Primary Thyroid Dysfunction Is Prevalent in Hidradenitis Suppurativa and Marked by a Signature of Hypothyroid Graves’ Disease: A Case–Control Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Patients and Data Collection
2.3. Laborarory Investigation
2.4. Calculation of Structural Parameters of Thyroid Homeostasis
2.5. Statistical Analysis
3. Results
3.1. Personal and Clinical Characteristics of HS Patients
3.2. Comparison of HS Patients with Control Group
3.3. Comparison of Characteristics from HS Patients with and without Hypothyroidism
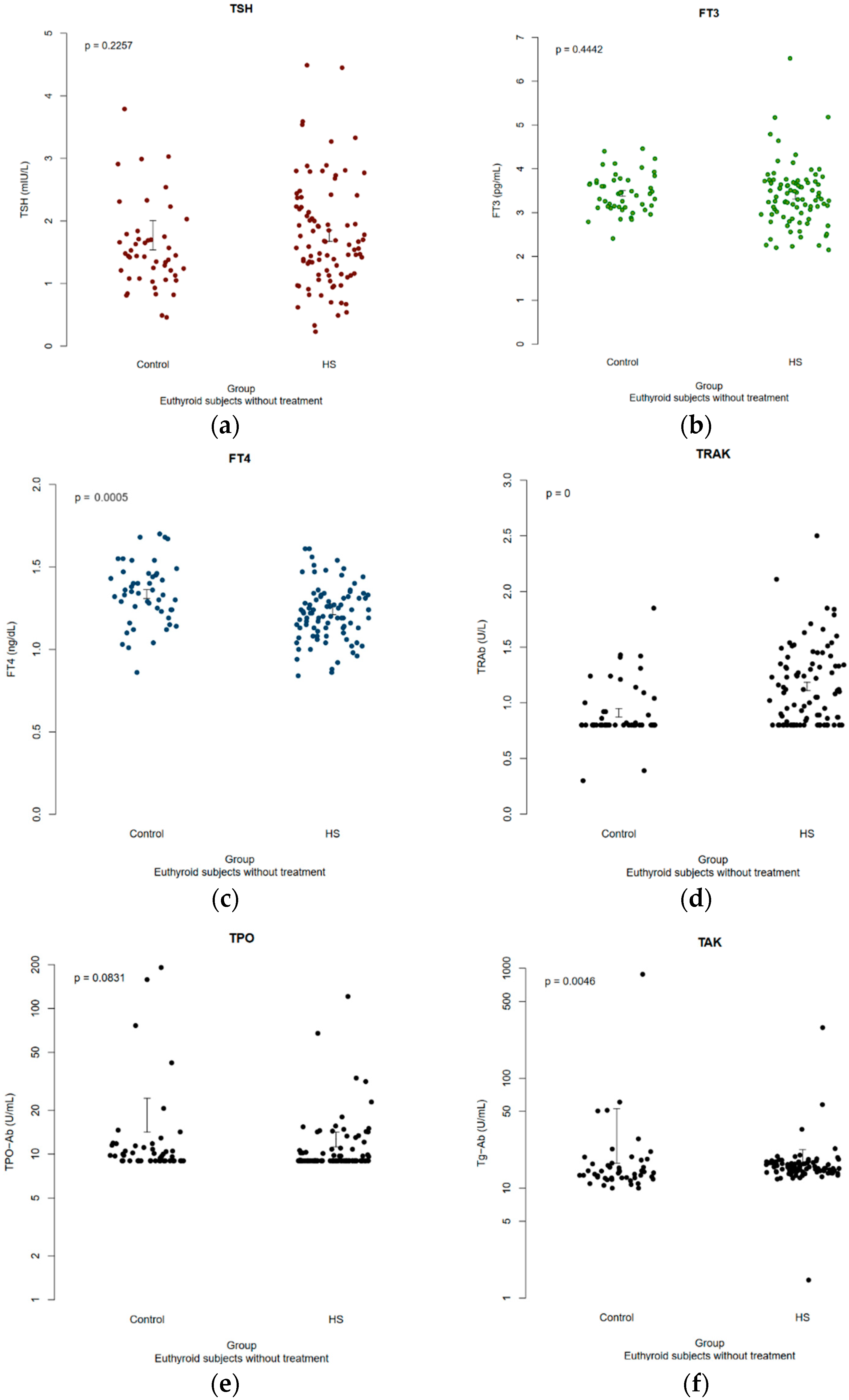
3.4. Comparison of Euthyroid HS Patients with Healthy Controls
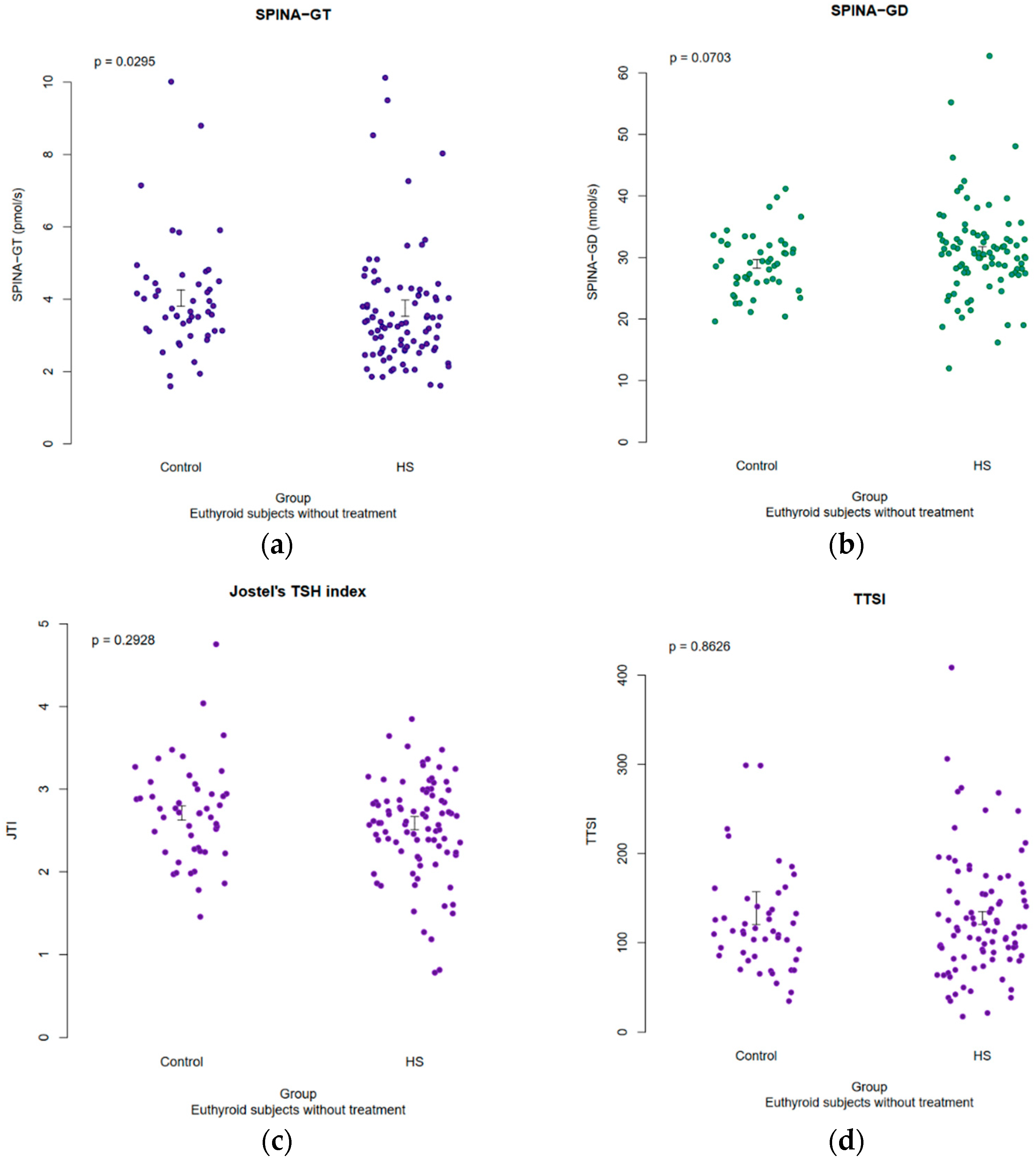
3.5. Results of Structural Parameters of Thyroid Homeostasis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A

References
- Sabat, R.; Jemec, G.B.E.; Matusiak, Ł.; Kimball, A.B.; Prens, E.; Wolk, K. Hidradenitis suppurativa. Nat. Rev. Dis. Primers 2020, 6, 18. [Google Scholar] [CrossRef] [PubMed]
- Scala, E.; Cacciapuoti, S.; Garzorz-Stark, N.; Megna, M.; Marasca, C.; Seiringer, P.; Volz, T.; Eyerich, K.; Fabbrocini, G. Hidradenitis Suppurativa: Where We Are and Where We Are Going. Cells 2021, 10, 2094. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.V.; Damiani, G.; Orenstein, L.A.V.; Hamzavi, I.; Jemec, G.B. Hidradenitis suppurativa: An update on epidemiology, phenotypes, diagnosis, pathogenesis, comorbidities and quality of life. J. Eur. Acad. Dermatol. Venereol. 2021, 35, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Abu Rached, N.; Gambichler, T.; Dietrich, J.W.; Ocker, L.; Seifert, C.; Stockfleth, E.; Bechara, F.G. The Role of Hormones in Hidradenitis Suppurativa: A Systematic Review. Int. J. Mol. Sci. 2022, 23, 15250. [Google Scholar] [CrossRef] [PubMed]
- Ingram, J.R. The Genetics of Hidradenitis Suppurativa. Dermatol. Clin. 2016, 34, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Vossen, A.R.J.V.; van der Zee, H.H.; Prens, E.P. Hidradenitis Suppurativa: A Systematic Review Integrating Inflammatory Pathways Into a Cohesive Pathogenic Model. Front. Immunol. 2018, 9, 2965. [Google Scholar] [CrossRef] [PubMed]
- Kelly, G.; Sweeney, C.M.; Tobin, A.-M.; Kirby, B. Hidradenitis suppurativa: The role of immune dysregulation. Int. J. Dermatol. 2014, 53, 1186–1196. [Google Scholar] [CrossRef]
- Prens, E.; Deckers, I. Pathophysiology of hidradenitis suppurativa: An update. J. Am. Acad. Dermatol. 2015, 73, S8–S11. [Google Scholar] [CrossRef]
- Amat-Samaranch, V.; Agut-Busquet, E.; Vilarrasa, E.; Puig, L. New perspectives on the treatment of hidradenitis suppurativa. Ther. Adv. Chronic Dis. 2021, 12, 20406223211055920. [Google Scholar] [CrossRef]
- Markota Čagalj, A.; Marinović, B.; Bukvić Mokos, Z. New and Emerging Targeted Therapies for Hidradenitis Suppurativa. Int. J. Mol. Sci. 2022, 23, 3753. [Google Scholar] [CrossRef]
