Tick-Borne Encephalitis—Review of the Current Status
Abstract
1. Introduction
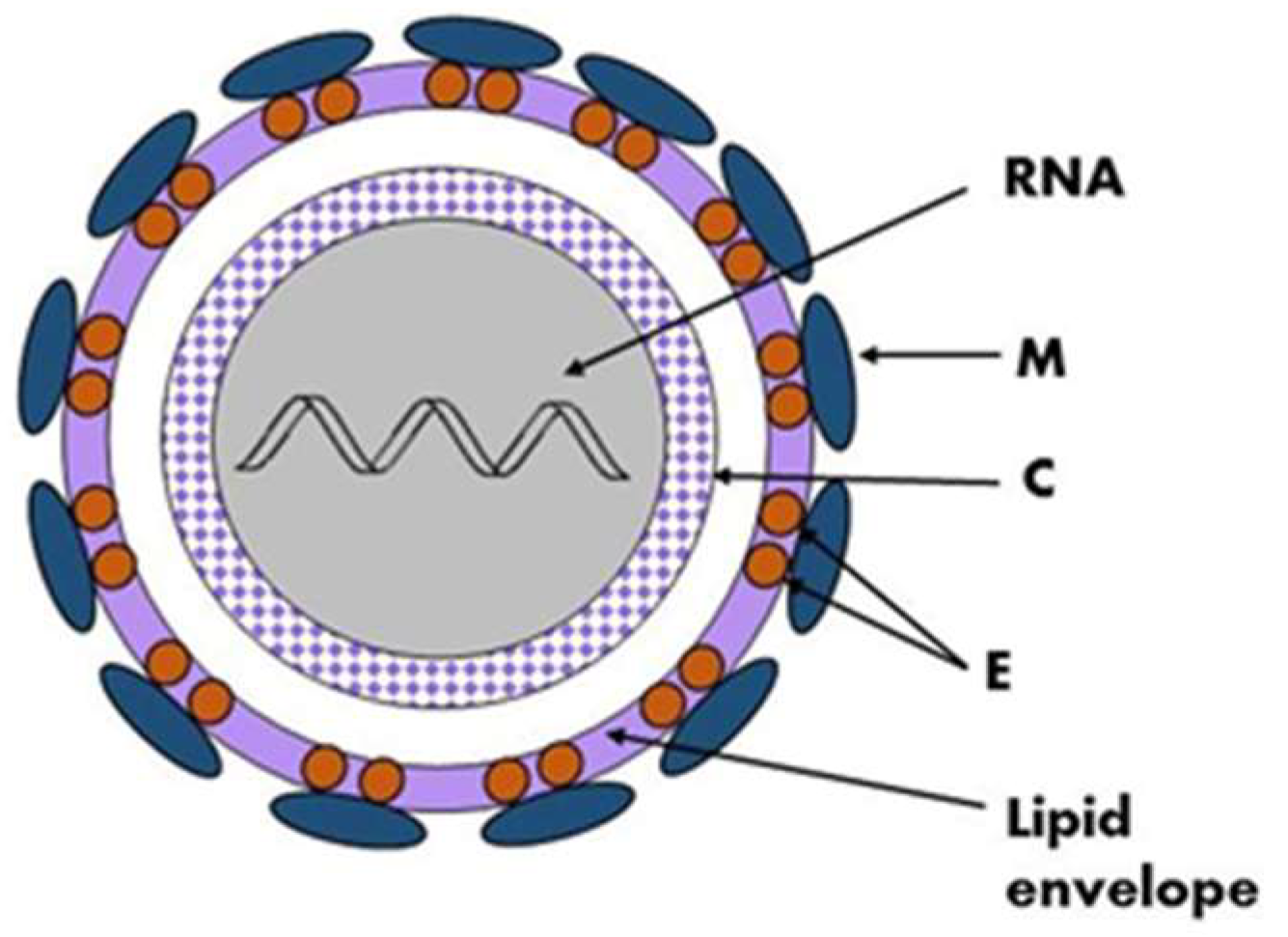
2. Virus Structure
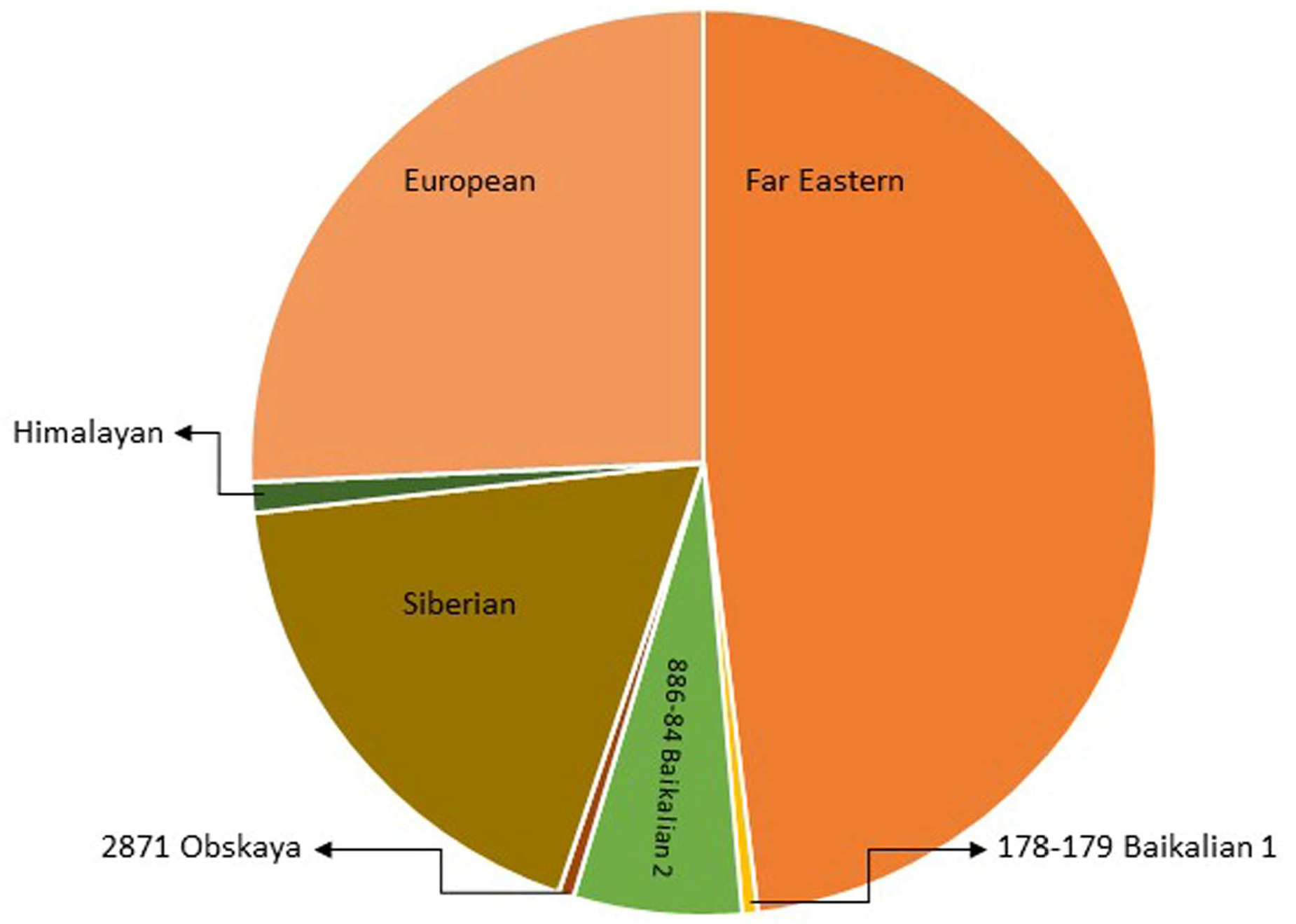
3. Phylogenetic Analysis of Circulating Virus Subtypes
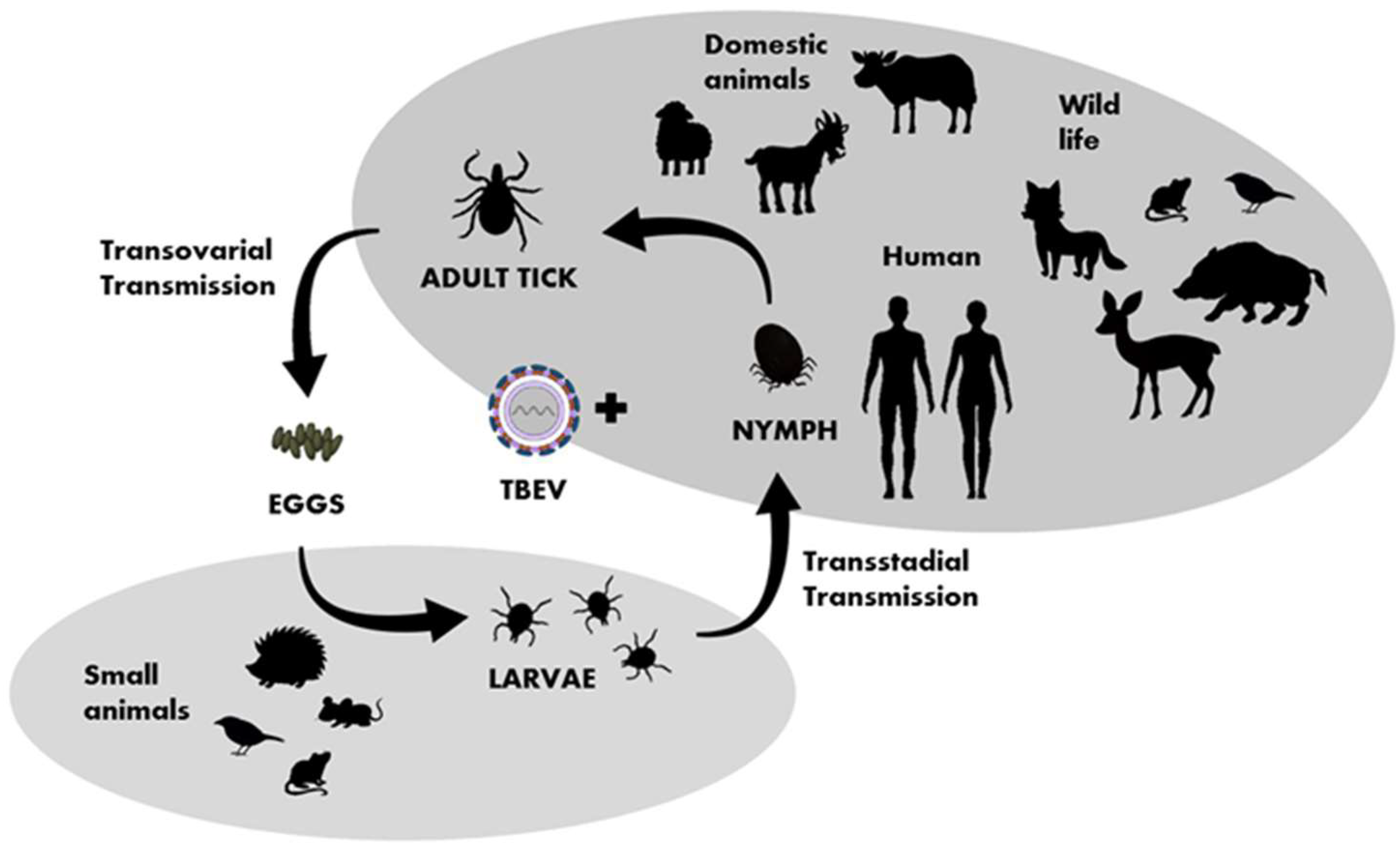
4. TBEV Reservoirs, Vectors, and Transmission
5. The Geographical Range and Frequency of Infections
6. Clinical Symptoms and Diagnosis
7. Impact of Climate and Land Use Changes
8. TBE and the COVID-19 Pandemic
9. Vaccination, Treatment, and Prevention
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gritsun, T.S.; Lashkevich, V.A.; Gould, E.A. Tick-Borne Encephalitis. Antivir. Res. 2003, 57, 129–146. [Google Scholar] [CrossRef] [PubMed]
- Chiffi, G.; Grandgirard, D.; Leib, S.L.; Chrdle, A.; Růžek, D. Tick-borne Encephalitis: A Comprehensive Review of the Epidemiology, Virology, and Clinical Picture. Rev. Med. Virol. 2023, 33, e2470. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Tick-Borne Encephalitis. In Annual Epidemiological Report for 2020; ECDC: Stockholm, Sweden, 2022. [Google Scholar]
- Socha, W.; Kwasnik, M.; Larska, M.; Rola, J.; Rozek, W. Vector-Borne Viral Diseases as a Current Threat for Human and Animal Health—One Health Perspective. J. Clin. Med. 2022, 11, 3026. [Google Scholar] [CrossRef]
- Ličková, M.; Fumačová Havlíková, S.; Sláviková, M.; Klempa, B. Alimentary Infections by Tick-Borne Encephalitis Virus. Viruses 2021, 14, 56. [Google Scholar] [CrossRef]
- Bakhvalova, V.N.; Panov, V.V.; Morozova, O.V. Tick-Borne Encephalitis Virus Quasispecies Rearrangements in Ticks and Mammals. In Flavivirus Encephalitis; Ruzek, D., Ed.; InTech: Houston, TX, USA, 2011; ISBN 978-953-307-669-0. [Google Scholar]
- Stadler, K.; Allison, S.L.; Schalich, J.; Heinz, F.X. Proteolytic Activation of Tick-Borne Encephalitis Virus by Furin. J. Virol. 1997, 71, 8475–8481. [Google Scholar] [CrossRef]
- Pulkkinen, L.I.A.; Barrass, S.V.; Domanska, A.; Överby, A.K.; Anastasina, M.; Butcher, S.J. Molecular Organisation of Tick-Borne Encephalitis Virus. Viruses 2022, 14, 792. [Google Scholar] [CrossRef] [PubMed]
- Füzik, T.; Formanová, P.; Růžek, D.; Yoshii, K.; Niedrig, M.; Plevka, P. Structure of Tick-Borne Encephalitis Virus and Its Neutralization by a Monoclonal Antibody. Nat. Commun. 2018, 9, 436. [Google Scholar] [CrossRef] [PubMed]
- Deviatkin, A.A.; Karganova, G.G.; Vakulenko, Y.A.; Lukashev, A.N. TBEV Subtyping in Terms of Genetic Distance. Viruses 2020, 12, 1240. [Google Scholar] [CrossRef]
- Upstone, L.; Colley, R.; Harris, M.; Goonawardane, N. Functional Characterization of 5′ Untranslated Region (UTR) Secondary RNA Structures in the Replication of Tick-Borne Encephalitis Virus in Mammalian Cells. PLoS Negl. Trop. Dis. 2023, 17, e0011098. [Google Scholar] [CrossRef]
- Ng, W.; Soto-Acosta, R.; Bradrick, S.; Garcia-Blanco, M.; Ooi, E. The 5′ and 3′ Untranslated Regions of the Flaviviral Genome. Viruses 2017, 9, 137. [Google Scholar] [CrossRef]
- Morozova, O.V.; Bakhvalova, V.N.; Morozov, I.V. Heterogeneity of 3′-Untraslated Region of Genome RNA of the Tick-Borne Encephalitis Virus (TBEV) Strains Isolated from Ticks in the Western Siberia, Russia. Int. J. Biomed. Sci. 2007, 3, 206–210. [Google Scholar] [PubMed]
- Hirano, M.; Muto, M.; Sakai, M.; Kondo, H.; Kobayashi, S.; Kariwa, H.; Yoshii, K. Dendritic Transport of Tick-Borne Flavivirus RNA by Neuronal Granules Affects Development of Neurological Disease. Proc. Natl. Acad. Sci. USA 2017, 114, 9960–9965. [Google Scholar] [CrossRef]
