Updates on Pathophysiology of Discogenic Back Pain
Abstract
1. Introduction
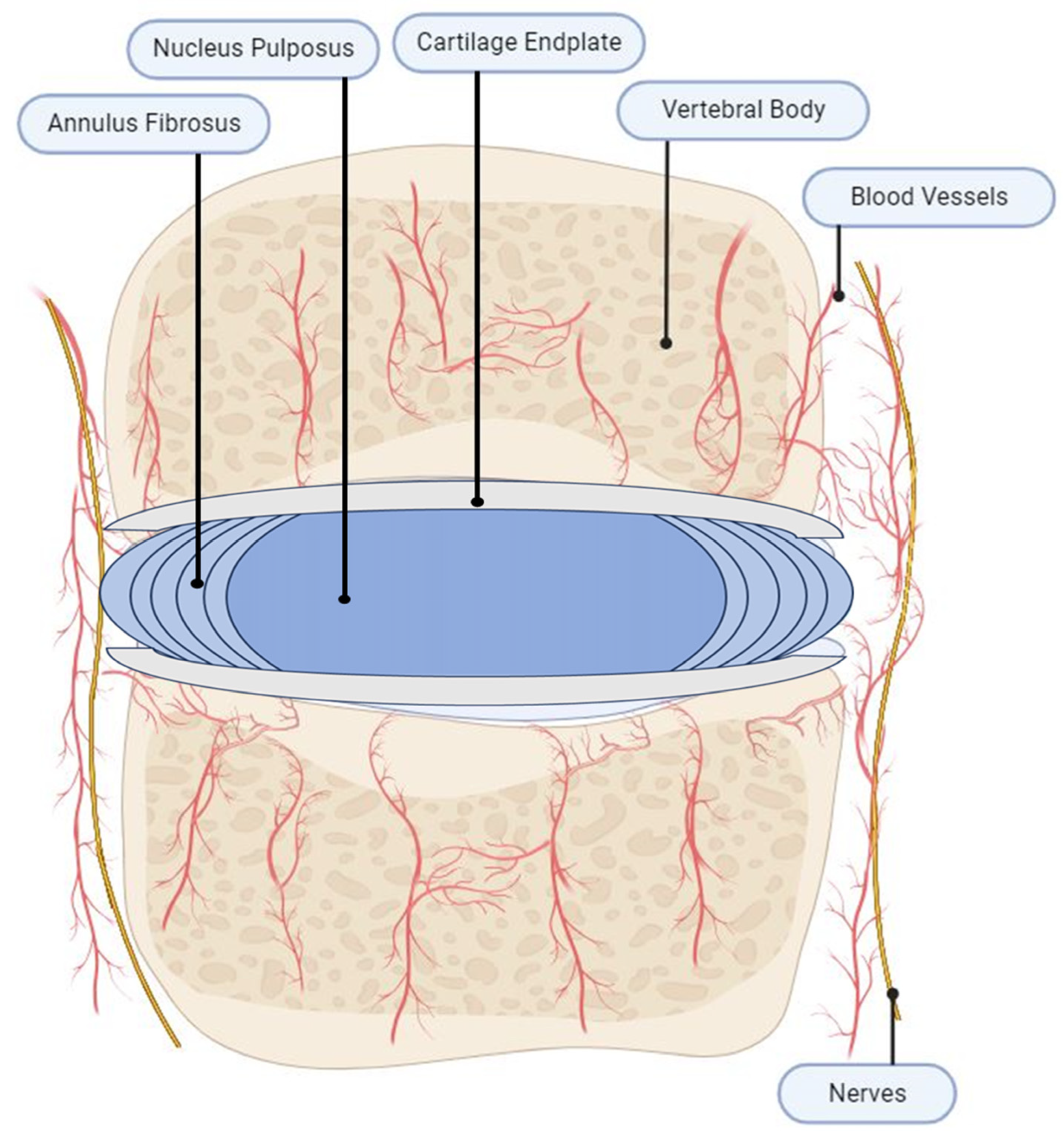
2. IVD Anatomy
3. Intervertebral Disc Damage and Degeneration
3.1. Risk Factors for Degeneration
3.2. Process of IVD Degeneration
4. Inflammatory Activation in Degenerated IVDs
4.1. Initial Inflammatory Activation
4.2. Immune Cell Recruitment
4.3. Inflammation Stimulates Further IVD Degradation
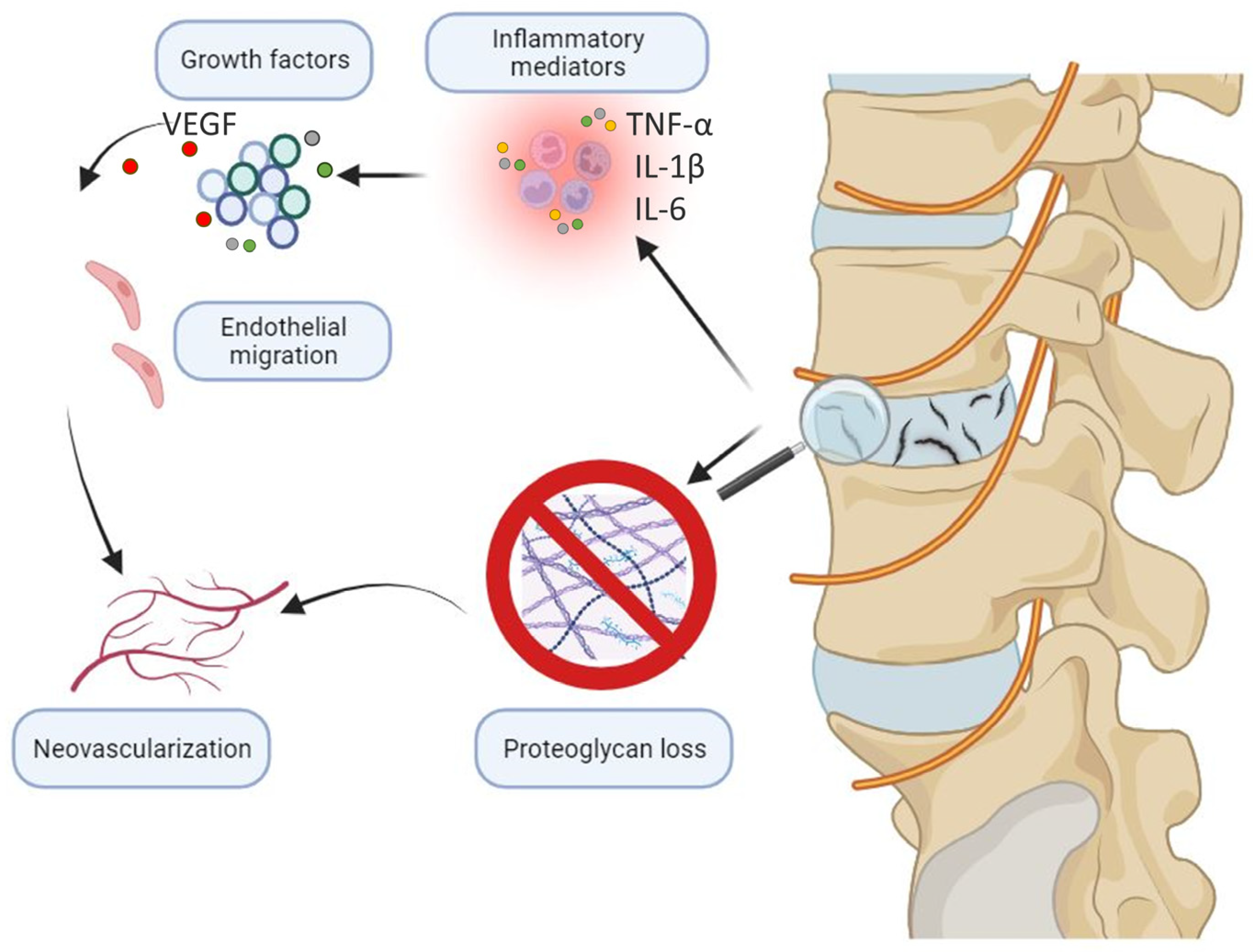
5. Neovascularization and Aberrant Healing
6. Pathological Innervation and Sensitization
6.1. Innervation of the Non-Degenerated IVD
6.2. Inflammation and Neovascularization Promotes New Nerve Growth
6.3. Neurotrophins (NTs) Induce New Nerve Growth
6.4. Nociceptive Sensitization Occurs in IVDs
7. Emerging Pathophysiology
8. Therapeutic Approaches
8.1. Emerging Targeted Therapies
8.2. Emerging Regenerative Therapies
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Andersson, G.B. Epidemiological features of chronic low-back pain. Lancet 1999, 354, 581–585. [Google Scholar] [CrossRef] [PubMed]
- Global Burden of Disease Study 2013 Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 386, 743–800. [Google Scholar] [CrossRef]
- Belitskaya-Levy, I.; Clark, J.D.; Shih, M.-C.; Bair, M.J. Treatment Preferences for Chronic Low Back Pain: Views of Veterans and Their Providers. J. Pain Res. 2021, 14, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Isa, I.L.M.; Teoh, S.L.; Nor, N.H.M.; Mokhtar, S.A. Discogenic Low Back Pain: Anatomy, Pathophysiology and Treatments of Intervertebral Disc Degeneration. Int. J. Mol. Sci. 2022, 24, 208. [Google Scholar] [CrossRef]
- Peng, B.-G. Pathophysiology, diagnosis, and treatment of discogenic low back pain. World J. Orthop. 2013, 4, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Minetama, M.; Kawakami, M.; Teraguchi, M.; Matsuo, S.; Enyo, Y.; Nakagawa, M.; Yamamoto, Y.; Nakatani, T.; Sakon, N.; Nagata, W.; et al. MRI grading of spinal stenosis is not associated with the severity of low back pain in patients with lumbar spinal stenosis. BMC Musculoskelet. Disord. 2022, 23, 857. [Google Scholar] [CrossRef]
- Teraguchi, M.; Yoshimura, N.; Hashizume, H.; Muraki, S.; Yamada, H.; Minamide, A.; Oka, H.; Ishimoto, Y.; Nagata, K.; Kagotani, R.; et al. Prevalence and distribution of intervertebral disc degeneration over the entire spine in a population-based cohort: The Wakayama Spine Study. Osteoarthr. Cartil. 2013, 22, 104–110. [Google Scholar] [CrossRef]
- Teraguchi, M.; Yoshimura, N.; Hashizume, H.; Muraki, S.; Yamada, H.; Oka, H.; Minamide, A.; Nakagawa, H.; Ishimoto, Y.; Nagata, K.; et al. The association of combination of disc degeneration, end plate signal change, and Schmorl node with low back pain in a large population study: The Wakayama Spine Study. Spine J. 2014, 15, 622–628. [Google Scholar] [CrossRef]
- Chou, D.; Samartzis, D.; Bellabarba, C.; Patel, A.; Luk, K.D.K.; Kisser, J.M.S.; Skelly, A.C. Degenerative magnetic resonance imaging changes in patients with chronic low back pain: A systematic re-view. Spine 2011, 36, S43–S53. [Google Scholar] [CrossRef]
- García-Cosamalón, J.; Del Valle, M.E.; Calavia, M.G.; García-Suárez, O.; López-Muñiz, A.; Otero, J.; Vega, J.A. Intervertebral disc, sensory nerves and neurotrophins: Who is who in discogenic pain? J. Anat. 2010, 217, 1–15. [Google Scholar] [CrossRef]
- Hancock, M.J.; Maher, C.G.; Latimer, J.; Spindler, M.F.; McAuley, J.H.; Laslett, M.; Bogduk, N. Systematic review of tests to identify the disc, SIJ or facet joint as the source of low back pain. Eur. Spine J. 2007, 16, 1539–1550. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Lv, F.-J. Symptomatic versus Asymptomatic Intervertebral Disc Degeneration: Is Inflammation the Key? Crit. Rev. Eukaryot. Gene Expr. 2015, 25, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Raj, P.P. Intervertebral disc: Anatomy-physiology-pathophysiology-treatment. Pain Pract. 2008, 8, 18–44. [Google Scholar] [CrossRef] [PubMed]
- Vital, J.-M.; Cawley, D.T. Spinal Anatomy: Modern Concepts; Springer: Cham, Switzerland, 2020. [Google Scholar]
- Mohanty, S.; Pinelli, R.; Pricop, P.; Albert, T.J.; Dahia, C.L. Chondrocyte-like nested cells in the aged intervertebral disc are late-stage nucleus pulposus cells. Aging Cell 2019, 18, e13006. [Google Scholar] [CrossRef]
- Molladavoodi, S.; McMorran, J.; Gregory, D. Mechanobiology of annulus fibrosus and nucleus pulposus cells in intervertebral discs. Cell Tissue Res. 2019, 379, 429–444. [Google Scholar] [CrossRef]
- Newell, N.; Little, J.; Christou, A.; Adams, M.; Adam, C.; Masouros, S. Biomechanics of the human intervertebral disc: A review of testing techniques and results. J. Mech. Behav. Biomed. Mater. 2017, 69, 420–434. [Google Scholar] [CrossRef]
- Choi, Y.-S. Pathophysiology of Degenerative Disc Disease. Asian Spine J. 2009, 3, 39–44. [Google Scholar] [CrossRef]
- Hadjipavlou, A.G.; Tzermiadianos, M.N.; Bogduk, N.; Zindrick, M.R. The pathophysiology of disc degeneration: A critical review. J. Bone Jt. Surg. 2008, 90, 1261–1270. [Google Scholar] [CrossRef]
- Navone, S.E.; Marfia, G.; Giannoni, A.; Beretta, M.; Guarnaccia, L.; Gualtierotti, R.; Nicoli, D.; Rampini, P.; Campanella, R. Inflammatory mediators and signalling pathways controlling intervertebral disc degeneration. Histol. Histopathol. 2017, 32, 523–542. [Google Scholar] [CrossRef]
- Urits, I.; Burshtein, A.; Sharma, M.; Testa, L.; Gold, P.A.; Orhurhu, V.; Viswanath, O.; Jones, M.R.; Sidransky, M.A.; Spektor, B.; et al. Low Back Pain, a Comprehensive Review: Pathophysiology, Diagnosis, and Treatment. Curr. Pain Headache Rep. 2019, 23, 23. [Google Scholar] [CrossRef]