- Abu Rached, N.; Gambichler, T.; Ocker, L.; Dietrich, J.W.; Quast, D.R.; Sieger, C.; Seifert, C.; Scheel, C.; Bechara, F.G. Screening for Diabetes Mellitus in Patients with Hidradenitis Suppurativa-A Monocentric Study in Germany. Int. J. Mol. Sci. 2023, 24, 6596. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-T.; Chi, C.-C. Association of Hidradenitis Suppurativa with Inflammatory Bowel Disease: A Systematic Review and Meta-analysis. JAMA Dermatol. 2019, 155, 1022–1027. [Google Scholar] [CrossRef] [PubMed]
- Wertenteil, S.; Strunk, A.; Garg, A. Overall and subgroup prevalence of acne vulgaris among patients with hidradenitis suppurativa. J. Am. Acad. Dermatol. 2019, 80, 1308–1313. [Google Scholar] [CrossRef] [PubMed]
- Richette, P.; Molto, A.; Viguier, M.; Dawidowicz, K.; Hayem, G.; Nassif, A.; Wendling, D.; Aubin, F.; Lioté, F.; Bachelez, H. Hidradenitis suppurativa associated with spondyloarthritis—Results from a multicenter national prospective study. J. Rheumatol. 2014, 41, 490–494. [Google Scholar] [CrossRef] [PubMed]
- Kridin, K.; Shavit, E.; Damiani, G.; Cohen, A.D. Hidradenitis suppurativa and rheumatoid arthritis: Evaluating the bidirectional association. Immunol. Res. 2021, 69, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Phan, K.; Huo, Y.R.; Charlton, O.; Smith, S.D. Hidradenitis Suppurativa and Thyroid Disease: Systematic Review and Meta-Analysis. J. Cutan. Med. Surg. 2020, 24, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Acharya, P.; Mathur, M. Thyroid disorders in patients with hidradenitis suppurativa: A systematic review and meta-analysis. J. Am. Acad. Dermatol. 2020, 82, 491–493. [Google Scholar] [CrossRef]
- Mancini, A.; Di Segni, C.; Raimondo, S.; Olivieri, G.; Silvestrini, A.; Meucci, E.; Currò, D. Thyroid Hormones, Oxidative Stress, and Inflammation. Mediat. Inflamm. 2016, 2016, 6757154. [Google Scholar] [CrossRef]
- Jara, E.L.; Muñoz-Durango, N.; Llanos, C.; Fardella, C.; González, P.A.; Bueno, S.M.; Kalergis, A.M.; Riedel, C.A. Modulating the function of the immune system by thyroid hormones and thyrotropin. Immunol. Lett. 2017, 184, 76–83. [Google Scholar] [CrossRef]
- De Vito, P.; Incerpi, S.; Pedersen, J.Z.; Luly, P.; Davis, F.B.; Davis, P.J. Thyroid hormones as modulators of immune activities at the cellular level. Thyroid 2011, 21, 879–890. [Google Scholar] [CrossRef]
- Abu Rached, N.; Gambichler, T.; Ocker, L.; Skrygan, M.; Seifert, C.; Scheel, C.H.; Stockfleth, E.; Bechara, F.G. Haptoglobin is an independent marker for disease severity and risk for metabolic complications in hidradenitis suppurativa: A prospective study. J. Eur. Acad. Dermatol. Venereol. 2023. [Google Scholar] [CrossRef] [PubMed]
