Immediate Effects of Instrument-Assisted Soft Tissue Mobilization on Hydration Content in Lumbar Myofascial Tissues: A Quasi-Experiment
Abstract
1. Introduction
2. Materials and Methods
2.1. Setting and Participants
2.2. Study Flow
2.3. Intervention
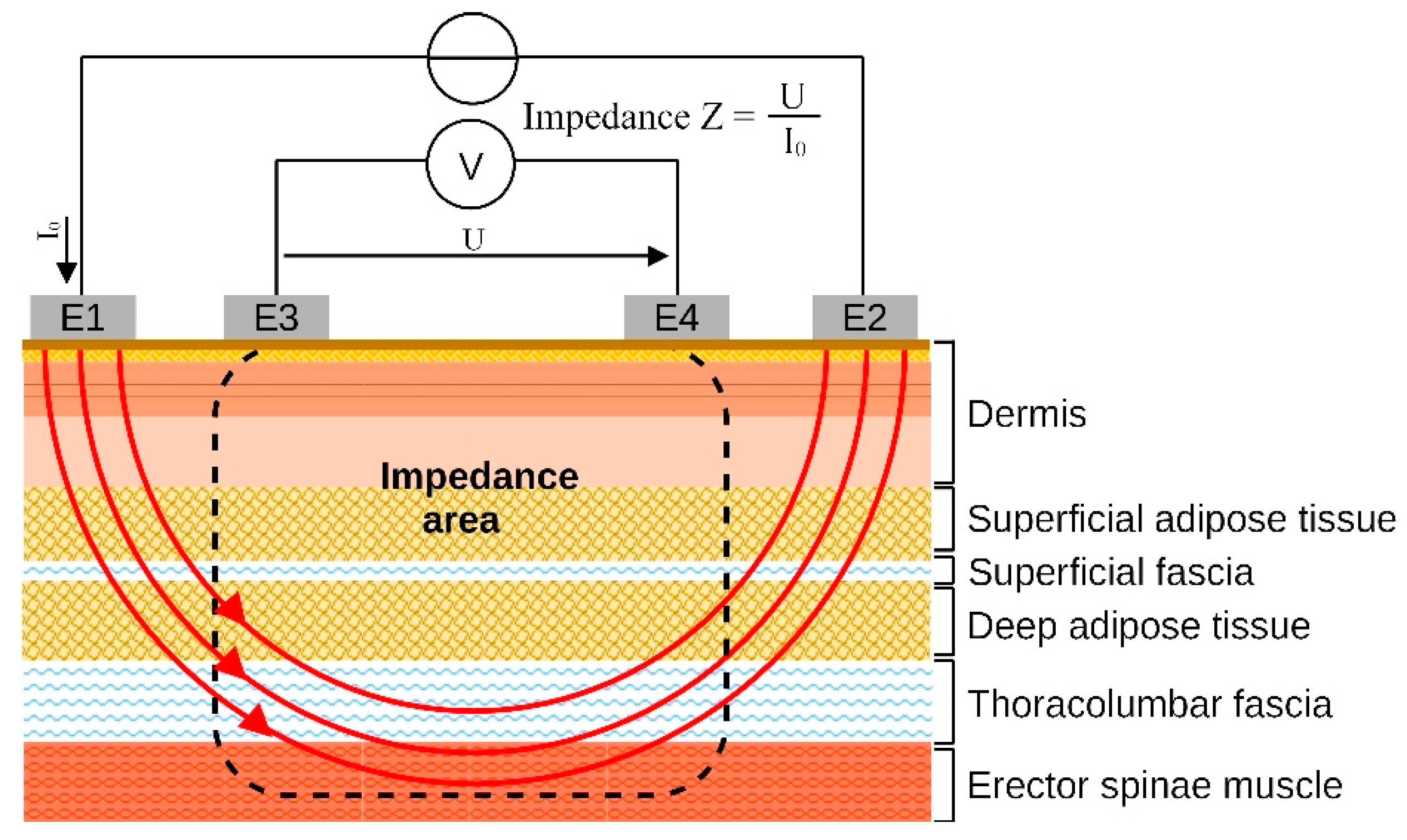
2.4. Outcomes
2.5. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Willard, F.H.; Vleeming, A.; Schuenke, M.D.; Danneels, L.; Schleip, R. The Thoracolumbar Fascia: Anatomy, Function and Clinical Considerations. J. Anat. 2012, 221, 507–536. [Google Scholar] [CrossRef] [PubMed]
- Langevin, H.M.; Fox, J.R.; Koptiuch, C.; Badger, G.J.; Greenan- Naumann, A.C.; Bouffard, N.A.; Konofagou, E.E.; Lee, W.-N.; Triano, J.J.; Henry, S.M. Reduced Thoracolumbar Fascia Shear Strain in Human Chronic Low Back Pain. BMC Musculoskelet. Disord. 2011, 12, 203. [Google Scholar] [CrossRef] [PubMed]
- Schleip, R.; Gabbiani, G.; Wilke, J.; Naylor, I.; Hinz, B.; Zorn, A.; Jäger, H.; Breul, R.; Schreiner, S.; Klingler, W. Fascia Is Able to Actively Contract and May Thereby Influence Musculoskeletal Dynamics: A Histochemical and Mechanographic Investigation. Front. Physiol. 2019, 10, 336. [Google Scholar] [CrossRef] [PubMed]
- Brandl, A.; Egner, C.; Schleip, R. Immediate Effects of Myofascial Release on the Thoracolumbar Fascia and Osteopathic Treatment for Acute Low Back Pain on Spine Shape Parameters: A Randomized, Placebo-Controlled Trial. Life 2021, 11, 845. [Google Scholar] [CrossRef]
- Mense, S. Innervation of the Thoracolumbar Fascia. Eur. J. Transl. Myol. 2019, 29, 151–158. [Google Scholar] [CrossRef]
- Tesarz, J.; Hoheisel, U.; Wiedenhöfer, B.; Mense, S. Sensory Innervation of the Thoracolumbar Fascia in Rats and Humans. Neuroscience 2011, 194, 302–308. [Google Scholar] [CrossRef]
- Fede, C.; Petrelli, L.; Guidolin, D.; Porzionato, A.; Pirri, C.; Fan, C.; De Caro, R.; Stecco, C. Evidence of a New Hidden Neural Network into Deep Fasciae. Sci. Rep. 2021, 11, 12623. [Google Scholar] [CrossRef]
- Wilke, J.; Schleip, R.; Klingler, W.; Stecco, C. The Lumbodorsal Fascia as a Potential Source of Low Back Pain: A Narrative Review. BioMed Res. Int. 2017, 2017, 5349620. [Google Scholar] [CrossRef]
- Wilke, J.; Schleip, R.; Yucesoy, C.A.; Banzer, W. Not Merely a Protective Packing Organ? A Review of Fascia and Its Force Transmission Capacity. J. Appl. Physiol. 2018, 124, 234–244. [Google Scholar] [CrossRef]