- Grard, G.; Moureau, G.; Charrel, R.N.; Lemasson, J.-J.; Gonzalez, J.-P.; Gallian, P.; Gritsun, T.S.; Holmes, E.C.; Gould, E.A.; de Lamballerie, X. Genetic Characterization of Tick-Borne Flaviviruses: New Insights into Evolution, Pathogenetic Determinants and Taxonomy. Virology 2007, 361, 80–92. [Google Scholar] [CrossRef]
- Dai, X.; Shang, G.; Lu, S.; Yang, J.; Xu, J. A New Subtype of Eastern Tick-Borne Encephalitis Virus Discovered in Qinghai-Tibet Plateau, China. Emerg. Microbes Infect. 2018, 7, 1–9. [Google Scholar] [CrossRef]
- Kovalev, S.Y.; Mukhacheva, T.A. Reconsidering the Classification of Tick-Borne Encephalitis Virus within the Siberian Subtype Gives New Insights into Its Evolutionary History. Infect. Genet. Evol. 2017, 55, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Kutschera, L.S.; Wolfinger, M.T. Evolutionary Traits of Tick-Borne Encephalitis Virus: Pervasive Non-Coding RNA Structure Conservation and Molecular Epidemiology. Virus Evol. 2022, 8, veac051. [Google Scholar] [CrossRef]
- Sukhorukov, G.A.; Paramonov, A.I.; Lisak, O.V.; Kozlova, I.V.; Bazykin, G.A.; Neverov, A.D.; Karan, L.S. The Baikal Subtype of Tick-Borne Encephalitis Virus Is Evident of Recombination between Siberian and Far-Eastern Subtypes. PLoS Negl. Trop. Dis. 2023, 17, e0011141. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, R. The Clinical and Epidemiological Profile of Tick-Borne Encephalitis in Southern Germany 1994–98. Brain 1999, 122, 2067–2078. [Google Scholar] [CrossRef]
- Mandl, C.W. Steps of the Tick-Borne Encephalitis Virus Replication Cycle That Affect Neuropathogenesis. Virus Res. 2005, 111, 161–174. [Google Scholar] [CrossRef] [PubMed]
- Mansfield, K.L.; Johnson, N.; Phipps, L.P.; Stephenson, J.R.; Fooks, A.R.; Solomon, T. Tick-Borne Encephalitis Virus—A Review of an Emerging Zoonosis. J. Gen. Virol. 2009, 90, 1781–1794. [Google Scholar] [CrossRef] [PubMed]
- Charrel, R.N.; Attoui, H.; Butenko, A.M.; Clegg, J.C.; Deubel, V.; Frolova, T.V.; Gould, E.A.; Gritsun, T.S.; Heinz, F.X.; Labuda, M.; et al. Tick-Borne Virus Diseases of Human Interest in Europe. Clin. Microbiol. Infect. 2004, 10, 1040–1055. [Google Scholar] [CrossRef]
- Tkachev, S.E.; Chicherina, G.S.; Golovljova, I.; Belokopytova, P.S.; Tikunov, A.Y.; Zadora, O.V.; Glupov, V.V.; Tikunova, N.V. New Genetic Lineage within the Siberian Subtype of Tick-Borne Encephalitis Virus Found in Western Siberia, Russia. Infect. Genet. Evol. 2017, 56, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Kozlova, I.V.; Verkhozina, M.M.; Demina, T.V.; Dzhioev, Y.P.; Tkachev, S.E.; Karan, L.S.; Doroshchenko, E.K.; Lisak, O.V.; Suntsova, O.V.; Paramonov, A.I.; et al. Genetic and Biological Properties of Original TBEV Strains Group Circulating in Eastern Siberia. In Encephalitis; Tkachev, S., Ed.; InTech: Houston, TX, USA, 2013; ISBN 978-953-51-0925-9. [Google Scholar]
- Tkachev, S.E.; Babkin, I.V.; Chicherina, G.S.; Kozlova, I.V.; Verkhozina, M.M.; Demina, T.V.; Lisak, O.V.; Doroshchenko, E.K.; Dzhioev, Y.P.; Suntsova, O.V.; et al. Genetic Diversity and Geographical Distribution of the Siberian Subtype of the Tick-Borne Encephalitis Virus. Ticks Tick-Borne Dis. 2020, 11, 101327. [Google Scholar] [CrossRef]
- Mandl, C.W.; Heinz, F.X.; Holzmann, H.; Kunz, C.; Ecker, M. Infectious CDNA Clones of Tick-Borne Encephalitis Virus European Subtype Prototypic Strain Neudoerfl and High Virulence Strain Hypr. J. Gen. Virol. 1997, 78, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Dumpis, U.; Crook, D.; Oksi, J. Tick-Borne Encephalitis. Clin. Infect. Dis. 1999, 28, 882–890. [Google Scholar] [CrossRef] [PubMed]
- Chitimia-Dobler, L.; Lemhöfer, G.; Król, N.; Bestehorn, M.; Dobler, G.; Pfeffer, M. Repeated Isolation of Tick-Borne Encephalitis Virus from Adult Dermacentor Reticulatus Ticks in an Endemic Area in Germany. Parasites Vectors 2019, 12, 90. [Google Scholar] [CrossRef] [PubMed]