- Samartzis, D.; Karppinen, J.; Mok, F.; Fong, D.Y.; Luk, K.D.; Cheung, K.M. A population-based study of juvenile disc degeneration and its association with overweight and obesity, low back pain, and diminished functional status. J. Bone Jt. Surg. 2011, 93, 662–670. [Google Scholar] [CrossRef] [PubMed]
- Dowdell, J.; Erwin, M.; Choma, T.; Vaccaro, A.; Iatridis, J.; Cho, S.K. Intervertebral Disk Degeneration and Repair. Neurosurgery 2017, 80, S46–S54, Erratum in Neurosurgery 2018, 83, 1084. [Google Scholar] [CrossRef] [PubMed]
- Kalichman, L.; Hunter, D.J. The genetics of intervertebral disc degeneration. Familial predisposition and heritability estimation. Jt. Bone Spine 2008, 75, 383–387. [Google Scholar] [CrossRef]
- Ikuno, A.; Akeda, K.; Takebayashi, S.-I.; Shimaoka, M.; Okumura, K.; Sudo, A. Genome-wide analysis of DNA methylation profile identifies differentially methylated loci associated with human intervertebral disc degeneration. PLoS ONE 2019, 14, e0222188. [Google Scholar] [CrossRef] [PubMed]
- Sakai, D.; Nakamura, Y.; Nakai, T.; Mishima, T.; Kato, S.; Grad, S.; Alini, M.; Risbud, M.V.; Chan, D.; Cheah, K.S.; et al. Exhaustion of nucleus pulposus progenitor cells with ageing and degeneration of the intervertebral disc. Nat. Commun. 2012, 3, 1264. [Google Scholar] [CrossRef] [PubMed]
- Risbud, M.V.; Schaer, T.P.; Shapiro, I.M. Toward an understanding of the role of notochordal cells in the adult intervertebral disc: From discord to accord. Dev. Dyn. 2010, 239, 2141–2148. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.N.; Jacobsen, H.E.; Khan, J.; Filippi, C.G.; Levine, M.; Lehman, R.A.; Riew, K.D.; Lenke, L.G.; Chahine, N.O. Inflammatory biomarkers of low back pain and disc degeneration: A review. Ann. N. Y. Acad. Sci. 2017, 1410, 68–84. [Google Scholar] [CrossRef]
- Binch, A.L.A.; Fitzgerald, J.C.; Growney, E.A.; Barry, F. Cell-based strategies for IVD repair: Clinical progress and translational obstacles. Nat. Rev. Rheumatol. 2021, 17, 158–175. [Google Scholar] [CrossRef]
- Peng, B.; Wu, W.; Hou, S.; Li, P.; Zhang, C.; Yang, Y. The pathogenesis of discogenic low back pain. J. Bone Jt. Surg. 2005, 87, 62–67. [Google Scholar] [CrossRef]
- Tang, S.; Richards, J.; Khan, S.; Hoyland, J.; Gallego-Perez, D.; Higuita-Castro, N.; Walter, B.; Purmessur, D. Nonviral Transfection with Brachyury Reprograms Human Intervertebral Disc Cells to a Pro-Anabolic Anti-Catabolic/Inflammatory Phenotype: A Proof of Concept Study. J. Orthop. Res. 2019, 37, 2389–2400. [Google Scholar] [CrossRef]
- Doita, M.; Kanatani, T.; Harada, T.; Mizuno, K. Immunohistologic Study of the Ruptured Intervertebral Disc of the Lumbar Spine. Spine 1996, 21, 235–241. [Google Scholar] [CrossRef]
- Vo, N.V.; Hartman, R.A.; Patil, P.R.; Risbud, M.V.; Kletsas, D.; Iatridis, J.C.; Hoyland, J.A.; Le Maitre, C.L.; Sowa, G.A.; Kang, J.D. Molecular mechanisms of biological aging in intervertebral discs. J. Orthop. Res. 2016, 34, 1289–1306. [Google Scholar] [CrossRef]
- Fields, A.J.; Berg-Johansen, B.; Metz, L.N.; Miller, S.; La, B.; Liebenberg, E.C.; Coughlin, D.G.; Graham, J.L.; Stanhope, K.L.; Havel, P.J.; et al. Alterations in intervertebral disc composition, matrix homeostasis and biomechanical behavior in the UCD-T2DM rat model of type 2 diabetes. J. Orthop. Res. 2015, 33, 738–746. [Google Scholar] [CrossRef]
- Iskra, D.A. Comorbidity of type 2 diabetes mellitus and low back pain. Zhurnal Nevrol. Psikhiatrii Im. S. S. Korsakova 2018, 118, 126–130. [Google Scholar] [CrossRef]
- Zhang, W.; Li, G.; Luo, R.; Lei, J.; Song, Y.; Wang, B.; Ma, L.; Liao, Z.; Ke, W.; Liu, H.; et al. Cytosolic escape of mitochondrial DNA triggers cGAS-STING-NLRP3 axis-dependent nucleus pulposus cell pyroptosis. Exp. Mol. Med. 2022, 54, 129–142. [Google Scholar] [CrossRef]
- Ding, F.; Shao, Z.-W.; Xiong, L.-M. Cell death in intervertebral disc degeneration. Apoptosis 2013, 18, 777–785. [Google Scholar] [CrossRef]
- Kirnaz, S.; Capadona, C.; Wong, T.; Goldberg, J.L.; Medary, B.; Sommer, F.; McGrath, L.B.; Härtl, R. Fundamentals of Intervertebral Disc Degeneration. World Neurosurg. 2022, 157, 264–273. [Google Scholar] [CrossRef]
- Bogduk, N.; Mercer, S. Biomechanics of the cervical spine. I: Normal kinematics. Clin. Biomech. 2000, 15, 633–648. [Google Scholar] [CrossRef]
- Shankar, H.; Scarlett, J.A.; Abram, S.E. Anatomy and pathophysiology of intervertebral disc disease. Tech. Reg. Anesth. Pain Manag. 2009, 13, 67–75. [Google Scholar] [CrossRef]
- Horner, H.A.; Urban, J.P.G. 2001 Volvo Award Winner in Basic Science Studies: Effect of nutrient supply on the viability of cells from the nucleus pulposus of the intervertebral disc. Spine 2001, 26, 2543–2549. [Google Scholar] [CrossRef]
- Grunhagen, T.; Shirazi-Adl, A.; Fairbank, J.C.; Urban, J.P. Intervertebral disk nutrition: A review of factors influencing concentrations of nutrients and metabolites. Orthop. Clin. N. Am. 2011, 42, 465–477. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Molinos, M.; Almeida, C.R.; Caldeira, J.; Cunha, C.; Goncalves, R.M.; Barbosa, M.A. Inflammation in intervertebral disc degeneration and regeneration. J. R. Soc. Interface 2015, 12, 20141191. [Google Scholar] [CrossRef]
- Marom, B.; Rahat, M.A.; Lahat, N.; Weiss-Cerem, L.; Kinarty, A.; Bitterman, H. Native and fragmented fibronectin oppositely modulate monocyte secretion of MMP-9. J. Leukoc. Biol. 2007, 81, 1466–1476. [Google Scholar] [CrossRef]
