Gender Difference in the Effects of COVID-19 Pandemic on Mechanical Reperfusion and 30-Day Mortality for STEMI: Results of the ISACS-STEMI COVID-19 Registry
Abstract
1. Introduction
2. Methods
Study Design and Population
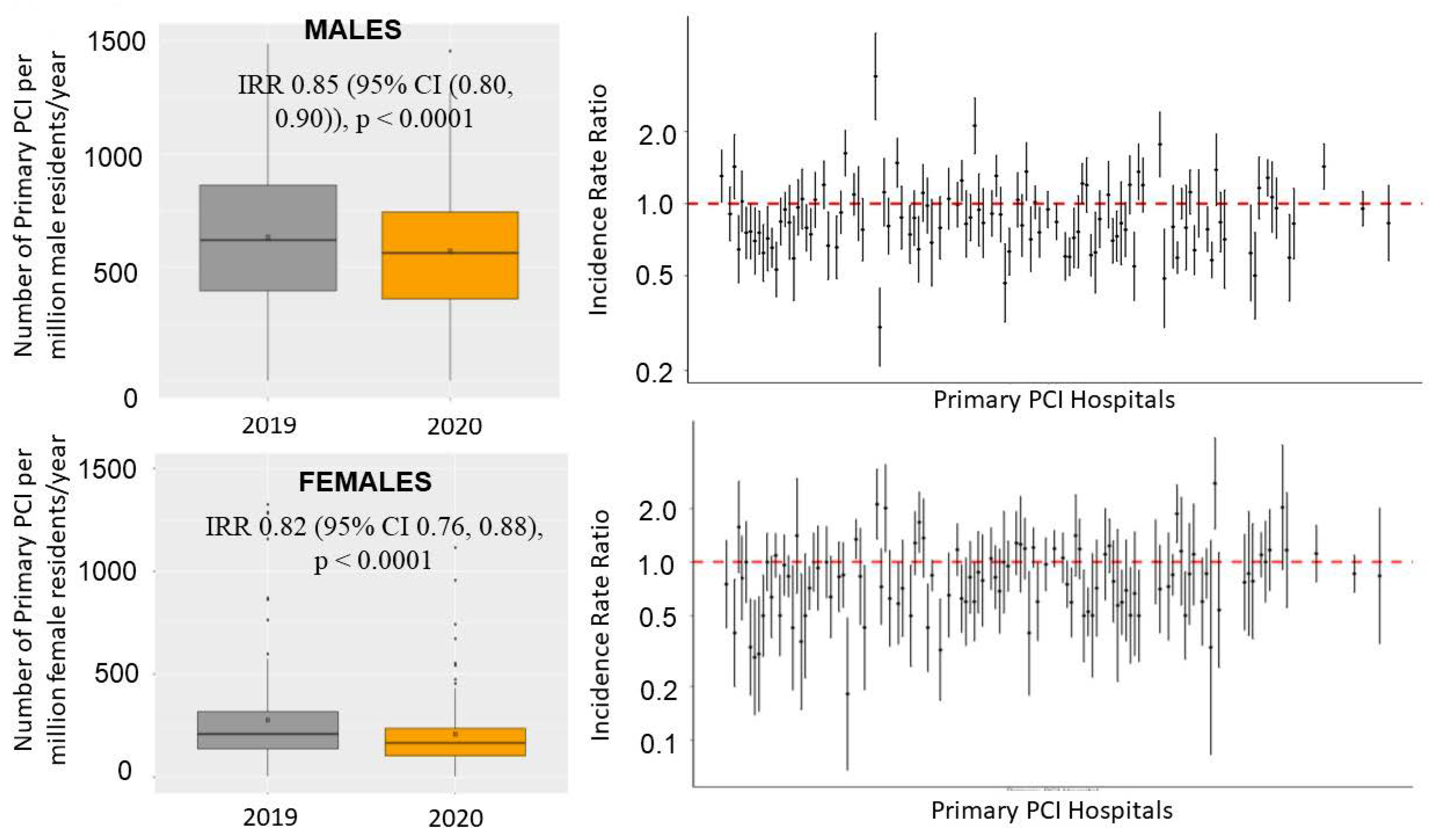
3. Results
4. Baseline Demographic and Clinical Characteristics
5. Procedural Characteristics
6. In-Hospital and 30-Day Mortality
7. Discussion
8. Limitations
9. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Percutaneous coronary intervention | (PCI) |
| ST-segment elevation myocardial infarction | (STEMI) |
| Door-to-balloon time | (DTB) |
| Incidence rate ratio | (IRR) |
| Acute coronary syndrome | (ACS) |
| Drug-eluting stent | (DES) |
| Renin-angiotensin system inhibitors | (RASIs) |
References
- Axenhus, M.; Schedin-Weiss, S.; Winblad, B.; Wimo, A. Changes in mortality trends amongst common diseases during the COVID-19 pandemic in Sweden. Scand. J. Public Health 2021, 50, 748–755. [Google Scholar] [CrossRef] [PubMed]
- Coronavirus (COVID-19) Deaths. Available online: https://ourworldindata.org/covid-deaths (accessed on 31 January 2022).