- Krivanec, K.; Kopecký, J.; Tomková, E.; Grubhoffer, L. Isolation of TBE Virus from the Tick Ixodes Hexagonus. Folia Parasitol. 1988, 35, 273–276. [Google Scholar]
- Lichard, M.; Kozuch, O. Persistence of Tick-Borne Encephalitis Virus in Nymphs and Adults of Ixodes Arboricola and Its Transmission to White Mice. Acta Virol. 1967, 11, 480. [Google Scholar] [PubMed]
- Hubálek, Z.; Rudolf, I. Tick-Borne Viruses in Europe. Parasitol. Res. 2012, 111, 9–36. [Google Scholar] [CrossRef] [PubMed]
- Riedl, H.; Kozuch, O.; Sixl, W.; Schmeller, E.; Nosek, J. Isolation of the tick-borne encephalitis virus (TBE-virus) from the tick Haemaphysalis concinna Koch. Arch. Hyg. Bakteriol. 1971, 154, 610–611. [Google Scholar]
- Kozuch, O.; Nosek, J. Transmission of Tick-Borne Encephalitis (TBE) Virus by Dermacentor Marginatus and D. Reticulatus Ticks. Acta Virol. 1971, 15, 334. [Google Scholar]
- Nosek, J.; Ciampor, F.; Kozuch, O.; Rajcáni, J. Localization of Tick-Borne Encephalitis Virus in Alveolar Cells of Salivary Glands of Dermacentor Marginatus and Haemaphysalis Inermis Ticks. Acta Virol. 1972, 16, 493–497. [Google Scholar]
- Bakhvalova, V.N.; Dobrotvorsky, A.K.; Panov, V.V.; Matveeva, V.A.; Tkachev, S.E.; Morozova, O.V. Natural Tick-Borne Encephalitis Virus Infection among Wild Small Mammals in the Southeastern Part of Western Siberia, Russia. Vector Borne Zoonotic Dis. 2006, 6, 32–41. [Google Scholar] [CrossRef]
- Bakhvalova, V.N.; Potapova, O.F.; Panov, V.V.; Morozova, O.V. Vertical Transmission of Tick-Borne Encephalitis Virus between Generations of Adapted Reservoir Small Rodents. Virus Res. 2009, 140, 172–178. [Google Scholar] [CrossRef]
- Wilhelmsson, P.; Jaenson, T.G.T.; Olsen, B.; Waldenström, J.; Lindgren, P.-E. Migratory Birds as Disseminators of Ticks and the Tick-Borne Pathogens Borrelia Bacteria and Tick-Borne Encephalitis (TBE) Virus: A Seasonal Study at Ottenby Bird Observatory in South-Eastern Sweden. Parasites Vectors 2020, 13, 607. [Google Scholar] [CrossRef] [PubMed]
- Michelitsch, A.; Wernike, K.; Klaus, C.; Dobler, G.; Beer, M. Exploring the Reservoir Hosts of Tick-Borne Encephalitis Virus. Viruses 2019, 11, 669. [Google Scholar] [CrossRef]
- Holding, M.; Dowall, S.D.; Medlock, J.M.; Carter, D.P.; McGinley, L.; Curran-French, M.; Pullan, S.T.; Chamberlain, J.; Hansford, K.M.; Baylis, M.; et al. Detection of New Endemic Focus of Tick-Borne Encephalitis Virus (TBEV), Hampshire/Dorset Border, England, September 2019. Euro Surveill. 2019, 24, 1900658. [Google Scholar] [CrossRef] [PubMed]
- Jääskeläinen, A.; Tonteri, E.; Pieninkeroinen, I.; Sironen, T.; Voutilainen, L.; Kuusi, M.; Vaheri, A.; Vapalahti, O. Siberian Subtype Tick-Borne Encephalitis Virus in Ixodes Ricinus in a Newly Emerged Focus, Finland. Ticks Tick Borne Dis. 2016, 7, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y. Multiple Transmissions of Tick-Borne Encephalitis Virus between Japan and Russia. Genes. Genet. Syst. 2007, 82, 187–195. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cerný, V. The Role of Mammals in Natural Foci of Tick-Borne Encephalitis in Central Europe. Folia Parasitol. 1975, 22, 271–273. [Google Scholar]
- Klaus, C.; Gethmann, J.; Hoffmann, B.; Ziegler, U.; Heller, M.; Beer, M. Tick Infestation in Birds and Prevalence of Pathogens in Ticks Collected from Different Places in Germany. Parasitol. Res. 2016, 115, 2729–2740. [Google Scholar] [CrossRef]
- Hofmeester, T.R.; Sprong, H.; Jansen, P.A.; Prins, H.H.T.; van Wieren, S.E. Deer Presence Rather than Abundance Determines the Population Density of the Sheep Tick, Ixodes Ricinus, in Dutch Forests. Parasit. Vectors 2017, 10, 433. [Google Scholar] [CrossRef]
