COVID-19 in Pediatric Intensive Care Units in Poland, PAPITCO-19 Study (Polish Analysis of PICU Trends during COVID-19)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Source
2.2. Reporting and Outcome
2.3. Statistical Analysis
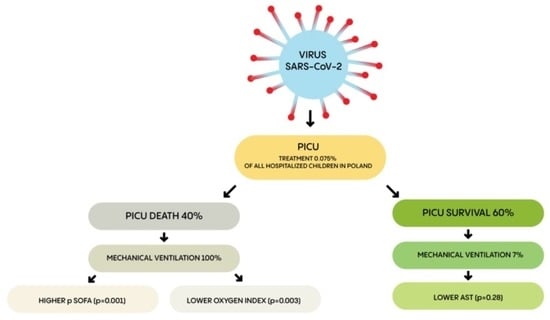
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Borrelli, M.; Corcione, A.; Castellano, F.; Fiori Nastro, F.; Santamaria, F. Coronavirus Disease 2019 in Children. Front. Pediatr. 2021, 9, 668484. [Google Scholar] [CrossRef] [PubMed]
- Leidman, E.; Duca, L.M.; Omura, J.D.; Proia, K.; Stephens, J.W.; Sauber-Schatz, E.K. COVID-19 Trends Among Persons Aged 0–24 Years—United States, March 1–December 12, 2020. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Graff, K.; Smith, C.; Silveira, L.; Jung, S.; Curran-Hays, S.M.; Jarjour, J.; Carpenter, L.B.; Pickard, K.B.; Mattiucci, M.; Fresia, J.B.; et al. Risk Factors for Severe COVID-19 in Children. Pediatr. Infect. Dis. J. 2021, 40, e137–e145. [Google Scholar] [CrossRef] [PubMed]
- Rowley, A.H. Understanding SARS-CoV-2-related multisystem inflammatory syndrome in children. Nat. Rev. Immunol. 2020, 20, 453–454. [Google Scholar] [CrossRef] [PubMed]
- Clark, B.C.; Sanchez-de-Toledo, J.; Bautista-Rodriguez, C.; Choueiter, N.; Lara, D.; Kang, H.; Mohsin, S.; Fraisse, A.; Cesar, S.; Sattar Shaikh, A.; et al. Cardiac abnormalities seen in pediatric patients during the SARS-CoV2 pandemic: An international experience. J. Am. Heart Assoc. 2020, 9, e018007. [Google Scholar] [CrossRef] [PubMed]
- Gov.pl Serwis Rzeczpospolitej Polskiej. Available online: https://www.gov.pl/web/koronawirus/wykaz-zarazen-koronawirusem-sars-cov-2 (accessed on 1 March 2023).
- Derespina, K.R.; Kaushik, S.; Plichta, A.; Conway, E.E., Jr.; Bercow, A.; Choi, J.; Eisenberg, R.; Gillen, J.; Sen, A.I.; Hennigan, C.M.; et al. Clinical manifestations and outcomes of critically ill children and adolescents with coronavirus disease 2019 in New York City. J. Pediatr. 2020, 226, 55–63.e2. [Google Scholar] [CrossRef]
- Simon, K.; Rorat, M.; Janocha-Litwin, J.; Pazgan-Simon, M. SARS-CoV-2 infection: Etiopathogenesis, clinical picture, current therapeutic options—The author’s observations. Nowotw. J. Oncol. 2021, 71, 38–41. [Google Scholar] [CrossRef]
- García-Salido, A.; Vicente, J.C.D.C.; Hofheinz, S.B.; Ramírez, J.B.; Barrio, M.S.; Gordillo, I.L.; Yuste, A.H.; Pardellans, C.G.; Tejedor, M.C.-M.; Labarga, B.H.; et al. Severe manifestations of SARS-CoV-2 in children and adolescents: From COVID-19 pneumonia to multisystem inflammatory syndrome: A multicentre study in pediatric intensive care units in Spain. Crit. Care 2020, 24, 666. [Google Scholar] [CrossRef]
- Shekerdemian, L.S.; Mahmood, N.R.; Wolfe, K.K.; Riggs, B.J.; Ross, C.E.; McKiernan, C.A.; Heidemann, S.M.; Kleinman, L.C.; Sen, A.I.; Hall, M.W.; et al. Characteristics and Outcomes of Children with Coronavirus Disease 2019 (COVID-19) Infection Admitted to US and Canadian Pediatric Intensive Care Units. JAMA Pediatr. 2020, 174, 868–873. [Google Scholar] [CrossRef]
- Adeyinka, A.; Bailey, K.; Pierre, L.; Kondamudi, N. COVID 19 infection: Pediatric perspectives. J. Am. Coll. Emerg. Phys. Open 2021, 2, e12375. [Google Scholar] [CrossRef]
- Krzych, Ł.; Bartkowska-Śniatkowska, A.; Knapik, P.; Zielińska, M.; Maciejewski, D.; Cettler, M.; Owczuk, R.; Kusza, K. Guidelines of the Polish Society of Anaesthesiology and Intensive Therapy defining the rules of qualification and criteria for admitting patients to anaesthesiology and intensive care units. Anaesthesiol. Intensiv. Ther. 2022, 54, 219–225. [Google Scholar] [CrossRef]