- Arroyo, A.G.; Iruela-Arispe, M.L. Extracellular matrix, inflammation, and the angiogenic response. Cardiovasc. Res. 2010, 86, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Quero, L.; Klawitter, M.; Schmaus, A.; Rothley, M.; Sleeman, J.; Tiaden, A.N.; Klasen, J.; Boos, N.; Hottiger, M.O.; Wuertz, K.; et al. Hyaluronic acid fragments enhance the inflammatory and catabolic response in human intervertebral disc cells through modulation of toll-like receptor 2 signalling pathways. Arthritis Res. Ther. 2013, 15, R94. [Google Scholar] [CrossRef]
- Wallach, D.; Arumugam, T.U.; Boldin, M.P.; Cantarella, G.; Ganesh, K.A.; Goltsev, Y.; Goncharov, T.M.; Kovalenko, A.V.; Rajput, A.; Varfolomeev, E.E.; et al. How are the regulators regulated? The search for mechanisms that impose specificity on induction of cell death and NF-κB activation by members of the TNF/NGF receptor family. Thromb. Haemost. 2002, 4 (Suppl. S3), S189–S196. [Google Scholar] [CrossRef]
- Bonini, S.; Rasi, G.; Bracci-Laudiero, M.; Procoli, A.; Aloe, L. Nerve growth factor: Neurotrophin or cytokine? Int. Arch. Allergy Immunol. 2003, 131, 80–84. [Google Scholar] [CrossRef]
- Liu, M.-C.; Chen, W.-H.; Wu, L.-C.; Hsu, W.-C.; Lo, W.-C.; Yeh, S.-D.; Wang, M.-F.; Zeng, R.; Deng, W.-P.; Molinos, M.; et al. Establishment of a promising human nucleus pulposus cell line for intervertebral disc tissue engineering. Tissue Eng. Part C Methods 2014, 20, 1–10. [Google Scholar] [CrossRef]
- Johnson, Z.; Schoepflin, Z.; Choi, H.; Shapiro, I.; Risbud, M. Disc in flames: Roles of TNF-α and IL-1β in intervertebral disc degeneration. Eur. Cells Mater. 2015, 30, 104–117; discussion 116–117. [Google Scholar] [CrossRef]
- Kepler, C.K.M.; Markova, D.Z.; Dibra, F.B.; Yadla, S.; Vaccaro, A.R.; Risbud, M.V.; Albert, T.J.; Anderson, D.G. Expression and Relationship of Proinflammatory Chemokine RANTES/CCL5 and Cytokine IL-1β in Painful Human Intervertebral Discs. Spine 2013, 38, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Risbud, M.V.; Shapiro, I.M. Role of cytokines in intervertebral disc degeneration: Pain and disc content. Nat. Rev. Rheumatol. 2013, 10, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Tian, Y.; Phillips, K.L.E.; Chiverton, N.; Haddock, G.; Bunning, R.A.; Cross, A.K.; Shapiro, I.M.; Le Maitre, C.L.; Risbud, M.V. Tumor necrosis factor α– and interleukin-1β–dependent induction of CCL3 expression by nucleus pulposus cells promotes macrophage migration through CCR1. Arthritis Rheum. 2013, 65, 832–842. [Google Scholar] [CrossRef] [PubMed]
- Miyagi, M.; Uchida, K.; Takano, S.; Nakawaki, M.; Sekiguchi, H.; Nakazawa, T.; Imura, T.; Saito, W.; Shirasawa, E.; Kawakubo, A.; et al. Role of CD14-positive cells in inflammatory cytokine and pain-related molecule expression in human degenerated intervertebral discs. J. Orthop. Res. 2021, 39, 1755–1762. [Google Scholar] [CrossRef] [PubMed]
- Miyagi, M.; Uchida, K.; Takano, S.; Fujimaki, H.; Aikawa, J.; Sekiguchi, H.; Nagura, N.; Ohtori, S.; Inoue, G.; Takaso, M. Macrophage-derived inflammatory cytokines regulate growth factors and pain-related molecules in mice with intervertebral disc injury. J. Orthop. Res. 2018, 36, 2274–2279. [Google Scholar] [CrossRef] [PubMed]
- Richards, J.; Tang, S.; Gunsch, G.; Sul, P.; Wiet, M.; Flanigan, D.C.; Khan, S.N.; Moore, S.; Walter, B.; Purmessur, D. Mast Cell/Proteinase Activated Receptor 2 (PAR2) Mediated Interactions in the Pathogenesis of Discogenic Back Pain. Front. Cell. Neurosci. 2019, 13, 294. [Google Scholar] [CrossRef] [PubMed]
- Wiet, M.G.; Piscioneri, A.; Khan, S.N.; Ballinger, M.N.; Hoyland, J.A.; Purmessur, D. Mast Cell-Intervertebral disc cell interactions regulate inflammation, catabolism and angiogenesis in Discogenic Back Pain. Sci. Rep. 2017, 7, 12492. [Google Scholar] [CrossRef]
- Sobajima, S.; Kompel, J.F.; Kim, J.S.; Wallach, C.J.; Robertson, D.D.; Vogt, M.T.; Kang, J.D.; Gilbertson, L.G. A slowly progressive and reproducible animal model of intervertebral disc degeneration characterized by MRI, X-ray, and histology. Spine 2005, 30, 15–24. [Google Scholar] [CrossRef]
- Le Maitre, C.; Freemont, A.J.; Hoyland, J. The role of interleukin-1 in the pathogenesis of human Intervertebral disc degeneration. Arthritis Res. Ther. 2005, 7, R732–R745. [Google Scholar] [CrossRef]
- Wang, J.; Markova, D.; Anderson, D.G.; Zheng, Z.; Shapiro, I.M.; Risbud, M.V. TNF-α and IL-1β promote a disintegrin-like and metalloprotease with thrombospondin type I mo-tif-5-mediated aggrecan degradation through syndecan-4 in intervertebral disc. J. Biol. Chem. 2011, 286, 39738–39749. [Google Scholar] [CrossRef]
- Wang, Y.; Che, M.; Xin, J.; Zheng, Z.; Li, J.; Zhang, S. The role of IL-1β and TNF-α in intervertebral disc degeneration. Biomed. Pharmacother. 2020, 131, 110660. [Google Scholar] [CrossRef] [PubMed]
- Joyce, K.; Isa, I.M.; Krouwels, A.; Creemers, L.; Devitt, A.; Pandit, A. The role of altered glycosylation in human nucleus pulposus cells in inflammation and degeneration. Eur. Cells Mater. 2021, 41, 401–420. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.M.; Doyle, P.; Minogue, B.M.; Gnanalingham, K.; Hoyland, J.A. Increased expression of matrix metalloproteinase-10, nerve growth factor and substance P in the painful degenerate intervertebral disc. Arthritis Res. Ther. 2009, 11, R126. [Google Scholar] [CrossRef] [PubMed]
- Marfia, G.; Navone, S.E.; Di Vito, C.; Tabano, S.; Giammattei, L.; Di Cristofori, A.; Gualtierotti, R.; Tremolada, C.; Zavanone, M.; Caroli, M.; et al. Gene expression profile analysis of human mesenchymal stem cells from herniated and degenerated inter-vertebral discs reveals different expression of osteopontin. Stem Cells Dev. 2015, 24, 320–328. [Google Scholar] [CrossRef]