- Krzysiak, M.K.; Anusz, K.; Konieczny, A.; Rola, J.; Salat, J.; Strakova, P.; Olech, W.; Larska, M. The European Bison (Bison Bonasus) as an Indicatory Species for the Circulation of Tick-Borne Encephalitis Virus (TBEV) in Natural Foci in Poland. Ticks Tick-Borne Dis. 2021, 12, 101799. [Google Scholar] [CrossRef]
- Salat, J.; Ruzek, D. Tick-Borne Encephalitis in Domestic Animals. Acta Virol. 2020, 64, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Böhm, B.; Schade, B.; Bauer, B.; Hoffmann, B.; Hoffmann, D.; Ziegler, U.; Beer, M.; Klaus, C.; Weissenböck, H.; Böttcher, J. Tick-Borne Encephalitis in a Naturally Infected Sheep. BMC Vet. Res. 2017, 13, 267. [Google Scholar] [CrossRef] [PubMed]
- Holzmann, H.; Aberle, S.W.; Stiasny, K.; Werner, P.; Mischak, A.; Zainer, B.; Netzer, M.; Koppi, S.; Bechter, E.; Heinz, F.X. Tick-Borne Encephalitis from Eating Goat Cheese in a Mountain Region of Austria. Emerg. Infect. Dis. 2009, 15, 1671–1673. [Google Scholar] [CrossRef] [PubMed]
- Buczek, A.M.; Buczek, W.; Buczek, A.; Wysokińska-Miszczuk, J. Food-Borne Transmission of Tick-Borne Encephalitis Virus-Spread, Consequences, and Prophylaxis. Int. J. Environ. Res. Public Health 2022, 19, 1812. [Google Scholar] [CrossRef]
- Levkovich, E.N.; Pogodina, V.V. Infection through the alimentary tract with tick-borne encephalitis. Vopr. Virusol. 1958, 3, 145–150. [Google Scholar] [PubMed]
- Bogovic, P.; Strle, F. Tick-Borne Encephalitis: A Review of Epidemiology, Clinical Characteristics, and Management. World J. Clin. Cases 2015, 3, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Avšič-Županc, T.; Poljak, M.; Matičič, M.; Radšel-Medvešček, A.; LeDuc, J.W.; Stiasny, K.; Kunz, C.; Heinz, F.X. Laboratory Acquired Tick-Borne Meningoencephalitis: Characterisation of Virus Strains. Clin. Diagn. Virol. 1995, 4, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Wahlberg, P.; Saikku, P.; Brummer-Korvenkontio, M. Tick-Borne Viral Encephalitis in Finland. The Clinical Features of Kumlinge Disease during 1959-1987. J. Intern. Med. 1989, 225, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Lipowski, D.; Popiel, M.; Perlejewski, K.; Nakamura, S.; Bukowska-Ośko, I.; Rzadkiewicz, E.; Dzieciątkowski, T.; Milecka, A.; Wenski, W.; Ciszek, M.; et al. A Cluster of Fatal Tick-Borne Encephalitis Virus Infection in Organ Transplant Setting. J. Infect. Dis. 2017, 215, 896–901. [Google Scholar] [CrossRef] [PubMed]
- Kerlik, J.; Avdičová, M.; Musilová, M.; Bérešová, J.; Mezencev, R. Breast Milk as Route of Tick-Borne Encephalitis Virus Transmission from Mother to Infant. Emerg. Infect. Dis. 2022, 28, 1060–1061. [Google Scholar] [CrossRef] [PubMed]
- Beauté, J.; Spiteri, G.; Warns-Petit, E.; Zeller, H. Tick-Borne Encephalitis in Europe, 2012 to 2016. Euro Surveill. 2018, 23, 1800201. [Google Scholar] [CrossRef] [PubMed]
- Yoshii, K.; Song, J.Y.; Park, S.-B.; Yang, J.; Schmitt, H.-J. Tick-Borne Encephalitis in Japan, Republic of Korea and China. Emerg. Microbes Infect. 2017, 6, e82. [Google Scholar] [CrossRef]
- Chen, X.; Li, F.; Yin, Q.; Liu, W.; Fu, S.; He, Y.; Lei, W.; Xu, S.; Liang, G.; Wang, S.; et al. Epidemiology of Tick-Borne Encephalitis in China, 2007–2018. PLoS ONE 2019, 14, e0226712. [Google Scholar] [CrossRef]
- Uchida, L.; Hayasaka, D.; Ngwe Tun, M.M.; Morita, K.; Muramatsu, Y.; Hagiwara, K. Survey of Tick-Borne Zoonotic Viruses in Wild Deer in Hokkaido, Japan. J. Vet. Med. Sci. 2018, 80, 985–988. [Google Scholar] [CrossRef]