- Garcia, S.; Albaghdadi, M.S.; Meraj, P.M.; Schmidt, C.; Garberich, R.; Jaffer, F.A.; Dixon, S.; Rade, J.J.; Tannenbaum, M.; Chambers, J.; et al. Reduction in ST-Segment Elevation Cardiac Catheterization Laboratory Activations in the United States during COVID-19 Pandemic. J. Am. Coll. Cardiol. 2020, 75, 2871–2872. [Google Scholar] [CrossRef]
- Tam, C.F.; Cheung, K.S.; Lam, S.; Wong, A.; Yung, A.; Sze, M.; Lam, Y.M.; Chan, C.; Tsang, T.C.; Tsui, M.; et al. Impact of Coronavirus Disease 2019 (COVID-19) Outbreak on ST-Segment-Elevation Myocardial Infarction Care in Hong Kong, China. Circ. Cardiovasc Qual. Outcomes 2020, 13, e006631. [Google Scholar] [CrossRef] [PubMed]
- Piccolo, R.; Bruzzese, D.; Mauro, C.; Aloia, A.; Baldi, C.; Boccalatte, M.; Bottiglieri, G.; Briguori, C.; Caiazzo, G.; Calabrò, P.; et al. Population Trends in Rates of Percutaneous Coronary Revascularization for Acute Coronary Syndromes Associated with the COVID-19. Outbreak Circ. 2020, 141, 2035–2037. [Google Scholar] [CrossRef] [PubMed]
- Stefanini, G.G.; Montorfano, M.; Trabattoni, D.; Andreini, D.; Ferrante, G.; Ancona, M.; Metra, M.; Curello, S.; Maffeo, D.; Pero, G.; et al. ST-Elevation Myocardial Infarction in Patients with COVID-19: Clinical and Angiographic. Outcomes Circ. 2020, 141, 2113–2116. [Google Scholar] [CrossRef]
- De Luca, G.; Suryapranata, H.; Ottervanger, J.P.; Antman, E.M. Time delay to treatment and mortality in primary angioplasty for acute myocardial infarction: Every minute of delay counts. Circulation 2004, 109, 1223–1225. [Google Scholar] [CrossRef]
- Jain, V.; Gupta, K.; Bhatia, K.; Bansal, A.; Arora, S.; Khandelwal, A.K.; Rosenberg, J.R.; Levisay, J.P.; Tommaso, C.L.; Ricciardi, M.J.; et al. Management of STEMI during the COVID-19 pandemic: Lessons learned in 2020 to prepare for 2021. Trends Cardiovasc. Med. 2021, 31, 135–140. [Google Scholar] [CrossRef] [PubMed]
- De Luca, G.; Verdoia, M.; Cercek, M.; Jensen, L.O.; Vavlukis, M.; Calmac, L.; Johnson, T.; Ferrer, G.R.; Ganyukov, V.; Wojakowski, W.; et al. Impact of COVID-19 Pandemic on Mechanical Reperfusion for Patients With STEMI. J. Am. Coll. Cardiol. 2020, 76, 2321–2330. [Google Scholar] [CrossRef]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 2020, 323, 1061–1069. [Google Scholar] [CrossRef]
- Cai, H. Sex difference and smoking predisposition in patients with COVID-19. Lancet Respir Med. 2020, 8, e20. [Google Scholar] [CrossRef]