- Wilke, J.; Macchi, V.; De Caro, R.; Stecco, C. Fascia Thickness, Aging and Flexibility: Is There an Association? J. Anat. 2019, 234, 43–49. [Google Scholar] [CrossRef]
- Lancerotto, L.; Stecco, C.; Macchi, V.; Porzionato, A.; Stecco, A.; De Caro, R. Layers of the Abdominal Wall: Anatomical Investigation of Subcutaneous Tissue and Superficial Fascia. Surg. Radiol. Anat. 2011, 33, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Stecco, C.; Fede, C.; Macchi, V.; Porzionato, A.; Petrelli, L.; Biz, C.; Stern, R.; De Caro, R. The Fasciacytes: A New Cell Devoted to Fascial Gliding Regulation. Clin. Anat. 2018, 31, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Streďanská, A.; Nečas, D.; Vrbka, M.; Křupka, I.; Hartl, M.; Toropitsyn, E.; Husby, J. Development of Tribological Model of Human Fascia: The Influence of Material Hardness and Motion Speed. Biotribology 2022, 30, 100209. [Google Scholar] [CrossRef]
- Paryavi, E.; Jobin, C.M.; Ludwig, S.C.; Zahiri, H.; Cushman, J. Acute Exertional Lumbar Paraspinal Compartment Syndrome. Spine 2010, 35, E1529. [Google Scholar] [CrossRef]
- Roman, M.; Chaudhry, H.; Bukiet, B.; Stecco, A.; Findley, T.W. Mathematical Analysis of the Flow of Hyaluronic Acid Around Fascia During Manual Therapy Motions. JOM 2013, 113, 600–610. [Google Scholar] [CrossRef]
- Stanek, J.; Sullivan, T.; Davis, S. Comparison of Compressive Myofascial Release and the Graston Technique for Improving Ankle-Dorsiflexion Range of Motion. J. Athl. Train. 2018, 53, 160–167. [Google Scholar] [CrossRef]
- Bertuccioli, A.; Cardinali, M.; Benelli, P. Segmental Bioimpedance Analysis as a Predictor of Injury and Performance Status in Professional Basketball Players: A New Application Potential? Life 2022, 12, 1062. [Google Scholar] [CrossRef]
- Fish, R.M.; Geddes, L.A. Conduction of Electrical Current to and through the Human Body: A Review. Eplasty 2009, 9, e44. [Google Scholar]
- Khalil, S.F.; Mohktar, M.S.; Ibrahim, F. The Theory and Fundamentals of Bioimpedance Analysis in Clinical Status Monitoring and Diagnosis of Diseases. Sensors 2014, 14, 10895–10928. [Google Scholar] [CrossRef]
- Buendia, R.; Seoane, F.; Lindecrantz, K.; Bosaeus, I.; Gil-Pita, R.; Johannsson, G.; Ellegård, L.; Ward, L.C. Estimation of Body Fluids with Bioimpedance Spectroscopy: State of the Art Methods and Proposal of Novel Methods. Physiol. Meas. 2015, 36, 2171–2187. [Google Scholar] [CrossRef]
- Martinsen, O.G.; Grimnes, S. Bioimpedance and Bioelectricity Basics; Academic Press: Cambridge, MA, USA, 2011; ISBN 978-0-08-056880-5. [Google Scholar]