- Yoshii, K.; Takahashi-Iwata, I.; Shirai, S.; Kobayashi, S.; Yabe, I.; Sasaki, H. A Retrospective Epidemiological Study of Tick-Borne Encephalitis Virus in Patients with Neurological Disorders in Hokkaido, Japan. Microorganisms 2020, 8, 1672. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-Y.; Yun, S.-M.; Han, M.G.; Lee, I.Y.; Lee, N.Y.; Jeong, Y.E.; Lee, B.C.; Ju, Y.R. Isolation of Tick-Borne Encephalitis Viruses from Wild Rodents, South Korea. Vector-Borne Zoonotic Dis. 2008, 8, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Holding, M.; Dowall, S.D.; Medlock, J.M.; Carter, D.P.; Pullan, S.T.; Lewis, J.; Vipond, R.; Rocchi, M.S.; Baylis, M.; Hewson, R. Tick-Borne Encephalitis Virus, United Kingdom. Emerg. Infect. Dis. 2020, 26, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Jahfari, S.; de Vries, A.; Rijks, J.M.; Van Gucht, S.; Vennema, H.; Sprong, H.; Rockx, B. Tick-Borne Encephalitis Virus in Ticks and Roe Deer, the Netherlands. Emerg. Infect. Dis. 2017, 23, 1028–1030. [Google Scholar] [CrossRef]
- Ternovoi, V.A.; Kurzhukov, G.P.; Sokolov, Y.V.; Ivanov, G.Y.; Ivanisenko, V.A.; Loktev, A.V.; Ryder, R.W.; Netesov, S.V.; Loktev, V.B. Tick-Borne Encephalitis with Hemorrhagic Syndrome, Novosibirsk Region, Russia, 1999. Emerg. Infect. Dis. 2003, 9, 743–746. [Google Scholar] [CrossRef]
- Bogovic, P.; Lotric-Furlan, S.; Strle, F. What Tick-Borne Encephalitis May Look like: Clinical Signs and Symptoms. Travel Med. Infect. Dis. 2010, 8, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Ergunay, K.; Tkachev, S.; Kozlova, I.; Růžek, D. A Review of Methods for Detecting Tick-Borne Encephalitis Virus Infection in Tick, Animal, and Human Specimens. Vector Borne Zoonotic Dis. 2016, 16, 4–12. [Google Scholar] [CrossRef]
- Taba, P.; Schmutzhard, E.; Forsberg, P.; Lutsar, I.; Ljøstad, U.; Mygland, Å.; Levchenko, I.; Strle, F.; Steiner, I. EAN Consensus Review on Prevention, Diagnosis and Management of Tick-Borne Encephalitis. Eur. J. Neurol. 2017, 24, 1214-e61. [Google Scholar] [CrossRef] [PubMed]
- Holzmann, H. Diagnosis of Tick-Borne Encephalitis. Vaccine 2003, 21, S36–S40. [Google Scholar] [CrossRef] [PubMed]
- Niedrig, M.; Vaisviliene, D.; Teichmann, A.; Klockmann, U.; Biel, S.S. Comparison of Six Different Commercial IgG-ELISA Kits for the Detection of TBEV-Antibodies. J. Clin. Virol. 2001, 20, 179–182. [Google Scholar] [CrossRef] [PubMed]
- Günther, G.; Haglund, M.; Lindquist, L.; Sköldenberg, B.; Forsgren, M. Intrathecal IgM, IgA and IgG Antibody Response in Tick-Borne Encephalitis. Long-Term Follow-up Related to Clinical Course and Outcome. Clin. Diagn. Virol. 1997, 8, 17–29. [Google Scholar] [CrossRef]
- Reusken, C.; Boonstra, M.; Rugebregt, S.; Scherbeijn, S.; Chandler, F.; Avšič-Županc, T.; Vapalahti, O.; Koopmans, M.; GeurtsvanKessel, C.H. An Evaluation of Serological Methods to Diagnose Tick-Borne Encephalitis from Serum and Cerebrospinal Fluid. J. Clin. Virol. 2019, 120, 78–83. [Google Scholar] [CrossRef]
- Donoso Mantke, O.; Aberle, S.W.; Avšič-Županc, T.; Labuda, M.; Niedrig, M. Quality Control Assessment for the PCR Diagnosis of Tick-Borne Encephalitis Virus Infections. J. Clin. Virol. 2007, 38, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Saksida, A.; Duh, D.; Lotric-Furlan, S.; Strle, F.; Petrovec, M.; Avsic-Zupanc, T. The Importance of Tick-Borne Encephalitis Virus RNA Detection for Early Differential Diagnosis of Tick-Borne Encephalitis. J. Clin. Virol. 2005, 33, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Caracciolo, I.; Bassetti, M.; Paladini, G.; Luzzati, R.; Santon, D.; Merelli, M.; Sabbata, G.D.; Carletti, T.; Marcello, A.; D’Agaro, P. Persistent Viremia and Urine Shedding of Tick-Borne Encephalitis Virus in an Infected Immunosuppressed Patient from a New Epidemic Cluster in North-Eastern Italy. J. Clin. Virol. 2015, 69, 48–51. [Google Scholar] [CrossRef] [PubMed]
