Maximal Effort Cytoreduction in Epithelial Ovarian Cancer: Perioperative Complications and Survival Outcomes from a Retrospective Cohort
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Definitions
2.3. Statistical Analysis
3. Results
3.1. Patient and Tumor Characteristics
3.2. Tumor Dissemination, Extent of Resectability and Perioperative Complications
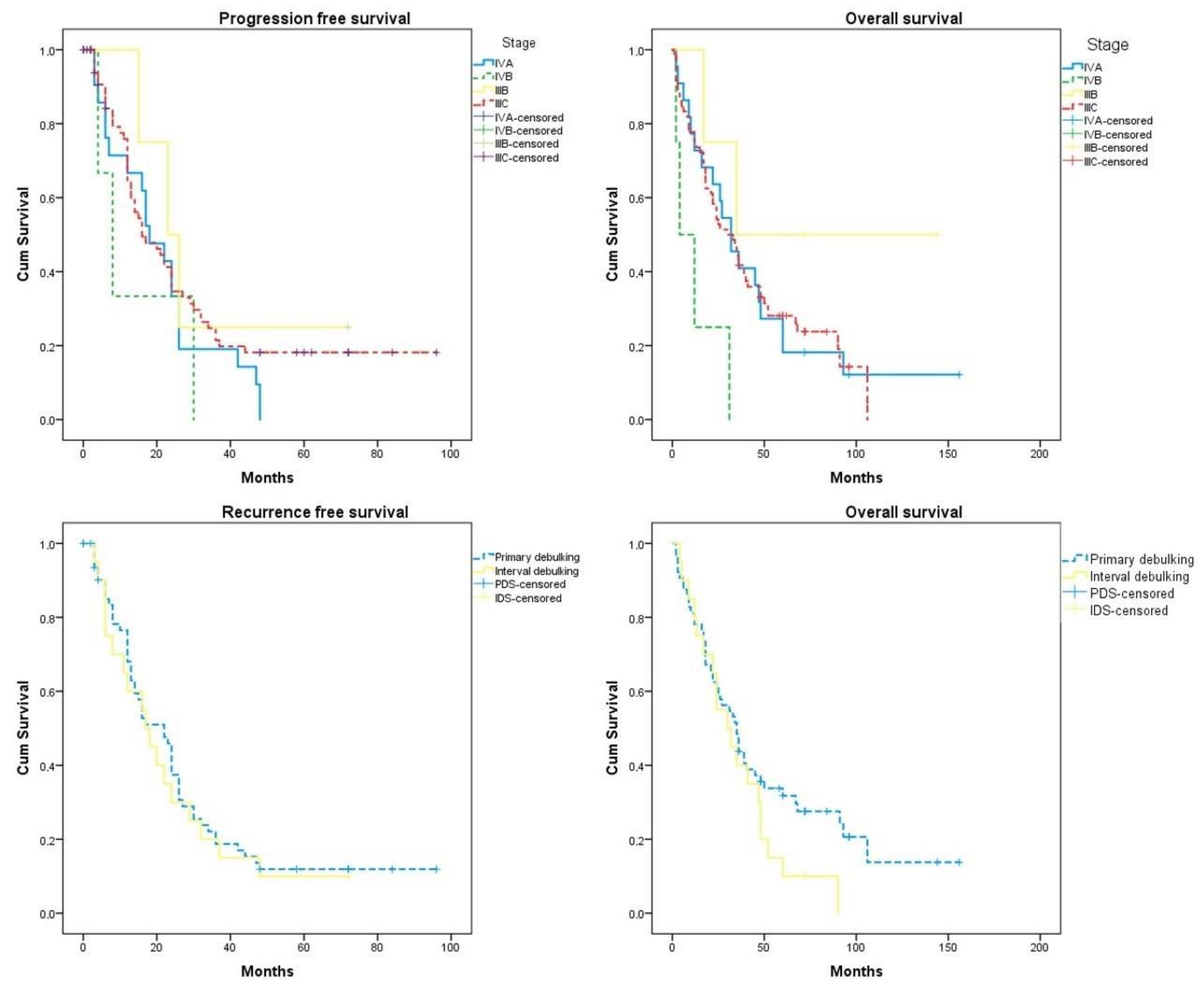