- Ferrante, G.; Barbieri, L.; Sponzilli, C.; Lucreziotti, S.; Salerno Uriarte, D.; Centola, M.; Verdoia, M.; Carugo, S. Predictors of Mortality and Long-Term Outcome in Patients with Anterior STEMI: Results from a Single Center Study. J. Clin. Med. 2021, 10, 5634. [Google Scholar] [CrossRef] [PubMed]
- Verdoia, M.; Suryapranata, H.; Damen, S.; Camaro, C.; Benit, E.; Barbieri, L.; Rasoul, S.; Liew, H.B.; Polad, J.; Ahmad, W.A.W.; et al. Gender differences with short-term vs 12 months dual antiplatelet therapy in patients with acute coronary syndrome treated with the COMBO dual therapy stent: 2-years follow-up results of the REDUCE trial. J. Thromb. Thrombolysis 2021, 52, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Silverio, A.; Di Maio, M.; Citro, R.; Esposito, L.; Iuliano, G.; Bellino, M.; Baldi, C.; De Luca, G.; Ciccarelli, M.; Vecchione, C.; et al. Cardiovascular risk factors and mortality in hospitalized patients with COVID-19: Systematic review and meta-analysis of 45 studies and 18,300 patients. BMC Cardiovasc. Disord. 2021, 21, 23. [Google Scholar] [CrossRef] [PubMed]
- Gelman, A.; Hill, J. Data Analysis Using Regression and Multilevel/Hierarchical Models; Cambridge University Press: New York, NY, USA, 2007. [Google Scholar]
- De Luca, G.; Suryapranata, H.; Dambrink, J.H.; Ottervanger, J.P.; van ‘t Hof, A.W.; Zijlstra, F.; Hoorntje, J.C.; Gosselink, A.T.; de Boer, M.J. Sex-related differences in outcome after ST-segment elevation myocardial infarction treated by primary angioplasty: Data from the Zwolle Myocardial Infarction study. Am. Heart J. 2004, 148, 852–856. [Google Scholar] [CrossRef]
- Paradossi, U.; Taglieri, N.; Massarelli, G.; Palmieri, C.; De Caterina, A.R.; Bruno, A.G.; Taddei, A.; Nardi, E.; Ghetti, G.; Palmerini, T.; et al. Female gender and mortality in ST-segment-elevation myocardial infarction treated with primary PCI. J. Cardiovasc. Med. 2022, 23, 234–241. [Google Scholar] [CrossRef]
- De Luca, G.; Parodi, G.; Sciagrà, R.; Bellandi, B.; Verdoia, M.; Vergara, R.; Migliorini, A.; Valenti, R.; Antoniucci, D. Relation of gender to infarct size in patients with ST-segment elevation myocardial infarction undergoing primary angioplasty. Am. J. Cardiol. 2013, 111, 936–940. [Google Scholar] [CrossRef]
- De Luca, G.; Gibson, C.M.; Gyöngyösi, M.; Zeymer, U.; Dudek, D.; Arntz, H.R.; Bellandi, F.; Maioli, M.; Noc, M.; Zorman, S.; et al. Gender-related differences in outcome after ST-segment elevation myocardial infarction treated by primary angioplasty and glycoprotein IIb-IIIa inhibitors: Insights from the EGYPT cooperation. J. Thromb. Thrombolysis 2010, 30, 342–346. [Google Scholar] [CrossRef]
