutolo, C.; Setola, S.V.; Grassi, R.; Grassi, F.; Ottaiano, A.; Nasti, G.; Tatangelo, F.; et al. Radiomics textural features by MR imaging to assess clinical outcomes following liver resection in colorectal liver metastases. Radiol. Med. 2022, 127, 461–470. [Google Scholar] [CrossRef]
- Matsoukas, S.; Scaggiante, J.; Schuldt, B.R.; Smith, C.J.; Chennareddy, S.; Kalagara, R.; Majidi, S.; Bederson, J.B.; Fifi, J.T.; Mocco, J.; et al. Accuracy of artificial intelligence for the detection of intracranial hemorrhage and chronic cerebral microbleeds: A systematic review and pooled analysis. Radiol. Med. 2022. ahead of print. [Google Scholar] [CrossRef]
- Cozzi, D.; Bicci, E.; Cavigli, E.; Danti, G.; Bettarini, S.; Tortoli, P.; Mazzoni, L.N.; Busoni, S.; Pradella, S.; Miele, V. Radiomics in pulmonary neuroendocrine tumours (NETs). Radiol. Med. 2022, 127, 609–615. [Google Scholar] [CrossRef]
- Xue, K.; Liu, L.; Liu, Y.; Guo, Y.; Zhu, Y.; Zhang, M. Radiomics model based on multi-sequence MR images for predicting preoperative immunoscore in rectal cancer. Radiol. Med. 2022, 127, 702–713. [Google Scholar] [CrossRef]
- Gao, W.; Wang, W.; Song, D.; Yang, C.; Zhu, K.; Zeng, M.; Rao, S.X.; Wang, M. A predictive model integrating deep and radiomics features based on gadobenate dimeglumine-enhanced MRI for postoperative early recurrence of hepatocellular carcinoma. Radiol. Med. 2022, 127, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Fusco, R.; De Muzio, F.; Cutolo, C.; Setola, S.V.; Dell’Aversana, F.; Grassi, F.; Belli, A.; Silvestro, L.; Ottaiano, A.; et al. Radiomics and machine learning analysis based on magnetic resonance imaging in the assessment of liver mucinous colorectal metastases. Radiol. Med. 2022, 127, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Fusco, R.; De Muzio, F.; Cutolo, C.; Setola, S.V.; Dell’Aversana, F.; Belli, A.; Romano, C.; Ottaiano, A.; Nasti, G.; et al. Magnetic Resonance Features of Liver Mucinous Colorectal Metastases: What the Radiologist Should Know. J. Clin. Med. 2022, 11, 2221. [Google Scholar] [CrossRef] [PubMed]
- Caruso, D.; Polici, M.; Rinzivillo, M.; Zerunian, M.; Nacci, I.; Marasco, M.; Magi, L.; Tarallo, M.; Gargiulo, S.; Iannicelli, E.; et al. CT-based radiomics for prediction of therapeutic response to Everolimus in metastatic neuroendocrine tumors. Radiol. Med. 2022, 127, 691–701. [Google Scholar] [CrossRef]
- Vicini, S.; Bortolotto, C.; Rengo, M.; Ballerini, D.; Bellini, D.; Carbone, I.; Preda, L.; Laghi, A.; Coppola, F.; Faggioni, L. A narrative review on current imaging applications of artificial intelligence and radiomics in oncology: Focus on the three most common cancers. Radiol. Med. 2022, 127, 819–836. [Google Scholar] [CrossRef]
- Yao, F.; Bian, S.; Zhu, D.; Yuan, Y.; Pan, K.; Pan, Z.; Feng, X.; Tang, K.; Yang, Y. Machine learning-based radiomics for multiple primary prostate cancer biological characteristics prediction with 18F-PSMA-1007 PET: Comparison among different volume segmentation thresholds. Radiol. Med. 2022. ahead of print. [Google Scholar] [CrossRef]
- Tagliafico, A.S.; Campi, C.; Bianca, B.; Bortolotto, C.; Buccicardi, D.; Francesca, C.; Prost, R.; Rengo, M.; Faggioni, L. Blockchain in radiology research and clinical practice: Current trends and future directions. Radiol. Med. 2022, 127, 391–397. [Google Scholar] [CrossRef]