- Bartkowska-Śniatkowska, A.; Byrska-Maciejasz, E.; Cettler, M.; Damps, M.; Jarosz, K.; Mierzewska-Schmidt, M.; Migdał, M.; Ożóg-Zabolska, I.; Piotrowski, A.; Rawicz, M.; et al. Guidelines regarding ineffective maintenance of organ functions (futile therapy) in paediatric intensive care units. Anaesthesiol. Intensiv. Ther. 2021, 53, 369–375. [Google Scholar] [CrossRef]
- Damps, M.; Gajda, M.; Stołtny, L.; Kowalska, M.; Kucewicz-Czech, E. Limiting futile therapy as part of end-of-life care in intensive care units. Anaesthesiol. Intensiv. Ther. 2022, 54, 279–284. [Google Scholar] [CrossRef]
- Kucewicz-Czech, E.; Damps, M. Triage during the COVID-19 pandemic. Anaesthesiol. Intensiv. Ther. 2020, 52, 312–315. [Google Scholar] [CrossRef]
- Choi, J.H.; Choi, S.-H.; Yun, K.W. Risk Factors for Severe COVID-19 in Children: A Systematic Review and Meta-Analysis. J. Korean Med. Sci. 2022, 37, e35. [Google Scholar] [CrossRef]
- Kim, L.; Whitaker, M.; O’Halloran, A.; Kambhampati, A.; Chai, S.J.; Reingold, A.; Armistead, I.; Kawasaki, B.; Meek, J.; Yousey-Hindes, K.; et al. COVID-NET Surveillance Team. Hospitalization rates and characteristics of children aged <18 years hospitalized with laboratory-confirmed COVID-19—COVID-NET, 14 States, March1-July 25, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1081–1088. [Google Scholar]
- Götzinger, F.; Santiago-García, B.; Noguera-Julián, A.; Lanaspa, M.; Lancella, L.; Calò Carducci, F.I.; Gabrovska, N.; Velizarova, S.; Prunk, P.; Osterman, V.; et al. COVID-19 in children and adolescents in Europe: A multinational, multicentre cohort study. Lancet Child Adolesc. Health 2020, 4, 653–661. [Google Scholar] [CrossRef]
- Swann, O.V.; Holden, K.A.; Turtle, L.; Pollock, L.; Fairfield, C.J.; Drake, T.M.; Seth, S.; Egan, C.; Hardwick, H.E.; Halpin, S.; et al. Clinical characteristics of children and young people admitted to hospital with COVID-19 in United Kingdom: Prospective multicentre observational cohort study. BMJ 2020, 370, m3249. [Google Scholar] [CrossRef]
- Bellino, S.; Punzo, O.; Rota, M.C.; Del Manso, M.; Urdiales, A.M.; Andrianou, X.; Fabiani, M.; Boros, S.; Vescio, F.; Riccardo, F.; et al. COVID-19 disease severity risk factors for pediatric patients in Italy. Pediatrics 2020, 146, e2020009399. [Google Scholar] [CrossRef]
- Tang, Y.; Li, W.; Baskota, M.; Zhou, Q.; Fu, Z.; Luo, Z.; Shi, Y.; Chen, Y.; Liu, E. Multisystem inflammatory syndrome in children during the coronavirus disease 2019 (COVID-19) pandemic: A systematic review of published case studies. Transl. Pediatr. 2021, 10, 121–135. [Google Scholar] [CrossRef]
- Hoste, L.; Van Paemel, R.; Haerynck, F. Multisystem inflammatory syndrome in children related to COVID-19: A systematic review. Eur. J. Pediatr. 2021, 180, 2019–2034. [Google Scholar] [CrossRef] [PubMed]
- Lippi, G.; Plebani, M. The critical role of laboratory medicine during coronavirus disease 2019 (COVID-19) and other viral outbreaks. Clin. Chem. Lab. Med. 2020, 58, 1063–1069. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoang, A.; Chorath, K.; Moreira, A.; Evans, M.; Burmeister-Morton, F.; Burmeister, F.; Naqvi, R.; Petershack, M.; Moreira, A. COVID-19 in 7780 pediatric patients: A systematic review. EClinicalMedicine 2020, 24, 100433. [Google Scholar] [CrossRef] [PubMed]