- Kohorst, J.J.; Kimball, A.B.; Davis, M.D.P. Systemic associations of hidradenitis suppurativa. J. Am. Acad. Dermatol. 2015, 73, S27–S35. [Google Scholar] [CrossRef] [PubMed]
- Frew, J.W. Hidradenitis suppurativa is an autoinflammatory keratinization disease: A review of the clinical, histologic, and molecular evidence. JAAD Int. 2020, 1, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Zouboulis, C.C.; Del Marmol, V.; Mrowietz, U.; Prens, E.P.; Tzellos, T.; Jemec, G.B.E. Hidradenitis Suppurativa/Acne Inversa: Criteria for Diagnosis, Severity Assessment, Classification and Disease Evaluation. Dermatology 2015, 231, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Hessam, S.; Scholl, L.; Sand, M.; Schmitz, L.; Reitenbach, S.; Bechara, F.G. A Novel Severity Assessment Scoring System for Hidradenitis Suppurativa. JAMA Dermatol. 2018, 154, 330–335. [Google Scholar] [CrossRef] [PubMed]
- Sartorius, K.; Lapins, J.; Emtestam, L.; Jemec, G.B.E. Suggestions for uniform outcome variables when reporting treatment effects in hidradenitis suppurativa. Br. J. Dermatol. 2003, 149, 211–213. [Google Scholar] [CrossRef]
- Roenigk, R.K.; Roenigk, H.H. Roenigk & Roenigk’s Dermatologic Surgery: Principles and Practice, 2nd ed.; M. Dekker: New York, NY, USA, 1996. [Google Scholar]
- Finlay, A.Y.; Khan, G.K. Dermatology Life Quality Index (DLQI)—A simple practical measure for routine clinical use. Clin. Exp. Dermatol. 1994, 19, 210–216. [Google Scholar] [CrossRef]
- Mahdavi, M.; Amouzegar, A.; Mehran, L.; Madreseh, E.; Tohidi, M.; Azizi, F. Investigating the prevalence of primary thyroid dysfunction in obese and overweight individuals: Tehran thyroid study. BMC Endocr. Disord. 2021, 21, 89. [Google Scholar] [CrossRef]
- Wang, X.; Ji, X. Sample Size Estimation in Clinical Research: From Randomized Controlled Trials to Observational Studies. Chest 2020, 158, S12–S20. [Google Scholar] [CrossRef]
- Ji, X.; Wang, X. Sample Size Formulas for Different Study Designs. Available online: https://riskcalc.org/samplesize/SampleSizeEstimation_Supplement_Chest2020.pdf (accessed on 3 November 2023).
- Dietrich, J.W.; Landgrafe-Mende, G.; Wiora, E.; Chatzitomaris, A.; Klein, H.H.; Midgley, J.E.M.; Hoermann, R. Calculated Parameters of Thyroid Homeostasis: Emerging Tools for Differential Diagnosis and Clinical Research. Front. Endocrinol. 2016, 7, 57. [Google Scholar] [CrossRef]
- Hoermann, R.; Midgley, J.E.M.; Larisch, R.; Dietrich, J.W. Individualised requirements for optimum treatment of hypothyroidism: Complex needs, limited options. Drugs Context 2019, 8, 212597. [Google Scholar] [CrossRef]
