Magnetic Resonance Imaging in Clinical Trials of Diabetic Kidney Disease
Abstract
1. Introduction
2. Diagnosing DKD
3. DKD Phenotypes and Stages of Kidney Disease
4. Endpoints for Clinical Trials in DKD
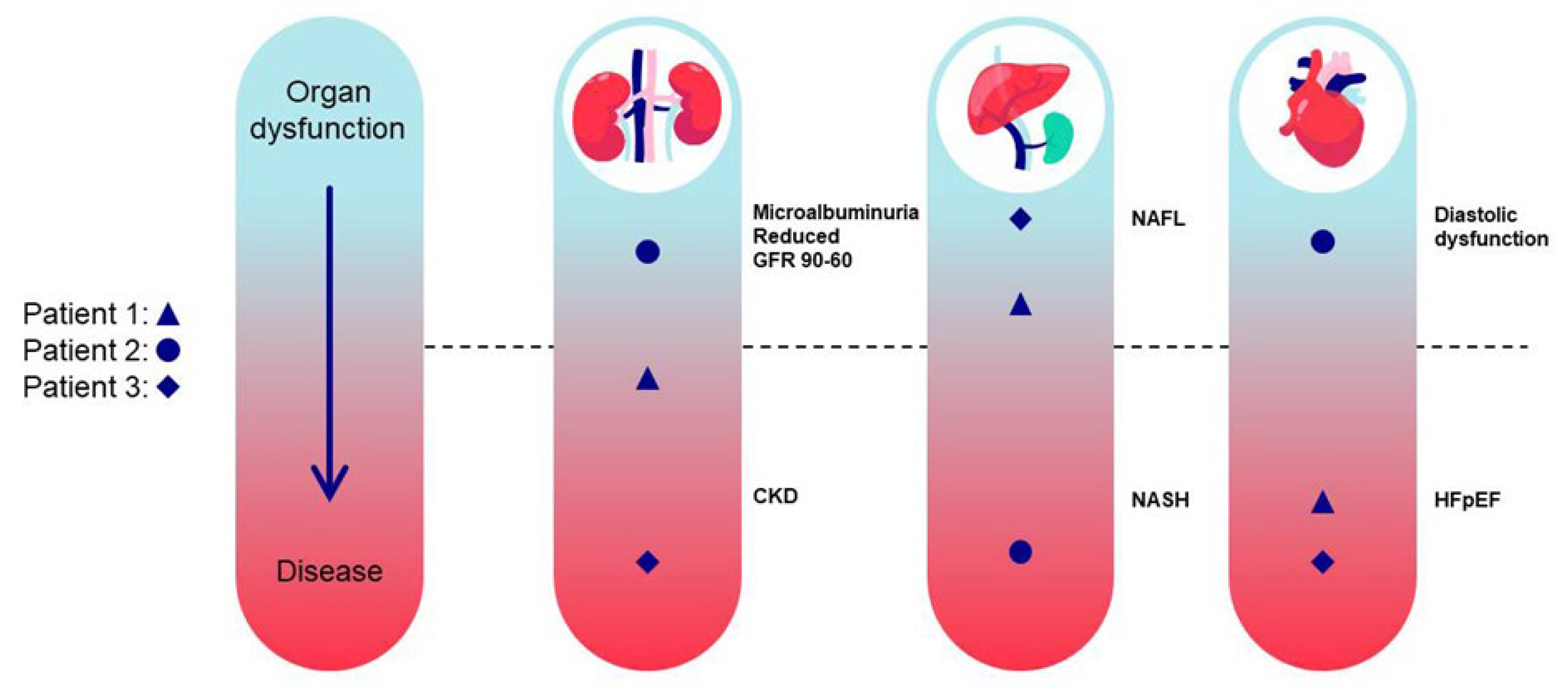
5. A Potential Role for and Opportunities with MRI
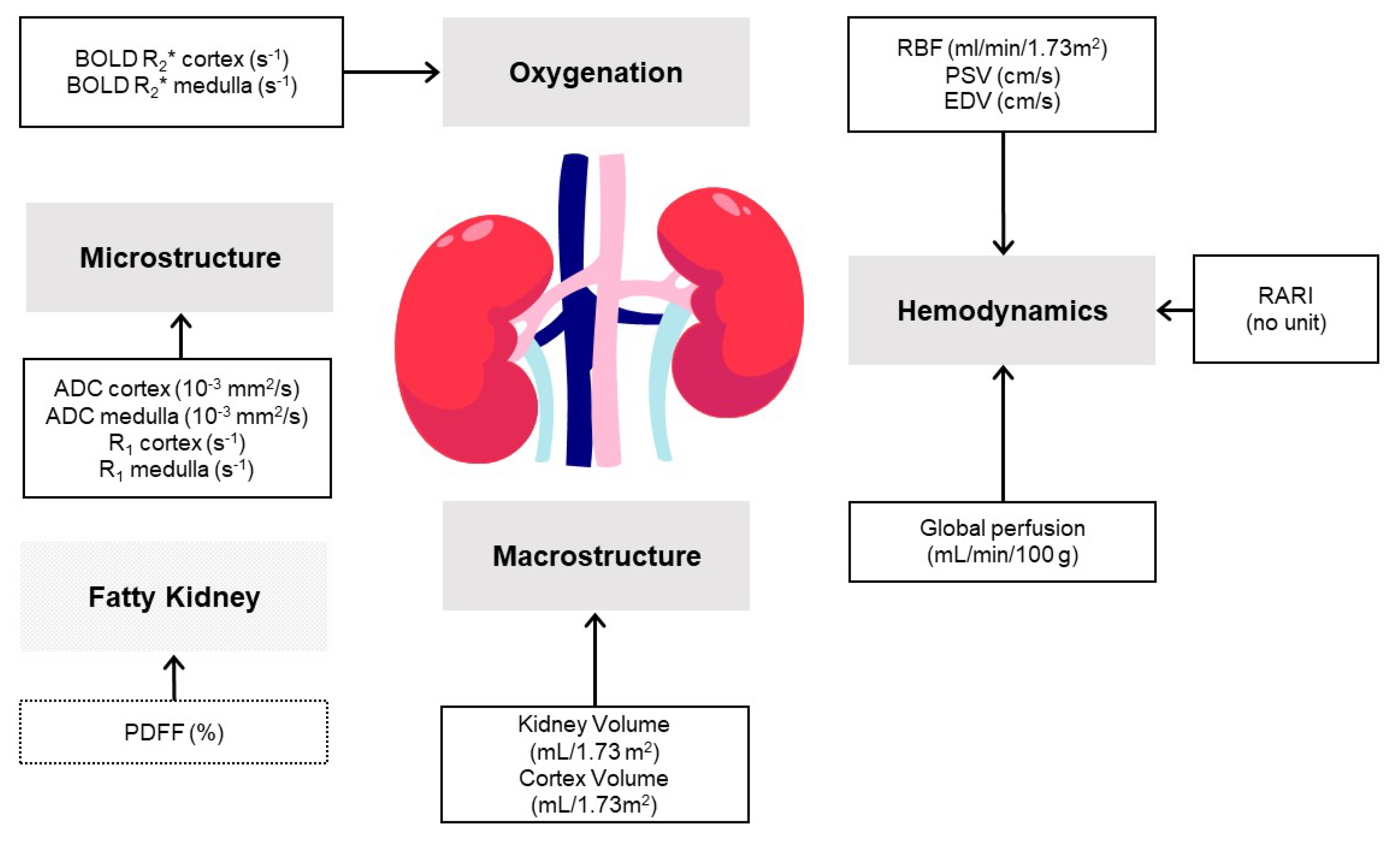
6. Employing MRI Endpoints in Clinical Trials
7. MRI Methods
7.1. Kidney Macrostructure
7.2. Kidney Haemodynamics
7.2.1. Phase-Contrast-MRI
7.2.2. Arterial Spin Labelling
7.2.3. Dynamic Contrast Enhanced-MRI
7.3. Oxygenation (BOLD)
7.4. Kidney Microstructure
7.4.1. The T1 Relaxation Time
7.4.2. DWI
7.5. Magnetic Resonance Elastography
7.6. Fatty Kidney
7.7. Multiparametric Imaging
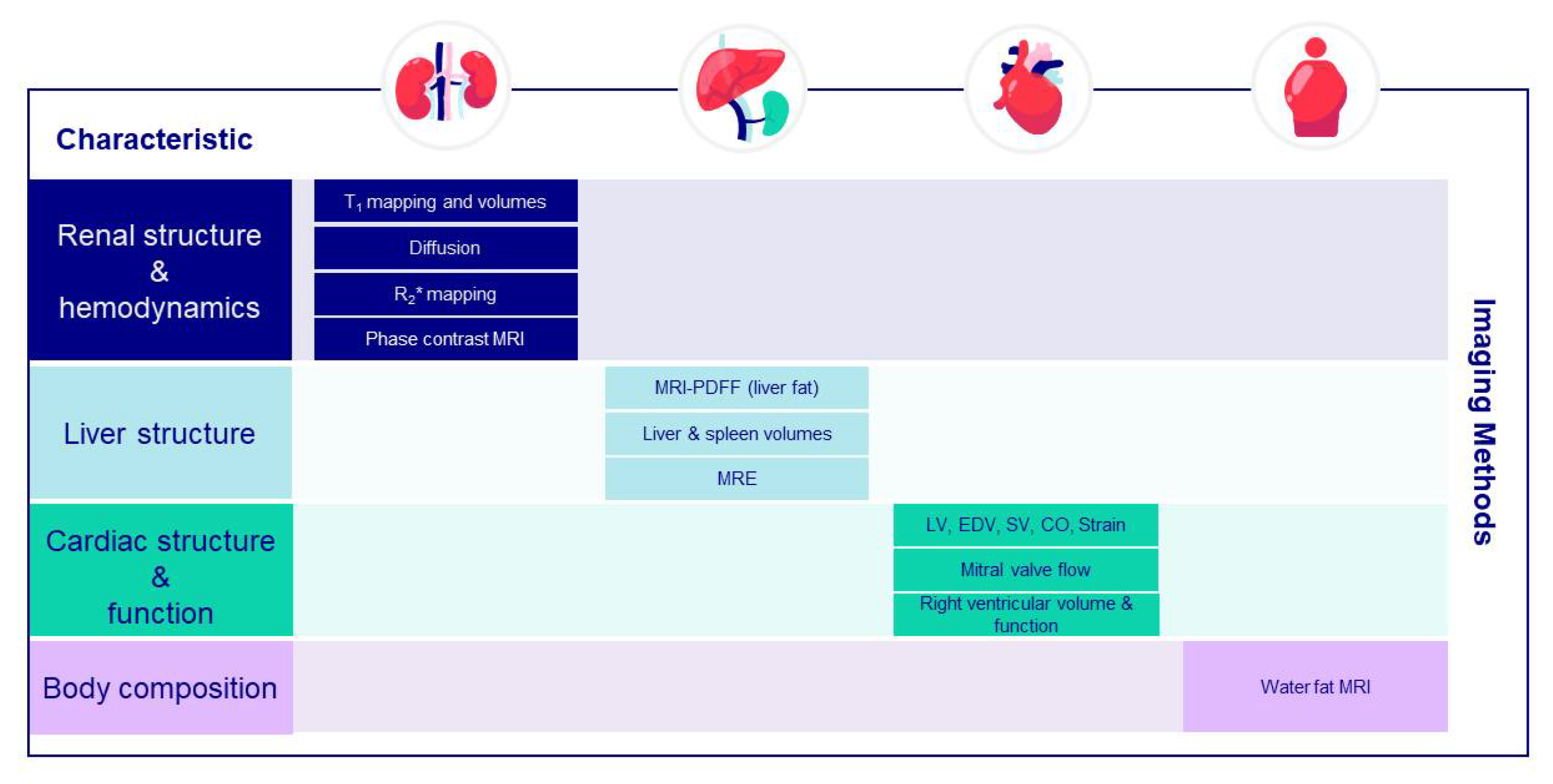
7.8. Multi-Organ Imaging
8. Challenges of Multi-Site Studies
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alicic, R.Z.; Rooney, M.T.; Tuttle, K.R. Diabetic Kidney Disease: Challenges, Progress, and Possibilities. Clin. J. Am. Soc. Nephrol. 2017, 12, 2032–2045. [Google Scholar] [CrossRef] [PubMed]
- Fu, H.; Liu, S.; Bastacky, S.I.; Wang, X.; Tian, X.J.; Zhou, D. Diabetic kidney diseases revisited: A new perspective for a new era. Mol. Metab. 2019, 30, 250–263. [Google Scholar] [CrossRef] [PubMed]
- Luis-Lima, S.; Porrini, E. An Overview of Errors and Flaws of Estimated GFR versus True GFR in Patients with Diabetes Mellitus. Nephron 2017, 136, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Stevens, L.A.; Levey, A.S. Measured GFR as a confirmatory test for estimated GFR. J. Am. Soc. Nephrol. 2009, 20, 2305–2313. [Google Scholar] [CrossRef] [PubMed]
- Waikar, S.S.; Rebholz, C.M.; Zheng, Z.; Hurwitz, S.; Hsu, C.Y.; Feldman, H.I.; Xie, D.; Liu, K.D.; Mifflin, T.E.; Eckfeldt, J.H.; et al. Biological Variability of Estimated GFR and Albuminuria in CKD. Am. J. Kidney Dis. 2018, 72, 538–546. [Google Scholar] [CrossRef]
- Endre, Z.H. Assessing Renal Recovery after Acute Kidney Injury: Can Biomarkers Help? Nephron 2018, 140, 86–89. [Google Scholar] [CrossRef]
- González, J.; Jatem, E.; Roig, J.; Valtierra, N.; Ostos, E.; Abó, A.; Santacana, M.; García, A.; Segarra, A. Usefulness of urinary biomarkers to estimate the interstitial fibrosis surface in diabetic nephropathy with normal kidney function. Nephrol. Dial. Transpl. 2022, 37, 2102–2110. [Google Scholar] [CrossRef]
- Stanton, R.C. Clinical challenges in diagnosis and management of diabetic kidney disease. Am. J. Kidney Dis. 2014, 63, S3–S21. [Google Scholar] [CrossRef]
- Luciano, R.L.; Moeckel, G.W. Update on the Native Kidney Biopsy: Core Curriculum 2019. Am. J. Kidney Dis. 2019, 73, 404–415. [Google Scholar] [CrossRef]
- Levin, A.; Stevens, P.E. Summary of KDIGO 2012 CKD Guideline: Behind the scenes, need for guidance, and a framework for moving forward. Kidney Int. 2014, 85, 49–61. [Google Scholar] [CrossRef]
- Jufar, A.H.; Lankadeva, Y.R.; May, C.N.; Cochrane, A.D.; Bellomo, R.; Evans, R.G. Renal functional reserve: From physiological phenomenon to clinical biomarker and beyond. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2020, 319, R690–R702. [Google Scholar] [CrossRef] [PubMed]
- D’Ambrosio, V.; Moochhala, S.; Unwin, R.J.; Ferraro, P.M. Why is diagnosis, investigation, and improved management of kidney stone disease important? Non-pharmacological and pharmacological treatments for nephrolithiasis. Expert. Rev. Clin. Pharm. 2022, 15, 407–414. [Google Scholar] [CrossRef]
- KDIGO Clinical Guideline Working Group. Chapter 1: Definition and classification of CKD. Kidney Int. Suppl. 2013, 3, 19–62. [Google Scholar] [CrossRef] [PubMed]
- Mogensen, C.E.; Christensen, C.K.; Vittinghus, E. The stages in diabetic renal disease: With emphasis on the stage of incipient diabetic nephropathy. Diabetes 1983, 32 (Suppl. S2), 64–78. [Google Scholar] [CrossRef] [PubMed]
- Koye, D.N.; Magliano, D.J.; Reid, C.M.; Jepson, C.; Feldman, H.I.; Herman, W.H.; Shaw, J.E. Risk of Progression of Nonalbuminuric CKD to End-Stage Kidney Disease in People with Diabetes: The CRIC (Chronic Renal Insufficiency Cohort) Study. Am. J. Kidney Dis. 2018, 72, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Pugliese, G.; Penno, G.; Natali, A.; Barutta, F.; Di Paolo, S.; Reboldi, G.; Gesualdo, L.; De Nicola, L. Diabetic kidney disease: New clinical and therapeutic issues. Joint position statement of the Italian Diabetes Society and the Italian Society of Nephrology on “The natural history of diabetic kidney disease and treatment of hyperglycemia in patients with type 2 diabetes and impaired renal function”. Nutr. Metab. Cardiovasc. Dis. 2019, 29, 1127–1150. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Ni, L.; Gao, L.; Wu, X. Comparison of Nonalbuminuric and Albuminuric Diabetic Kidney Disease Among Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis. Front. Endocrinol. 2022, 13, 871272. [Google Scholar] [CrossRef]
- Ekinci, E.I.; Jerums, G.; Skene, A.; Crammer, P.; Power, D.; Cheong, K.Y.; Panagiotopoulos, S.; McNeil, K.; Baker, S.T.; Fioretto, P.; et al. Renal structure in normoalbuminuric and albuminuric patients with type 2 diabetes and impaired renal function. Diabetes Care 2013, 36, 3620–3626. [Google Scholar] [CrossRef]
- Li, X.; Liang, Q.; Zhong, J.; Gan, L.; Zuo, L. The Effect of Metabolic Syndrome and Its Individual Components on Renal Function: A Meta-Analysis. J. Clin. Med. 2023, 12, 1614. [Google Scholar] [CrossRef]