- Park, H.; Kim, K.A.; Jung, J.H.; Rhie, J.; Choi, S.Y. MRI features and texture analysis for the early prediction of therapeutic response to neoadjuvant chemoradiotherapy and tumor recurrence of locally advanced rectal cancer. Eur. Radiol. 2020, 30, 4201–4211. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Yu, N.; Yu, Y.; He, T.; Duan, X. Performance of CT radiomics in predicting the overall survival of patients with stage III clear cell renal carcinoma after radical nephrectomy. Radiol. Med. 2022, 127, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Autorino, R.; Gui, B.; Panza, G.; Boldrini, L.; Cusumano, D.; Russo, L.; Nardangeli, A.; Persiani, S.; Campitelli, M.; Ferrandina, G.; et al. Radiomics-based prediction of two-year clinical outcome in locally advanced cervical cancer patients undergoing neoadjuvant chemoradiotherapy. Radiol. Med. 2022, 127, 498–506. [Google Scholar] [CrossRef] [PubMed]
- Masci, G.M.; Iafrate, F.; Ciccarelli, F.; Pambianchi, G.; Panebianco, V.; Pasculli, P.; Ciardi, M.R.; Mastroianni, C.M.; Ricci, P.; Catalano, C.; et al. Tocilizumab effects in COVID-19 pneumonia: Role of CT texture analysis in quantitative assessment of response to therapy. Radiol. Med. 2021, 126, 1170–1180. [Google Scholar] [CrossRef]
- Gurgitano, M.; Angileri, S.A.; Rodà, G.M.; Liguori, A.; Pandolfi, M.; Ierardi, A.M.; Wood, B.J.; Carrafiello, G. Interventional Radiology ex-machina: Impact of Artificial Intelligence on practice. Radiol. Med. 2021, 126, 998–1006. [Google Scholar] [CrossRef]
- Giurazza, F.; Contegiacomo, A.; Calandri, M.; Mosconi, C.; Modestino, F.; Corvino, F.; Scrofani, A.R.; Marra, P.; Coniglio, G.; Failla, G.; et al. IVC filter retrieval: A multicenter proposal of two score systems to predict application of complex technique and procedural outcome. Radiol. Med. 2021, 126, 1007–1016. [Google Scholar] [CrossRef]
- Barile, A. Some thoughts and greetings from the new Editor-in-Chief. Radiol. Med. 2021, 126, 3–4, Erratum in Radiol. Med. 2021, 126, 1377. [Google Scholar] [CrossRef]
- Cappabianca, S.; Granata, V.; Di Grezia, G.; Mandato, Y.; Reginelli, A.; Di Mizio, V.; Grassi, R.; Rotondo, A. The role of nasoenteric intubation in the MR study of patients with Crohn’s disease: Our experience and literature review. Radiol. Med. 2011, 116, 389–406. [Google Scholar] [CrossRef]
- De Filippo, M.; Puglisi, S.; D’Amuri, F.; Gentili, F.; Paladini, I.; Carrafiello, G.; Maestroni, U.; Del Rio, P.; Ziglioli, F.; Pagnini, F. CT-guided percutaneous drainage of abdominopelvic collections: A pictorial essay. Radiol. Med. 2021, 126, 1561–1570. [Google Scholar] [CrossRef]
- Petralia, G.; Zugni, F.; Summers, P.E.; Colombo, A.; Pricolo, P.; Grazioli, L.; Colagrande, S.; Giovagnoni, A.; Padhani, A.R.; Italian Working Group on Magnetic Resonance. Whole-body magnetic resonance imaging (WB-MRI) for cancer screening: Recommendations for use. Radiol. Med. 2021, 126, 1434–1450. [Google Scholar] [CrossRef]
- Palmisano, A.; Scotti, G.M.; Ippolito, D.; Morelli, M.J.; Vignale, D.; Gandola, D.; Sironi, S.; De Cobelli, F.; Ferrante, L.; Spessot, M.; et al. Chest CT in the emergency department for suspected COVID-19 pneumonia. Radiol. Med. 2021, 126, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, A.F.; Afsahi, A.M.; Gupta, A.; Gholamrezanezhad, A. Severe acute respiratory syndrome (SARS), Middle East respiratory syndrome (MERS), influenza, and COVID-19, beyond the lungs: A review article. Radiol. Med. 2021, 126, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Gabelloni, M.; Faggioni, L.; Cioni, D.; Mendola, V.; Falaschi, Z.; Coppola, S.; Corradi, F.; Isirdi, A.; Brandi, N.; Coppola, F.; et al. Extracorporeal membrane oxygenation (ECMO) in COVID-19 patients: A pocket guide for radiologists. Radiol. Med. 2022, 13, 369–382. [Google Scholar] [CrossRef] [PubMed]
- Grassi, R.; Cappabianca, S.; Urraro, F.; Feragalli, B.; Montanelli, A.; Patelli, G.; Granata, V.; Giacobbe, G.; Russo, G.M.; Grillo, A.; et al. Chest CT Computerized Aided Quantification of PNEUMONIA Lesions in COVID-19 Infection: A Comparison among Three Commercial Software. Int. J. Environ. Res. Public Health 2020, 17, 6914. [Google Scholar] [CrossRef] [PubMed]