- Norman, K.; Stobäus, N.; Pirlich, M.; Bosy-Westphal, A. Bioelectrical Phase Angle and Impedance Vector Analysis—Clinical Relevance and Applicability of Impedance Parameters. Clin. Nutr. 2012, 31, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Arnold, W.D.; Taylor, R.S.; Li, J.; Nagy, J.A.; Sanchez, B.; Rutkove, S.B. Electrical Impedance Myography Detects Age-Related Muscle Change in Mice. PLoS ONE 2017, 12, e0185614. [Google Scholar] [CrossRef]
- Patil, P.M.; Kamat, D.K. Chapter 11—Embedded healthcare system based on bioimpedance analysis for identification and classification of skin diseases in Indian context. In U-Healthcare Monitoring Systems; Dey, N., Ashour, A.S., Fong, S.J., Borra, S., Eds.; Advances in Ubiquitous Sensing Applications for Healthcare; Academic Press: Cambridge, MA, USA, 2019; pp. 261–288. [Google Scholar]
- Das, L.; Das, S.; Chatterjee, J. Electrical Bioimpedance Analysis: A New Method in Cervical Cancer Screening. J. Med. Eng. 2015, 2015, e636075. [Google Scholar] [CrossRef] [PubMed]
- Dennenmoser, S.; Schleip, R.; Klingler, W. Clinical Mechanistic Research: Manual and Movement Therapy Directed at Fascia Electrical Impedance and Sonoelastography as a Tool for the Examination of Changes in Lumbar Fascia after Tissue Manipulation. J. Bodyw. Mov. Ther. 2016, 20, 145. [Google Scholar] [CrossRef]
- Schleip, R.; Duerselen, L.; Vleeming, A.; Naylor, I.L.; Lehmann-Horn, F.; Zorn, A.; Jaeger, H.; Klingler, W. Strain Hardening of Fascia: Static Stretching of Dense Fibrous Connective Tissues Can Induce a Temporary Stiffness Increase Accompanied by Enhanced Matrix Hydration. J. Bodyw. Mov. Ther. 2012, 16, 94–100. [Google Scholar] [CrossRef]
- Chan, A.-W.; Tetzlaff, J.M.; Gøtzsche, P.C.; Altman, D.G.; Mann, H.; Berlin, J.A.; Dickersin, K.; Hróbjartsson, A.; Schulz, K.F.; Parulekar, W.R. SPIRIT 2013 Explanation and Elaboration: Guidance for Protocols of Clinical Trials. BMJ 2013, 346, e7586. [Google Scholar] [CrossRef]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical Principles for Medical Research Involving Human Subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef] [PubMed]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.-G. Statistical Power Analyses Using G*Power 3.1: Tests for Correlation and Regression Analyses. Behav. Res. Methods 2009, 41, 1149–1160. [Google Scholar] [CrossRef]