- Pustijanac, E.; Buršić, M.; Talapko, J.; Škrlec, I.; Meštrović, T.; Lišnjić, D. Tick-Borne Encephalitis Virus: A Comprehensive Review of Transmission, Pathogenesis, Epidemiology, Clinical Manifestations, Diagnosis, and Prevention. Microorganisms 2023, 11, 1634. [Google Scholar] [CrossRef] [PubMed]
- Pukhovskaya, N.M.; Morozova, O.V.; Vysochina, N.P.; Belozerova, N.B.; Bakhmetyeva, S.V.; Zdanovskaya, N.I.; Seligman, S.J.; Ivanov, L.I. Tick-Borne Encephalitis Virus in Arthropod Vectors in the Far East of Russia. Ticks Tick. Borne Dis. 2018, 9, 824–833. [Google Scholar] [CrossRef]
- Bakhvalova, V.N.; Chicherina, G.S.; Potapova, O.F.; Panov, V.V.; Glupov, V.V.; Potapov, M.A.; Seligman, S.J.; Morozova, O.V. Tick-Borne Encephalitis Virus Diversity in Ixodid Ticks and Small Mammals in South-Western Siberia, Russia. Vector Borne Zoonotic Dis. 2016, 16, 541–549. [Google Scholar] [CrossRef]
- Gray, J.S.; Dautel, H.; Estrada-Peña, A.; Kahl, O.; Lindgren, E. Effects of Climate Change on Ticks and Tick-Borne Diseases in Europe. Interdiscip. Perspect. Infect. Dis. 2009, 2009, 593232. [Google Scholar] [CrossRef]
- Heine, P.; Hausen, J.; Ottermanns, R.; Schäffer, A.; Roß-Nickoll, M. Forest Conversion from Norway Spruce to European Beech Increases Species Richness and Functional Structure of Aboveground Macrofungal Communities. For. Ecol. Manag. 2019, 432, 522–533. [Google Scholar] [CrossRef]
- Palo, R.T. Tick-Borne Encephalitis Transmission Risk: Its Dependence on Host Population Dynamics and Climate Effects. Vector-Borne Zoonotic Dis. 2014, 14, 346–352. [Google Scholar] [CrossRef]
- Randolph, S.E.; On Behalf of The Eden-Tbd Sub-Project Team, C. Human Activities Predominate in Determining Changing Incidence of Tick-Borne Encephalitis in Europe. Eurosurveillance 2010, 15, 19606. [Google Scholar] [CrossRef]
- Čabanová, V.; Kerlik, J.; Kirschner, P.; Rosochová, J.; Klempa, B.; Sláviková, M.; Ličková, M. Co-Circulation of West Nile, Usutu, and Tick-Borne Encephalitis Viruses in the Same Area: A Great Challenge for Diagnostic and Blood and Organ Safety. Viruses 2023, 15, 366. [Google Scholar] [CrossRef]
- Tardy, O.; Acheson, E.S.; Bouchard, C.; Chamberland, É.; Fortin, A.; Ogden, N.H.; Leighton, P.A. Mechanistic Movement Models to Predict Geographic Range Expansions of Ticks and Tick-Borne Pathogens: Case Studies with Ixodes Scapularis and Amblyomma Americanum in Eastern North America. Ticks Tick-Borne Dis. 2023, 14, 102161. [Google Scholar] [CrossRef] [PubMed]
- Nah, K.; Bede-Fazekas, Á.; Trájer, A.J.; Wu, J. The Potential Impact of Climate Change on the Transmission Risk of Tick-Borne Encephalitis in Hungary. BMC Infect. Dis. 2020, 20, 34. [Google Scholar] [CrossRef] [PubMed]
- Chow, E.J.; Uyeki, T.M.; Chu, H.Y. The Effects of the COVID-19 Pandemic on Community Respiratory Virus Activity. Nat. Rev. Microbiol. 2022, 21, 195–210. [Google Scholar] [CrossRef] [PubMed]
- Zając, Z.; Bartosik, K.; Kulisz, J.; Woźniak, A. Incidence of Tick-Borne Encephalitis during the COVID-19 Pandemic in Selected European Countries. J. Clin. Med. 2022, 11, 803. [Google Scholar] [CrossRef]
- Ullrich, A.; Schranz, M.; Rexroth, U.; Hamouda, O.; Schaade, L.; Diercke, M.; Boender, T.S. Impact of the COVID-19 Pandemic and Associated Non-Pharmaceutical Interventions on Other Notifiable Infectious Diseases in Germany: An Analysis of National Surveillance Data during Week 1–2016—Week 32–2020. Lancet Reg. Health Eur. 2021, 6, 100103. [Google Scholar] [CrossRef]