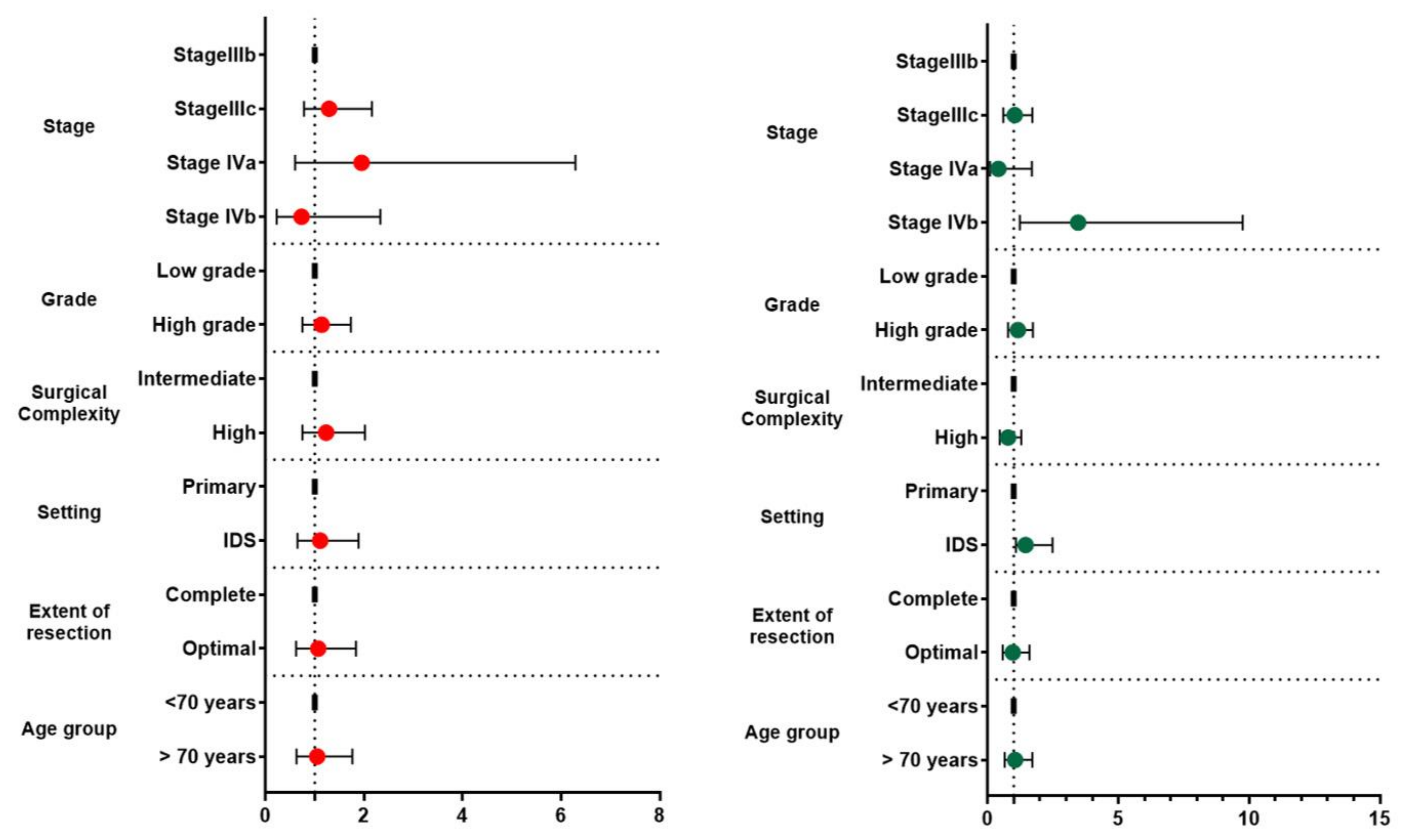
3.3. Survival Rates
4. Discussion
4.1. Strengths and Limitations of Our Study
4.2. Implications for Future Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- National Cancer Institute. Surveillance, Epidemiology and End Results. Cancer Stat Facts: Ovarian Cancer. Available online: https://seer.cancer.gov/statfacts/html/ovary.html (accessed on 22 July 2022).