- Fusco, R.; Grassi, R.; Granata, V.; Setola, S.V.; Grassi, F.; Cozzi, D.; Pecori, B.; Izzo, F.; Petrillo, A. Artificial Intelligence and COVID-19 Using Chest CT Scan and Chest X-ray Images: Machine Learning and Deep Learning Approaches for Diagnosis and Treatment. J. Pers. Med. 2021, 11, 993. [Google Scholar] [CrossRef]
- Özel, M.; Aslan, A.; Araç, S. Use of the COVID-19 Reporting and Data System (CO-RADS) classification and chest computed tomography involvement score (CT-IS) in COVID-19 pneumonia. Radiol. Med. 2021, 126, 679–687. [Google Scholar] [CrossRef]
- Ippolito, D.; Giandola, T.; Maino, C.; Pecorelli, A.; Capodaglio, C.; Ragusi, M.; Porta, M.; Gandola, D.; Masetto, A.; Drago, S.; et al. Acute pulmonary embolism in hospitalized patients with SARS-CoV-2-related pneumonia: Multicentric experience from Italian endemic area. Radiol. Med. 2021, 126, 669–678. [Google Scholar] [CrossRef]
- Fusco, R.; Granata, V.; Petrillo, A. Introduction to Special Issue of Radiology and Imaging of Cancer. Cancers 2020, 12, 2665. [Google Scholar] [CrossRef]
- Scapicchio, C.; Gabelloni, M.; Barucci, A.; Cioni, D.; Saba, L.; Neri, E. A deep look into radiomics. Radiol. Med. 2021, 126, 1296–1311. [Google Scholar] [CrossRef]
- Morin, O.; Vallières, M.; Jochems, A.; Woodruff, H.C.; Valdes, G.; Braunstein, S.E.; Wildberger, J.E.; Villanueva-Meyer, J.E.; Kearney, V.; Yom, S.S.; et al. A Deep Look into the Future of Quantitative Imaging in Oncology: A Statement of Working Principles and Proposal for Change. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 1074–1082. [Google Scholar] [CrossRef]
- Cellina, M.; Pirovano, M.; Ciocca, M.; Gibelli, D.; Floridi, C.; Oliva, G. Radiomic analysis of the optic nerve at the first episode of acute optic neuritis: An indicator of optic nerve pathology and a predictor of visual recovery? Radiol. Med. 2021, 126, 698–706. [Google Scholar] [CrossRef] [PubMed]
- Santone, A.; Brunese, M.C.; Donnarumma, F.; Guerriero, P.; Mercaldo, F.; Reginelli, A.; Miele, V.; Giovagnoni, A.; Brunese, L. Radiomic features for prostate cancer grade detection through formal verification. Radiol. Med. 2021, 126, 688–697. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Fusco, R.; Barretta, M.L.; Picone, C.; Avallone, A.; Belli, A.; Patrone, R.; Ferrante, M.; Cozzi, D.; Grassi, R.; et al. Radiomics in hepatic metastasis by colorectal cancer. Infect. Agent. Cancer 2021, 16, 39. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Grassi, R.; Fusco, R.; Galdiero, R.; Setola, S.V.; Palaia, R.; Belli, A.; Silvestro, L.; Cozzi, D.; Brunese, L.; et al. Pancreatic cancer detection and characterization: State of the art and radiomics. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 3684–3699. [Google Scholar] [CrossRef] [PubMed]
- Agazzi, G.M.; Ravanelli, M.; Roca, E.; Medicina, D.; Balzarini, P.; Pessina, C.; Vermi, W.; Berruti, A.; Maroldi, R.; Farina, D. CT texture analysis for prediction of EGFR mutational status and ALK rearrangement in patients with non-small cell lung cancer. Radiol. Med. 2021, 126, 786–794. [Google Scholar] [CrossRef] [PubMed]
- Agüloğlu, N.; Aksu, A.; Akyol, M.; Katgı, N.; Doksöz, T.Ç. Importance of pretreatment 18F-FDG PET/CT texture analysis in predicting Egfr and Alk mutation in Patients with non-small cell lung cancer. Nuklearmedizin 2022, 61, 433–439. [Google Scholar] [CrossRef]
- Mayerhoefer, M.E.; Materka, A.; Langs, G.; Häggström, I.; Szczypiński, P.; Gibbs, P.; Cook, G. Introduction to Radiomics. J. Nucl. Med. 2020, 61, 488–495. [Google Scholar] [CrossRef]
- Yip, S.S.; Aerts, H.J. Applications and limitations of radiomics. Phys. Med. Biol. 2016, 61, R150–R166. [Google Scholar] [CrossRef]