- Persani, L.; Brabant, G.; Dattani, M.; Bonomi, M.; Feldt-Rasmussen, U.; Fliers, E.; Gruters, A.; Maiter, D.; Schoenmakers, N.; van Trotsenburg, A.S.P. 2018 European Thyroid Association (ETA) Guidelines on the Diagnosis and Management of Central Hypothyroidism. Eur. Thyroid J. 2018, 7, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Jostel, A.; Ryder, W.D.J.; Shalet, S.M. The use of thyroid function tests in the diagnosis of hypopituitarism: Definition and evaluation of the TSH Index. Clin. Endocrinol. 2009, 71, 529–534. [Google Scholar] [CrossRef] [PubMed]
- Khattak, R.M.; Ittermann, T.; Nauck, M.; Below, H.; Völzke, H. Monitoring the prevalence of thyroid disorders in the adult population of Northeast Germany. Popul. Health Metr. 2016, 14, 39. [Google Scholar] [CrossRef] [PubMed]
- Garmendia Madariaga, A.; Santos Palacios, S.; Guillén-Grima, F.; Galofré, J.C. The incidence and prevalence of thyroid dysfunction in Europe: A meta-analysis. J. Clin. Endocrinol. Metab. 2014, 99, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Sherman, S.; Tzur Bitan, D.; Kridin, K.; Pavlovsky, L.; Hodak, E.; Cohen, A.D. Hidradenitis suppurativa is associated with hypothyroidism and hyperthyroidism: A large-scale population-based study. Int. J. Dermatol. 2021, 60, 321–326. [Google Scholar] [CrossRef] [PubMed]
- González-López, M.A.; Hernández, J.L.; Vilanova, I.; Mata, C.; López-Escobar, M.; González-Vela, M.C.; López-Hoyos, M.; González-Gay, M.A.; Blanco, R. Thyroid autoimmunity in patients with hidradenitis suppurativa: A case-control study. Clin. Exp. Dermatol. 2017, 42, 642–644. [Google Scholar] [CrossRef] [PubMed]
- Quadbeck, B.; Roggenbuck, U.; Janssen, O.E.; Hahn, S.; Mann, K.; Hoermann, R. Impact of smoking on the course of Graves’ disease after withdrawal of antithyroid drugs. Exp. Clin. Endocrinol. Diabetes 2006, 114, 406–411. [Google Scholar] [CrossRef]
- Takasu, N.; Oshiro, C.; Akamine, H.; Komiya, I.; Nagata, A.; Sato, Y.; Yoshimura, H.; Ito, K. Thyroid-stimulating antibody and TSH-binding inhibitor immunoglobulin in 277 Graves’ patients and in 686 normal subjects. J. Endocrinol. Investig. 1997, 20, 452–461. [Google Scholar] [CrossRef]
- Miao, L.Y.; Kim, H.J.; Whitlatch, K.; Jaiswal, D.; Navarro, A.; Egan, R.; Olivo, P.D. A rapid homogenous bioassay for detection of thyroid-stimulating antibodies based on a luminescent cyclic AMP biosensor. J. Immunol. Methods 2022, 501, 113199. [Google Scholar] [CrossRef]
- Frank, C.U.; Braeth, S.; Dietrich, J.W.; Wanjura, D.; Loos, U. Bridge Technology with TSH Receptor Chimera for Sensitive Direct Detection of TSH Receptor Antibodies Causing Graves’ Disease: Analytical and Clinical Evaluation. Horm. Metab. Res. 2015, 47, 880–888. [Google Scholar] [CrossRef]