- Kollaritsch, H.; Paulke-Korinek, M.; Holzmann, H.; Hombach, J.; Bjorvatn, B.; Barrett, A. Vaccines and Vaccination against Tick-Borne Encephalitis. Expert Rev. Vaccines 2012, 11, 1103–1119. [Google Scholar] [CrossRef]
- Šmit, R.; Postma, M.J. Review of Tick-Borne Encephalitis and Vaccines: Clinical and Economical Aspects. Expert Rev. Vaccines 2015, 14, 737–747. [Google Scholar] [CrossRef]
- Tuchynskaya, K.; Volok, V.; Illarionova, V.; Okhezin, E.; Polienko, A.; Belova, O.; Rogova, A.; Chernokhaeva, L.; Karganova, G. Experimental Assessment of Possible Factors Associated with Tick-Borne Encephalitis Vaccine Failure. Microorganisms 2021, 9, 1172. [Google Scholar] [CrossRef] [PubMed]
- Hills, S.L.; Broussard, K.R.; Broyhill, J.C.; Shastry, L.G.; Cossaboom, C.M.; White, J.L.; Machesky, K.D.; Kosoy, O.; Girone, K.; Klena, J.D.; et al. Tick-Borne Encephalitis among US Travellers, 2010–2020. J. Travel Med. 2022, 29, taab167. [Google Scholar] [CrossRef]
- Rumyantsev, A.A.; Chanock, R.M.; Murphy, B.R.; Pletnev, A.G. Comparison of Live and Inactivated Tick-Borne Encephalitis Virus Vaccines for Safety, Immunogenicity and Efficacy in Rhesus Monkeys. Vaccine 2006, 24, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Ershova, A.S.; Gra, O.A.; Lyaschuk, A.M.; Grunina, T.M.; Tkachuk, A.P.; Bartov, M.S.; Savina, D.M.; Sergienko, O.V.; Galushkina, Z.M.; Gudov, V.P.; et al. Recombinant Domains III of Tick-Borne Encephalitis Virus Envelope Protein in Combination with Dextran and CpGs Induce Immune Response and Partial Protectiveness against TBE Virus Infection in Mice. BMC Infect. Dis. 2016, 16, 544. [Google Scholar] [CrossRef] [PubMed]
- Wollner, C.J.; Richner, J.M. MRNA Vaccines against Flaviviruses. Vaccines 2021, 9, 148. [Google Scholar] [CrossRef] [PubMed]
- VanBlargan, L.A.; Himansu, S.; Foreman, B.M.; Ebel, G.D.; Pierson, T.C.; Diamond, M.S. An MRNA Vaccine Protects Mice against Multiple Tick-Transmitted Flavivirus Infections. Cell Rep. 2018, 25, 3382–3392.e3. [Google Scholar] [CrossRef] [PubMed]
- Mandl, C.W.; Aberle, J.H.; Aberle, S.W.; Holzmann, H.; Allison, S.L.; Heinz, F.X. In Vitro-Synthesized Infectious RNA as an Attenuated Live Vaccine in a Flavivirus Model. Nat. Med. 1998, 4, 1438–1440. [Google Scholar] [CrossRef]
- Kofler, R.M.; Aberle, J.H.; Aberle, S.W.; Allison, S.L.; Heinz, F.X.; Mandl, C.W. Mimicking Live Flavivirus Immunization with a Noninfectious RNA Vaccine. Proc. Natl. Acad. Sci. USA 2004, 101, 1951–1956. [Google Scholar] [CrossRef] [PubMed]
- Eyer, L.; Nencka, R.; de Clercq, E.; Seley-Radtke, K.; Růžek, D. Nucleoside Analogs as a Rich Source of Antiviral Agents Active against Arthropod-Borne Flaviviruses. Antivir. Chem. Chemother. 2018, 26, 2040206618761299. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.-D.; Tang, H.-L.; Peng, H.-R.; Ren, R.-W.; Zhao, P.; Zhao, L.-J. Inhibition of Tick-Borne Encephalitis Virus in Cell Cultures by Ribavirin. Front. Microbiol. 2023, 14, 1182798. [Google Scholar] [CrossRef] [PubMed]
- VanBlargan, L.A.; Errico, J.M.; Kafai, N.M.; Burgomaster, K.E.; Jethva, P.N.; Broeckel, R.M.; Meade-White, K.; Nelson, C.A.; Himansu, S.; Wang, D.; et al. Broadly Neutralizing Monoclonal Antibodies Protect against Multiple Tick-Borne Flaviviruses. J. Exp. Med. 2021, 218, e20210174. [Google Scholar] [CrossRef] [PubMed]
- Eyer, L.; Seley-Radtke, K.; Ruzek, D. New Directions in the Experimental Therapy of Tick-Borne Encephalitis. Antivir. Res. 2023, 210, 105504. [Google Scholar] [CrossRef]
- Wardhaugh, K.G. Insecticidal Activity of Synthetic Pyrethroids, Organophosphates, Insect Growth Regulators, and Other Livestock Parasiticides: An Australian Perspective. Environ. Toxicol. Chem. 2005, 24, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Antwi, F.B.; Shama, L.M.; Peterson, R.K.D. Risk Assessments for the Insect Repellents DEET and Picaridin. Regul. Toxicol. Pharmacol. 2008, 51, 31–36. [Google Scholar] [CrossRef]