- Granata, V.; Bicchierai, G.; Fusco, R.; Cozzi, D.; Grazzini, G.; Danti, G.; De Muzio, F.; Maggialetti, N.; Smorchkova, O.; D’Elia, M.; et al. Diagnostic protocols in oncology: Workup and treatment planning. Part 2: Abbreviated MR protocol. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 6499–6528. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; Catalano, O.; Setola, S.V.; de Lutio di Castelguidone, E.; Piccirillo, M.; Palaia, R.; Grassi, R.; Granata, F.; Izzo, F.; et al. Multidetector computer tomography in the pancreatic adenocarcinoma assessment: An update. Infect. Agent. Cancer. 2016, 11, 57. [Google Scholar] [CrossRef]
- Wilson, R.; Devaraj, A. Radiomics of pulmonary nodules and lung cancer. Transl. Lung Cancer Res. 2017, 6, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Binczyk, F.; Prazuch, W.; Bozek, P.; Polanska, J. Radiomics and artificial intelligence in lung cancer screening. Transl. Lung Cancer Res. 2021, 10, 1186–1199. [Google Scholar] [CrossRef] [PubMed]
- Fiz, F.; Viganò, L.; Gennaro, N.; Costa, G.; La Bella, L.; Boichuk, A.; Cavinato, L.; Sollini, M.; Politi, L.S.; Chiti, A.; et al. Radiomics of Liver Metastases: A Systematic Review. Cancers 2020, 12, 2881. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; Avallone, A.; Catalano, O.; Piccirillo, M.; Palaia, R.; Nasti, G.; Petrillo, A.; Izzo, F. A radiologist’s point of view in the presurgical and intraoperative setting of colorectal liver metastases. Future Oncol. 2018, 14, 2189–2206. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, G.; Mori, M.; Panzeri, M.M.; Barbera, M.; Palumbo, D.; Sini, C.; Muffatti, F.; Andreasi, V.; Steidler, S.; Doglioni, C.; et al. CT-derived radiomic features to discriminate histologic characteristics of pancreatic neuroendocrine tumors. Radiol. Med. 2021, 126, 745–760. [Google Scholar] [CrossRef] [PubMed]
- Stefanini, M.; Simonetti, G. Interventional Magnetic Resonance Imaging Suite (IMRIS): How to build and how to use. Radiol. Med. 2022. ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Fusco, R.; Avallone, A.; De Stefano, A.; Ottaiano, A.; Sbordone, C.; Brunese, L.; Izzo, F.; Petrillo, A. Radiomics-Derived Data by Contrast Enhanced Magnetic Resonance in RAS Mutations Detection in Colorectal Liver Metastases. Cancers 2021, 13, 453. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; Costa, M.; Picone, C.; Cozzi, D.; Moroni, C.; La Casella, G.V.; Montanino, A.; Monti, R.; Mazzoni, F.; et al. Preliminary Report on Computed Tomography Radiomics Features as Biomarkers to Immunotherapy Selection in Lung Adenocarcinoma Patients. Cancers 2021, 13, 3992. [Google Scholar] [CrossRef]
- Taha, B.; Boley, D.; Sun, J.; Chen, C.C. State of Radiomics in Glioblastoma. Neurosurgery 2021, 89, 177–184. [Google Scholar] [CrossRef]
- Yu, Y.; He, Z.; Ouyang, J.; Tan, Y.; Chen, Y.; Gu, Y.; Mao, L.; Ren, W.; Wang, J.; Lin, L.; et al. Magnetic resonance imaging radiomics predicts preoperative axillary lymph node metastasis to support surgical decisions and is associated with tumor microenvironment in invasive breast cancer: A machine learning, multicenter study. EBioMedicine 2021, 69, 103460. [Google Scholar] [CrossRef]
- Taha, B.; Boley, D.; Sun, J.; Chen, C. Potential and limitations of radiomics in neuro-oncology. J. Clin. Neurosci. 2021, 90, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Frix, A.N.; Cousin, F.; Refaee, T.; Bottari, F.; Vaidyanathan, A.; Desir, C.; Vos, W.; Walsh, S.; Occhipinti, M.; Lovinfosse, P.; et al. Radiomics in Lung Diseases Imaging: State-of-the-Art for Clinicians. J. Pers. Med. 2021, 11, 602. [Google Scholar] [CrossRef] [PubMed]