- Krouse, R.S.; Royal, R.E.; Heywood, G.; Weintraub, B.D.; White, D.E.; Steinberg, S.M.; Rosenberg, S.A.; Schwartzentruber, D.J. Thyroid dysfunction in 281 patients with metastatic melanoma or renal carcinoma treated with interleukin-2 alone. J. Immunother. Emphas. Tumor Immunol. 1995, 18, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Koh, L.K.; Greenspan, F.S.; Yeo, P.P. Interferon-alpha induced thyroid dysfunction: Three clinical presentations and a review of the literature. Thyroid 1997, 7, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Roti, E.; Minelli, R.; Giuberti, T.; Marchelli, S.; Schianchi, C.; Gardini, E.; Salvi, M.; Fiaccadori, F.; Ugolotti, G.; Neri, T.M.; et al. Multiple changes in thyroid function in patients with chronic active HCV hepatitis treated with recombinant interferon-alpha. Am. J. Med. 1996, 101, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Schwartzentruber, D.J.; White, D.E.; Zweig, M.H.; Weintraub, B.D.; Rosenberg, S.A. Thyroid dysfunction associated with immunotherapy for patients with cancer. Cancer 1991, 68, 2384–2390. [Google Scholar] [CrossRef]
- De Luca, R.; Davis, P.J.; Lin, H.-Y.; Gionfra, F.; Percario, Z.A.; Affabris, E.; Pedersen, J.Z.; Marchese, C.; Trivedi, P.; Anastasiadou, E.; et al. Thyroid Hormones Interaction with Immune Response, Inflammation and Non-thyroidal Illness Syndrome. Front. Cell Dev. Biol. 2020, 8, 614030. [Google Scholar] [CrossRef] [PubMed]
- González-López, M.A.; Blanco, R.; Mata, C.; López-Escobar, M.; Lacalle, M.; Consuegra, G.; González-Vela, M.C.; González-Gay, M.A. Coexistence of Hidradenitis Suppurativa with Autoimmune Thyroiditis: Report of Three Cases. Dermatology 2016, 232, 162–164. [Google Scholar] [CrossRef]
- Der Sarkissian, S.; Hessam, S.; Kirby, J.S.; Lowes, M.A.; Mintoff, D.; Naik, H.B.; Ring, H.C.; Chandran, N.S.; Frew, J.W. Identification of Biomarkers and Critical Evaluation of Biomarker Validation in Hidradenitis Suppurativa: A Systematic Review. JAMA Dermatol. 2022, 158, 300–313. [Google Scholar] [CrossRef]
- Hessam, S.; Sand, M.; Gambichler, T.; Bechara, F.G. Correlation of inflammatory serum markers with disease severity in patients with hidradenitis suppurativa (HS). J. Am. Acad. Dermatol. 2015, 73, 998–1005. [Google Scholar] [CrossRef]
- Gambichler, T.; Hessam, S.; Cramer, P.; Abu Rached, N.; Bechara, F.G. Complete blood collection-based systemic inflammation biomarkers for patients with hidradenitis suppurativa. J. Eur. Acad. Dermatol. Venereol. 2022, 36, 1593–1596. [Google Scholar] [CrossRef]
- Lim, D.-W.; Choi, M.-S.; Kim, S.-M. Bioinformatics and Connectivity Map Analysis Suggest Viral Infection as a Critical Causative Factor of Hashimoto’s Thyroiditis. Int. J. Mol. Sci. 2023, 24, 1157. [Google Scholar] [CrossRef] [PubMed]
- Janegova, A.; Janega, P.; Rychly, B.; Kuracinova, K.; Babal, P. The role of Epstein-Barr virus infection in the development of autoimmune thyroid diseases. Endokrynol. Pol. 2015, 66, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Dittfeld, A.; Gwizdek, K.; Michalski, M.; Wojnicz, R. A possible link between the Epstein-Barr virus infection and autoimmune thyroid disorders. Cent. Eur. J. Immunol. 2016, 41, 297–301. [Google Scholar] [CrossRef]