- De Luca, G.; Verdoia, M.; Dirksen, M.T.; Spaulding, C.; Kelbæk, H.; Schalij, M.; Thuesen, L.; Hoeven Bv Vink, M.A.; Kaiser, C.; Musto, C.; et al. Gender-related differences in outcome after BMS or DES implantation in patients with ST-segment elevation myocardial infarction treated by primary angioplasty: Insights from the DESERT cooperation. Atherosclerosis 2013, 230, 3056. [Google Scholar] [CrossRef]
- Chew, N.W.; Sia, C.H.; Wee, H.L.; Benedict, L.J.; Rastogi, S.; Kojodjojo, P.; Chor, W.P.D.; Leong, B.S.; Koh, B.C.; Tam, H.; et al. Impact of the COVID-19 Pandemic on Door-to-Balloon Time for Primary Percutaneous Coronary Intervention–Results From the Singapore Western STEMI Network. Circ. J. 2021, 85, 139–149. [Google Scholar] [CrossRef]
- Xiang, D.; Xiang, X.; Zhang, W.; Yi, S.; Zhang, J.; Gu, X.; Xu, Y.; Huang, K.; Su, X.; Yu, B.; et al. Management and Outcomes of Patients With STEMI During the COVID-19 Pandemic in China. J. Am. Coll. Cardiol. 2020, 76, 1318–1324. [Google Scholar] [CrossRef]
- De Luca, G.; Cercek, M.; Jensen, L.O.; Vavlukis, M.; Calmac, L.; Johnson, T.; Roura IFerrer, G.; Ganyukov, V.; Wojakowski, W.; von Birgelen, C.; et al. Impact of COVID-19 pandemic and diabetes on mechanical reperfusion in patients with STEMI: Insights from the ISACS STEMI COVID 19 Registry. Cardiovasc. Diabetol. 2020, 19, 215. [Google Scholar] [CrossRef] [PubMed]
- Rangé, G.; Hakim, R.; Beygui, F.; Angoulvant, D.; Marcollet, P.; Godin, M.; Deballon, R.; Bonnet, P.; Fichaux, O.; Barbey, C.; et al. Incidence, delays, and outcomes of STEMI during COVID-19 outbreak: Analysis from the France PCI registry. J. Am. Coll. Emerg. Physicians Open 2020, 1, 1168–1176. [Google Scholar] [CrossRef] [PubMed]
- Meisel, S.R.; Tal, O.; Kobo, O.; Saada, M.; Nashed, H.; Fanne, R.A.; Alcalai, R.; Amsalem, N.; Levi, Y.; Mohsen, J.; et al. Impact of the Admission Pathway on the Gender-Related Mortality of Patients With ST-Elevation Myocardial Infarction. Am. J. Cardiol. 2022, 166, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Verdoia, M.; Viglione, F.; Boggio, A.; Stefani, D.; Panarotto, N.; Malabaila, A.; Rolla, R.; Soldà, P.L.; De Luca, G. Novara Atherosclerosis Study Group (NAS). Vitamin D deficiency is associated with impaired reperfusion in STEMI patients undergoing primary percutaneous coronary intervention. Vasc. Pharmacol. 2021, 140, 106897. [Google Scholar] [CrossRef]
