Normonatremic Transient Renal Salt Wasting (TRSW) Is Not Rare in a Department of Internal Medicine
Abstract
1. Introduction
2. Materials and Methods
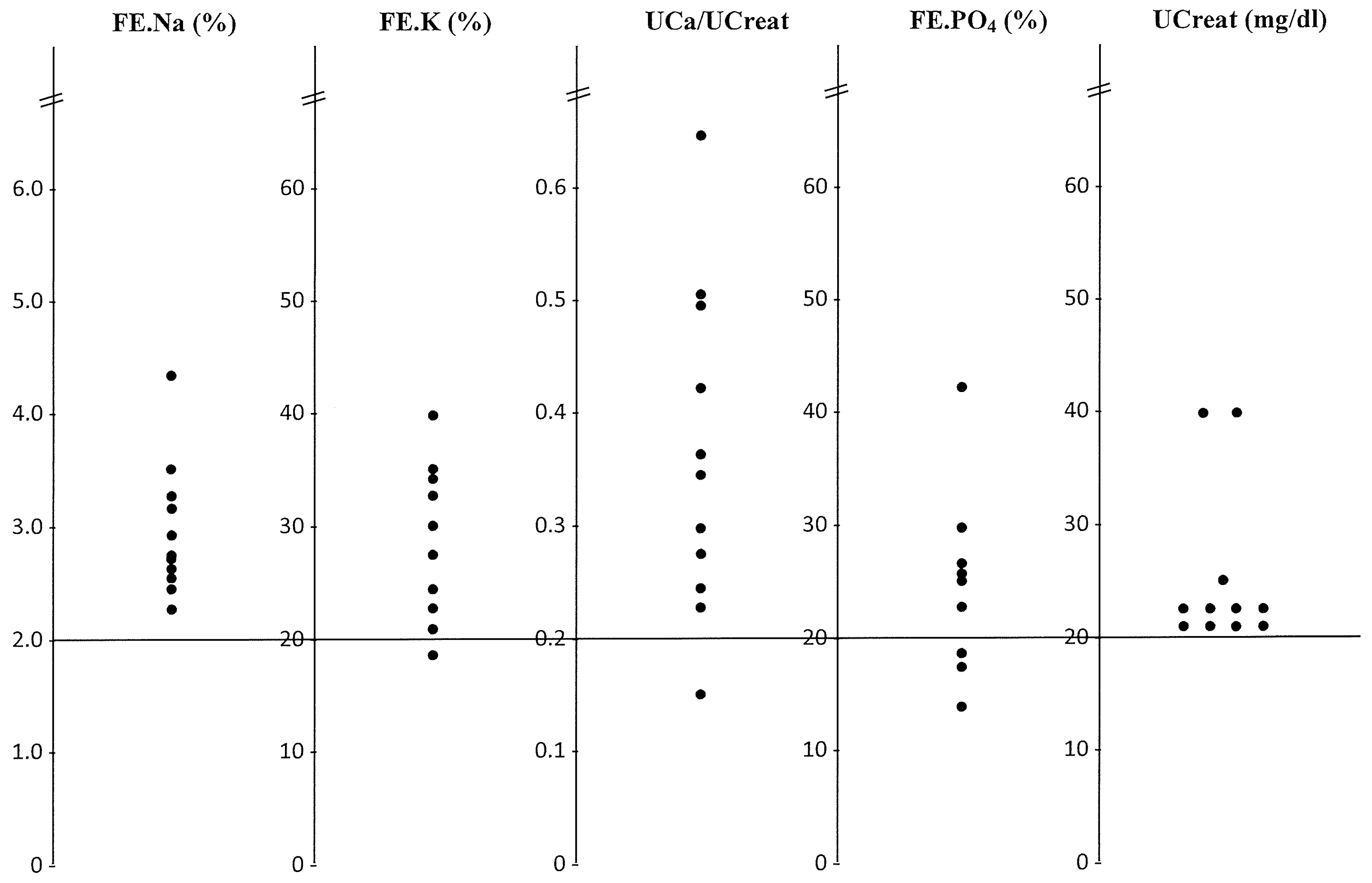
3. Result
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Musch, W.; Decaux, G. Utility and limitations of biochemical parameters in the evolution of hyponatremia in the elderly. Int. Urol. Nephrol. 2001, 32, 475–793. [Google Scholar] [CrossRef] [PubMed]
- Musch, W.; Decaux, G. Hyponatremia secondary to transient renal salt wasting (TRSW): A not so uncommon observation in the elderly. Clin. Nephrol. 2019, 91, 344–352. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.P. A salt wasting syndrome associated with cerebral disease. Trans. Assoc. Am. Physicians 1950, 63, 57–64. [Google Scholar] [PubMed]
- Golber, M.; Handler, J.S. Hyponatremia and renal wasting of sodium in patients with malfunction of the central nervous system. N. Engl. J. Med. 1960, 263, 1037–1043. [Google Scholar]
- Mangos, J.; Lobeck, C.C. Studies of sustained hyponatremia due to central nervous system infection. Pediatrics 1964, 34, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Maesaka, J.; Imbriano, L.; Mattan, J.; Gallagher, D.; Bade, N.; Sharif, S. Differentiating SIADH from Cerebral/Renal salt wasting: Failure of the volume approach and need for a new approach to hyponatremia. J. Clin. Med. 2014, 3, 1373–1385. [Google Scholar] [CrossRef] [PubMed]
- Costello-Boerrigter, L.; Boerrigter, G.; Burnett, J.C., Jr. Revisiting salt and water retention: New diuretics, aquaretics and natriuretics. Med. Clin. N. Am. 2003, 87, 475–491. [Google Scholar] [CrossRef] [PubMed]
- Goetz, K.L. Physiology and pathophysiology of atrial peptides. Am. J. Physiol. Endocrinol. Metab. 1988, 254, E1–E5. [Google Scholar] [CrossRef] [PubMed]
- Richards, A.; Tonolo, G.; Montorsi, P.; Finlayson, J.; Fraser, R.; Inglis, G.; Towrie, A.; Morton, J.J. Low dose infusions of 26- and 28-amino acid human atrial natriuretic peptides in normal man. J. Clin. Endocrinol. Metab. 1988, 66, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Maesaka, J.; Imbriano, L.; Ali, N.; Ilamathi, E. Is it cerebral or renal salt wasting? Kidney. Int. 2009, 76, 934–938. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Lewicki, J.; Johnson, L.; Cogan, M.C. Renal mechanism of action of rat atrial natriuretic factor. J. Clin. Investig. 1985, 75, 769–773. [Google Scholar] [CrossRef] [PubMed]