- Miyazono, K.; Kamiya, Y.; Morikawa, M. Bone morphogenetic protein receptors and signal transduction. J. Biochem. 2009, 147, 35–51. [Google Scholar] [CrossRef]
- Wang, H.; Tian, Y.; Wang, J.; Phillips, K.L.E.; Binch, A.L.A.; Dunn, S.; Cross, A.; Chiverton, N.; Zheng, Z.; Shapiro, I.M.; et al. Inflammatory cytokines induce NOTCH signaling in nucleus pulposus cells: Implications in intervertebral disc degeneration. J. Biol. Chem. 2013, 288, 16761–16774. [Google Scholar] [CrossRef]
- Purmessur, D.; Walter, B.; Roughley, P.; Laudier, D.; Hecht, A.; Iatridis, J. A role for TNFα in intervertebral disc degeneration: A non-recoverable catabolic shift. Biochem. Biophys. Res. Commun. 2013, 433, 151–156. [Google Scholar] [CrossRef]
- Muñoz-Espín, D.; Serrano, M. Cellular senescence: From physiology to pathology. Nat. Rev. Mol. Cell Biol. 2014, 15, 482–496. [Google Scholar] [CrossRef]
- Sun, K.; Zhu, J.; Yan, C.; Li, F.; Kong, F.; Sun, J.; Sun, X.; Shi, J.; Wang, Y. CGRP Regulates Nucleus Pulposus Cell Apoptosis and Inflammation via the MAPK/NF-κB Signaling Pathways during Intervertebral Disc Degeneration. Oxidative Med. Cell. Longev. 2021, 2021, 2958584. [Google Scholar] [CrossRef]
- Gruber, H.E.; Hoelscher, G.L.; Ingram, J.A.; Bethea, S.; Hanley, E.N. Autophagy in the Degenerating Human Intervertebral Disc: In Vivo Molecular and Morphological Evidence, and Induction of Autophagy in Cultured Annulus Cells Exposed to Proinflammatory Cytokines-Implications for Disc Degeneration. Spine 2015, 40, 773–782. [Google Scholar] [CrossRef]
- Wang, B.; Wang, D.; Yan, T.; Yuan, H. MiR-138-5p promotes TNF-α-induced apoptosis in human intervertebral disc degeneration by targeting SIRT1 through PTEN/PI3K/Akt signaling. Exp. Cell Res. 2016, 345, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Shirazi-Adl, A.; Taheri, M.; Urban, J. Analysis of cell viability in intervertebral disc: Effect of endplate permeability on cell population. J. Biomech. 2010, 43, 1330–1336. [Google Scholar] [CrossRef] [PubMed]
- Stefanakis, M.; Al-Abbasi, M.; Harding, I.; Pollintine, P.; Dolan, P.; Tarlton, J.; Adams, M.A. Annulus fissures are mechanically and chemically conducive to the ingrowth of nerves and blood vessels. Spine 2012, 37, 1883–1891. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, A.; Yoon, S.T. Update on the pathophysiology of degenerative disc disease and new developments in treatment strategies. Open Access J. Sports Med. 2010, 1, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Sato, J.; Inage, K.; Miyagi, M.; Sakuma, Y.; Yamauchi, K.; Suzuki, M.; Koda, M.; Furuya, T.; Nakamura, J.; Eguchi, Y.; et al. Vascular endothelial growth factor in degenerating intervertebral discs of rat caudal vertebrae. Spine Surg. Relat. Res. 2018, 2, 42–47. [Google Scholar] [CrossRef]
- Lee, J.M.; Song, J.Y.; Baek, M.; Jung, H.-Y.; Kang, H.; Han, I.B.; Kwon, Y.D.; Shin, D.E. Interleukin-1β induces angiogenesis and innervation in human intervertebral disc degeneration. J. Orthop. Res. 2011, 29, 265–269. [Google Scholar] [CrossRef]
- Lee, T.Y.; Chin, G.S.; Kim, W.J.; Chau, D.; Gittes, G.K.; Longaker, M.T. Expression of transforming growth factor beta 1, 2, and 3 proteins in keloids. Ann. Plast. Surg. 1999, 43, 179–184. [Google Scholar] [CrossRef]
- Fu, X.; Shen, Z.; Chen, Y.; Xie, J.; Guo, Z.; Zhang, M.; Sheng, Z. Randomised placebo-controlled trial of use of topical recombinant bovine basic fibroblast growth factor for second-degree burns. Lancet 1998, 352, 1661–1664. [Google Scholar] [CrossRef]
- Tolonen, J.; Grönblad, M.; Virri, J.; Seitalo, S.; Rytömaa, T.; Karaharju, E. Basic fibroblast growth factor immunoreactivity in blood vessels and cells of disc herniations. Spine 1995, 20, 271–276. [Google Scholar] [CrossRef]
- Zhang, Y.; Chee, A.; Shi, P.D.; Adams, S.L.; Markova, D.Z.; Anderson, D.G.; Smith, H.E.; Deng, Y.; Plastaras, C.T.; An, H.S. Intervertebral Disc Cells Produce Interleukins Found in Patients with Back Pain. Am. J. Phys. Med. Rehabil. 2016, 95, 407–415. [Google Scholar] [CrossRef]
- Johnson, W.E.B.; Caterson, B.; Eisenstein, S.M.; Hynds, D.L.; Snow, D.M.; Roberts, S. Human intervertebral disc aggrecan inhibits nerve growth in vitro. Arthritis Rheum. 2002, 46, 2658–2664. [Google Scholar] [CrossRef] [PubMed]
- Melrose, J.; Roberts, S.; Smith, S.; Menage, J.; Ghosh, P. Increased nerve and blood vessel ingrowth associated with proteoglycan depletion in an ovine anular lesion model of experimental disc degeneration. Spine 2002, 27, 1278–1285. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Pang, J.; Sun, H.; Zheng, G.; Lin, Y.; Ge, W. Overexpression of TIMP3 inhibits discogenic pain by suppressing angiogenesis and the expression of substance P in nucleus pulposus. Mol. Med. Rep. 2020, 21, 1163–1171. [Google Scholar] [CrossRef] [PubMed]
- Nico, B.; Mangieri, D.; Benagiano, V.; Crivellato, E.; Ribatti, D. Nerve growth factor as an angiogenic factor. Microvasc. Res. 2008, 75, 135–141. [Google Scholar] [CrossRef]
- Cattin, A.-L.; Burden, J.J.; Van Emmenis, L.; Mackenzie, F.E.; Hoving, J.J.; Calavia, N.G.; Guo, Y.; McLaughlin, M.; Rosenberg, L.H.; Quereda, V.; et al. Macrophage-Induced Blood Vessels Guide Schwann Cell-Mediated Regeneration of Peripheral Nerves. Cell 2015, 162, 1127–1139. [Google Scholar] [CrossRef]
- Freemont, A.J.; Watkins, A.; Le Maitre, C.; Baird, P.; Jeziorska, M.; Knight, M.T.N.; Ross, E.R.S.; O’Brien, J.P.; Hoyland, J.A. Nerve growth factor expression and innervation of the painful intervertebral disc. J. Pathol. 2002, 197, 286–292. [Google Scholar] [CrossRef]
- Groh, A.M.R.; E Fournier, D.; Battié, M.C.; Séguin, C.A. Innervation of the Human Intervertebral Disc: A Scoping Review. Pain Med. 2021, 22, 1281–1304. [Google Scholar] [CrossRef]