- Mo, P.; Xing, Y.; Xiao, Y.; Deng, L.; Zhao, Q.; Wang, H.; Xiong, Y.; Cheng, Z.; Gao, S.; Liang, K.; et al. Clinical characteristics of refractory COVID-19 pneumonia in Wuhan, China. Clin. Infect. Dis. 2021, 73, e4208–e4213. [Google Scholar] [CrossRef]
- Gebhard, C.; Regitz-Zagrosek, V.; Neuhauser, H.K.; Morgan, R.; Klein, S.L. Impact of sex and gender on COVID-19 outcomes in Europe. Biol. Sex. Differ. 2020, 11, 29. [Google Scholar] [CrossRef]
- Galloway, J.B.; Norton, S.; Barker, R.D.; Brookes, A.; Carey, I.; Clarke, B.D.; Jina, R.; Reid, C.; Russell, M.D.; Sneep, R.; et al. A clinical risk score to identify patients with COVID-19 at high risk of critical care admission or death: An observational cohort study. J. Infect. 2020, 81, 282–288. [Google Scholar] [CrossRef]
- Megiorni, F.; Pontecorvi, P.; Gerini, G.; Anastasiadou, E.; Marchese, C.; Ceccarelli, S. Sex-Related Factors in Cardiovascular Complications Associated to COVID-19; Sex differences underlying preexisting cardiovascular disease and cardiovascular injury in COVID-19. Biomolecules 2021, 12, 21. [Google Scholar] [CrossRef]
- Fischer, M.; Baessler, A.; Schunkert, H. Renin angiotensin system and gender differences in the cardiovascular system. Cardiovasc. Res. 2002, 53, 672–677. [Google Scholar] [CrossRef]
- Chappell, M.C.; Marshall, A.C.; Alzayadneh, E.M.; Shaltout, H.A.; Diz, D.I. Update on the angiotensin converting enzyme 2-angiotensin (1-7)-Mas receptor axis: Fetal programing, sex differences, and intracellular pathways. Front. Endocrinol. 2014, 4, 201. [Google Scholar] [CrossRef]
- De Rosa, S.; Spaccarotella, C.; Basso, C.; Calabrò, M.P.; Curcio, A.; Filardi, P.P.; Mancone, M.; Mercuro, G.; Muscoli, S.; Nodari, S.; et al. Reduction of hospitalizations for myocardial infarction in Italy in the COVID-19 era. Società Italiana di Cardiologia and the CCU Academy investigators group. Eur. Heart J. 2020, 41, 2083–2088. [Google Scholar] [CrossRef] [PubMed]
- Barbero, U.; Moncalvo, C.; Trabattoni, D.; Pavani, M.; Amoroso, G.R.; Bocchino, P.P.; Giachet, A.T.; Saglietto, A.; Monticone, S.; Secco, G.G.; et al. Gender differences in acute coronary syndromes patterns during the COVID-19 outbreak. Am. J. Cardiovasc. Dis. 2020, 10, 506–513. [Google Scholar] [PubMed]
- Huynh, J.; Barmano, N.; Karlsson, J.E.; Stomby, A. Sex and age differences in the incidence of acute myocardial infarction during the COVID-19 pandemic in a Swedish health-care region without lockdown: A retrospective cohort study. Lancet 2021, 2, e283–e289. [Google Scholar] [CrossRef] [PubMed]