- Colombo, N.; Sessa, C.; Du Bois, A.; Ledermann, J.; McCluggage, W.G.; McNeish, I.; Morice, P.; Pignata, S.; Ray-Coquard, I.; Vergote, I.; et al. ESMO–ESGO consensus conference recommendations on ovarian cancer: Pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Ann. Oncol. 2019, 30, 672–705. [Google Scholar] [CrossRef] [PubMed]
- Colombo, N.; Sessa, C.; Du Bois, A.; Ledermann, J.; McCluggage, W.G.; McNeish, I.; Morice, P.; Pignata, S.; Ray-Coquard, I.; Vergote, I.; et al. ESMO–ESGO consensus conference recommendations on ovarian cancer: Pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Int. J. Gynecol. Cancer 2019, 29, 728–760. [Google Scholar] [CrossRef]
- Banerjee, S.; Gonzalez-Martin, A.; Harter, P.; Lorusso, D.; Moore, K.N.; Oaknin, A.; Ray-Coquard, I. First-line PARP inhibitors in ovarian cancer: Summary of an ESMO Open—Cancer Horizons round-table discussion. ESMO Open 2020, 5, e001110. [Google Scholar] [CrossRef] [PubMed]
- Monk, B.; Minion, L.; Coleman, R. Anti-angiogenic agents in ovarian cancer: Past, present, and future. Ann. Oncol. 2016, 27 (Suppl. S1), i33–i39. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Trabert, B.; DeSantis, C.E.; Miller, K.D.; Samimi, G.; Runowicz, C.D.; Gaudet, M.M.; Jemal, A.; Siegel, R.L. Ovarian cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 284–296. [Google Scholar] [CrossRef] [PubMed]
- Du Bois, A.; Reuss, A.; Pujade-Lauraine, E.; Harter, P.; Ray-Coquard, I.; Pfisterer, J. Role of surgical outcome as prognostic factor in advanced epithelial ovarian cancer: A combined exploratory analysis of 3 prospectively randomized phase 3 multicenter trials: By the Arbeitsgemeinschaft Gynaekologische Onkologie Studiengruppe Ovarialkarzinom (AGO-OVAR) and the Groupe d’Investigateurs Nationaux Pour les Etudes des Cancers de l’Ovaire (GINECO. Cancer 2009, 115, 1234–1244. [Google Scholar] [CrossRef] [PubMed]
- Wimberger, P.; Wehling, M.; Lehmann, N.; Kimmig, R.; Schmalfeldt, B.; Burges, A.; Harter, P.; Pfisterer, J.; Du Bois, A. Influence of Residual Tumor on Outcome in Ovarian Cancer Patients with FIGO Stage IV Disease: An exploratory analysis of the AGO-OVAR (Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group). Ann. Surg. Oncol. 2010, 17, 1642–1648. [Google Scholar] [CrossRef]
- Ghirardi, V.; Moruzzi, M.; Bizzarri, N.; Vargiu, V.; D’Indinosante, M.; Garganese, G.; Pasciuto, T.; Loverro, M.; Scambia, G.; Fagotti, A. Minimal residual disease at primary debulking surgery versus complete tumor resection at interval debulking surgery in advanced epithelial ovarian cancer: A survival analysis. Gynecol. Oncol. 2020, 157, 209–213. [Google Scholar] [CrossRef]
- Fagotti, A.; Ferrandina, M.G.; Vizzielli, G.; Pasciuto, T.; Fanfani, F.; Gallotta, V.; Margariti, P.A.; Chiantera, V.; Costantini, B.; Alletti, S.G.; et al. Randomized trial of primary debulking surgery versus neoadjuvant chemotherapy for advanced epithelial ovarian cancer (SCORPION-NCT01461850). Int. J. Gynecol. Cancer 2020, 30, 1657–1664. [Google Scholar] [CrossRef]