- Edgar, M.A. The nerve supply of the lumbar intervertebral disc. J. Bone Jt. Surg. 2007, 89, 1135–1139. [Google Scholar] [CrossRef]
- Aoki, Y.; Takahashi, Y.; Takahashi, K.; Chiba, T.; Kurokawa, M.; Ozawa, T.; Moriya, H. Sensory innervation of the lateral portion of the lumbar intervertebral disc in rats. Spine J. 2004, 4, 275–280. [Google Scholar] [CrossRef]
- Lyu, F.-J.; Cui, H.; Pan, H.; MC Cheung, K.; Cao, X.; Iatridis, J.C.; Zheng, Z. Painful intervertebral disc degeneration and inflammation: From laboratory evidence to clinical interventions. Bone Res. 2021, 9, 7. [Google Scholar] [CrossRef]
- Freemont, A.; Peacock, T.; Goupille, P.; Hoyland, J.; O’Brien, J.; Jayson, M. Nerve ingrowth into diseased intervertebral disc in chronic back pain. Lancet 1997, 350, 178–181. [Google Scholar] [CrossRef] [PubMed]
- Suseki, K.; Takahashi, Y.; Takahashi, K.; Chiba, T.; Yamagata, M.; Moriya, H. Sensory nerve fibres from lumbar intervertebral discs pass through rami communicantes. A possible pathway for discogenic low back pain. J. Bone Jt. Surg. 1998, 80, 737–742. [Google Scholar] [CrossRef]
- Takahashi, Y.; Ohtori, S.; Takahashi, K. Peripheral Nerve Pathways of Afferent Fibers Innervating the Lumbar Spine in Rats. J. Pain 2009, 10, 416–425. [Google Scholar] [CrossRef]
- Kim, H.S.; Wu, P.H.; Jang, I.-T. Lumbar Degenerative Disease Part 1: Anatomy and Pathophysiology of Intervertebral Discogenic Pain and Radiofrequency Ablation of Basivertebral and Sinuvertebral Nerve Treatment for Chronic Discogenic Back Pain: A Prospective Case Series and Review of Literature. Int. J. Mol. Sci. 2020, 21, 1483. [Google Scholar] [CrossRef]
- Purmessur, D.; Freemont, A.J.; Hoyland, J.A. Expression and regulation of neurotrophins in the nondegenerate and degenerate human intervertebral disc. Arthritis. Res. Ther. 2008, 10, R99. [Google Scholar] [CrossRef] [PubMed]
- Reichardt, L.F. Neurotrophin-regulated signalling pathways. Philos. Trans. R. Soc. B Biol. Sci. 2006, 361, 1545–1564. [Google Scholar] [CrossRef]
- Abe, Y.; Akeda, K.; An, H.S.; Aoki, Y.; Pichika, R.; Muehleman, C.; Kimura, T.; Masuda, K. Proinflammatory cytokines stimulate the expression of nerve growth factor by human intervertebral disc cells. Spine 2007, 32, 635–642. [Google Scholar] [CrossRef]
- Boyd, L.M.; Richardson, W.J.; Chen, J.; Kraus, V.B.; Tewari, A.; Setton, L.A. Osmolarity regulates gene expression in intervertebral disc cells determined by gene array and real-time quantitative RT-PCR. Ann. Biomed. Eng. 2005, 33, 1071–1077. [Google Scholar] [CrossRef]
- Vega, J.A.; Garcia-Suarez, O.; Hannestad, J.; Perez-Perez, M.; Germana, A. Neurotrophins and the immune system. J. Anat. 2003, 203, 1–19. [Google Scholar] [CrossRef]
- Lee, S.; Moon, C.S.; Sul, D.; Lee, J.; Bae, M.; Hong, Y.; Lee, M.; Choi, S.; Derby, R.; Kim, B.-J.; et al. Comparison of growth factor and cytokine expression in patients with degenerated disc disease and herniated nucleus pulposus. Clin. Biochem. 2009, 42, 1504–1511. [Google Scholar] [CrossRef]
- Zheng, B.; Li, S.; Xiang, Y.; Zong, W.; Ma, Q.; Wang, S.; Wu, H.; Song, H.; Ren, H.; Chen, J.; et al. Netrin-1 mediates nerve innervation and angiogenesis leading to discogenic pain. J. Orthop. Transl. 2023, 39, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Bach, F.C.; Poramba-Liyanage, D.W.; Riemers, F.M.; Guicheux, J.; Camus, A.; Iatridis, J.C.; Chan, D.; Ito, K.; Le Maitre, C.L.; Tryfonidou, M.A. Notochordal Cell-Based Treatment Strategies and Their Potential in Intervertebral Disc Regeneration. Front. Cell Dev. Biol. 2021, 9, 780749. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Hu, B.; Liu, W.; Wang, P.; Lv, X.; Chen, S.; Shao, Z. The role of structure and function changes of sensory nervous system in intervertebral disc-related low back pain. Osteoarthr. Cartil. 2021, 29, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Choi, J.Y.; Kim, R.U.; Lee, Y.S.; Cho, H.J.; Kim, D.S. Downregulation of voltage-gated potassium channel alpha gene expression by axotomy and neurotrophins in rat dorsal root ganglia. Mol. Cells 2003, 16, 256–259. [Google Scholar]
- Priestley, J.V.; Michael, G.J.; Averill, S.; Liu, M.; Willmott, N. Regulation of nociceptive neurons by nerve growth factor and glial cell line derived neurotrophic factor. Can. J. Physiol. Pharmacol. 2002, 80, 495–505. [Google Scholar] [CrossRef]
- Nojima, D.; Inage, K.; Sakuma, Y.; Sato, J.; Orita, S.; Yamauchi, K.; Eguchi, Y.; Ochiai, N.; Kuniyoshi, K.; Aoki, Y.; et al. Efficacy of Anti-NaV1.7 Antibody on the Sensory Nervous System in a Rat Model of Lumbar Intervertebral Disc Injury. Yonsei Med. J. 2016, 57, 748–753. [Google Scholar] [CrossRef] [PubMed]
- Snider, W.D.; McMahon, S.B. Tackling pain at the source: New ideas about nociceptors. Neuron 1998, 20, 629–632. [Google Scholar] [CrossRef]
- Yamada, J.; Akeda, K.; Sano, T.; Iwasaki, T.; Takegami, N.; Sudo, A. Expression of Glial Cell Line-derived Neurotrophic Factor in the Human Intervertebral Disc. Spine 2020, 45, E768–E775. [Google Scholar] [CrossRef]
- Leimer, E.M.; Gayoso, M.G.; Jing, L.; Tang, S.Y.; Gupta, M.C.; Setton, L.A. Behavioral Compensations and Neuronal Remodeling in a Rodent Model of Chronic Intervertebral Disc Degeneration. Sci. Rep. 2019, 9, 3759. [Google Scholar] [CrossRef]
- Zhang, X.; Huang, J.; McNaughton, P.A. NGF rapidly increases membrane expression of TRPV1 heat-gated ion channels. EMBO J. 2005, 24, 4211–4223. [Google Scholar] [CrossRef]
- Merighi, A.; Salio, C.; Ghirri, A.; Lossi, L.; Ferrini, F.; Betelli, C.; Bardoni, R. BDNF as a pain modulator. Prog. Neurobiol. 2008, 85, 297–317. [Google Scholar] [CrossRef]