- Godoy-Matos, A.F.; Silva Júnior, W.S.; Valerio, C.M. NAFLD as a continuum: From obesity to metabolic syndrome and diabetes. Diabetol. Metab. Syndr. 2020, 12, 60. [Google Scholar] [CrossRef]
- Gutiérrez-Cuevas, J.; Sandoval-Rodriguez, A.; Meza-Rios, A.; Monroy-Ramírez, H.C.; Galicia-Moreno, M.; García-Bañuelos, J.; Santos, A.; Armendariz-Borunda, J. Molecular Mechanisms of Obesity-Linked Cardiac Dysfunction: An Up-Date on Current Knowledge. Cells 2021, 10, 629. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Liu, S.; Zhang, C. The Related Metabolic Diseases and Treatments of Obesity. Healthcare 2022, 10, 1616. [Google Scholar] [CrossRef] [PubMed]
- Zoccali, C.; Vanholder, R.; Massy, Z.A.; Ortiz, A.; Sarafidis, P.; Dekker, F.W.; Fliser, D.; Fouque, D.; Heine, G.H.; Jager, K.J.; et al. The systemic nature of CKD. Nat. Rev. Nephrol. 2017, 13, 344–358. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; He, J.C. AKI-to-CKD transition is a potential mechanism for non-albuminuric diabetic kidney disease. Fac. Rev. 2022, 11, 21. [Google Scholar] [CrossRef] [PubMed]
- KDIGO CKD Work Group. KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. Suppl. 2013, 3, 5–14. [Google Scholar]
- Sato, Y.; Takahashi, M.; Yanagita, M. Pathophysiology of AKI to CKD progression. Semin. Nephrol. 2020, 40, 206–215. [Google Scholar] [CrossRef]
- Tanaka, S.; Tanaka, T.; Nangaku, M. Hypoxia as a key player in the AKI-to-CKD transition. Am. J. Physiol. Ren. Physiol. 2014, 307, F1187–F1195. [Google Scholar] [CrossRef]
- Kovesdy, C.P. Epidemiology of chronic kidney disease: An update 2022. Kidney Int. Suppl. 2022, 12, 7–11. [Google Scholar] [CrossRef]
- Kovesdy, C.P. Clinical trials in end-stage renal disease-priorities and challenges. Nephrol. Dial. Transpl. 2019, 34, 1084–1089. [Google Scholar] [CrossRef]
- Biomarkers Definitions Working Group. Biomarkers and surrogate endpoints: Preferred definitions and conceptual framework. Clin. Pharm. 2001, 69, 89–95. [Google Scholar] [CrossRef]
- Levey, A.S.; Gansevoort, R.T.; Coresh, J.; Inker, L.A.; Heerspink, H.L.; Grams, M.E.; Greene, T.; Tighiouart, H.; Matsushita, K.; Ballew, S.H.; et al. Change in Albuminuria and GFR as End Points for Clinical Trials in Early Stages of CKD: A Scientific Workshop Sponsored by the National Kidney Foundation in Collaboration with the US Food and Drug Administration and European Medicines Agency. Am. J. Kidney Dis. 2020, 75, 84–104. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.J.L.; Greene, T.; Tighiouart, H.; Gansevoort, R.T.; Coresh, J.; Simon, A.L.; Chan, T.M.; Hou, F.F.; Lewis, J.B.; Locatelli, F.; et al. Change in albuminuria as a surrogate endpoint for progression of kidney disease: A meta-analysis of treatment effects in randomised clinical trials. Lancet Diabetes Endocrinol. 2019, 7, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Inker, L.A.; Heerspink, H.J.L.; Tighiouart, H.; Levey, A.S.; Coresh, J.; Gansevoort, R.T.; Simon, A.L.; Ying, J.; Beck, G.J.; Wanner, C.; et al. GFR Slope as a Surrogate End Point for Kidney Disease Progression in Clinical Trials: A Meta-Analysis of Treatment Effects of Randomized Controlled Trials. J. Am. Soc. Nephrol. 2019, 30, 1735–1745. [Google Scholar] [CrossRef] [PubMed]
- Holtkamp, F.; Gudmundsdottir, H.; Maciulaitis, R.; Benda, N.; Thomson, A.; Vetter, T. Change in Albuminuria and Estimated GFR as End Points for Clinical Trials in Early Stages of CKD: A Perspective from European Regulators. Am. J. Kidney Dis. 2020, 75, 6–8. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.; Smith, K.; Lawrence, J. Change in Estimated GFR and Albuminuria as End Points in Clinical Trials: A Viewpoint from the FDA. Am. J. Kidney Dis. 2020, 75, 4–5. [Google Scholar] [CrossRef]
- Palmer, B.F. Change in albuminuria as a surrogate endpoint for cardiovascular and renal outcomes in patients with diabetes. Diabetes Obes. Metab. 2023, 25, 1434–1443. [Google Scholar] [CrossRef]
- Thompson, A.; Carroll, K.; Lesley, A.I.; Floege, J.; Perkovic, V.; Boyer-Suavet, S.; Rupert, W.M.; Judith, I.S.; Barratt, J.; Cattran, D.C.; et al. Proteinuria Reduction as a Surrogate End Point in Trials of IgA Nephropathy. Clin. J. Am. Soc. Nephrol. 2019, 14, 469–481. [Google Scholar] [CrossRef]
- Bolignano, D.; Zoccali, C. Non-proteinuric rather than proteinuric renal diseases are the leading cause of end-stage kidney disease. Nephrol. Dial. Transpl. 2017, 32, ii194–ii199. [Google Scholar] [CrossRef]
- Rossing, P.; Caramori, M.L.; Chan, J.C.; Heerspink, H.J.; Hurst, C.; Khunti, K.; Liew, A.; Michos, E.D.; Navaneethan, S.D.; Olowu, W.A.; et al. KDIGO 2022 Clinical Practice Guideline for Diabetes Management in Chronic Kidney Disease. Kidney Int. 2022, 102, S1–S127. [Google Scholar] [CrossRef]
- FDA-NIH Biomarker Working Group. BEST (Biomarkers, EndpointS, and Other Tools) Resource. Available online: https://www.ncbi.nlm.nih.gov/books/NBK326791/ (accessed on 13 May 2023).
- Fliser, D.; Wanner, C. Precision medicine in diabetic nephropathy and chronic kidney disease. Nephrol. Dial. Transpl. 2021, 36, 10–13. [Google Scholar] [CrossRef]
- Andersen, U.B.; Haddock, B.; Asmar, A. Multiparametric magnetic resonance imaging: A robust tool to test pathogenesis and pathophysiology behind nephropathy in humans. Clin. Physiol. Funct. Imaging 2023, 43, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Selby, N.M.; Blankestijn, P.J.; Boor, P.; Combe, C.; Eckardt, K.U.; Eikefjord, E.; Garcia-Fernandez, N.; Golay, X.; Gordon, I.; Grenier, N.; et al. Magnetic resonance imaging biomarkers for chronic kidney disease: A position paper from the European Cooperation in Science and Technology Action PARENCHIMA. Nephrol. Dial. Transpl. 2018, 33, ii4–ii14. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.L.; Morrell, G.; Rusinek, H.; Sigmund, E.E.; Chandarana, H.; Lerman, L.O.; Prasad, P.V.; Niles, D.; Artz, N.; Fain, S.; et al. New magnetic resonance imaging methods in nephrology. Kidney Int. 2014, 85, 768–778. [Google Scholar] [CrossRef] [PubMed]