| 2019 Males (n = 6571) | 2020 Males (n = 5593) | p-Value | 2019 Females (n = 2127) | 2020 Females (n = 1792) | p-Value | ||
|---|---|---|---|---|---|---|---|
| Age—median [IQR] | 61 [53–69] | 61 [53–70] | 0.85 | 68 [59–79] | 67 [58–78] | 0.01 * | |
| Elderly (>75 y)—n (%) | 967 (14.7) | 805 (14.4) | 0.624 | 715 (33.6) | 560 (31.3) | 0.615 | |
| Medical History | |||||||
| Hypertension—n (%) | 3375 (51.4) | 2896 (51.8) | 0.647 | 1370 (64.4) | 1172 (65.4) | 0.517 | |
| Diabetes mellitus—n (%) | 1458 (22.2) | 1228 (22) | 0.758 | 580 (27.3) | 546 (30.5) | 0.027 | |
| Hypercholesterolemia—n (%) | 2591 (39.4) | 2150 (38.4) | 0.265 | 854 (40.2) | 758 (42.3) | 0.173 | |
| Smoker—n (%) | 3972 (60.4) | 3233 (57.8) | 0.003 | 857 (40.3) | 724 (40.4) | 0.944 | |
| Family history of CAD—n (%) | 1409 (21.4) | 1112 (19.9) | 0.034 | 426 (20) | 351 (19.6) | 0.730 | |
| Previous STEMI—n (%) | 680 (10.3) | 575 (10.3) | 0.903 | 152 (7.1) | 136 (7.6) | 0.596 | |
| Previous PCI—n (%) | 855 (13) | 781 (14) | 0.125 | 183 (8.6) | 174 (9.7) | 0.231 | |
| Previous CABG—n (%) | 120 (1.8) | 98 (1.8) | 0.759 | 24 (1.1) | 30 (1.7) | 0.144 | |
| Geographic area | 0.001 | 0.089 | |||||
| Europe—n (%) | 5201 (79.2) | 4378 (78.3) | 1782 (83.8) | 1453 (81.1) | |||
| Latin America—n (%) | 446 (6.8) | 523 (9.4) | 184 (8.7) | 197 (11) | |||
| Southeast Asia—n (%) | 613 (9.3) | 506 (9) | 93 (4.4) | 81 (4.5) | |||
| North Africa—n (%) | 311 (4.7) | 186 (3.3) | 68 (3.2) | 61 (3.4) | |||
| Referral to Primary PCI Hospital | |||||||
| Type | |||||||
| Ambulance (from community)—n (%) | 3111 (47.3) | 2680 (47.9) | 0.782 | 1051 (49.4) | 896 (50) | 0.616 | |
| Direct access to hub—n (%) | 1896 (28.9) | 1585 (28.3) | 553 (26) | 479 (26.7) | |||
| Transfer from spoke—n (%) | 1564 (23.8) | 1328 (23.7) | 523 (24.6) | 417 (23.3) | |||
| Time delays | |||||||
| Ischemia time—median [25–75th] | 190 [120–340] | 213 [129–390] | <0.001 | 215 [130–390] | 240 [140–433] | <0.001 * | |
| Total ischemia time | <0.001 | 0.015 | |||||
| <6 h—n (%) | 5078 (77.3) | 4063 (72.6) | 1544 (72.6) | 1237 (69) | |||
| 6–12 h—n (%) | 931 (14.2) | 881 (15.8) | 353 (16.6) | 334 (18.6) | |||
| 12–24 h—n (%) | 376 (5.7) | 417 (7.5) | 161 (7.6) | 134 (7.5) | |||
| >24 h—n (%) | 186 (2.8) | 232 (4.1) | 69 (3.2) | 87 (4.9) | |||
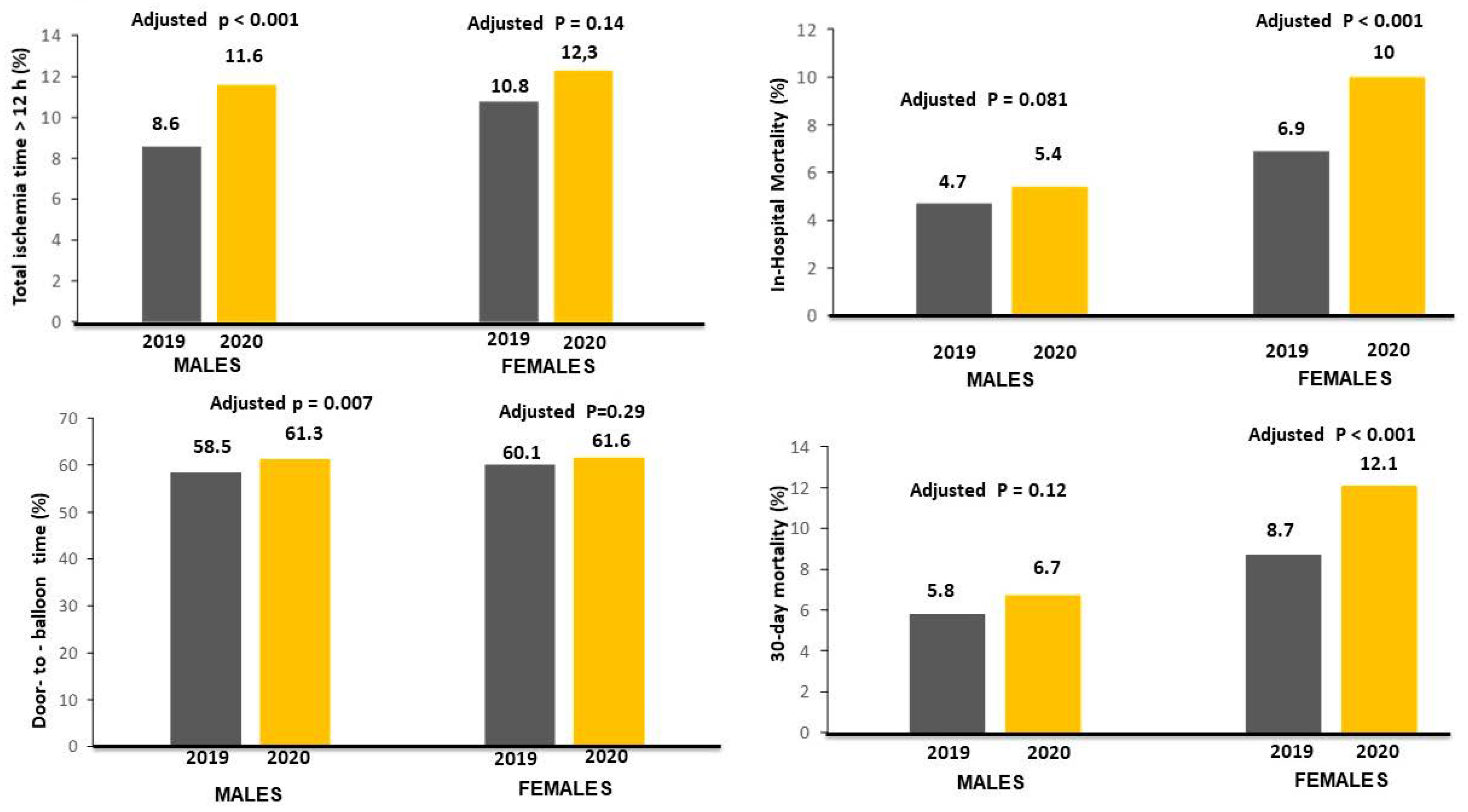
| Total ischemia time > 12 h—n (%) | 562 (8.6) | 649 (11.6) | <0.001 | 230 (10.8) | 221 (12.3) | 0.138 | |
| Door-to-balloon time—median [25—75th] | 39 [25–62] | 40 [25–70] | <0.001 | 40 [25–70] | 40 [25–70] | 0.2 * | |