- Vergote, I.; Tropé, C.G.; Amant, F.; Kristensen, G.B.; Ehlen, T.; Johnson, N.; Verheijen, R.H.M.; van der Burg, M.E.L.; Lacave, A.J.; Panici, P.B.; et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N. Engl. J. Med. 2010, 363, 943–953. [Google Scholar] [CrossRef]
- Reuss, A.; Du Bois, A.; Harter, P.; Fotopoulou, C.; Sehouli, J.; Aletti, G.; Guyon, F.; Greggi, S.; Mosgaard, B.J.; Reinthaller, A.; et al. TRUST: Trial of Radical Upfront Surgical Therapy in advanced ovarian cancer (ENGOT ov33/AGO-OVAR OP7). Int. J. Gynecol. Cancer 2019, 29, 1327–1331. [Google Scholar] [CrossRef] [PubMed]
- Fotopoulou, C.; Jones, B.P.; Savvatis, K.; Campbell, J.; Kyrgiou, M.; Farthing, A.; Brett, S.; Roux, R.; Hall, M.; Rustin, G.; et al. Maximal effort cytoreductive surgery for disseminated ovarian cancer in a UK setting: Challenges and possibilities. Arch. Gynecol. Obstet. 2016, 294, 607–614. [Google Scholar] [CrossRef] [PubMed]
- Chi, D.S.; Franklin, C.C.; Levine, D.A.; Akselrod, F.; Sabbatini, P.; Jarnagin, W.R.; DeMatteo, R.; Poynor, E.A.; Abu-Rustum, N.R.; Barakat, R.R. Improved optimal cytoreduction rates for stages IIIC and IV epithelial ovarian, fallopian tube, and primary peritoneal cancer: A change in surgical approach. Gynecol. Oncol. 2004, 94, 650–654. [Google Scholar] [CrossRef] [PubMed]
- Hall, M.; Savvatis, K.; Nixon, K.; Kyrgiou, M.; Hariharan, K.; Padwick, M.; Owens, O.; Cunnea, P.; Campbell, J.; Farthing, A.; et al. Maximal-Effort Cytoreductive Surgery for Ovarian Cancer Patients with a High Tumor Burden: Variations in Practice and Impact on Outcome. Ann. Surg. Oncol. 2019, 26, 2943–2951. [Google Scholar] [CrossRef] [PubMed]
- Tewarie, N.B.; van Driel, W.; van Ham, M.; Wouters, M.; Kruitwagen, R.; Kruse, A.; Yigit, R.; van der Aa, M.; Mens, J.; Stam, T.; et al. Postoperative outcomes of primary and interval cytoreductive surgery for advanced ovarian cancer registered in the Dutch Gynecological Oncology Audit (DGOA). Gynecol. Oncol. 2021, 162, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Narasimhulu, D.M.; Thannickal, A.; Kumar, A.; Weaver, A.L.; McGree, M.E.; Langstraat, C.L.; Cliby, W.A. Appropriate triage allows aggressive primary debulking surgery with rates of morbidity and mortality comparable to interval surgery after chemotherapy. Gynecol. Oncol. 2021, 160, 681–687. [Google Scholar] [CrossRef]
- Aletti, G.D.; Dowdy, S.C.; Gostout, B.S.; Jones, M.B.; Stanhope, R.C.; Wilson, T.O.; Podratz, K.C.; Cliby, W.A. Quality Improvement in the Surgical Approach to Advanced Ovarian Cancer: The Mayo Clinic Experience. J. Am. Coll. Surg. 2009, 208, 614–620. [Google Scholar] [CrossRef]
- Sehouli, J.; Könsgen, D.; Mustea, A.; Oskay-Özcelik, G.; Katsares, I.; Weidemann, H.; Lichtenegger, W. „IMO”—Intraoperatives Mapping des Ovarialkarzinoms. Zentralbl Gynakol. 2003, 125, 129–135. [Google Scholar] [CrossRef]
- Onda, T.; Satoh, T.; Ogawa, G.; Saito, T.; Kasamatsu, T.; Nakanishi, T.; Mizutani, T.; Takehara, K.; Okamoto, A.; Ushijima, K.; et al. Comparison of survival between primary debulking surgery and neoadjuvant chemotherapy for stage III/IV ovarian, tubal and peritoneal cancers in phase III randomised trial. Eur. J. Cancer 2020, 130, 114–125. [Google Scholar] [CrossRef]