- Qiu, S.; Shi, C.; Anbazhagan, A.N.; Das, V.; Arora, V.; Kc, R.; Li, X.; O-Sullivan, I.; van Wijnen, A.; Chintharlapalli, S.; et al. Absence of VEGFR-1/Flt-1 signaling pathway in mice results in insensitivity to discogenic low back pain in an established disc injury mouse model. J. Cell. Physiol. 2020, 235, 5305–5317. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Stefanoska, D.; Grad, S.; Alini, M.; Peroglio, M. Direct and Intervertebral DiscMediated Sensitization of Dorsal Root Ganglion Neurons by Hypoxia and Low pH. Neurospine 2020, 17, 42–59. [Google Scholar] [CrossRef] [PubMed]
- Park, E.; Moon, S.; Suh, H.; Hochman, S.; Lee, M.-G.; Kim, Y.; Jang, I.; Han, H. Disc degeneration induces a mechano-sensitization of disc afferent nerve fibers that associates with low back pain. Osteoarthr. Cartil. 2019, 27, 1608–1617. [Google Scholar] [CrossRef] [PubMed]
- Stover, J.D.; Lawrence, B.; Bowles, R.D. Degenerative IVD conditioned media and acidic pH sensitize sensory neurons to cyclic tensile strain. J. Orthop. Res. 2020, 39, 1192–1203. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-G.; Strong, J.A.; Xie, W.; Zhang, J.-M. Local Inflammation in Rat Dorsal Root Ganglion Alters Excitability and Ion Currents in Small-diameter Sensory Neurons. Anesthesiology 2007, 107, 322–332. [Google Scholar] [CrossRef]
- Burke, J.G.; Watson, R.W.G.; McCormack, D.; Dowling, F.E.; Walsh, M.G.; Fitzpatrick, J.M. Intervertebral discs which cause low back pain secrete high levels of proinflammatory mediators. J. Bone Jt. Surg. 2002, 84, 196–201. [Google Scholar] [CrossRef]
- Shamji, M.F.; Setton, L.A.; Jarvis, W.; So, S.; Chen, J.; Jing, L.; Bullock, R.; Isaacs, R.E.; Brown, C.; Richardson, W.J. Proinflammatory cytokine expression profile in degenerated and herniated human intervertebral disc tissues. Arthritis Rheum. 2010, 62, 1974–1982. [Google Scholar] [CrossRef] [PubMed]
- Ebbinghaus, M.; Uhlig, B.; Richter, F.; von Banchet, G.S.; Gajda, M.; Bräuer, R.; Schaible, H.-G. The role of interleukin-1β in arthritic pain: Main involvement in thermal, but not mechanical, hyperalgesia in rat antigen-induced arthritis. Arthritis Rheum. 2012, 64, 3897–3907. [Google Scholar] [CrossRef]
- Kepler, C.K.; Markova, D.Z.; Hilibrand, A.S.; Vaccaro, A.R.; Risbud, M.V.; Albert, T.J.; Anderson, D.G. Substance P stimulates production of inflammatory cytokines in human disc cells. Spine 2013, 38, E1291–E1299. [Google Scholar] [CrossRef]
- Mannion, R.J.; Costigan, M.; Decosterd, I.; Amaya, F.; Ma, Q.-P.; Holstege, J.C.; Ji, R.-R.; Acheson, A.; Lindsay, R.M.; Wilkinson, G.A.; et al. Neurotrophins: Peripherally and centrally acting modulators of tactile stimulus-induced inflammatory pain hypersensitivity. Proc. Natl. Acad. Sci. USA 1999, 96, 9385–9390. [Google Scholar] [CrossRef]
- Neumann, S.; Doubell, T.P.; Leslie, T.; Woolf, C.J. Inflammatory pain hypersensitivity mediated by phenotypic switch in myelinated primary sensory neurons. Nature 1996, 384, 360–364. [Google Scholar] [CrossRef]
- Ganko, R.; Rao, P.J.; Phan, K.; Mobbs, R.J. Can bacterial infection by low virulent organisms be a plausible cause for symptomatic disc degeneration? A systematic review. Spine 2015, 40, E587–E592. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.J.; Maharaj, M.; Chau, C.; Taylor, P.; Phan, K.; Choy, W.J.; Scherman, D.; Mews, P.; Scholsem, M.; Coughlan, M.; et al. Degenerate-disc infection study with contaminant control (DISC): A multicenter prospective case-control trial. Spine J. 2020, 20, 1544–1553. [Google Scholar] [CrossRef] [PubMed]
- Albert, H.B.; Manniche, C. Modic changes following lumbar disc herniation. Eur. Spine J. 2007, 16, 977–982. [Google Scholar] [CrossRef]
- Jiao, Y.; Yuan, Y.; Lin, Y.; Zhou, Z.; Zheng, Y.; Wu, W.; Tang, G.; Chen, Y.; Xiao, J.; Li, C.; et al. Propionibacterium acnes induces discogenic low back pain via stimulating nucleus pulposus cells to secrete pro-algesic factor of IL-8/CINC-1 through TLR2–NF-κB p65 pathway. J. Mol. Med. 2019, 97, 25–35. [Google Scholar] [CrossRef]
- Schmid, B.; Hausmann, O.; Hitzl, W.; Achermann, Y.; Wuertz-Kozak, K. The Role of Cutibacterium acnes in Intervertebral Disc Inflammation. Biomedicines 2020, 8, 186. [Google Scholar] [CrossRef]
- Jiao, Y.; Lin, Y.; Zheng, J.; Shi, L.; Zheng, Y.; Zhang, Y.; Li, J.; Chen, Z.; Cao, P. Propionibacterium acnes contributes to low back pain via upregulation of NGF in TLR2-NF-κB/JNK or ROS pathway. Microbes Infect. 2022, 24, 104980. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Liao, W.; Shen, M.; Mei, H. Insight into neural mechanisms underlying discogenic back pain. J. Int. Med. Res. 2018, 46, 4427–4436. [Google Scholar] [CrossRef]
- Yang, G.; Chen, L.; Gao, Z.; Wang, Y. Implication of microglia activation and CSF1/CSF1R pathway in lumbar disc degeneration-related back pain. Mol. Pain 2018, 14, 1744806918811238. [Google Scholar] [CrossRef]
- Li, Z.; Yu, X.; Shen, J.; Chan, M.T.; Wu, W.K.K. MicroRNA in intervertebral disc degeneration. Cell Prolif. 2015, 48, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Cui, L.; Gu, Q.; Guo, S.; Zhu, B.; Liu, X.; Li, Y.; Liu, X.; Wang, D.; Li, S. The Mechanism and Function of miRNA in Intervertebral Disc Degeneration. Orthop. Surg. 2022, 14, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhang, J.; Yao, Y.; Weixuan, Y.; Bernstock, J.D.; Feng, T.; Chen, B.; Guo, H.; Lu, Y. Selective inhibition of Colony Stimulation Factor 1 Receptor pathway modulates intervertebral disc degeneration and alleviates discogenic back pain. Manuscript in preparation.