- Bortolotto, C.; Lancia, A.; Stelitano, C.; Montesano, M.; Merizzoli, E.; Agustoni, F.; Stella, G.; Preda, L.; Filippi, A.R. Radiomics features as predictive and prognostic biomarkers in NSCLC. Expert Rev. Anticancer Ther. 2021, 21, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Fusco, R.; Granata, V.; Grazzini, G.; Pradella, S.; Borgheresi, A.; Bruno, A.; Palumbo, P.; Bruno, F.; Grassi, R.; Giovagnoni, A.; et al. Radiomics in medical imaging: Pitfalls and challenges in clinical management. Jpn. J. Radiol. 2022, 40, 919–929. [Google Scholar] [CrossRef]
- Nakamura, Y.; Higaki, T.; Honda, Y.; Tatsugami, F.; Tani, C.; Fukumoto, W.; Narita, K.; Kondo, S.; Akagi, M.; Awai, K. Advanced CT techniques for assessing hepatocellular carcinoma. Radiol. Med. 2021, 126, 925–935. [Google Scholar] [CrossRef]
- Chianca, V.; Albano, D.; Messina, C.; Vincenzo, G.; Rizzo, S.; Del Grande, F.; Sconfienza, L.M. An update in musculoskeletal tumors: From quantitative imaging to radiomics. Radiol. Med. 2021, 126, 1095–1105. [Google Scholar] [CrossRef]
- Halefoglu, A.M.; Ozagari, A.A. Tumor grade estımatıon of clear cell and papıllary renal cell carcınomas usıng contrast-enhanced MDCT and FSE T2 weıghted MR ımagıng: Radıology-pathology correlatıon. Radiol. Med. 2021, 126, 1139–1148. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; Risi, C.; Ottaiano, A.; Avallone, A.; De Stefano, A.; Grimm, R.; Grassi, R.; Brunese, L.; Izzo, F.; et al. Diffusion-Weighted MRI and Diffusion Kurtosis Imaging to Detect RAS Mutation in Colorectal Liver Metastasis. Cancers 2020, 12, 2420. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; Setola, S.V.; Simonetti, I.; Cozzi, D.; Grazzini, G.; Grassi, F.; Belli, A.; Miele, V.; Izzo, F.; et al. An update on radiomics techniques in primary liver cancers. Infect. Agent. Cancer 2022, 17, 6. [Google Scholar] [CrossRef]
- Avanzo, M.; Stancanello, J.; El Naqa, I. Beyond imaging: The promise of radiomics. Phys. Med. 2017, 38, 122–139. [Google Scholar] [CrossRef]
- Fornacon-Wood, I.; Faivre-Finn, C.; O’Connor, J.P.B.; Price, G.J. Radiomics as a personalized medicine tool in lung cancer: Separating the hope from the hype. Lung Cancer 2020, 146, 197–208. [Google Scholar] [CrossRef]
- Bogowicz, M.; Vuong, D.; Huellner, M.W.; Pavic, M.; Andratschke, N.; Gabrys, H.S.; Guckenberger, M.; Tanadini-Lang, S. CT radiomics and PET radiomics: Ready for clinical implementation? Q J. Nucl. Med. Mol. Imaging 2019, 63, 355–370. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Fusco, R.; Sansone, M.; Grassi, R.; Maio, F.; Palaia, R.; Tatangelo, F.; Botti, G.; Grimm, R.; Curley, S.; et al. Magnetic resonance imaging in the assessment of pancreatic cancer with quantitative parameter extraction by means of dynamic contrast-enhanced magnetic resonance imaging, diffusion kurtosis imaging and intravoxel incoherent motion diffusion-weighted imaging. Therap. Adv. Gastroenterol. 2020, 13, 1756284819885052. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Fusco, R.; Setola, S.V.; Picone, C.; Vallone, P.; Belli, A.; Incollingo, P.; Albino, V.; Tatangelo, F.; Izzo, F.; et al. Microvascular invasion and grading in hepatocellular carcinoma: Correlation with major and ancillary features according to LIRADS. Abdom. Radiol. 2019, 44, 2788–2800. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Fusco, R.; Setola, S.V.; De Muzio, F.; Dell’ Aversana, F.; Cutolo, C.; Faggioni, L.; Miele, V.; Izzo, F.; Petrillo, A. CT-Based Radiomics Analysis to Predict Histopathological Outcomes Following Liver Resection in Colorectal Liver Metastases. Cancers 2022, 14, 1648. [Google Scholar] [CrossRef]
- Sun, J.; Li, H.; Gao, J.; Li, J.; Li, M.; Zhou, Z.; Peng, Y. Performance evaluation of a deep learning image reconstruction (DLIR) algorithm in “double low” chest CTA in children: A feasibility study. Radiol. Med. 2021, 126, 1181–1188. [Google Scholar] [CrossRef]