- Villa, G.; Ringgaard, S.; Hermann, I.; Noble, R.; Brambilla, P.; Khatir, D.S.; Zöllner, F.G.; Francis, S.T.; Selby, N.M.; Remuzzi, A.; et al. Phase-contrast magnetic resonance imaging to assess renal perfusion: A systematic review and statement paper. Magma 2020, 33, 3–21. [Google Scholar] [CrossRef]
- Cox, E.F.; Buchanan, C.E.; Bradley, C.R.; Prestwich, B.; Mahmoud, H.; Taal, M.; Selby, N.M.; Francis, S.T. Multiparametric Renal Magnetic Resonance Imaging: Validation, Interventions, and Alterations in Chronic Kidney Disease. Front. Physiol. 2017, 8, 696. [Google Scholar] [CrossRef]
- Hirakawa, Y.; Nangaku, M. Visualizing fibrosis-hope for ideal markers beyond imaging. Kidney Int. 2020, 97, 437–438. [Google Scholar] [CrossRef] [PubMed]
- Makvandi, K.; Hockings, P.D.; Jensen, G.; Unnerstall, T.; Leonhardt, H.; Jarl, L.V.; Englund, C.; Francis, S.; Sundgren, A.K.; Hulthe, J.; et al. Multiparametric magnetic resonance imaging allows non-invasive functional and structural evaluation of diabetic kidney disease. Clin. Kidney J. 2022, 15, 1387–1402. [Google Scholar] [CrossRef]
- Caroli, A.; Pruijm, M.; Burnier, M.; Selby, N.M. Functional magnetic resonance imaging of the kidneys: Where do we stand? The perspective of the European COST Action PARENCHIMA. Nephrol. Dial. Transplant. 2018, 33, ii1–ii3. [Google Scholar] [CrossRef]
- Bane, O.; Mendichovszky, I.A.; Milani, B.; Dekkers, I.A.; Deux, J.F.; Eckerbom, P.; Grenier, N.; Hall, M.E.; Inoue, T.; Laustsen, C.; et al. Consensus-based technical recommendations for clinical translation of renal BOLD MRI. Magma 2020, 33, 199–215. [Google Scholar] [CrossRef]
- Caroli, A.; Schneider, M.; Friedli, I.; Ljimani, A.; De Seigneux, S.; Boor, P.; Gullapudi, L.; Kazmi, I.; Mendichovszky, I.A.; Notohamiprodjo, M.; et al. Diffusion-weighted magnetic resonance imaging to assess diffuse renal pathology: A systematic review and statement paper. Nephrol. Dial. Transplant. 2018, 33, ii29–ii40. [Google Scholar] [CrossRef]
- de Boer, A.; Villa, G.; Bane, O.; Bock, M.; Cox, E.F.; Dekkers, I.A.; Eckerbom, P.; Fernández-Seara, M.A.; Francis, S.T.; Haddock, B.; et al. Consensus-Based Technical Recommendations for Clinical Translation of Renal Phase Contrast MRI. J. Magn. Reson. Imaging 2022, 55, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Dekkers, I.A.; de Boer, A.; Sharma, K.; Cox, E.F.; Lamb, H.J.; Buckley, D.L.; Bane, O.; Morris, D.M.; Prasad, P.V.; Semple, S.I.K.; et al. Consensus-based technical recommendations for clinical translation of renal T1 and T2 mapping MRI. Magma 2020, 33, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Ljimani, A.; Caroli, A.; Laustsen, C.; Francis, S.; Mendichovszky, I.A.; Bane, O.; Nery, F.; Sharma, K.; Pohlmann, A.; Dekkers, I.A.; et al. Consensus-based technical recommendations for clinical translation of renal diffusion-weighted MRI. Magma 2020, 33, 177–195. [Google Scholar] [CrossRef] [PubMed]
- Nery, F.; Buchanan, C.E.; Harteveld, A.A.; Odudu, A.; Bane, O.; Cox, E.F.; Derlin, K.; Gach, H.M.; Golay, X.; Gutberlet, M.; et al. Consensus-based technical recommendations for clinical translation of renal ASL MRI. Magma 2020, 33, 141–161. [Google Scholar] [CrossRef]
- Odudu, A.; Nery, F.; Harteveld, A.A.; Evans, R.G.; Pendse, D.; Buchanan, C.E.; Francis, S.T.; Fernández-Seara, M.A. Arterial spin labelling MRI to measure renal perfusion: A systematic review and statement paper. Nephrol. Dial. Transpl. 2018, 33, ii15–ii21. [Google Scholar] [CrossRef]
- Pedersen, M.; Ursprung, S.; Jensen, J.D.; Jespersen, B.; Gallagher, F.; Laustsen, C. Hyperpolarised (13)C-MRI metabolic and functional imaging: An emerging renal MR diagnostic modality. Magma 2020, 33, 23–32. [Google Scholar] [CrossRef]
- Pruijm, M.; Mendichovszky, I.A.; Liss, P.; Van der Niepen, P.; Textor, S.C.; Lerman, L.O.; Krediet, C.T.P.; Caroli, A.; Burnier, M.; Prasad, P.V. Renal blood oxygenation level-dependent magnetic resonance imaging to measure renal tissue oxygenation: A statement paper and systematic review. Nephrol. Dial. Transpl. 2018, 33, ii22–ii28. [Google Scholar] [CrossRef]
- Wolf, M.; de Boer, A.; Sharma, K.; Boor, P.; Leiner, T.; Sunder-Plassmann, G.; Moser, E.; Caroli, A.; Jerome, N.P. Magnetic resonance imaging T1- and T2-mapping to assess renal structure and function: A systematic review and statement paper. Nephrol. Dial. Transpl. 2018, 33, ii41–ii50. [Google Scholar] [CrossRef]
- Zöllner, F.G.; Kociński, M.; Hansen, L.; Golla, A.K.; Trbalić, A.Š.; Lundervold, A.; Materka, A.; Rogelj, P. Kidney Segmentation in Renal Magnetic Resonance Imaging—Current Status and Prospects. IEEE Access 2021, 9, 71577–71605. [Google Scholar] [CrossRef]
- Zöllner, F.G.; Šerifović-Trbalić, A.; Kabelitz, G.; Kociński, M.; Materka, A.; Rogelj, P. Image registration in dynamic renal MRI-current status and prospects. Magma 2020, 33, 33–48. [Google Scholar] [CrossRef]
- FDA. Clinical Trial Imaging Endpoint Process. Standards Guidance for Industry. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/clinical-trial-imaging-endpoint-process-standards-guidance-industry (accessed on 13 May 2023).