- Urquhart, D.M.; Kelsall, H.L.; Hoe, V.C.; Cicuttini, F.M.; Forbes, A.B.; Sim, M.R. Are Psychosocial Factors Associated with Low Back Pain and Work Absence for Low Back Pain in an Occupational Cohort? Clin. J. Pain 2013, 29, 1015–1020. [Google Scholar] [CrossRef] [PubMed]
- Derby, R.; Lettice, J.J.; Kula, T.A.; Lee, S.-H.; Seo, K.-S.; Kim, B.-J. Single-level lumbar fusion in chronic discogenic low-back pain: Psychological and emotional status as a predictor of outcome measured using the 36-item Short Form. J. Neurosurg. Spine 2005, 3, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Benyamin, R.M.; Manchikanti, L.; Parr, A.T.; Diwan, S.; Singh, V.; Falco, F.J.E.; Datta, S.; Abdi, S.; Hirsch, J.A. The effectiveness of lumbar interlaminar epidural injections in managing chronic low back and lower extremity pain. Pain Physician 2012, 15, E363–E404. [Google Scholar] [CrossRef]
- Pasqualucci, A.; Varrassi, G.; Braschi, A.; Peduto, V.A.; Brunelli, A.; Marinangeli, F.; Gori, F.; Colò, F.; Paladini, A.; Mojoli, F. Epidural local anesthetic plus corticosteroid for the treatment of cervical brachial radicular pain: Single injection versus continuous infusion. Clin. J. Pain 2007, 23, 551–557. [Google Scholar] [CrossRef]
- Nakamura, S.; Takahashi, K.; Takahashi, Y.; Yamagata, M.; Moriya, H. The afferent pathways of discogenic low-back pain. Evaluation of L2 spinal nerve infiltration. J. Bone Joint. Surg. Br. 1996, 78, 606–612. [Google Scholar] [CrossRef]
- Kallewaard, J.W.; Edelbroek, C.; Terheggen, M.; Raza, A.; Geurts, J.W. A Prospective Study of Dorsal Root Ganglion Stimulation for Non-Operated Discogenic Low Back Pain. Neuromodulation Technol. Neural Interface 2019, 23, 196–202. [Google Scholar] [CrossRef]
- Peng, B.; Chen, J.; Kuang, Z.; Li, D.; Pang, X.; Zhang, X. Diagnosis and surgical treatment of back pain originating from endplate. Eur. Spine J. 2009, 18, 1035–1040. [Google Scholar] [CrossRef]
- Tobert, D.G.; Antoci, V.; Patel, S.P.; Saadat, E.; Bono, C.M. Adjacent Segment Disease in the Cervical and Lumbar Spine. Clin. Spine Surg. A Spine Publ. 2017, 30, 94–101. [Google Scholar] [CrossRef]
- Wang, Z.; Hutton, W.C.; Yoon, S.T. Bone morphogenetic protein-7 antagonizes tumor necrosis factor-α–induced activation of nuclear factor κB and up-regulation of the ADAMTS, leading to decreased degradation of disc matrix macromolecules aggrecan and collagen II. Spine J. 2014, 14, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Gorth, D.J.; Mauck, R.L.; Chiaro, J.A.; Mohanraj, B.; Hebela, N.M.; Dodge, G.R.; Elliott, D.M.; Smith, L.J. IL-1ra delivered from poly(lactic-co-glycolic acid) microspheres attenuates IL-1beta mediated degradation of nucleus pulposus in vitro. Arthritis Res. Ther. 2012, 14, R179. [Google Scholar] [CrossRef] [PubMed]
- Kobori, S.; Miyagi, M.; Orita, S.; Gemba, T.; Ishikawa, T.; Kamoda, H.; Suzuki, M.; Hishiya, T.; Yamada, T.; Eguchi, Y.; et al. Inhibiting IκB Kinase-β Downregulates Inflammatory Cytokines in Injured Discs and Neuropeptides in Dorsal Root Ganglia Innervating Injured Discs in Rats. Spine 2014, 39, 1171–1177. [Google Scholar] [CrossRef] [PubMed]
- Kato, K.; Akeda, K.; Miyazaki, S.; Yamada, J.; Muehleman, C.; Miyamoto, K.; Asanuma, Y.; Asanuma, K.; Fujiwara, T.; Lenz, M.; et al. NF-kB decoy oligodeoxynucleotide preserves disc height in a rabbit anular-puncture model and reduces pain induction in a rat xenograft-radiculopathy model. Eur. Cells Mater. 2021, 42, 90–109. [Google Scholar] [CrossRef] [PubMed]
- Chou, P.-H.; Chee, A.; Shi, P.; Lin, C.-L.; Zhao, Y.; Zhang, L.; An, H.S. Small molecule antagonist of C-C chemokine receptor 1 (CCR1) reduces disc inflammation in the rabbit model. Spine J. 2020, 20, 2025–2036. [Google Scholar] [CrossRef]
- Sato, J.; Inage, K.; Miyagi, M.; Sakuma, Y.; Yamauchi, K.; Koda, M.; Furuya, T.; Nakamura, J.; Suzuki, M.; Kubota, G.; et al. Inhibiting Vascular Endothelial Growth Factor in Injured Intervertebral Discs Attenuates Pain-Related Neuropeptide Expression in Dorsal Root Ganglia in Rats. Asian Spine J. 2017, 11, 556–561. [Google Scholar] [CrossRef]
- Katz, N.; Borenstein, D.G.; Birbara, C.; Bramson, C.; Nemeth, M.A.; Smith, M.D.; Brown, M.T. Efficacy and safety of tanezumab in the treatment of chronic low back pain. Pain 2011, 152, 2248–2258. [Google Scholar] [CrossRef]
- Kivitz, A.J.; Gimbel, J.S.; Bramson, C.; Nemeth, M.A.; Keller, D.S.; Brown, M.T.; West, C.R.; Verburg, K.M. Efficacy and safety of tanezumab versus naproxen in the treatment of chronic low back pain. Pain 2013, 154, 1009–1021. [Google Scholar] [CrossRef]
- Sanga, P.; Polverejan, E.; Wang, S.; Kelly, K.M.; Thipphawong, J. Efficacy, Safety, and Tolerability of Fulranumab as an Adjunctive Therapy in Patients with Inadequately Controlled, Moderate-to-Severe Chronic Low Back Pain: A Randomized, Double-blind, Placebo-controlled, Dose-ranging, Dose-loading Phase II Study. Clin. Ther. 2016, 38, 1435–1450. [Google Scholar] [CrossRef][Green Version]
- Peng, B.; Pang, X.; Wu, Y.; Zhao, C.; Song, X. A randomized placebo-controlled trial of intradiscal methylene blue injection for the treatment of chronic discogenic low back pain. Pain 2010, 149, 124–129. [Google Scholar] [CrossRef]
- Deng, M.; Huang, H.; Ma, Y.-G.; Zhou, Y.; Chen, Q.; Xie, P. Intradiskal Injection of Methylene Blue for Discogenic Back Pain: A Meta-Analysis of Randomized Controlled Trials. J. Neurol. Surg. Part A Cent. Eur. Neurosurg. 2021, 82, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Roh, E.J.; Darai, A.; Kyung, J.W.; Choi, H.; Kwon, S.Y.; Bhujel, B.; Kim, K.T.; Han, I. Genetic Therapy for Intervertebral Disc Degeneration. Int. J. Mol. Sci. 2021, 22, 1579. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yu, T.; Ma, X.-X.; Xiang, H.-F.; Hu, Y.-G.; Chen, B.-H. Lentivirus-mediated TGF-β3, CTGF and TIMP1 gene transduction as a gene therapy for intervertebral disc degeneration in an in vivo rabbit model. Exp. Ther. Med. 2016, 11, 1399–1404. [Google Scholar] [CrossRef] [PubMed]
- Leckie, S.K.; Bechara, B.P.; Hartman, R.A.; Sowa, G.A.; Woods, B.I.; Coelho, J.P.; Witt, W.T.; Dong, Q.D.; Bowman, B.W.; Bell, K.M.; et al. Injection of AAV2-BMP2 and AAV2-TIMP1 into the nucleus pulposus slows the course of intervertebral disc degeneration in an in vivo rabbit model. Spine J. 2012, 12, 7–20. [Google Scholar] [CrossRef]