- Honorato, R.de.C.; Ferraz, A.S.M.; Kassiano, W.; Carvalho, D.P.; Ceccatto, V.M. Test-Retest Reliability of Electrical Impedance Myography in Hamstrings of Healthy Young Men. J. Electromyogr. Kinesiol. 2021, 56, 102511. [Google Scholar] [CrossRef] [PubMed]
- Koch, V.; Wilke, J. Reliability of a New Indentometer Device for Measuring Myofascial Tissue Stiffness. J. Clin. Med. 2022, 11, 5194. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, N.; Miller, P.; Gemmell, H. A Theoretical Framework for the Role of Fascia in Manual Therapy. J. Bodyw. Mov. Ther. 2012, 16, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Weber, P.; Graf, C.; Klingler, W.; Weber, N.; Schleip, R. The Feasibility and Impact of Instrument-Assisted Manual Therapy (IAMT) for the Lower Back on the Structural and Functional Properties of the Lumbar Area in Female Soccer Players: A Randomised, Placebo-Controlled Pilot Study Design. Pilot Feasibility Stud. 2020, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, N.; Otsuka, S.; Kawanishi, Y.; Kawakami, Y. Effects of Instrument-Assisted Soft Tissue Mobilization on Musculoskeletal Properties. Med. Sci. Sport. Exerc. 2019, 51, 2166–2172. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, A.; Knoblauch, N.T.M.; Dobos, G.J.; Michalsen, A.; Kaptchuk, T.J. The Effect of Gua Sha Treatment on the Microcirculation of Surface Tissue: A Pilot Study in Healthy Subjects. Explore 2007, 3, 456–466. [Google Scholar] [CrossRef]
- Gordon, C.-M.; Lindner, S.M.; Birbaumer, N.; Montoya, P. Correlation between Hydration and Fascia Stiffness during a Self-Help Treatment with a Myofascial Manipulation Tool a Bioimpedance Controlled, Clinical Trial. J. Bodyw. Mov. Ther. 2015, 19, 668. [Google Scholar] [CrossRef]
- Gordon, C.-M.; Schleip, R.; Montoya, P.; Andrasik, F.; Schweisthal, M. Vibration Based Shearing Technique (Vibro-Shearing) versus Rolling Technique in Terms of Tissue Hydration, Stiffness, Elasticity, and Thermography: A Double Controlled, Standardized Study. J. Bodyw. Mov. Ther. 2018, 22, 854. [Google Scholar] [CrossRef]
- Wakker, J.; Kratzer, W.; Schmidberger, J.; Graeter, T.; Boretzki, S.; Schulze, J.; Uhlemann, S.; Heizelmann, A.; Stumpf, S.; Jäger, H.; et al. Elasticity Standard Values of the Thoracolumbar Fascia Assessed with Acoustic Radiation Force Impulse Elastography on Healthy Volunteers: A Cross Section Study. J. Bodyw. Mov. Ther. 2021, 26, 530–537. [Google Scholar] [CrossRef]
- Brandl, A.; Egner, C.; Reer, R.; Schmidt, T.; Schleip, R. Associations between Deformation of the Thoracolumbar Fascia and Activation of the Erector Spinae and Multifidus Muscle in Patients with Acute Low Back Pain and Healthy Controls: A Matched Pair Case-Control Study. Life 2022, 12, 1735. [Google Scholar] [CrossRef]