- FDA. Qualification of Biomarker. Total Kidney Volume in Studies for Treatment of Autosomal Dominant Polycystic Kidney Disease. Guidance for Industry. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/qualification-biomarker-total-kidney-volume-studies-treatment-autosomal-dominant-polycystic-kidney (accessed on 13 May 2023).
- Chebib, F.T.; Torres, V.E. Assessing Risk of Rapid Progression in Autosomal Dominant Polycystic Kidney Disease and Special Considerations for Disease-Modifying Therapy. Am. J. Kidney Dis. 2021, 78, 282–292. [Google Scholar] [CrossRef]
- Moore, T.J.; Zhang, H.; Anderson, G.; Alexander, G.C. Estimated Costs of Pivotal Trials for Novel Therapeutic Agents Approved by the US Food and Drug Administration, 2015–2016. JAMA Intern. Med. 2018, 178, 1451–1457. [Google Scholar] [CrossRef]
- Prasad, P.V.; Edelman, R.R.; Epstein, F.H. Noninvasive evaluation of intrarenal oxygenation with BOLD MRI. Circulation 1996, 94, 3271–3275. [Google Scholar] [CrossRef] [PubMed]
- Manotham, K.; Ongvilawan, B.; Urusopone, P.; Chetsurakarn, S.; Tanamai, J.; Limkuansuwan, P.; Tungsanga, K.; Eiam-Ong, S. Angiotensin II receptor blocker partially ameliorated intrarenal hypoxia in chronic kidney disease patients: A pre-/post-study. Intern. Med. J. 2012, 42, e33–e37. [Google Scholar] [CrossRef] [PubMed]
- Prasad, P.V.; Thacker, J.; Li, L.P.; Haque, M.; Li, W.; Koenigs, H.; Zhou, Y.; Sprague, S.M. Multi-Parametric Evaluation of Chronic Kidney Disease by MRI: A Preliminary Cross-Sectional Study. PLoS ONE 2015, 10, e0139661. [Google Scholar] [CrossRef] [PubMed]
- Vakilzadeh, N.; Muller, M.E.; Forni, V.; Milani, B.; Hoffman, L.; Piskunowicz, M.; Maillard, M.; Zweiacker, C.; Pruijm, M.; Burnier, M. Comparative Effect of a Renin Inhibitor and a Thiazide Diuretic on Renal Tissue Oxygenation in Hypertensive Patients. Kidney Blood Press. Res. 2015, 40, 542–554. [Google Scholar] [CrossRef] [PubMed]
- Vink, E.E.; de Boer, A.; Hoogduin, H.J.; Voskuil, M.; Leiner, T.; Bots, M.L.; Joles, J.A.; Blankestijn, P.J. Renal BOLD-MRI relates to kidney function and activity of the renin-angiotensin-aldosterone system in hypertensive patients. J. Hypertens. 2015, 33, 597–603, discussion 594–603. [Google Scholar] [CrossRef]
- Vakilzadeh, N.; Zanchi, A.; Milani, B.; Ledoux, J.B.; Braconnier, P.; Burnier, M.; Pruijm, M. Acute hyperglycemia increases renal tissue oxygenation as measured by BOLD-MRI in healthy overweight volunteers. Diabetes Res. Clin. Pract. 2019, 150, 138–143. [Google Scholar] [CrossRef]
- Khatir, D.S.; Pedersen, M.; Ivarsen, P.; Christensen, K.L.; Jespersen, B.; Buus, N.H. Effects of additional vasodilatory or nonvasodilatory treatment on renal function, vascular resistance and oxygenation in chronic kidney disease: A randomized clinical trial. J. Hypertens. 2019, 37, 116–124. [Google Scholar] [CrossRef]
- Laursen, J.C.; Søndergaard-Heinrich, N.; de Melo, J.M.L.; Haddock, B.; Rasmussen, I.K.B.; Safavimanesh, F.; Hansen, C.S.; Størling, J.; Larsson, H.B.W.; Groop, P.H.; et al. Acute effects of dapagliflozin on renal oxygenation and perfusion in type 1 diabetes with albuminuria: A randomised, double-blind, placebo-controlled crossover trial. EClinicalMedicine 2021, 37, 100895. [Google Scholar] [CrossRef]
- Lee, M.M.Y.; Gillis, K.A.; Brooksbank, K.J.M.; Allwood-Spiers, S.; Hall Barrientos, P.; Wetherall, K.; Roditi, G.; AlHummiany, B.; Berry, C.; Campbell, R.T.; et al. Effect of Empagliflozin on Kidney Biochemical and Imaging Outcomes in Patients with Type 2 Diabetes, or Prediabetes, and Heart Failure with Reduced Ejection Fraction (SUGAR-DM-HF). Circulation 2022, 146, 364–367. [Google Scholar] [CrossRef]
- Lin, L.; Dekkers, I.A.; Tao, Q.; Paiman, E.H.M.; Bizino, M.B.; Jazet, I.M.; Lamb, H.J. Effect of glucose regulation on renal parenchyma and sinus fat volume in patients with type 2 diabetes. Diabetes Metab. 2023, 49, 101408. [Google Scholar] [CrossRef] [PubMed]
- Gullaksen, S.; Vernstrøm, L.; Sørensen, S.S.; Ringgaard, S.; Laustsen, C.; Funck, K.L.; Poulsen, P.L.; Laugesen, E. Separate and combined effects of semaglutide and empagliflozin on kidney oxygenation and perfusion in people with type 2 diabetes: A randomised trial. Diabetologia 2023, 66, 813–825. [Google Scholar] [CrossRef] [PubMed]
- Koratala, A.; Teodorescu, V.; Niyyar, V.D. The Nephrologist as an Ultrasonographer. Adv. Chronic Kidney Dis. 2020, 27, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Cheung, C.M.; Shurrab, A.E.; Buckley, D.L.; Hegarty, J.; Middleton, R.J.; Mamtora, H.; Kalra, P.A. MR-derived renal morphology and renal function in patients with atherosclerotic renovascular disease. Kidney Int. 2006, 69, 715–722. [Google Scholar] [CrossRef]
- Notohamiprodjo, M.; Goepfert, M.; Will, S.; Lorbeer, R.; Schick, F.; Rathmann, W.; Martirosian, P.; Peters, A.; Müller-Peltzer, K.; Helck, A.; et al. Renal and renal sinus fat volumes as quantified by magnetic resonance imaging in subjects with prediabetes, diabetes, and normal glucose tolerance. PLoS ONE 2020, 15, e0216635. [Google Scholar] [CrossRef]
- Johansson, L.L.; Taro; Hockings, P.; Jarl, L.; Maldonis, L.; Fall, T.; Ahlström, H.; Kullberg, J. The Aging Kidney: Renal Parenchymal Volumes from MRI—A Comparison Between T2D and Non-T2D in 37,450 UK Biobank Participants. J. Am. Soc. Nephrol. 2020, 31, 330. [Google Scholar]
- Buchanan, C.E.; Mahmoud, H.; Cox, E.F.; McCulloch, T.; Prestwich, B.L.; Taal, M.W.; Selby, N.M.; Francis, S.T. Quantitative assessment of renal structural and functional changes in chronic kidney disease using multi-parametric magnetic resonance imaging. Nephrol. Dial. Transpl. 2020, 35, 955–964. [Google Scholar] [CrossRef]
- Buchanan, C.; Mahmoud, H.; Cox, E.; Noble, R.; Prestwich, B.; Kasmi, I.; Taal, M.W.; Francis, S.; Selby, N.M. Multiparametric MRI assessment of renal structure and function in acute kidney injury and renal recovery. Clin. Kidney J. 2021, 14, 1969–1976. [Google Scholar] [CrossRef]
- Semelka, R.C.; Corrigan, K.; Ascher, S.M.; Brown, J.J.; Colindres, R.E. Renal corticomedullary differentiation: Observation in patients with differing serum creatinine levels. Radiology 1994, 190, 149–152. [Google Scholar] [CrossRef]
- Otsuka, T.; Kaneko, Y.; Sato, Y.; Kaseda, R.; Aoyagi, R.; Yamamoto, S.; Goto, S.; Narita, I. Kidney morphological parameters measured using noncontrast-enhanced steady-state free precession MRI with spatially selective inversion recovery pulse correlate with eGFR in patients with advanced CKD. Clin. Exp. Nephrol. 2018, 22, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Coresh, J. Chronic kidney disease. Lancet 2012, 379, 165–180. [Google Scholar] [CrossRef] [PubMed]
- Cortinovis, M.; Perico, N.; Ruggenenti, P.; Remuzzi, A.; Remuzzi, G. Glomerular hyperfiltration. Nat. Rev. Nephrol. 2022, 18, 435–451. [Google Scholar] [CrossRef] [PubMed]
- Premen, A.J. Potential mechanisms mediating postprandial renal hyperemia and hyperfiltration. FASEB J. 1988, 2, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Battilana, C.; Zhang, H.P.; Olshen, R.A.; Wexler, L.; Myers, B.D. PAH extraction and estimation of plasma flow in diseased human kidneys. Am. J. Physiol. 1991, 261, F726–F733. [Google Scholar] [CrossRef] [PubMed]
- Alhummiany, B.A.; Shelley, D.; Saysell, M.; Olaru, M.A.; Kühn, B.; Buckley, D.L.; Bailey, J.; Wroe, K.; Coupland, C.; Mansfield, M.W.; et al. Bias and Precision in Magnetic Resonance Imaging-Based Estimates of Renal Blood Flow: Assessment by Triangulation. J. Magn. Reson. Imaging 2022, 55, 1241–1250. [Google Scholar] [CrossRef] [PubMed]
- Eaton, D.C.; Pooler; John, P. Vander’s Renal Physiology, 7th ed.; Mc Graw Hill Medical: New York, NY, USA, 2009. [Google Scholar]