| Door-to-balloon time | <0.001 | 0.556 | |||||
| <30 min—n (%) | 2730 (41.5) | 2165 (38.7) | 849 (39.9) | 689 (38.4) | |||
| 30–60 min—n (%) | 2161 (32.9) | 1836 (32.8) | 684 (32.2) | 578 (32.3) | |||
| >60 min—n (%) | 1680 (25.6) | 1592 (28.5) | 594 (27.9) | 525 (29.3) | |||
| Door-to-balloon time > 30 min—n (%) | 3841 (58.5) | 3428 (61.3) | 0.001 | 1278 (60.1) | 1103 (61.6) | 0.349 | |
| Clinical Presentation | |||||||
| Anterior STEMI—n (%) | 3036 (46.2) | 2657 (47.5) | 0.151 | 950 (44.7) | 803 (44.8) | 0.927 | |
| Out-of-hospital cardiac arrest—n (%) | 400 (6.1) | 346 (6.2) | 0.821 | 115 (5.4) | 95 (5.3) | 0.547 | |
| Cardiogenic shock—n (%) | 460 (7) | 396 (7.1) | 0.551 | 165 (7.8) | 148 (8.3) | 0.564 | |
| Rescue PCI for failed thrombolysis—n (%) | 468 (7.1) | 364 (6.5) | 0.181 | 137 (6.4) | 130 (7.3) | 0.314 | |
| 2019 Males (n = 6571) | 2020 Males (n = 5593) | p-Value | 2019 Females (n = 2127) | 2020 Females (n = 1792) | p-Value | |
|---|---|---|---|---|---|---|
| Radial access—n (%) | 4960 (75.5) | 4438 (79.3) | <0.001 | 1563 (73.5) | 1307 (72.9) | 0.699 |
| Culprit vessel | 0.395 | 0.820 | ||||
| Left main—n (%) | 110 (1.7) | 88 (1.6) | 31 (1.5) | 23 (1.3) | ||
| Left anterior descending artery—n (%) | 3025 (46.1) | 2585 (46.2) | 964 (45.3) | 786 (43.9) | ||
| Circumflex—n (%) | 967 (14.7) | 867 (15.5) | 279 (13.1) | 237 (13.2) | ||
| Right coronary artery—n (%) | 2416 (36.8) | 2007 (35.9) | 844 (39.7) | 734 (41) | ||
| Anterolateral branch—n (%) | 23 (0.4) | 13 (0.2) | 2 (0.1) | 3 (0.2) | ||
| SVG—n (%) | 30 (0.5) | 33 (0.6) | 7 (0.3) | 9 (0.5) | ||
| In-stent thrombosis—n (%) | 274 (4.2) | 227 (4.1) | 0.758 | 65 (3.1) | 66 (3.7) | 0.277 |
| Vessel disease—n (%) | 0.216 | 0.526 | ||||
| 1 | 3311 (50.4) | 2788 (49.8) | 1151 (54.1) | 947 (52.8) | ||
| 2 | 1902 (28.9) | 1695 (30.3) | 572 (26.9) | 511 (28.5) | ||
| 3 | 1358 (20.7) | 1110 (19.8) | 404 (19) | 334 (18.6) | ||
| Preprocedural TIMI 0 flow—n (%) | 4422 (67.3) | 3784 (67.7) | 0.673 | 1344 (63.2) | 1181 (65.9) | 0.077 |
| Thrombectomy—n (%) | 1115 (17) | 921 (16.5) | 0.460 | 287 (13.5) | 240 (13.4) | 0.927 |
| Stenting—n (%) | 6056 (92.2) | 5141 (91.9) | 0.620 | 1942 (91.3) | 1627 (90.8) | 0.487 |
| Drug-eluting stent—n (%) | 5814 (88.5) | 5027 (89.9) | 0.013 | 1842 (86.6) | 1571 (87.7) | 0.321 |
| Postprocedural TIMI 3 flow—n (%) | 6104 (92.9) | 5188 (92.8) | 0.775 | 1926 (90.6) | 1603 (89.5) | 0.253 |
| Gp IIb-IIIa inhibitors/cangrelor—n (%) | 1426 (21.7) | 1180 (21.1) | 0.419 | 327 (15.4) | 334 (18.6) | 0.007 |