- Dubinskaya, V.A.; Eng, L.S.; Rebrow, L.B.; Bykov, V.A. Comparative Study of the State of Water in Various Human Tissues. Bull. Exp. Biol. Med. 2007, 144, 294–297. [Google Scholar] [CrossRef]



| Study Group (n = 21) | |||||||
|---|---|---|---|---|---|---|---|
| Quantitative Variable | Descriptive Statistics | ||||||
| M | Me | Min | Max | Q1 | Q3 | SD | |
| Age (years) | 41.9 | 42.0 | 18.0 | 75.0 | 36.0 | 49.0 | 14.3 |
| Height (cm) | 173.3 | 172.0 | 162.0 | 191.0 | 166.0 | 179.0 | 8.1 |
| Weight (kg) | 77.1 | 76.0 | 60.0 | 105.0 | 65.0 | 85.0 | 13.7 |
| Body Mass Index (kg/m2) | 25.5 | 25.0 | 20.0 | 33.0 | 24.0 | 27.0 | 3.4 |
| Qualitative variable | n | % | |||||
| Gender | Female | 16 | 76 | ||||
| Male | 5 | 24 | |||||
| Study Group (n = 21) | |||||||
|---|---|---|---|---|---|---|---|
| Bioimpedance (Ω) | Descriptive Statistics | ||||||
| M | Me | Min | Max | Q1 | Q3 | SD | |
| t0 | 58.3 | 57.3 | 43.0 | 86.7 | 48.5 | 66.0 | 11.1 |
| t1 | 60.0 | 58.0 | 44.3 | 86.9 | 52.1 | 66.0 | 10.3 |
| t2 | 60.0 | 58.0 | 44.8 | 86.9 | 53.0 | 66.0 | 10.1 |
| t3 | 60.4 | 58.6 | 44.9 | 87.0 | 53.0 | 67.0 | 10.2 |
| p-value (main effect) * | p < 0.001 | ||||||
| p-value (multiple comparisons) ** | t0:t1—p = 0.20 | ||||||
| t0:t2—p = 0.07 | |||||||
| t0:t3—p < 0.001 | |||||||
| t1:t2—p = 0.99 | |||||||
| t1:t3—p = 0.05 | |||||||
| t2:t3—p = 0.15 | |||||||
| Study Group (n = 21) | |||||||
|---|---|---|---|---|---|---|---|
| Tissue Stiffness (N/mm) | Descriptive Statistics | ||||||
| M | Me | Min | Max | Q1 | Q3 | SD | |
| t0 | 0.69 | 0.71 | 0.13 | 1.40 | 0.41 | 0.89 | 0.38 |
| t1 | 0.68 | 0.62 | 0.13 | 1.44 | 0.39 | 0.91 | 0.41 |
| t2 | 0.67 | 0.56 | 0.12 | 1.43 | 0.37 | 0.93 | 0.39 |
| t3 | 0.65 | 0.55 | 0.13 | 1.52 | 0.33 | 0.94 | 0.38 |
| p-value (main effect) * | p = 0.84 | ||||||
| p-value (multiple comparisons) ** | - | ||||||
| Study Group (n = 21) | |||||||
|---|---|---|---|---|---|---|---|
| Temperature (Celsius) | Descriptive Statistics | ||||||
| M | Me | Min | Max | Q1 | Q3 | SD | |
| t0 | 36.1 | 36.5 | 34.2 | 38.4 | 34.6 | 36.6 | 1.2 |
| t1 | 36.3 | 36.4 | 34.0 | 37.8 | 36.2 | 36.7 | 0.8 |
| t2 | 36.6 | 36.6 | 34.4 | 38.0 | 36.3 | 36.9 | 0.7 |
| t3 | 36.4 | 36.6 | 34.2 | 38.0 | 36.2 | 36.9 | 1.0 |
| p-value (main effect) * | p = 0.011 | ||||||
| p-value (multiple comparisons) ** | t0:t1—p = 0.99 | ||||||