- Khatir, D.S.; Pedersen, M.; Jespersen, B.; Buus, N.H. Evaluation of Renal Blood Flow and Oxygenation in CKD Using Magnetic Resonance Imaging. Am. J. Kidney Dis. 2015, 66, 402–411. [Google Scholar] [CrossRef]
- Khatir, D.S.; Pedersen, M.; Jespersen, B.; Buus, N.H. Reproducibility of MRI renal artery blood flow and BOLD measurements in patients with chronic kidney disease and healthy controls. J. Magn. Reson. Imaging 2014, 40, 1091–1098. [Google Scholar] [CrossRef]
- Liss, P.; Cox, E.F.; Eckerbom, P.; Francis, S.T. Imaging of intrarenal haemodynamics and oxygen metabolism. Clin. Exp. Pharm. Physiol. 2013, 40, 158–167. [Google Scholar] [CrossRef]
- van der Bel, R.; Coolen, B.F.; Nederveen, A.J.; Potters, W.V.; Verberne, H.J.; Vogt, L.; Stroes, E.S.; Krediet, C.T. Magnetic Resonance Imaging-Derived Renal Oxygenation and Perfusion During Continuous, Steady-State Angiotensin-II Infusion in Healthy Humans. J. Am. Heart Assoc. 2016, 5, e003185. [Google Scholar] [CrossRef]
- Taso, M.; Aramendía-Vidaurreta, V.; Englund, E.K.; Francis, S.; Franklin, S.; Madhuranthakam, A.J.; Martirosian, P.; Nayak, K.S.; Qin, Q.; Shao, X.; et al. Update on state-of-the-art for arterial spin labeling (ASL) human perfusion imaging outside of the brain. Magn. Reson. Med. 2023, 89, 1754–1776. [Google Scholar] [CrossRef] [PubMed]
- Bradley, C.R.; Bragg, D.D.; Cox, E.F.; El-Sharkawy, A.M.; Buchanan, C.E.; Chowdhury, A.H.; Macdonald, I.A.; Francis, S.T.; Lobo, D.N. A randomized, controlled, double-blind crossover study on the effects of isoeffective and isovolumetric intravenous crystalloid and gelatin on blood volume, and renal and cardiac hemodynamics. Clin. Nutr. 2020, 39, 2070–2079. [Google Scholar] [CrossRef] [PubMed]
- Haddock, B.; Kristensen, K.B.; Tayyab, M.; Larsson, H.B.W.; Lindberg, U.; Vestergaard, M.; Francis, S.; Jensen, B.L.; Andersen, U.B.; Asmar, A. GLP-1 Promotes Cortical and Medullary Perfusion in the Human Kidney and Maintains Renal Oxygenation During NaCl Loading. J. Am. Heart Assoc. 2023, 12, e027712. [Google Scholar] [CrossRef] [PubMed]
- Roberts, D.A.; Detre, J.A.; Bolinger, L.; Insko, E.K.; Lenkinski, R.E.; Pentecost, M.J.; Leigh, J.S., Jr. Renal perfusion in humans: MR imaging with spin tagging of arterial water. Radiology 1995, 196, 281–286. [Google Scholar] [CrossRef]
- Grattan-Smith, J.D.; Chow, J.; Kurugol, S.; Jones, R.A. Quantitative renal magnetic resonance imaging: Magnetic resonance urography. Pediatr. Radiol. 2022, 52, 228–248. [Google Scholar] [CrossRef] [PubMed]
- Notohamiprodjo, M.; Reiser, M.F.; Sourbron, S.P. Diffusion and perfusion of the kidney. Eur. J. Radiol. 2010, 76, 337–347. [Google Scholar] [CrossRef]
- Pedersen, M.; Irrera, P.; Dastrù, W.; Zöllner, F.G.; Bennett, K.M.; Beeman, S.C.; Bretthorst, G.L.; Garbow, J.R.; Longo, D.L. Dynamic Contrast Enhancement (DCE) MRI-Derived Renal Perfusion and Filtration: Basic Concepts. Methods Mol. Biol. 2021, 2216, 205–227. [Google Scholar] [CrossRef]
- Do, C.; DeAguero, J.; Brearley, A.; Trejo, X.; Howard, T.; Escobar, G.P.; Wagner, B. Gadolinium-Based Contrast Agent Use, Their Safety, and Practice Evolution. Kidney360 2020, 1, 561–568. [Google Scholar] [CrossRef]
- Rudnick, M.R.; Wahba, I.M.; Leonberg-Yoo, A.K.; Miskulin, D.; Litt, H.I. Risks and Options with Gadolinium-Based Contrast Agents in Patients with CKD: A Review. Am. J. Kidney Dis. 2021, 77, 517–528. [Google Scholar] [CrossRef]
- Mendichovszky, I.; Pedersen, M.; Frøkiaer, J.; Dissing, T.; Grenier, N.; Anderson, P.; McHugh, K.; Yang, Q.; Gordon, I. How accurate is dynamic contrast-enhanced MRI in the assessment of renal glomerular filtration rate? A critical appraisal. J. Magn. Reson. Imaging 2008, 27, 925–931. [Google Scholar] [CrossRef]
- Eikefjord, E.; Andersen, E.; Hodneland, E.; Zöllner, F.; Lundervold, A.; Svarstad, E.; Rørvik, J. Use of 3D DCE-MRI for the estimation of renal perfusion and glomerular filtration rate: An intrasubject comparison of FLASH and KWIC with a comprehensive framework for evaluation. AJR Am. J. Roentgenol. 2015, 204, W273–W281. [Google Scholar] [CrossRef]
- Lietzmann, F.; Zöllner, F.G.; Attenberger, U.I.; Haneder, S.; Michaely, H.J.; Schad, L.R. DCE-MRI of the human kidney using BLADE: A feasibility study in healthy volunteers. J. Magn. Reson. Imaging 2012, 35, 868–874. [Google Scholar] [CrossRef]
- Zhang, Y.D.; Wu, C.J.; Zhang, J.; Wang, X.N.; Liu, X.S.; Shi, H.B. Feasibility study of high-resolution DCE-MRI for glomerular filtration rate (GFR) measurement in a routine clinical modal. Magn. Reson. Imaging 2015, 33, 978–983. [Google Scholar] [CrossRef] [PubMed]
- de Boer, A.; Leiner, T.; Vink, E.E.; Blankestijn, P.J.; van den Berg, C.A.T. Modified dixon-based renal dynamic contrast-enhanced MRI facilitates automated registration and perfusion analysis. Magn. Reson. Med. 2018, 80, 66–76. [Google Scholar] [CrossRef]
- El-Melegy, M.; Kamel, R.; Abou El-Ghar, M.; Alghamdi, N.S.; El-Baz, A. Level-Set-Based Kidney Segmentation from DCE-MRI Using Fuzzy Clustering with Population-Based and Subject-Specific Shape Statistics. Bioengineering 2022, 9, 654. [Google Scholar] [CrossRef]
- Khalifa, F.; Abou El-Ghar, M.; Abdollahi, B.; Frieboes, H.B.; El-Diasty, T.; El-Baz, A. A comprehensive non-invasive framework for automated evaluation of acute renal transplant rejection using DCE-MRI. NMR Biomed. 2013, 26, 1460–1470. [Google Scholar] [CrossRef] [PubMed]
- Klepaczko, A.; Eikefjord, E.; Lundervold, A. Healthy Kidney Segmentation in the Dce-Mr Images Using a Convolutional Neural Network and Temporal Signal Characteristics. Sensors 2021, 21, 6714. [Google Scholar] [CrossRef] [PubMed]
- Tofts, P.S.; Cutajar, M.; Mendichovszky, I.A.; Peters, A.M.; Gordon, I. Precise measurement of renal filtration and vascular parameters using a two-compartment model for dynamic contrast-enhanced MRI of the kidney gives realistic normal values. Eur. Radiol. 2012, 22, 1320–1330. [Google Scholar] [CrossRef]
- Bokacheva, L.; Rusinek, H.; Zhang, J.L.; Chen, Q.; Lee, V.S. Estimates of glomerular filtration rate from MR renography and tracer kinetic models. J. Magn. Reson. Imaging 2009, 29, 371–382. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; Chang, D.; Zhang, S.J.; Wang, Q.; Ju, S. The Accuracy of Renal Function Measurements in Obstructive Hydronephrosis Using Dynamic Contrast-Enhanced MR Renography. AJR Am. J. Roentgenol. 2019, 213, 859–866. [Google Scholar] [CrossRef]
- Taton, B.; De La Faille, R.; Asselineau, J.; Perez, P.; Merville, P.; Colin, T.; Combe, C.; Sourbron, S.; Grenier, N. A prospective comparison of dynamic contrast-enhanced MRI and (51)Cr-EDTA clearance for glomerular filtration rate measurement in 42 kidney transplant recipients. Eur. J. Radiol. 2019, 117, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Bane, O.; Wagner, M.; Zhang, J.L.; Dyvorne, H.A.; Orton, M.; Rusinek, H.; Taouli, B. Assessment of renal function using intravoxel incoherent motion diffusion-weighted imaging and dynamic contrast-enhanced MRI. J. Magn. Reson. Imaging 2016, 44, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Lee, V.S.; Rusinek, H.; Bokacheva, L.; Huang, A.J.; Oesingmann, N.; Chen, Q.; Kaur, M.; Prince, K.; Song, T.; Kramer, E.L.; et al. Renal function measurements from MR renography and a simplified multicompartmental model. Am. J. Physiol. Ren. Physiol. 2007, 292, F1548–F1559. [Google Scholar] [CrossRef] [PubMed]
- Cutajar, M.; Thomas, D.L.; Hales, P.W.; Banks, T.; Clark, C.A.; Gordon, I. Comparison of ASL and DCE MRI for the non-invasive measurement of renal blood flow: Quantification and reproducibility. Eur. Radiol. 2014, 24, 1300–1308. [Google Scholar] [CrossRef]