| Bivalirudin—n (%) | 25 (0.4) | 14 (0.3) | 0.206 | 9 (0.4) | 4 (0.2) | 0.278 |
| Mechanical support—n (%) | 190 (2.9) | 192 (3.4) | 0.088 | 56 (2.6) | 59 (3.3) | 0.223 |
| Additional PCI | 0.009 | 0.046 | ||||
| During index procedure—n (%) | 605 (9.2) | 599 (10.7) | 182 (8.6) | 190 (10.6) | ||
| Staged—n (%) | 702 (10.7) | 629 (11.2) | 184 (8.7) | 171 (9.5) | ||
| DAPT therapy—n (%) | 6492 (98.8) | 5539 (99) | 0.211 | 2101 (98.8) | 1773 (98.9) | 0.635 |
| In-hospital RASI—n (%) | 3552 (54.1) | 3293 (58.9) | <0.001 | 1074 (50.5) | 978 (54.6) | 0.011 |
| In-hospital mortality—n (%) | 310 (4.7) | 302 (5.4) | 0.086 | 147 (6.9) | 179 (10) | 0.001 |
| 30-day mortality—n (%) | 343 (5.8) | 332 (6.7) | 0.074 | 161 (8.7) | 191 (12.1) | 0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Luca, G.; Manzo-Silberman, S.; Algowhary, M.; Uguz, B.; Oliveira, D.C.; Ganyukov, V.; Busljetik, O.; Cercek, M.; Okkels, L.; Loh, P.H.; et al. Gender Difference in the Effects of COVID-19 Pandemic on Mechanical Reperfusion and 30-Day Mortality for STEMI: Results of the ISACS-STEMI COVID-19 Registry. J. Clin. Med. 2023, 12, 896. https://doi.org/10.3390/jcm12030896
De Luca G, Manzo-Silberman S, Algowhary M, Uguz B, Oliveira DC, Ganyukov V, Busljetik O, Cercek M, Okkels L, Loh PH, et al. Gender Difference in the Effects of COVID-19 Pandemic on Mechanical Reperfusion and 30-Day Mortality for STEMI: Results of the ISACS-STEMI COVID-19 Registry. Journal of Clinical Medicine. 2023; 12(3):896. https://doi.org/10.3390/jcm12030896
Chicago/Turabian StyleDe Luca, Giuseppe, Stephane Manzo-Silberman, Magdy Algowhary, Berat Uguz, Dinaldo C. Oliveira, Vladimir Ganyukov, Oliver Busljetik, Miha Cercek, Lisette Okkels, Poay Huan Loh, and et al. 2023. "Gender Difference in the Effects of COVID-19 Pandemic on Mechanical Reperfusion and 30-Day Mortality for STEMI: Results of the ISACS-STEMI COVID-19 Registry" Journal of Clinical Medicine 12, no. 3: 896. https://doi.org/10.3390/jcm12030896
APA StyleDe Luca, G., Manzo-Silberman, S., Algowhary, M., Uguz, B., Oliveira, D. C., Ganyukov, V., Busljetik, O., Cercek, M., Okkels, L., Loh, P. H., Calmac, L., Ferrer, G. R. i., Quadros, A., Milewski, M., Scotto di Uccio, F., von Birgelen, C., Versaci, F., Ten Berg, J., Casella, G., ... Verdoia, M. (2023). Gender Difference in the Effects of COVID-19 Pandemic on Mechanical Reperfusion and 30-Day Mortality for STEMI: Results of the ISACS-STEMI COVID-19 Registry. Journal of Clinical Medicine, 12(3), 896. https://doi.org/10.3390/jcm12030896