| Sex/Age | SNa | SK | Urea (mg/dL) | Creat (mg/dL) | Uric (mg/dL) | Ht (%) | Prot (g/dL) | Ca (mg/dL) | PO4 (mg/dL) | UOsm | FE.Na (%) | FE.K (%) | UCa/UCr | UCreat (mg/dL) | FePO4 (%) | FE.Uric (%) | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ♂ 69 | 139 | 4.3 | 39 | 0.9 | 7.3 | 42 | 7.0 | 9.4 | 3.9 | 249 | 2.5 | 32 | 0.165 | 20 | 25.5 | -- | Angina |
| ♀ 87 | 142 | 3.7 | 41 | 1 | 5.9 | 48.6 | 8.4 | 10 | 3.3 | 290 | 2.2 | 23 | 0.3 | 20 | 30 | 11 | Angina/HBP |
| ♂ 87 | 141 | 3.6 | 56 | 0.8 | 3.9 | 33.4 | 7.0 | 10.2 | 3.2 | 538 | 4.4 | 21.1 | 0.5 | 21 | 43 | 22.5 | Rectal bleeding HBP |
| ♀ 67 | 140 | 4.8 | 72 | 0.8 | 6.2 | 47.3 | 8.6 | 10.3 | 3.3 | 455 | 2.8 | 17.5 | 0.35 | 20 | 26.6 | 13 | Syncope/HBP |
| ♀ 84 | 136 | 4.2 | 23 | 0.6 | -- | 40.1 | -- | 9.2 | 3.6 | 422 | 2.7 | 25 | 0.64 | 21 | 27.5 | -- | Stroke |
| ♀ 75 | 133 | 4.8 | 51 | 0.9 | 5.4 | 33.1 | 6.6 | 8.7 | -- | 406 | 3.1 | 23 | 0.23 | 19 | -- | 9.2 | Epileptic crisis |
| ♀ 77 | 140 | 4.1 | 35 | 0.9 | 4.6 | 37.6 | 7.9 | 10.1 | 2.9 | 320 | 2.4 | 35 | 0.30 | 20 | 18.6 | 14.7 | Stroke |
| ♀ 80 | 138 | 3.6 | 34 | 0.9 | 2.5 | 39.4 | 7.1 | 9.6 | 3.5 | 476 | 3.2 | 27 | 0.36 | 20 | 14 | 23.4 | Angina |
| ♀ 86 | 137 | 4.1 | 37 | 0.9 | 4.3 | 40.2 | 8.1 | 9.8 | 4.5 | 403 | 2.4 | 30 | 0.5 | 40 | 24 | 14.6 | Transient ischemic attack |
| ♂ 76 | 138 | 3.7 | 38 | 0.9 | 7.6 | 38.7 | 7.9 | 9.8 | 2.8 | 469 | 3.5 | 35 | 0.28 | 40 | 18 | 11 | Stroke atrial fibrillation |
| ♂ 68 | 140 | 4 | 34 | 0.7 | 4 | -- | 6.1 | 8.7 | 2.8 | 394 | 2.6 | 39 | 0.43 | 26 | -- | 15.5 | Pneumothorax COPD |
| Angina | J0 | J8hr | J24hr | J48hr | J96hr |
|---|---|---|---|---|---|
| + | - | - | - | No clinical pain | |
| PNa (mEq/L) | 138 | 137 | 139 | 139 | |
| PK (mEq/L) | 3.6 | 4.6 | 3.9 | 3.7 | |
| PUrea (mg/dL) | 34 | 30 | 35 | 34 | |
| PCreat (mg/dL) | 0.9 | 0.8 | 0.8 | 0.8 | |
| PUric acid (mg/dL) | 2.5 | 3.2 | 2.7 | 3.2 | |
| Haematocrit (%) | 39.4 | 39 | 35.3 | 35 | |