- Eikefjord, E.; Andersen, E.; Hodneland, E.; Hanson, E.A.; Sourbron, S.; Svarstad, E.; Lundervold, A.; Rørvik, J.T. Dynamic contrast-enhanced MRI measurement of renal function in healthy participants. Acta Radiol. 2017, 58, 748–757. [Google Scholar] [CrossRef]
- de Boer, A.; Harteveld, A.A.; Stemkens, B.; Blankestijn, P.J.; Bos, C.; Franklin, S.L.; Froeling, M.; Joles, J.A.; Verhaar, M.C.; van den Berg, N.; et al. Multiparametric Renal MRI: An Intrasubject Test-Retest Repeatability Study. J. Magn. Reson. Imaging 2021, 53, 859–873. [Google Scholar] [CrossRef]
- Prasad, P.V.; Li, L.-P.; Hack, B.K.; Leloudas, N.; Sprague, S.M. Quantitative BOLD MRI for Estimating Intra-renal Oxygen Availability Demonstrates Kidneys Are Hypoxemic in Human CKD. Kidney Int. Rep. 2023, 8, 1057–1067. [Google Scholar] [CrossRef]
- Hirakawa, Y.; Tanaka, T.; Nangaku, M. Renal Hypoxia in CKD; Pathophysiology and Detecting Methods. Front. Physiol. 2017, 8, 99. [Google Scholar] [CrossRef]
- Evans, R.G.; Smith, D.W.; Lee, C.J.; Ngo, J.P.; Gardiner, B.S. What Makes the Kidney Susceptible to Hypoxia? Anat. Rec. 2020, 303, 2544–2552. [Google Scholar] [CrossRef]
- Li, Z.C.; Cai, Y.Z.; Tang, Z.G.; Zuo, P.L.; Liu, R.B.; Liu, F. Lipo-prostaglandin E1 improves renal hypoxia evaluated by BOLD-MRI in patients with diabetic kidney disease. Clin. Imaging 2018, 50, 239–242. [Google Scholar] [CrossRef]
- Hesp, A.C.; Schaub, J.A.; Prasad, P.V.; Vallon, V.; Laverman, G.D.; Bjornstad, P.; van Raalte, D.H. The role of renal hypoxia in the pathogenesis of diabetic kidney disease: A promising target for newer renoprotective agents including SGLT2 inhibitors? Kidney Int. 2020, 98, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Zhang, Y.; Wang, T.; Huang, S.; Gong, S.; Wang, J.; Yu, P. Canagliflozin could improve the levels of renal oxygenation in newly diagnosed type 2 diabetes patients with normal renal function. Diabetes Metab. 2021, 47, 101274. [Google Scholar] [CrossRef] [PubMed]
- Evans, R.G.; Ince, C.; Joles, J.A.; Smith, D.W.; May, C.N.; O’Connor, P.M.; Gardiner, B.S. Haemodynamic influences on kidney oxygenation: Clinical implications of integrative physiology. Clin. Exp. Pharm. Physiol. 2013, 40, 106–122. [Google Scholar] [CrossRef] [PubMed]
- Niendorf, T.; Pohlmann, A.; Arakelyan, K.; Flemming, B.; Cantow, K.; Hentschel, J.; Grosenick, D.; Ladwig, M.; Reimann, H.; Klix, S.; et al. How bold is blood oxygenation level-dependent (BOLD) magnetic resonance imaging of the kidney? Opportunities, challenges and future directions. Acta Physiol. 2015, 213, 19–38. [Google Scholar] [CrossRef]
- Pruijm, M.; Milani, B.; Burnier, M. Blood Oxygenation Level-Dependent MRI to Assess Renal Oxygenation in Renal Diseases: Progresses and Challenges. Front. Physiol. 2016, 7, 667. [Google Scholar] [CrossRef]
- Inoue, T.; Kozawa, E.; Ishikawa, M.; Fukaya, D.; Amano, H.; Watanabe, Y.; Tomori, K.; Kobayashi, N.; Niitsu, M.; Okada, H. Comparison of multiparametric magnetic resonance imaging sequences with laboratory parameters for prognosticating renal function in chronic kidney disease. Sci. Rep. 2021, 11, 22129. [Google Scholar] [CrossRef]
- Michaely, H.J.; Metzger, L.; Haneder, S.; Hansmann, J.; Schoenberg, S.O.; Attenberger, U.I. Renal BOLD-MRI does not reflect renal function in chronic kidney disease. Kidney Int. 2012, 81, 684–689. [Google Scholar] [CrossRef]
- Prasad, P.V.; Li, L.P.; Thacker, J.M.; Li, W.; Hack, B.; Kohn, O.; Sprague, S.M. Cortical Perfusion and Tubular Function as Evaluated by Magnetic Resonance Imaging Correlates with Annual Loss in Renal Function in Moderate Chronic Kidney Disease. Am. J. Nephrol. 2019, 49, 114–124. [Google Scholar] [CrossRef]
- Pruijm, M.; Milani, B.; Pivin, E.; Podhajska, A.; Vogt, B.; Stuber, M.; Burnier, M. Reduced cortical oxygenation predicts a progressive decline of renal function in patients with chronic kidney disease. Kidney Int. 2018, 93, 932–940. [Google Scholar] [CrossRef]
- Sugiyama, K.; Inoue, T.; Kozawa, E.; Ishikawa, M.; Shimada, A.; Kobayashi, N.; Tanaka, J.; Okada, H. Reduced oxygenation but not fibrosis defined by functional magnetic resonance imaging predicts the long-term progression of chronic kidney disease. Nephrol. Dial. Transpl. 2020, 35, 964–970. [Google Scholar] [CrossRef]
- Messroghli, D.R.; Radjenovic, A.; Kozerke, S.; Higgins, D.M.; Sivananthan, M.U.; Ridgway, J.P. Modified Look-Locker inversion recovery (MOLLI) for high-resolution T1 mapping of the heart. Magn. Reson. Med. 2004, 52, 141–146. [Google Scholar] [CrossRef]
- Bradley, C.R.; Cox, E.F.; Scott, R.A.; James, M.W.; Kaye, P.; Aithal, G.P.; Francis, S.T.; Guha, I.N. Multi-organ assessment of compensated cirrhosis patients using quantitative magnetic resonance imaging. J. Hepatol. 2018, 69, 1015–1024. [Google Scholar] [CrossRef] [PubMed]
- Graham-Brown, M.P.; Singh, A.; Wormleighton, J.; Brunskill, N.J.; McCann, G.P.; Barratt, J.; Burton, J.O.; Xu, G. Association between native T1 mapping of the kidney and renal fibrosis in patients with IgA nephropathy. BMC Nephrol. 2019, 20, 256. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Shi, Z.; Zhang, Y.; Yan, J.; Shang, F.; Wang, Y.; Lu, H.; Gu, H.; Dou, W.; Wang, X.; et al. Native T1 Mapping in Assessing Kidney Fibrosis for Patients with Chronic Glomerulonephritis. Front. Med. 2021, 8, 772326. [Google Scholar] [CrossRef] [PubMed]
- Bane, O.; Hectors, S.J.; Gordic, S.; Kennedy, P.; Wagner, M.; Weiss, A.; Khaim, R.; Yi, Z.; Zhang, W.; Delaney, V.; et al. Multiparametric magnetic resonance imaging shows promising results to assess renal transplant dysfunction with fibrosis. Kidney Int. 2020, 97, 414–420. [Google Scholar] [CrossRef]
- Berchtold, L.; Friedli, I.; Crowe, L.A.; Martinez, C.; Moll, S.; Hadaya, K.; de Perrot, T.; Combescure, C.; Martin, P.Y.; Vallée, J.P.; et al. Validation of the corticomedullary difference in magnetic resonance imaging-derived apparent diffusion coefficient for kidney fibrosis detection: A cross-sectional study. Nephrol. Dial. Transpl. 2020, 35, 937–945. [Google Scholar] [CrossRef]
- Beck-Tölly, A.; Eder, M.; Beitzke, D.; Eskandary, F.; Agibetov, A.; Lampichler, K.; Hamböck, M.; Regele, H.; Kläger, J.; Nackenhorst, M.; et al. Magnetic Resonance Imaging for Evaluation of Interstitial Fibrosis in Kidney Allografts. Transpl. Direct 2020, 6, e577. [Google Scholar] [CrossRef]
- Hockings, P.; Makvandi, K.; Hulthe, J.; Baid-Agrawal, S. Multiparametric Magnetic Resonance Imaging biomarkers outperform lab biomarkers for predicting progression of diabetic kidney disease. Nephrol. Dial. Transplant. 2023, 38 (Suppl. S1), gfad063c_4204. [Google Scholar] [CrossRef]
- Echeverria-Chasco, R.; Martin-Moreno, P.L.; Garcia-Fernandez, N.; Vidorreta, M.; Aramendia-Vidaurreta, V.; Cano, D.; Villanueva, A.; Bastarrika, G.; Fernández-Seara, M.A. Multiparametric renal magnetic resonance imaging: A reproducibility study in renal allografts with stable function. NMR Biomed. 2022, 36, e4832. [Google Scholar] [CrossRef]
- Rankin, A.J.; Allwood-Spiers, S.; Lee, M.M.Y.; Zhu, L.; Woodward, R.; Kuehn, B.; Radjenovic, A.; Sattar, N.; Roditi, G.; Mark, P.B.; et al. Comparing the interobserver reproducibility of different regions of interest on multi-parametric renal magnetic resonance imaging in healthy volunteers, patients with heart failure and renal transplant recipients. Magma 2020, 33, 103–112. [Google Scholar] [CrossRef]
- Friedli, I.; Crowe, L.A.; Berchtold, L.; Moll, S.; Hadaya, K.; de Perrot, T.; Vesin, C.; Martin, P.Y.; de Seigneux, S.; Vallée, J.P. New Magnetic Resonance Imaging Index for Renal Fibrosis Assessment: A Comparison between Diffusion-Weighted Imaging and T1 Mapping with Histological Validation. Sci. Rep. 2016, 6, 30088. [Google Scholar] [CrossRef] [PubMed]