- Sørensen, S.M.; Høgdall, C.; Mosgaard, B.J.; Dalgaard, M.I.R.; Jensen, M.P.; Fuglsang, K.; Schnack, T.H. Residual tumor and primary debulking surgery vs interval debulking surgery in stage IV epithelial ovarian cancer. Acta Obstet. Gynecol. Scand. 2022, 101, 334–343. [Google Scholar] [CrossRef]
- Pinelli, C.; Morotti, M.; Casarin, J.; Tozzi, R.; Ghezzi, F.; Mavroeidis, V.K.; Alazzam, M.; Majd, H.S. Interval Debulking Surgery for Advanced Ovarian Cancer in Elderly Patients (≥70 y): Does the Age Matter? J. Investig. Surg. 2021, 34, 1023–1030. [Google Scholar] [CrossRef] [PubMed]
- Kuusela, K.; Norppa, N.; Auranen, A.; Saarelainen, S. Maximal surgical effort increases the risk of postoperative complications in the treatment of advanced ovarian cancer. Eur. J. Surg. Oncol. (EJSO) 2022, 48, 2525–2530. [Google Scholar] [CrossRef] [PubMed]
- Aletti, G.; Dowdy, S.C.; Podratz, K.C.; Cliby, W.A. Relationship among surgical complexity, short-term morbidity, and overall survival in primary surgery for advanced ovarian cancer. Am. J. Obstet. Gynecol. 2007, 197, 676.e1–676.e7. [Google Scholar] [CrossRef] [PubMed]
- Haidopoulos, D.; Pergialiotis, V.; Aggelou, K.; Thomakos, N.; Alexakis, N.; Stamatakis, E.; Rodolakis, A. Pelvic exenteration for gynecologic malignancies: The experience of a tertiary center from Greece. Surg. Oncol. 2022, 40, 101702. [Google Scholar] [CrossRef]
- Fotopoulou, C.; Planchamp, F.; Aytulu, T.; Chiva, L.; Cina, A.; Ergönül, Ö.; Fagotti, A.; Haidopoulos, D.; Hasenburg, A.; Hughes, C.; et al. European Society of Gynaecological Oncology guidelines for the peri-operative management of advanced ovarian cancer patients undergoing debulking surgery. Int. J. Gynecol. Cancer 2021, 31, 1199–1206. [Google Scholar] [CrossRef]
- Sundar, S.; Cummins, C.; Kumar, S.; Long, J.; Arora, V.; Balega, J.; Broadhead, T.; Duncan, T.; Edmondson, R.; Fotopoulou, C.; et al. Quality of life from cytoreductive surgery in advanced ovarian cancer: Investigating the association between disease burden and surgical complexity in the international, prospective, SOCQER-2 cohort study. BJOG Int. J. Obstet. Gynaecol. 2022, 129, 1122–1132. [Google Scholar] [CrossRef]
| Age | 62 (23–84) |
|---|---|
| Stage | |
| IIIb | 4 |
| IIIc | 76 |
| IVa | 23 |
| IVb | 4 |
| Histological grade | |
| Low | 11 |
| High | 96 |
| Histological type | |
| Serous | 90 |
| Mucinous | 2 |
| Clear cell | 3 |
| Endometrioid | 3 |
| Carcinosarcoma | 9 |
| Interval debulking | 20 |
| Primary debulking | 87 |
| Ascites | |
| None | 24 |
| <500 mL | 40 |
| >500 mL | 42 |
| ECOG 0/1 | 88 |
| ECOG 2/3 | 19 |
| Hysterectomy | 106 |
| Omentectomy | 105 |
| Pelvic lymphadenectomy | 78 |
| Paraortic lymphadenectomy | 35 |
| Large bowel resection | 31 |
| Large bowel stoma | 16 |
| Small bowel resection | 10 |
| Small bowel stoma | 12 |
| Splenectomy | 26 |
| Cholocystectomy | 3 |
| Sphenoid liver resection | 9 |