| t0:t2—p = 0.06 | |||||||
| t0:t3—p = 0.58 | |||||||
| t1:t2—p = 0.029 | |||||||
| t1:t3—p = 0.41 | |||||||
| t2:t3—p = 0.86 | |||||||
| Variable | Timepoint | Bioimpedance (Ω) | Tissue Stiffness (N/mm) | Temperature (Celsius) | |||
|---|---|---|---|---|---|---|---|
| rs | p-Value | rs | p-Value | rs | p-Value | ||
| Age | t0 | 0.14 | 0.54 | 0.08 | 0.75 | −0.23 | 0.31 |
| t1 | 0.09 | 0.68 | 0.04 | 0.88 | −0.19 | 0.41 | |
| t2 | 0.12 | 0.59 | 0.14 | 0.56 | −0.12 | 0.59 | |
| t3 | 0.12 | 0.61 | 0.08 | 0.75 | −0.14 | 0.56 | |
| Height (cm) | t0 | −0.18 | 0.43 | 0.21 | 0.36 | 0.09 | 0.70 |
| t1 | −0.30 | 0.18 | 0.16 | 0.49 | 0.07 | 0.76 | |
| t2 | −0.34 | 0.13 | 0.24 | 0.30 | −0.04 | 0.87 | |
| t3 | −0.34 | 0.13 | 0.31 | 0.18 | −0.12 | 0.60 | |
| Weight (kg) | t0 | 0.08 | 0.74 | 0.07 | 0.77 | −0.17 | 0.47 |
| t1 | 0.05 | 0.83 | −0.01 | 0.96 | 0.12 | 0.59 | |
| t2 | −0.01 | 0.96 | 0.07 | 0.77 | 0.05 | 0.84 | |
| t3 | −0.01 | 0.96 | 0.11 | 0.64 | 0.03 | 0.89 | |
| Body Mass Index (kg/m2) | t0 | 0.16 | 0.50 | 0.02 | 0.91 | −0.27 | 0.23 |
| t1 | 0.20 | 0.39 | −0.05 | 0.82 | 0.16 | 0.50 | |
| t2 | 0.15 | 0.52 | −0.01 | 0.97 | 0.12 | 0.60 | |
| t3 | 0.15 | 0.53 | 0.02 | 0.95 | 0.16 | 0.49 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brandl, A.; Egner, C.; Schwarze, M.; Reer, R.; Schmidt, T.; Schleip, R. Immediate Effects of Instrument-Assisted Soft Tissue Mobilization on Hydration Content in Lumbar Myofascial Tissues: A Quasi-Experiment. J. Clin. Med. 2023, 12, 1009. https://doi.org/10.3390/jcm12031009
Brandl A, Egner C, Schwarze M, Reer R, Schmidt T, Schleip R. Immediate Effects of Instrument-Assisted Soft Tissue Mobilization on Hydration Content in Lumbar Myofascial Tissues: A Quasi-Experiment. Journal of Clinical Medicine. 2023; 12(3):1009. https://doi.org/10.3390/jcm12031009
Chicago/Turabian StyleBrandl, Andreas, Christoph Egner, Monique Schwarze, Rüdiger Reer, Tobias Schmidt, and Robert Schleip. 2023. "Immediate Effects of Instrument-Assisted Soft Tissue Mobilization on Hydration Content in Lumbar Myofascial Tissues: A Quasi-Experiment" Journal of Clinical Medicine 12, no. 3: 1009. https://doi.org/10.3390/jcm12031009
APA StyleBrandl, A., Egner, C., Schwarze, M., Reer, R., Schmidt, T., & Schleip, R. (2023). Immediate Effects of Instrument-Assisted Soft Tissue Mobilization on Hydration Content in Lumbar Myofascial Tissues: A Quasi-Experiment. Journal of Clinical Medicine, 12(3), 1009. https://doi.org/10.3390/jcm12031009