| PProtein (g/dL) | 7.1 | 6.9 | 6.2 | 6.5 | |
| PCalcium (mg/dL) | 9.6 | 9.6 | 8.5 | 9.1 | |
| PPhosphore (mg/dL) | 3.5 | 3.5 | 3.8 | 4.2 | |
| FE.Na (%) | 3.2 | 0.59 | 0.57 | 0.31 | 3.6 |
| FE.K (%) | 27.3 | 8.8 | 9.9 | 12 | 30 |
| FE.Urea (%) | 66 | 57 | 41 | 46 | 67 |
| FE.Uric acid (%) | 23.4 | 18 | 9.4 | 14 | 21 |
| UCa/UCreat | 0.36 | 0.25 | 0.13 | 0.14 | 0.2 |
| Fe.PO4 (%) | 14.4 | 14.8 | 11.4 | 10.7 | 21 |
| UCr/PCr | 22 | 44 | 50 | 50 | 25 |
| UOsm | 476 | -- | -- | -- | 527 |
| ADH (0–7 pg/mL) | 0.6 | ||||
| REN (7.5–40 pg/mL) | 2 | ||||
| Aldo (0–310 pg/mL) | 52 | ||||
| ANP (25–65 ng/L) | 185 |
| Controls | TRSW (n = 11) | Under Furosemide (n = 9) | Before Furosemide (n = 5) |
|---|---|---|---|
| BW (kg) | 63.6 ± 13 | 59.4 ± 11 | NA |
| SNa (135–145 mEq/L) | 138 ± 2.4 | 139 ± 3.6 | 138 ± 3 |
| SK (3.5–5.1 mEq/L) | 4.1 ± 0.45 | 4.1 ± 0.4 | 4.2 ± 0.3 |
| Urea (17–48 mg/dL) | 42 ± 12.5 | 46 ± 12 | 36 ± 17 |
| Creatinin (0.7–1.1 mg/dL) | 0.85 ± 0.1 | 0.9 ± 0.1 | 0.9 ± 0.1 |
| FE.Osm (<3%) | 5.1 ± 1.4 | --- | --- |
| FE.Na (<1.6%) | 2.9 ± 0.6 | 3.0 ± 0.8 | 0.23 ± 0.2 * |
| FE.K (<18%) | 28 ± 6.4 | 22 ± 5.5 | 12.5 ± 5 ** |
| UCa/UCr (<0.20) | 0.37 ± 0.13 | 0.35 ± 0.12 | 0.10 ± 0.06 * |
| FE.PO4 (N < 20%) | 23.2 ± 9.6 | 18.1 ± 6.2 | 11.5 ± 4.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Musch, W.; Decaux, G. Normonatremic Transient Renal Salt Wasting (TRSW) Is Not Rare in a Department of Internal Medicine. J. Clin. Med. 2023, 12, 397. https://doi.org/10.3390/jcm12020397
Musch W, Decaux G. Normonatremic Transient Renal Salt Wasting (TRSW) Is Not Rare in a Department of Internal Medicine. Journal of Clinical Medicine. 2023; 12(2):397. https://doi.org/10.3390/jcm12020397
Chicago/Turabian StyleMusch, Wim, and Guy Decaux. 2023. "Normonatremic Transient Renal Salt Wasting (TRSW) Is Not Rare in a Department of Internal Medicine" Journal of Clinical Medicine 12, no. 2: 397. https://doi.org/10.3390/jcm12020397
APA StyleMusch, W., & Decaux, G. (2023). Normonatremic Transient Renal Salt Wasting (TRSW) Is Not Rare in a Department of Internal Medicine. Journal of Clinical Medicine, 12(2), 397. https://doi.org/10.3390/jcm12020397