- Mohammadi, B.; Dua, K.; Saghafi, M.; Singh, S.K.; Heydarifard, Z.; Zandi, M. COVID-19-induced autoimmune thyroiditis: Exploring molecular mechanisms. J. Med. Virol. 2023, 95, e29001. [Google Scholar] [CrossRef]
- Kotwal, A.; Kottschade, L.; Ryder, M. PD-L1 Inhibitor-Induced Thyroiditis Is Associated with Better Overall Survival in Cancer Patients. Thyroid 2020, 30, 177–184. [Google Scholar] [CrossRef]
- Álvarez-Sierra, D.; Marín-Sánchez, A.; Ruiz-Blázquez, P.; de Jesús Gil, C.; Iglesias-Felip, C.; González, Ó.; Casteras, A.; Costa, R.F.; Nuciforo, P.; Colobran, R.; et al. Analysis of the PD-1/PD-L1 axis in human autoimmune thyroid disease: Insights into pathogenesis and clues to immunotherapy associated thyroid autoimmunity. J. Autoimmun. 2019, 103, 102285. [Google Scholar] [CrossRef]
- Ferrari, S.M.; Fallahi, P.; Galetta, F.; Citi, E.; Benvenga, S.; Antonelli, A. Thyroid disorders induced by checkpoint inhibitors. Rev. Endocr. Metab. Disord. 2018, 19, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Muir, C.A.; Menzies, A.M.; Clifton-Bligh, R.; Tsang, V.H.M. Thyroid Toxicity Following Immune Checkpoint Inhibitor Treatment in Advanced Cancer. Thyroid 2020, 30, 1458–1469. [Google Scholar] [CrossRef]
- Deligiorgi, M.V.; Sagredou, S.; Vakkas, L.; Trafalis, D.T. The Continuum of Thyroid Disorders Related to Immune Checkpoint Inhibitors: Still Many Pending Queries. Cancers 2021, 13, 5277. [Google Scholar] [CrossRef]
- Agrawal, L.; Bacal, A.; Jain, S.; Singh, V.; Emanuele, N.; Emanuele, M.; Meah, F. Immune checkpoint inhibitors and endocrine side effects, a narrative review. Postgrad. Med. 2020, 132, 206–214. [Google Scholar] [CrossRef]
- Ippolito, S.; Di Dalmazi, G.; Pani, F.; Sabini, E.; Caturegli, P. Distinct Cytokine Signatures in Thyroiditis Induced by PD-1 or CTLA-4 Blockade: Insights from a New Mouse Model. Thyroid 2021, 31, 1839–1849. [Google Scholar] [CrossRef] [PubMed]
- Muir, C.A.; Clifton-Bligh, R.J.; Long, G.V.; Scolyer, R.A.; Lo, S.N.; Carlino, M.S.; Tsang, V.H.M.; Menzies, A.M. Thyroid Immune-related Adverse Events Following Immune Checkpoint Inhibitor Treatment. J. Clin. Endocrinol. Metab. 2021, 106, e3704–e3713. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Yao, J.; Yuan, G.; Gao, Y.; Zhang, J.; Guo, X. Immune Checkpoint Inhibitor-related New-onset Thyroid Dysfunction: A Retrospective Analysis Using the US FDA Adverse Event Reporting System. Oncologist 2022, 27, e126–e132. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Hodi, F.S.; Giobbie-Hurder, A.; Ott, P.A.; Buchbinder, E.I.; Haq, R.; Tolaney, S.; Barroso-Sousa, R.; Zhang, K.; Donahue, H.; et al. Characterization of Thyroid Disorders in Patients Receiving Immune Checkpoint Inhibition Therapy. Cancer Immunol. Res. 2017, 5, 1133–1140. [Google Scholar] [CrossRef]