- Granata, V.; Faggioni, L.; Grassi, R.; Fusco, R.; Reginelli, A.; Rega, D.; Maggialetti, N.; Buccicardi, D.; Frittoli, B.; Rengo, M.; et al. Structured reporting of computed tomography in the staging of colon cancer: A Delphi consensus proposal. Radiol. Med. 2022, 127, 21–29. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; De Muzio, F.; Cutolo, C.; Setola, S.V.; Dell’Aversana, F.; Ottaiano, A.; Nasti, G.; Grassi, R.; Pilone, V.; et al. EOB-MR Based Radiomics Analysis to Assess Clinical Outcomes following Liver Resection in Colorectal Liver Metastases. Cancers 2022, 14, 1239. [Google Scholar] [CrossRef]
- Arimura, H.; Soufi, M.; Kamezawa, H.; Ninomiya, K.; Yamada, M. Radiomics with artificial intelligence for precision medicine in radiation therapy. J. Radiat. Res. 2019, 60, 150–157. [Google Scholar] [CrossRef]
- Li, F.; Pan, D.; He, Y.; Wu, Y.; Peng, J.; Li, J.; Wang, Y.; Yang, H.; Chen, J. Using ultrasound features and radiomics analysis to predict lymph node metastasis in patients with thyroid cancer. BMC Surg. 2020, 20, 315. [Google Scholar] [CrossRef]
- Zanfardino, M.; Franzese, M.; Pane, K.; Cavaliere, C.; Monti, S.; Esposito, G.; Salvatore, M.; Aiello, M. Bringing radiomics into a multi-omics framework for a comprehensive genotype-phenotype characterization of oncological diseases. J. Transl. Med. 2019, 17, 337. [Google Scholar] [CrossRef]
- Izzo, F.; Piccirillo, M.; Albino, V.; Palaia, R.; Belli, A.; Granata, V.; Setola, S.; Fusco, R.; Petrillo, A.; Orlando, R.; et al. Prospective screening increases the detection of potentially curable hepatocellular carcinoma: Results in 8,900 high-risk patients. HPB 2013, 15, 985–990. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; De Muzio, F.; Cutolo, C.; Setola, S.V.; Dell’ Aversana, F.; Ottaiano, A.; Avallone, A.; Nasti, G.; Grassi, F.; et al. Contrast MR-Based Radiomics and Machine Learning Analysis to Assess Clinical Outcomes following Liver Resection in Colorectal Liver Metastases: A Preliminary Study. Cancers 2022, 14, 1110. [Google Scholar] [CrossRef]
- Liu, J.; Wang, C.; Guo, W.; Zeng, P.; Liu, Y.; Lang, N.; Yuan, H. A preliminary study using spinal MRI-based radiomics to predict high-risk cytogenetic abnormalities in multiple myeloma. Radiol. Med. 2021, 126, 1226–1235. [Google Scholar] [CrossRef] [PubMed]
- Qin, H.; Que, Q.; Lin, P.; Li, X.; Wang, X.R.; He, Y.; Chen, J.Q.; Yang, H. Magnetic resonance imaging (MRI) radiomics of papillary thyroid cancer (PTC): A comparison of predictive performance of multiple classifiers modeling to identify cervical lymph node metastases before surgery. Radiol. Med. 2021, 126, 1312–1327. [Google Scholar] [CrossRef] [PubMed]
- Fusco, R.; Di Bernardo, E.; Piccirillo, A.; Rubulotta, M.R.; Petrosino, T.; Barretta, M.L.; Mattace Raso, M.; Vallone, P.; Raiano, C.; Di Giacomo, R.; et al. Radiomic and Artificial Intelligence Analysis with Textural Metrics Extracted by Contrast-Enhanced Mammography and Dynamic Contrast Magnetic Resonance Imaging to Detect Breast Malignant Lesions. Curr. Oncol. 2022, 29, 1947–1966. [Google Scholar] [CrossRef] [PubMed]
- Neri, E.; Granata, V.; Montemezzi, S.; Belli, P.; Bernardi, D.; Brancato, B.; Caumo, F.; Calabrese, M.; Coppola, F.; Cossu, E.; et al. Structured reporting of x-ray mammography in the first diagnosis of breast cancer: A Delphi consensus proposal. Radiol. Med. 2022, 127, 471–483. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; De Muzio, F.; Cutolo, C.; Mattace Raso, M.; Gabelloni, M.; Avallone, A.; Ottaiano, A.; Tatangelo, F.; Brunese, M.C.; et al. Radiomics and Machine Learning Analysis Based on Magnetic Resonance Imaging in the Assessment of Colorectal Liver Metastases Growth Pattern. Diagnostics 2022, 12, 1115. [Google Scholar] [CrossRef] [PubMed]