| Diaphragmatic stripping | 34 |
| Diaphragmatic resection | 18 |
| Extent of debulking | |
| R0 | 81 |
| R1 | 20 |
| R2 | 6 |
| Parameter | Progression Free Survival (95% CI) | Overall Survival Months (95% CI) | Hazard Ratio 95% CI PFS | Hazard Ratio 95% CI OS |
|---|---|---|---|---|
| Stage | ||||
| IIIb | 34.00 (12.14, 55.86) | 85.00 (26.85, 143.15) | Ref | Ref |
| IIIc | 31.21 (23.19, 39.23) | 42.21 (33.55, 50.87) | 1.29 (0.78, 2.16) | 1.04 (0.60, 1.71) |
| IVa | 21.05 (14.70, 27.39) | 46.95 (27.29, 66.15) | 1.95 (0.60, 6.29) | 0.41 (0.10, 1.69) |
| IVb | 14.00 (2.00, 29.84) | 12.25 (2.00, 25.21 | 0.73 (0.23, 2.33) | 3.46 (1.23, 9.75) |
| Grade of differentiation | ||||
| Low grade | 26.25 (11.94, 40.56) | 48.20 (26.96, 69.44) | Ref | Ref |
| High grade | 25.77 (20.12, 31.41) | 46.82 (35.44, 58.20) | 1.14 (0.75, 1.73) | 1.16 (0.78, 1.73) |
| Surgical complexity | ||||
| Intermediate | 31.59 (23.59, 39.59) | 42.93 (31.04, 54.82) | Ref | Ref |
| High | 24.74 (15.60, 33.88) | 46.51 (34.16, 58.86) | 1.23 (0.75, 2.02) | 0.78 (0.47, 1.29) |
| Setting of operation | ||||
| Primary | 23.15 (14.40, 31.90) | 53.75 (39.98, 67.52) | Ref | Ref |
| Interval | 28.13 (21.15, 35.11) | 35.15 (24.21, 46.09) | 1.11 (0.65, 1.89) | 1.45 (1.08, 2.48) |
| Level of resection | ||||
| R0 | 28.99 (22.17, 35.81) | 46.14 (35.23, 57.05) | Ref | Ref |
| R1 | 24.91 (15.21, 34.61) | 47.92 (27.85, 67.98) | 1.07 (0.62, 1.84) | 0.96 (0.58, 1.60) |
| Patient age | ||||
| <70 years | 26.94 (21.22, 32.66) | 45.39 (34.89, 55.89) | Ref | Ref |
| >70 years | 29.48 (16.24, 42.72) | 45.60 (26.16, 56.04) | 1.05 (0.63, 1.76) | 1.05 (0.65, 1.71) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haidopoulos, D.; Pergialiotis, V.; Zachariou, E.; Sapantzoglou, I.; Thomakos, N.; Stamatakis, E.; Alexakis, N. Maximal Effort Cytoreduction in Epithelial Ovarian Cancer: Perioperative Complications and Survival Outcomes from a Retrospective Cohort. J. Clin. Med. 2023, 12, 622. https://doi.org/10.3390/jcm12020622
Haidopoulos D, Pergialiotis V, Zachariou E, Sapantzoglou I, Thomakos N, Stamatakis E, Alexakis N. Maximal Effort Cytoreduction in Epithelial Ovarian Cancer: Perioperative Complications and Survival Outcomes from a Retrospective Cohort. Journal of Clinical Medicine. 2023; 12(2):622. https://doi.org/10.3390/jcm12020622
Chicago/Turabian StyleHaidopoulos, Dimitrios, Vasilios Pergialiotis, Eleftherios Zachariou, Ioakim Sapantzoglou, Nikolaos Thomakos, Emmanouil Stamatakis, and Nikolaos Alexakis. 2023. "Maximal Effort Cytoreduction in Epithelial Ovarian Cancer: Perioperative Complications and Survival Outcomes from a Retrospective Cohort" Journal of Clinical Medicine 12, no. 2: 622. https://doi.org/10.3390/jcm12020622
APA StyleHaidopoulos, D., Pergialiotis, V., Zachariou, E., Sapantzoglou, I., Thomakos, N., Stamatakis, E., & Alexakis, N. (2023). Maximal Effort Cytoreduction in Epithelial Ovarian Cancer: Perioperative Complications and Survival Outcomes from a Retrospective Cohort. Journal of Clinical Medicine, 12(2), 622. https://doi.org/10.3390/jcm12020622