- Her, Y.F.; Kubrova, E.; Alvarez, G.A.M.; D’Souza, R.S. The Analgesic Efficacy of Intradiscal Injection of Bone Marrow Aspirate Concentrate and Culture-Expanded Bone Marrow Mesenchymal Stromal Cells in Discogenic Pain: A Systematic Review. J. Pain Res. 2022, 15, 3299–3318. [Google Scholar] [CrossRef]
- Miguélez-Rivera, L.; Pérez-Castrillo, S.; González-Fernández, M.L.; Prieto-Fernández, J.G.; López-González, M.E.; García-Cosamalón, J.; Villar-Suárez, V. Immunomodulation of mesenchymal stem cells in discogenic pain. Spine J. 2018, 18, 330–342. [Google Scholar] [CrossRef]
- Haines, C.M.; Bhatt, F.R.; Orosz, L.D.; Yamout, T.; Namian, S.; Bharara, N.; Bucci, A.; Schuler, T.C.; Jazini, E.; Good, C.R. Low Back Pain, Disability, and Quality of Life One Year following Intradiscal Injection of Autologous Bone Marrow Aspirate Concentrate. Stem Cells Int. 2022, 2022, 9617511. [Google Scholar] [CrossRef]
- Sharan, J.; Barmada, A.; Prodromos, C.; Candido, K. First Human Report of Relief of Lumbar and Cervical Discogenic and Arthritic Back Pain after Epidural and Facet Joint Mesenchymal Stem Cell Injection. Curr. Stem Cell Res. Ther. 2022, 18, 1013–1015. [Google Scholar] [CrossRef]
- Li, L.; Sheng, K.; Mannarino, M.; Jarzem, P.; Cherif, H.; Haglund, L. o-Vanillin Modulates Cell Phenotype and Extracellular Vesicles of Human Mesenchymal Stem Cells and Intervertebral Disc Cells. Cells 2022, 11, 3589. [Google Scholar] [CrossRef] [PubMed]
- Noriega, D.C.; Ardura, F.; Hernández-Ramajo, R.; Martín-Ferrero, M.Á.; Sánchez-Lite, I.; Toribio, B.; Alberca, M.; García, V.; Moraleda, J.M.; Sánchez, A.; et al. Intervertebral Disc Repair by Allogeneic Mesenchymal Bone Marrow Cells: A Randomized Controlled Trial. Transplantation 2017, 101, 1945–1951. [Google Scholar] [CrossRef]
- Wu, C.-C.; Chen, W.-H.; Zao, B.; Lai, P.-L.; Lin, T.-C.; Lo, H.-Y.; Shieh, Y.-H.; Wu, C.-H.; Deng, W.-P. Regenerative potentials of platelet-rich plasma enhanced by collagen in retrieving pro-inflammatory cytokine-inhibited chondrogenesis. Biomaterials 2011, 32, 5847–5854. [Google Scholar] [CrossRef] [PubMed]
- Akeda, K.; Ohishi, K.; Takegami, N.; Sudo, T.; Yamada, J.; Fujiwara, T.; Niimi, R.; Matsumoto, T.; Nishimura, Y.; Ogura, T.; et al. Platelet-Rich Plasma Releasate versus Corticosteroid for the Treatment of Discogenic Low Back Pain: A Double-Blind Randomized Controlled Trial. J. Clin. Med. 2022, 11, 304. [Google Scholar] [CrossRef] [PubMed]
- Tschugg, A.; Diepers, M.; Simone, S.; Michnacs, F.; Quirbach, S.; Strowitzki, M.; Meisel, H.J.; Thomé, C. A prospective randomized multicenter phase I/II clinical trial to evaluate safety and efficacy of NOVOCART disk plus autologous disk chondrocyte transplantation in the treatment of nucleotomized and degenerative lumbar disks to avoid secondary disease: Safety results of Phase I—A short report. Neurosurg. Rev. 2017, 40, 155–162. [Google Scholar] [CrossRef]
- Isa, I.L.M.; Mokhtar, S.A.; Abbah, S.A.; Fauzi, M.B.; Devitt, A.; Pandit, A. Intervertebral Disc Degeneration: Biomaterials and Tissue Engineering Strategies toward Precision Medicine. Adv. Health Mater. 2022, 11, e2102530. [Google Scholar] [CrossRef]
- Fujii, K.; Yamazaki, M.; Kang, J.D.; Risbud, M.V.; Cho, S.K.; Qureshi, S.A.; Hecht, A.C.; Iatridis, J.C. Discogenic Back Pain: Literature Review of Definition, Diagnosis, and Treatment. JBMR Plus 2019, 3, e10180. [Google Scholar] [CrossRef] [PubMed]
- Isa, I.L.M.; Abbah, S.A.; Kilcoyne, M.; Sakai, D.; Dockery, P.; Finn, D.P.; Pandit, A. Implantation of hyaluronic acid hydrogel prevents the pain phenotype in a rat model of intervertebral disc injury. Sci. Adv. 2018, 4, eaaq0597. [Google Scholar] [CrossRef]


| Initial IVD Degeneration | Cellular Loss |
| Oxidative stress products: free radicals, advanced glycation end products | |
| Apoptotic pathways | |
| Initial Inflammatory Activation | ECM breakdown phagocytosis → NALP3 → IL-1β |
| Fibronectin end products, hyaluronic acid fragments | |
| IL-1β, TNF-α | |
| IL-6, IL-7, IFN-γ | |
| NGF → NF-kB | |
| SP | |
| Immune Cell Recruitment | IL-1β, TNF-α |
| IL-6 | |
| Mast Cells | |
| Persistent Inflammatory Degradation | MMPs |
| ADAMTS | |
| SDC4, PHD-3 → NF-kB, ERK | |
| OPN, GDF5 | |
| IL-1β, TNF-α → positive feedback senescence | |
| CGRP → NF-kB, MAPK | |
| Neovascularization | VEGF |
| bFGF, TGF-β, CTGF, IL-8, Plieotrophin | |
| Loss of proteoglycans, ↓ TIMP3 | |
| Pathological Innervation | NTs (NGF, BDNF, NT-3, NT-4/5) |
| Netrin-1, Semaphorin 3a | |
| Nociceptive Sensitization | NGF → SP, CGRP, Ion Channels, ASIC receptors |
| GDNF, BDNF | |
| VEGF | |
| Mechanoreceptors | |
| Inflammatory cytokines | |
| Emerging Pathophysiology | Low virulence infections |
| Microglia metabolic activation | |
| miRNAs | |
| CSFR1 |
| Current Therapies | Conservative Management | Analgesics |
| Physical Therapy and Exercise | ||
| Invasive Measures | Epidural Anesthetics | |
| Steroid Injections | ||
| Dorsal Root Ganglion Stimulation | ||
| Surgical Discectomy and Fusion | ||
| Emerging Therapies | Targeted Therapies | TNF-α blockers |
| IL-1 inhibitors | ||
| NF-kB antagonists | ||
| VEGF inhibitors | ||
| Anti-NGF antibodies | ||
| Regenerative Therapies | Gene Therapy | |
| Mesenchymal Stem Cell Therapy | ||
| Platelet-Rich Plasma Therapy | ||
| Tissue Engineering Strategies |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jha, R.; Bernstock, J.D.; Chalif, J.I.; Hoffman, S.E.; Gupta, S.; Guo, H.; Lu, Y. Updates on Pathophysiology of Discogenic Back Pain. J. Clin. Med. 2023, 12, 6907. https://doi.org/10.3390/jcm12216907
Jha R, Bernstock JD, Chalif JI, Hoffman SE, Gupta S, Guo H, Lu Y. Updates on Pathophysiology of Discogenic Back Pain. Journal of Clinical Medicine. 2023; 12(21):6907. https://doi.org/10.3390/jcm12216907
Chicago/Turabian StyleJha, Rohan, Joshua D. Bernstock, Joshua I. Chalif, Samantha E. Hoffman, Saksham Gupta, Hong Guo, and Yi Lu. 2023. "Updates on Pathophysiology of Discogenic Back Pain" Journal of Clinical Medicine 12, no. 21: 6907. https://doi.org/10.3390/jcm12216907
APA StyleJha, R., Bernstock, J. D., Chalif, J. I., Hoffman, S. E., Gupta, S., Guo, H., & Lu, Y. (2023). Updates on Pathophysiology of Discogenic Back Pain. Journal of Clinical Medicine, 12(21), 6907. https://doi.org/10.3390/jcm12216907