- Chopra, D.; Arens, R.A.; Amornpairoj, W.; Lowes, M.A.; Tomic-Canic, M.; Strbo, N.; Lev-Tov, H.; Pastar, I. Innate immunity and microbial dysbiosis in hidradenitis suppurativa—Vicious cycle of chronic inflammation. Front. Immunol. 2022, 13, 960488. [Google Scholar] [CrossRef]
- Davies, T.F.; Amino, N. A new classification for human autoimmune thyroid disease. Thyroid 1993, 3, 331–333. [Google Scholar] [CrossRef]
- Nakano, Y.; Kurihara, H.; Sasaki, J. Graves’ disease following subacute thyroiditis. Tohoku J. Exp. Med. 2011, 225, 301–309. [Google Scholar] [CrossRef]
- Murugan, A.K.; Alzahrani, A.S. SARS-CoV-2 plays a pivotal role in inducing hyperthyroidism of Graves’ disease. Endocrine 2021, 73, 243–254. [Google Scholar] [CrossRef]
- Chatzitomaris, A.; Hoermann, R.; Midgley, J.E.; Hering, S.; Urban, A.; Dietrich, B.; Abood, A.; Klein, H.H.; Dietrich, J.W. Thyroid Allostasis-Adaptive Responses of Thyrotropic Feedback Control to Conditions of Strain, Stress, and Developmental Programming. Front. Endocrinol. 2017, 8, 163. [Google Scholar] [CrossRef]
| Parameter | HS Patient Value (s) | Control Group Value (s) | |
|---|---|---|---|
| sex, n (%) | female male | 50 (46.3) 58 (53.7) | 26 (50) 26 (50) |
| age, mean (±SD), y | 42 (±13) | 43.2 (±9.8) | |
| age of onset, median (IQR), y | 21 (17–31) | - | |
| disease duration, median (IQR), y | 13.5 (6–24.3) | - | |
| BMI, mean (±SD), kg/m2 | 31.4 (±6.5) | 27 (±4.9) | |
| family history of HS, n (%) | positive negative | 33 (30.6) 75 (69.4) | - |
| smoker, n (%) | current smoker ex-smoker non-smoker | 70 (64.8) 2 (1.9) 36 (33.3) | 5 (9.6) 0 (0) 47 (90.4) |
| hypothyroidism, n (%) | total | 19 (17.6) | 4 (7.7) |
| male female | 6 (10.3) 13 (26) | 0 (0) 4 (15.4) | |
| prevalence of hypothyroidism to Hurley classification, n (%) | Hurley I Hurley II Hurley III | 1 (12.5) 11 (22) 7 (14) | - |
| mHSS, median (IQR) | 40.5 (21–71.3) | - | |
| SAHS, median (IQR) | 8 (5–9) | - |
| Parameter | HS Patients (n = 108) | Control Group (n = 52) | p Value(s) |
|---|---|---|---|
| TSH, median (IQR) | 1.49 (0.99–2.15) | 1.44 (1.12–1.89) | 0.95 |
| FT3, median (IQR) | 3.25 (2.8–3.66) | 3.41 (3.12–3.71) | 0.11 |
| FT4, median (IQR) | 1.24 (1.13–1.37) | 1.34 (1.24–1.46) | 0.016 * |
| TRAb, median (IQR) | 1.08 (0.8–1.35) | 0.8 (0.8–0.92) | <0.001 * |
| TPO-Ab, median (IQR) | 9 (9–10.7) | 9.8 (9–11.4) | 0.05 |
| Tg-Ab, median (IQR) | 15.4 (14.05–17) | 14.2 (12.4–17.8) | 0.026 * |
| Parameter | HS Patients with Hypothyroidism (n = 19) | HS Patients without Hypothyroidism (n = 89) | p Value(s) |
|---|---|---|---|
| Disease duration of HS, median (ICR), y | 23 (12–30.5) | 12 (2–24) | 0.018 * |
| Initial manifestation of HS, median (ICR), y | 20 (18–28) | 21 (16–31) | 0.6 |
| BMI, mean (± SD), kg/m2 | 30.5 (26.3–36.3) | 30.9 (26.8–34.2) | 0.7 |