- Brunese, L.; Brunese, M.C.; Carbone, M.; Ciccone, V.; Mercaldo, F.; Santone, A. Automatic PI-RADS assignment by means of formal methods. Radiol. Med. 2022, 127, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Bellardita, L.; Colciago, R.R.; Frasca, S.; De Santis, M.C.; Gay, S.; Palorini, F.; La Rocca, E.; Valdagni, R.; Rancati, T.; Lozza, L. Breast cancer patient perspective on opportunities and challenges of a genetic test aimed to predict radio-induced side effects before treatment: Analysis of the Italian branch of the REQUITE project. Radiol. Med. 2021, 126, 1366–1373. [Google Scholar] [CrossRef] [PubMed]
- Caruso, D.; Pucciarelli, F.; Zerunian, M.; Ganeshan, B.; De Santis, D.; Polici, M.; Rucci, C.; Polidori, T.; Guido, G.; Bracci, B.; et al. Chest CT texture-based radiomics analysis in differentiating COVID-19 from other interstitial pneumonia. Radiol. Med. 2021, 126, 1415–1424. [Google Scholar] [CrossRef] [PubMed]
- Karmazanovsky, G.; Gruzdev, I.; Tikhonova, V.; Kondratyev, E.; Revishvili, A. Computed tomography-based radiomics approach in pancreatic tumors characterization. Radiol. Med. 2021. ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Danti, G.; Flammia, F.; Matteuzzi, B.; Cozzi, D.; Berti, V.; Grazzini, G.; Pradella, S.; Recchia, L.; Brunese, L.; Miele, V. Gastrointestinal neuroendocrine neoplasms (GI-NENs): Hot topics in morphological, functional, and prognostic imaging. Radiol. Med. 2021, 126, 1497–1507. [Google Scholar] [CrossRef] [PubMed]
- Satake, H.; Ishigaki, S.; Ito, R.; Naganawa, S. Radiomics in breast MRI: Current progress toward clinical application in the era of artificial intelligence. Radiol. Med. 2022, 127, 39–56. [Google Scholar] [CrossRef]
- Chiloiro, G.; Cusumano, D.; de Franco, P.; Lenkowicz, J.; Boldrini, L.; Carano, D.; Barbaro, B.; Corvari, B.; Dinapoli, N.; Giraffa, M.; et al. Does restaging MRI radiomics analysis improve pathological complete response prediction in rectal cancer patients? A prognostic model development. Radiol. Med. 2022, 127, 11–20. [Google Scholar] [CrossRef]
- Gregucci, F.; Fiorentino, A.; Mazzola, R.; Ricchetti, F.; Bonaparte, I.; Surgo, A.; Figlia, V.; Carbonara, R.; Caliandro, M.; Ciliberti, M.P.; et al. Radiomic analysis to predict local response in locally advanced pancreatic cancer treated with stereotactic body radiation therapy. Radiol. Med. 2022, 127, 100–107. [Google Scholar] [CrossRef]
- Hegde, P.S.; Karanikas, V.; Evers, S. The Where, the When, and the How of Immune Monitoring for Cancer Immunotherapies in the Era of Checkpoint Inhibition. Clin. Cancer Res. 2016, 22, 1865–1874. [Google Scholar] [CrossRef]
- Herbst, R.S.; Soria, J.C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 2014, 515, 563–567. [Google Scholar] [CrossRef]
- Wu, M.; Zhang, Y.; Zhang, Y.; Liu, Y.; Wu, M.; Ye, Z. Imaging-based Biomarkers for Predicting and Evaluating Cancer Immunotherapy Response. Radiol. Imaging Cancer 2019, 1, e190031. [Google Scholar] [CrossRef]
- Sun, R.; Limkin, E.J.; Vakalopoulou, M.; Dercle, L.; Champiat, S.; Han, S.R.; Verlingue, L.; Brandao, D.; Lancia, A.; Ferté, C.; et al. A radiomics approach to assess tumour-infiltrating CD8 cells and response to anti-PD-1 or anti-PD-L1 immunotherapy: An imaging biomarker, retrospective multicohort study. Lancet Oncol. 2018, 19, 1180–1191. [Google Scholar] [CrossRef]
- Tang, C.; Hobbs, B.; Amer, A.; Li, X.; Behrens, C.; Canales, J.R.; Cuentas, E.P.; Villalobos, P.; Fried, D.; Koay, E.J.; et al. Development of an Immune-Pathology Informed Radiomics Model for Non-Small Cell Lung Cancer. Sci. Rep. 2018, 8, 1922. [Google Scholar] [CrossRef] [PubMed]