- George, J.E.; Pound, J.M.; Davey, R.B. Chemical Control of Ticks on Cattle and the Resistance of These Parasites to Acaricides. Parasitology 2004, 129, S353–S366. [Google Scholar] [CrossRef]
- Van Wieren, S.E.; Braks, M.A.H.; Lahr, J. 19. Effectiveness and Environmental Hazards of Acaricides Applied to Large Mammals for Tick Control. In Ecology and Control of Vector-borne Diseases; Braks, M.A.H., Van Wieren, S.E., Takken, W., Sprong, H., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2016; Volume 4, pp. 265–278. ISBN 978-90-8686-293-1. [Google Scholar]
- Banumathi, B.; Vaseeharan, B.; Rajasekar, P.; Prabhu, N.M.; Ramasamy, P.; Murugan, K.; Canale, A.; Benelli, G. Exploitation of Chemical, Herbal and Nanoformulated Acaricides to Control the Cattle Tick, Rhipicephalus (Boophilus) Microplus—A Review. Vet. Parasitol. 2017, 244, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Nana, P.; Ekesi, S.; Nchu, F.; Maniania, N.K. Compatibility of Metarhizium anisopliae with Calpurnia aurea Leaf Extracts and Virulence against Rhipicephalus pulchellus. J. Appl. Entomol. 2016, 140, 590–597. [Google Scholar] [CrossRef]
- Adenubi, O.T.; Ahmed, A.S.; Fasina, F.O.; McGaw, L.J.; Eloff, J.N.; Naidoo, V. Pesticidal Plants as a Possible Alternative to Synthetic Acaricides in Tick Control: A Systematic Review and Meta-Analysis. Ind. Crops Prod. 2018, 123, 779–806. [Google Scholar] [CrossRef]
- Benelli, G.; Maggi, F.; Romano, D.; Stefanini, C.; Vaseeharan, B.; Kumar, S.; Higuchi, A.; Alarfaj, A.A.; Mehlhorn, H.; Canale, A. Nanoparticles as Effective Acaricides against Ticks—A Review. Ticks Tick-Borne Dis. 2017, 8, 821–826. [Google Scholar] [CrossRef] [PubMed]
- Rahlenbeck, S.; Fingerle, V.; Doggett, S. Prevention of Tick-Borne Diseases: An Overview. Br. J. Gen. Pract. 2016, 66, 492–494. [Google Scholar] [CrossRef]
- Kunze, M.; Banović, P.; Bogovič, P.; Briciu, V.; Čivljak, R.; Dobler, G.; Hristea, A.; Kerlik, J.; Kuivanen, S.; Kynčl, J.; et al. Recommendations to Improve Tick-Borne Encephalitis Surveillance and Vaccine Uptake in Europe. Microorganisms 2022, 10, 1283. [Google Scholar] [CrossRef] [PubMed]
| Vaccine | TBEV Strain | Adjuvant | Stabilizer | Distribution |
|---|---|---|---|---|
| FSME IMMUN/TICOVAC | Neudorfl TBEV-Eu | Al(OH)3 | HAS (human serum albumin) | Europe, USA |
| Encepur | K23 TBEV-Eu | Al(OH)3 | sucrose | Europe |
| EnceVir | 205 TBEV-Fe | Al(OH)3 | sucrose, HSA | Russian Federation |
| Tick-E-Vac | Sofjin TBEV-Fe | Al(OH)3 | sucrose, HSA | Russian Federation |
| TBE vaccine Moscow | Sofjin TBEV-Fe | Al(OH)3 | sucrose, HSA, gelatose | Russian Federation |
| SenTaiBao | Sen-Zhang TBEV-Fe | Al(OH)3 | HSA | China |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwasnik, M.; Rola, J.; Rozek, W. Tick-Borne Encephalitis—Review of the Current Status. J. Clin. Med. 2023, 12, 6603. https://doi.org/10.3390/jcm12206603
Kwasnik M, Rola J, Rozek W. Tick-Borne Encephalitis—Review of the Current Status. Journal of Clinical Medicine. 2023; 12(20):6603. https://doi.org/10.3390/jcm12206603
Chicago/Turabian StyleKwasnik, Malgorzata, Jerzy Rola, and Wojciech Rozek. 2023. "Tick-Borne Encephalitis—Review of the Current Status" Journal of Clinical Medicine 12, no. 20: 6603. https://doi.org/10.3390/jcm12206603
APA StyleKwasnik, M., Rola, J., & Rozek, W. (2023). Tick-Borne Encephalitis—Review of the Current Status. Journal of Clinical Medicine, 12(20), 6603. https://doi.org/10.3390/jcm12206603