| Female, n (%) | 13 (68.4) | 37 (41.6) | 0.033 * |
| mHSS, median (IQR) | 47 (19–70) | 40 (21–74) | 0.9 |
| SAHS, median (IQR) | 8 (5–11) | 8 (5–9) | 0.26 |
| DLQI, median (IQR) | 15 (9.5–24.5) | 13 (7–19) | 0.086 |
| Hurley III, n (%) | 7 (36.8) | 43 (48.3) | 0.5 |
| TSH, median (IQR) | 0.67 (0.36–1.43) | 1.62 (1.15–2.22) | 0.001 * |
| fT3, median (IQR) | 2.66 (2.54–3) | 3.34 (2.98–3.7) | <0.001 * |
| fT4, median (IQR) | 1.57 (1.32–1.76) | 1.22 (1.13–1.32) | <0.001 * |
| TRAb, median (IQR) | 0.97 (0.8–1.34) | 1.1 (0.8–1.34) | 0.7 |
| TPO-Ab, median (IQR) | 9 (9–10.25) | 9 (9–10.65) | 0.5 |
| Tg-Ab, median (IQR) | 14.8 (13.85–16.5) | 15.5 (14.1–17.02) | 0.5 |
| Parameter | Euthyroid HS Patients (n = 89) | Euthyroid Healthy Controls (n = 48) | p Value(s) |
|---|---|---|---|
| Age, mean (±SD), y | 40.6 (±13.1) | 42.8 (±10.1) | 0.24 |
| BMI, mean (±SD), kg/m2 | 30.9 (26.8–34.2) | 26.8 (24.4–29.4) | <0.001 * |
| Female, n (%) | 37 (41.6) | 22 (45.8) | 0.6 |
| TSH, median (IQR) | 1.62 (1.15–2.22) | 1.44 (1.12–1.76) | 0.23 |
| FT3, median (IQR) | 3.34 (2.98–3.7) | 3.43 (3.13–3.73) | 0.45 |
| FT4, median (IQR) | 1.22 (1.13–1.32) | 1.34 (1.24–1.45) | <0.001 * |
| TRAb, median (IQR) | 1.1 (0.8–1.34) | 0.8 (0.8–0.94) | <0.001 * |
| TPO-Ab, median (IQR) | 9 (9–10.65) | 9.75 (9–11.43) | 0.11 |
| Tg-Ab, median (IQR) | 15.5 (14.1–17.02) | 13.75 (12.4–16.7) | 0.005 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abu Rached, N.; Dietrich, J.W.; Ocker, L.; Quast, D.R.; Scheel, C.; Gambichler, T.; Bechara, F.G. Primary Thyroid Dysfunction Is Prevalent in Hidradenitis Suppurativa and Marked by a Signature of Hypothyroid Graves’ Disease: A Case–Control Study. J. Clin. Med. 2023, 12, 7490. https://doi.org/10.3390/jcm12237490
Abu Rached N, Dietrich JW, Ocker L, Quast DR, Scheel C, Gambichler T, Bechara FG. Primary Thyroid Dysfunction Is Prevalent in Hidradenitis Suppurativa and Marked by a Signature of Hypothyroid Graves’ Disease: A Case–Control Study. Journal of Clinical Medicine. 2023; 12(23):7490. https://doi.org/10.3390/jcm12237490
Chicago/Turabian StyleAbu Rached, Nessr, Johannes W. Dietrich, Lennart Ocker, Daniel R. Quast, Christina Scheel, Thilo Gambichler, and Falk G. Bechara. 2023. "Primary Thyroid Dysfunction Is Prevalent in Hidradenitis Suppurativa and Marked by a Signature of Hypothyroid Graves’ Disease: A Case–Control Study" Journal of Clinical Medicine 12, no. 23: 7490. https://doi.org/10.3390/jcm12237490
APA StyleAbu Rached, N., Dietrich, J. W., Ocker, L., Quast, D. R., Scheel, C., Gambichler, T., & Bechara, F. G. (2023). Primary Thyroid Dysfunction Is Prevalent in Hidradenitis Suppurativa and Marked by a Signature of Hypothyroid Graves’ Disease: A Case–Control Study. Journal of Clinical Medicine, 12(23), 7490. https://doi.org/10.3390/jcm12237490