- Chiti, G.; Grazzini, G.; Flammia, F.; Matteuzzi, B.; Tortoli, P.; Bettarini, S.; Pasqualini, E.; Granata, V.; Busoni, S.; Messserini, L.; et al. Gastroenteropancreatic neuroendocrine neoplasms (GEP-NENs): A radiomic model to predict tumor grade. Radiol. Med. 2022, 127, 928–938. [Google Scholar] [CrossRef] [PubMed]
- De Robertis, R.; Geraci, L.; Tomaiuolo, L.; Bortoli, L.; Beleù, A.; Malleo, G.; D’Onofrio, M. Liver metastases in pancreatic ductal adenocarcinoma: A predictive model based on CT texture analysis. Radiol. Med. 2022, 127, 1079–1084. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.H.; Zheng, H.L.; Li, J.T.; Li, P.; Zheng, C.H.; Chen, Q.Y.; Huang, C.M.; Xie, J.W. Prediction of recurrence-free survival and adjuvant therapy benefit in patients with gastrointestinal stromal tumors based on radiomics features. Radiol. Med. 2022, 127, 1085–1097. [Google Scholar] [CrossRef]
- McCarty, T.R.; Bazarbashi, A.N.; Hathorn, K.E.; Thompson, C.C.; Aihara, H. Endoscopic submucosal dissection (ESD) versus transanal endoscopic microsurgery (TEM) for treatment of rectal tumors: A comparative systematic review and meta-analysis. Surg. Endosc. 2020, 34, 1688–1695. [Google Scholar] [CrossRef] [PubMed]
- Sagae, V.M.T.; Ribeiro, I.B.; de Moura, D.T.H.; Brunaldi, V.O.; Logiudice, F.P.; Funari, M.P.; Baba, E.R.; Bernardo, W.M.; de Moura, E.G.H. Endoscopic submucosal dissection versus transanal endoscopic surgery for the treatment of early rectal tumor: A systematic review and meta-analysis. Surg. Endosc. 2020, 34, 1025–1034. [Google Scholar] [CrossRef] [PubMed]
- Baird, P.; Steinke, J.D.; Minnaar, H.S.; Stewart, A.J. Assessment of Quality of Life in Rectal Cancer with Organ-Preservation Treatment: Are We There yet? Clin. Oncol. 2022. ahead of print. [Google Scholar] [CrossRef]
- Gilbert, A.; Homer, V.; Brock, K.; Korsgen, S.; Geh, I.; Hill, J.; Gill, T.; Hainsworth, P.; Tutton, M.; TREC, collaborators; et al. Quality-of-life outcomes in older patients with early-stage rectal cancer receiving organ-preserving treatment with hypofractionated short-course radiotherapy followed by transanal endoscopic microsurgery (TREC): Non-randomised registry of patients unsuitable for total mesorectal excision. Lancet Healthy Longev. 2022. ahead of print. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Muzio, F.; Fusco, R.; Cutolo, C.; Giacobbe, G.; Bruno, F.; Palumbo, P.; Danti, G.; Grazzini, G.; Flammia, F.; Borgheresi, A.; et al. Post-Surgical Imaging Assessment in Rectal Cancer: Normal Findings and Complications. J. Clin. Med. 2023, 12, 1489. https://doi.org/10.3390/jcm12041489
De Muzio F, Fusco R, Cutolo C, Giacobbe G, Bruno F, Palumbo P, Danti G, Grazzini G, Flammia F, Borgheresi A, et al. Post-Surgical Imaging Assessment in Rectal Cancer: Normal Findings and Complications. Journal of Clinical Medicine. 2023; 12(4):1489. https://doi.org/10.3390/jcm12041489
Chicago/Turabian StyleDe Muzio, Federica, Roberta Fusco, Carmen Cutolo, Giuliana Giacobbe, Federico Bruno, Pierpaolo Palumbo, Ginevra Danti, Giulia Grazzini, Federica Flammia, Alessandra Borgheresi, and et al. 2023. "Post-Surgical Imaging Assessment in Rectal Cancer: Normal Findings and Complications" Journal of Clinical Medicine 12, no. 4: 1489. https://doi.org/10.3390/jcm12041489
APA StyleDe Muzio, F., Fusco, R., Cutolo, C., Giacobbe, G., Bruno, F., Palumbo, P., Danti, G., Grazzini, G., Flammia, F., Borgheresi, A., Agostini, A., Grassi, F., Giovagnoni, A., Miele, V., Barile, A., & Granata, V. (2023). Post-Surgical Imaging Assessment in Rectal Cancer: Normal Findings and Complications. Journal of Clinical Medicine, 12(4), 1489. https://doi.org/10.3390/jcm12041489