- Berchtold, L.; Crowe, L.A.; Friedli, I.; Legouis, D.; Moll, S.; de Perrot, T.; Martin, P.Y.; Vallée, J.P.; de Seigneux, S. Diffusion magnetic resonance imaging detects an increase in interstitial fibrosis earlier than the decline of renal function. Nephrol. Dial. Transpl. 2020, 35, 1274–1276. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, C.M.; Eirin, A.; Abumoawad, A.; Saad, A.; Jiang, K.; Hedayat, A.F.; Misra, S.; Glockner, J.; Textor, S.C.; Lerman, L.O. Renal fibrosis detected by diffusion-weighted magnetic resonance imaging remains unchanged despite treatment in subjects with renovascular disease. Sci. Rep. 2020, 10, 16300. [Google Scholar] [CrossRef] [PubMed]
- Mao, W.; Zhou, J.; Zeng, M.; Ding, Y.; Qu, L.; Chen, C.; Ding, X.; Wang, Y.; Fu, C.; Gu, F. Intravoxel incoherent motion diffusion-weighted imaging for the assessment of renal fibrosis of chronic kidney disease: A preliminary study. Magn. Reson. Imaging 2018, 47, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yu, Y.; Wen, J.; Zhang, M.; Chen, J.; Cheng, D.; Zhang, L.; Liu, Z. Combination of Functional Magnetic Resonance Imaging and Histopathologic Analysis to Evaluate Interstitial Fibrosis in Kidney Allografts. Clin. J. Am. Soc. Nephrol. 2019, 14, 1372–1380. [Google Scholar] [CrossRef]
- Zheng, Z.; Yan, T.; Jia, J.; Li, D.; Wei, L.; Shang, W.; Shi, H. Assessment of Renal Pathological Changes in Lupus Nephritis Using Diffusion Weighted Imaging: A Multiple Correspondence Analysis. Kidney Blood Press. Res. 2018, 43, 847–859. [Google Scholar] [CrossRef]
- Srivastava, A.; Cai, X.; Lee, J.; Li, W.; Larive, B.; Kendrick, C.; Gassman, J.J.; Middleton, J.P.; Carr, J.; Raphael, K.L.; et al. Kidney Functional Magnetic Resonance Imaging and Change in eGFR in Individuals with CKD. Clin. J. Am. Soc. Nephrol. 2020, 15, 776–783. [Google Scholar] [CrossRef]
- Gooding, K.M.; Lienczewski, C.; Papale, M.; Koivuviita, N.; Maziarz, M.; Dutius Andersson, A.M.; Sharma, K.; Pontrelli, P.; Garcia Hernandez, A.; Bailey, J.; et al. Prognostic imaging biomarkers for diabetic kidney disease (iBEAt): Study protocol. BMC Nephrol. 2020, 21, 242. [Google Scholar] [CrossRef]
- Mariappan, Y.K.; Glaser, K.J.; Ehman, R.L. Magnetic resonance elastography: A review. Clin. Anat. 2010, 23, 497–511. [Google Scholar] [CrossRef]
- Dittmann, F.; Tzschätzsch, H.; Hirsch, S.; Barnhill, E.; Braun, J.; Sack, I.; Guo, J. Tomoelastography of the abdomen: Tissue mechanical properties of the liver, spleen, kidney, and pancreas from single MR elastography scans at different hydration states. Magn. Reson. Med. 2017, 78, 976–983. [Google Scholar] [CrossRef]
- Ozturk, A.; Grajo, J.R.; Dhyani, M.; Anthony, B.W.; Samir, A.E. Principles of ultrasound elastography. Abdom. Radiol. 2018, 43, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Venkatesh, S.K.; Yin, M. Advances in Magnetic Resonance Elastography of Liver. Magn. Reson. Imaging Clin. N. Am. 2020, 28, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, E.A.; Mózes, F.E.; Jayaswal, A.N.A.; Zafarmand, M.H.; Vali, Y.; Lee, J.A.; Levick, C.K.; Young, L.A.J.; Palaniyappan, N.; Liu, C.H.; et al. Diagnostic accuracy of elastography and magnetic resonance imaging in patients with NAFLD: A systematic review and meta-analysis. J. Hepatol. 2021, 75, 770–785. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Yuen, D.A.; Leung, G.; Jothy, S.; Zaltzman, J.; Ramesh Prasad, G.V.; Prabhudesai, V.; Mnatzakanian, G.; Kirpalani, A. Role of Magnetic Resonance Elastography as a Noninvasive Measurement Tool of Fibrosis in a Renal Allograft: A Case Report. Transpl. Proc. 2017, 49, 1555–1559. [Google Scholar] [CrossRef]
- Kirpalani, A.; Hashim, E.; Leung, G.; Kim, J.K.; Krizova, A.; Jothy, S.; Deeb, M.; Jiang, N.N.; Glick, L.; Mnatzakanian, G.; et al. Magnetic Resonance Elastography to Assess Fibrosis in Kidney Allografts. Clin. J. Am. Soc. Nephrol. 2017, 12, 1671–1679. [Google Scholar] [CrossRef]
- Güven, A.T.; Idilman, I.S.; Cebrayilov, C.; Önal, C.; Kibar, M.; Sağlam, A.; Yıldırım, T.; Yılmaz, R.; Altun, B.; Erdem, Y.; et al. Evaluation of renal fibrosis in various causes of glomerulonephritis by MR elastography: A clinicopathologic comparative analysis. Abdom. Radiol. 2022, 47, 288–296. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, J.; Chen, H.; Su, Z. Evaluation of renal fibrosis in patients with chronic kidney disease by shear wave elastography: A comparative analysis with pathological findings. Abdom. Radiol. 2022, 47, 738–745. [Google Scholar] [CrossRef]
- Gandhi, D.; Kalra, P.; Raterman, B.; Mo, X.; Dong, H.; Kolipaka, A. Magnetic Resonance Elastography of kidneys: SE-EPI MRE reproducibility and its comparison to GRE MRE. NMR Biomed. 2019, 32, e4141. [Google Scholar] [CrossRef]
- Rouvière, O.; Souchon, R.; Pagnoux, G.; Ménager, J.M.; Chapelon, J.Y. Magnetic resonance elastography of the kidneys: Feasibility and reproducibility in young healthy adults. J. Magn. Reson. Imaging 2011, 34, 880–886. [Google Scholar] [CrossRef]
- Marticorena Garcia, S.R.; Grossmann, M.; Lang, S.T.; Tzschätzsch, H.; Dittmann, F.; Hamm, B.; Braun, J.; Guo, J.; Sack, I. Tomoelastography of the native kidney: Regional variation and physiological effects on in vivo renal stiffness. Magn. Reson. Med. 2018, 79, 2126–2134. [Google Scholar] [CrossRef]
- Lang, S.T.; Guo, J.; Bruns, A.; Dürr, M.; Braun, J.; Hamm, B.; Sack, I.; Marticorena Garcia, S.R. Multiparametric Quantitative MRI for the Detection of IgA Nephropathy Using Tomoelastography, DWI, and BOLD Imaging. Investig. Radiol. 2019, 54, 669–674. [Google Scholar] [CrossRef] [PubMed]
- de Vries, A.P.; Ruggenenti, P.; Ruan, X.Z.; Praga, M.; Cruzado, J.M.; Bajema, I.M.; D’Agati, V.D.; Lamb, H.J.; Pongrac Barlovic, D.; Hojs, R.; et al. Fatty kidney: Emerging role of ectopic lipid in obesity-related renal disease. Lancet Diabetes Endocrinol. 2014, 2, 417–426. [Google Scholar] [CrossRef] [PubMed]
- D’Marco, L.; Puchades, M.J.; Panizo, N.; Romero-Parra, M.; Gandía, L.; Giménez-Civera, E.; Pérez-Bernat, E.; Gonzalez-Rico, M.; Gorriz, J.L. Cardiorenal Fat: A Cardiovascular Risk Factor with Implications in Chronic Kidney Disease. Front. Med. 2021, 8, 640814. [Google Scholar] [CrossRef] [PubMed]
- Foster, M.C.; Hwang, S.J.; Porter, S.A.; Massaro, J.M.; Hoffmann, U.; Fox, C.S. Fatty kidney, hypertension, and chronic kidney disease: The Framingham Heart Study. Hypertension 2011, 58, 784–790. [Google Scholar] [CrossRef]
- Spit, K.A.; Muskiet, M.H.A.; Tonneijck, L.; Smits, M.M.; Kramer, M.H.H.; Joles, J.A.; de Boer, A.; van Raalte, D.H. Renal sinus fat and renal hemodynamics: A cross-sectional analysis. Magma 2020, 33, 73–80. [Google Scholar] [CrossRef]
- Reeder, S.B.; Hu, H.H.; Sirlin, C.B. Proton density fat-fraction: A standardized MR-based biomarker of tissue fat concentration. J. Magn. Reson. Imaging 2012, 36, 1011–1014. [Google Scholar] [CrossRef]
- Dekkers, I.A.; Bizino, M.B.; Paiman, E.H.M.; Smit, J.W.; Jazet, I.M.; de Vries, A.P.J.; Lamb, H.J. The Effect of Glycemic Control on Renal Triglyceride Content Assessed by Proton Spectroscopy in Patients with Type 2 Diabetes Mellitus: A Single-Center Parallel-Group Trial. J. Ren. Nutr. 2021, 31, 611–619. [Google Scholar] [CrossRef]
- Grigoraș, A.; Balan, R.A.; Căruntu, I.D.; Giușcă, S.E.; Lozneanu, L.; Avadanei, R.E.; Rusu, A.; Riscanu, L.A.; Amalinei, C. Perirenal Adipose Tissue-Current Knowledge and Future Opportunities. J. Clin. Med. 2021, 10, 1291. [Google Scholar] [CrossRef]
- Hall, M.E.; do Carmo, J.M.; da Silva, A.A.; Juncos, L.A.; Wang, Z.; Hall, J.E. Obesity, hypertension, and chronic kidney disease. Int. J. Nephrol. Renov. Dis. 2014, 7, 75–88. [Google Scholar] [CrossRef]
- Lin, L.; Dekkers, I.A.; Huang, L.; Tao, Q.; Paiman, E.H.M.; Bizino, M.B.; Jazet, I.M.; Lamb, H.J. Renal sinus fat volume in type 2 diabetes mellitus is associated with glycated hemoglobin and metabolic risk factors. J. Diabetes Complicat. 2021, 35, 107973. [Google Scholar] [CrossRef]
- Lin, L.; Dekkers, I.A.; Lamb, H.J. Fat accumulation around and within the kidney. In Visceral and Ectopic Fat. Risk Factors for Type 2 Diabetes, Atherosclerosis, and Cardiovascular Disease, 1st ed.; Lamb, H.J., Ed.; Elsevier: Amsterdam, The Netherlands, 2023; pp. 131–147. [Google Scholar]
- Favre, G.; Grangeon-Chapon, C.; Raffaelli, C.; François-Chalmin, F.; Iannelli, A.; Esnault, V. Perirenal fat thickness measured with computed tomography is a reliable estimate of perirenal fat mass. PLoS ONE 2017, 12, e0175561. [Google Scholar] [CrossRef]
- Manno, C.; Campobasso, N.; Nardecchia, A.; Triggiani, V.; Zupo, R.; Gesualdo, L.; Silvestris, F.; De Pergola, G. Relationship of para- and perirenal fat and epicardial fat with metabolic parameters in overweight and obese subjects. Eat. Weight. Disord. 2019, 24, 67–72. [Google Scholar] [CrossRef]
- Zelicha, H.; Schwarzfuchs, D.; Shelef, I.; Gepner, Y.; Tsaban, G.; Tene, L.; Yaskolka Meir, A.; Bilitzky, A.; Komy, O.; Cohen, N.; et al. Changes of renal sinus fat and renal parenchymal fat during an 18-month randomized weight loss trial. Clin. Nutr. 2018, 37, 1145–1153. [Google Scholar] [CrossRef]
- Gjela, M.; Askeland, A.; Frøkjær, J.B.; Mellergaard, M.; Handberg, A. MRI-based quantification of renal fat in obese individuals using different image analysis approaches. Abdom. Radiol. 2022, 47, 3546–3553. [Google Scholar] [CrossRef] [PubMed]
- Yokoo, T.; Clark, H.R.; Pedrosa, I.; Yuan, Q.; Dimitrov, I.; Zhang, Y.; Lingvay, I.; Beg, M.S.; Bobulescu, I.A. Quantification of renal steatosis in type II diabetes mellitus using dixon-based MRI. J. Magn. Reson. Imaging 2016, 44, 1312–1319. [Google Scholar] [CrossRef]
- Wang, Y.C.; Feng, Y.; Lu, C.Q.; Ju, S. Renal fat fraction and diffusion tensor imaging in patients with early-stage diabetic nephropathy. Eur. Radiol. 2018, 28, 3326–3334. [Google Scholar] [CrossRef]
- Sun, H.; Chen, J.; Hua, Y.; Zhang, Y.; Liu, Z. New insights into the role of empagliflozin on diabetic renal tubular lipid accumulation. Diabetol. Metab. Syndr. 2022, 14, 121. [Google Scholar] [CrossRef] [PubMed]
- Raunig, D.L.; Pennello, G.A.; Delfino, J.G.; Buckler, A.J.; Hall, T.J.; Guimaraes, A.R.; Wang, X.; Huang, E.P.; Barnhart, H.X.; de Souza, N.; et al. Multiparametric Quantitative Imaging Biomarker as a Multivariate Descriptor of Health: A Roadmap. Acad. Radiol. 2023, 30, 159–182. [Google Scholar] [CrossRef] [PubMed]
- Barentsz, J.O.; Weinreb, J.C.; Verma, S.; Thoeny, H.C.; Tempany, C.M.; Shtern, F.; Padhani, A.R.; Margolis, D.; Macura, K.J.; Haider, M.A.; et al. Synopsis of the PI-RADS v2 Guidelines for Multiparametric Prostate Magnetic Resonance Imaging and Recommendations for Use. Eur. Urol. 2016, 69, 41–49. [Google Scholar] [CrossRef] [PubMed]
- McCafferty, K.; Caplin, B.; Knight, S.; Hockings, P.; Wheeler, D.; Fan, S.L.; Hulthe, J.; Kleta, R.; Ashman, N.; Papastefanou, V.; et al. HEROIC: A 5-year observational cohort study aimed at identifying novel factors that drive diabetic kidney disease: Rationale and study protocol. BMJ Open 2020, 10, e033923. [Google Scholar] [CrossRef]
- Prasad, P.V.; Li, W.; Raj, D.S.; Carr, J.; Carr, M.; Thacker, J.; Li, L.P.; Wang, C.; Sprague, S.M.; Ix, J.H.; et al. Multicenter Study Evaluating Intrarenal Oxygenation and Fibrosis Using Magnetic Resonance Imaging in Individuals with Advanced CKD. Kidney Int. Rep. 2018, 3, 1467–1472. [Google Scholar] [CrossRef] [PubMed]
- Ford, R.; Mozley, P. Report of Task Force II: Best Practices in the Use of Medical Imaging Techniques in Clinical Trials. Drug Inf. J. 2008, 42, 515–523. [Google Scholar] [CrossRef]
- BEAt-DKD. Biomarker Enterprise to Attack Diabetic Kidney Disease. Available online: https://www.beat-dkd.eu/ (accessed on 13 May 2023).
- UKRIN. UK Renal Imaging Network. Enabling Clinical Translation of Functional MRI for Kidney Disease. Available online: https://gtr.ukri.org/projects?ref=MR%2FR02264X%2F1 (accessed on 13 May 2023).
- KPMP. Kidney Precision Medicine Project. Available online: https://www.kpmp.org/ (accessed on 13 May 2023).

| Reference | Intervention | Subjects | N | MR Intervention | Comments |
|---|---|---|---|---|---|
| Prasad et al., 1996 [66] | Furosemide; acetazolamide; water loading | HV | 7 | BOLD | Furosemide and water diuresis decreased medullary R2* |
| Manotham et al., 2012 [67] | Olmesartan | DKD; CKD; HV | 19 | BOLD | Olmesartan decreased R2* in both CKD groups, but not in HV after 60 min |
| Prasad et al., 2015 [68] | Furosemide | CKD | 59 | BOLD, DWI, Volume | CKD patients had lower renal volume, higher cortical R2*, and lower response to furosemide on medullary R2* |
| Vakilzadeh et al., 2015 [69] | Aliskiren; HCTZ | HT | 20 | BOLD | Aliskiren and HCTZ patients with a fall in systolic blood pressure > 10 mmHg decreased cortical R2* levels |
| Vink 2015 [70] | Captopril | HT | 75 | BOLD | The blood pressure-lowering effect of captopril was positively related to cortical and medullary R2* |
| Vakilzadeh et al., 2019 [71] | Glucose | Overweight | 19 | BOLD | Acute hyperglycemia decreased renal R2* |
| Khatir et al., 2019 [72] | RAS inhibitors/CCB; metoprolol | CKD | 75 | PC-MRI, BOLD | Vasodilation treatment reduced intrarenal vascular resistance but did not affect R2* |
| Laursen et al., 2021 [73] | Dapagliflozin; placebo | T1DM + albuminuria | 15 | BOLD, ASL, PC-MRI | A single dose of dapagliflozin decreased cortical R2* without changes in renal perfusion or blood flow |
| Lee et al., 2022 [74] | Empagliflozin; placebo | Heart failure | 105 | ASL, T1, apparent extracellular volume, post-contrast T1, TKV | Empagliflozin reduced perfusion (ASL) and kidney extracellular volume. No between-group differences in kidney T1, TKV, or eGFR |
| Lin et al., 2023 [75] | Liraglutide; placebo | T2DM | 96 | Renal volume, sinus fat volume | Renal enlargement in T2DM can be reversed by liraglutide treatment |
| Gullaksen et al., 2023 [76] | Semaglutide; empagliflozin; combination; or placebo | T2DM + CVD risk | 80 | BOLD, ASL | Empagliflozin increased medullary R2*; semaglutide decreased perfusion in cortex and medulla |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Friedli, I.; Baid-Agrawal, S.; Unwin, R.; Morell, A.; Johansson, L.; Hockings, P.D. Magnetic Resonance Imaging in Clinical Trials of Diabetic Kidney Disease. J. Clin. Med. 2023, 12, 4625. https://doi.org/10.3390/jcm12144625
Friedli I, Baid-Agrawal S, Unwin R, Morell A, Johansson L, Hockings PD. Magnetic Resonance Imaging in Clinical Trials of Diabetic Kidney Disease. Journal of Clinical Medicine. 2023; 12(14):4625. https://doi.org/10.3390/jcm12144625
Chicago/Turabian StyleFriedli, Iris, Seema Baid-Agrawal, Robert Unwin, Arvid Morell, Lars Johansson, and Paul D. Hockings. 2023. "Magnetic Resonance Imaging in Clinical Trials of Diabetic Kidney Disease" Journal of Clinical Medicine 12, no. 14: 4625. https://doi.org/10.3390/jcm12144625
APA StyleFriedli, I., Baid-Agrawal, S., Unwin, R., Morell, A., Johansson, L., & Hockings, P. D. (2023). Magnetic Resonance Imaging in Clinical Trials of Diabetic Kidney Disease. Journal of Clinical Medicine, 12(14), 4625. https://doi.org/10.3390/jcm12144625