- Christophers, B.; Gallo Marin, B.; Oliva, R.; Powell, W.T.; Savage, T.J.; Michelow, I.C. Trends in clinical presentation of children with COVID-19: A systematic review of individual participant data. Pediatr. Res. 2020, 91, 494–501. [Google Scholar] [CrossRef]
- Potere, N.; Batticciotto, A.; Vecchié, A.; Porreca, E.; Cappelli, A.; Abbate, A.; Dentali, F.; Bonaventura, A. The role of IL-6 and IL-6 blockade in COVID-19. Expert Rev. Clin. Immunol. 2021, 17, 601–618. [Google Scholar] [CrossRef]
- Mania, A.; Faltin, K.; Mazur-Melewska, K.; Małecki, P.; Jończyk-Potoczna, K.; Lubarski, K.; Lewandowska, Z.; Cwalińska, A.; Rosada-Kurasińska, J.; Bartkowska-Śniatkowska, A.; et al. Clinical Picture and Risk Factors of Severe Respiratory Symptoms in COVID-19 in Children. Viruses 2021, 13, 2366. [Google Scholar] [CrossRef]
- Henry, B.M.; Benoit, S.W.; de Oliveira, M.H.S.; Hsieh, W.C.; Benoit, J.; Ballout, R.A.; Plebani, M.; Lippi, G. Laboratory abnormalities in children with mild and severe coronavirus disease 2019 (COVID-19): A pooled analysis and review. Clin. Biochem. 2020, 81, 1–8. [Google Scholar] [CrossRef]
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Xia, J.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir. Med. 2020, 8, 475–481, Erratum in Lancet Respir. Med. 2020, 8, e26. [Google Scholar] [CrossRef] [Green Version]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Verdecchia, P.; Cavallini, C.; Spanevello, A.; Angeli, F. The pivotal link between ACE2 deficiency and SARS-CoV-2 infection. Eur. J. Intern. Med. 2020, 76, 14–20. [Google Scholar] [CrossRef]
- Marjot, T.; Webb, G.J.; Barritt, A.S., IV; Moon, A.M.; Stamataki, Z.; Wong, V.W.; Barnes, E. COVID-19 and liver disease: Mechanistic and clinical perspectives. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 348–364. [Google Scholar] [CrossRef]
- Oualha, M.; Bendavid, M.; Berteloot, L.; Corsia, A.; Lesage, F.; Vedrenne, M.; Salvador, E.; Grimaud, M.; Chareyre, J.; de Marcellus, C.; et al. Severe and fatal forms of COVID-19 in children. Arch. Pédiatrie 2020, 27, 235–238. [Google Scholar] [CrossRef]
- Kompaniyets, L.; Agathis, N.T.; Nelson, J.M.; Preston, L.E.; Ko, J.Y.; Belay, B.; Pennington, A.F.; Danielson, M.L.; DeSisto, C.L.; Chevinsky, J.R.; et al. Underlying Medical Conditions Associated with Severe COVID-19 Illness Among Children. JAMA Netw. Open 2021, 4, e2111182. [Google Scholar] [CrossRef]
- Delahoy, M.J.; Ujamaa, D.; Whitaker, M.; O’Halloran, A.; Anglin, O.; Burns, E.; Cummings, C.; Holstein, R.; Kambhampati, A.K.; Milucky, J.; et al. Hospitalizations Associated with COVID-19 Among Children and Adolescents—COVID-NET, 14 States, March 1, 2020–August 14, 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1255–1260. [Google Scholar] [CrossRef]
| Hospital Name, City | Number of Patients |
|---|---|
| Upper Silesian Child Health Centre, Katowice | 9 |
| University Child Hospital, Lublin | 9 |
| University Hospital, Poznan | 7 |
| University Hospital Nr 1, Szczecin | 4 |
| University Child Hospital, Białystok | 4 |
| University Hospital, Rzeszów | 3 |
| Copernicus Hospital, Gdansk | 2 |
| University Clinical Centre of the Medical University of Warsaw | 2 |
| University Children’s Hospital of Cracow | 2 |
| University Clinical Hospital in Wroclaw | 2 |
| Children’s Memorial Health Institute, Warsaw | 1 |
| Survived (n = 27) | Died (n = 18) | p | |
|---|---|---|---|
| Sex Females Males | n = 9 n = 18 | n = 11 n = 7 | 0.125 |
| Age [months] mean ± SD, median | 73.15 ± 75.93, 30.5 | 71.82 ± 75.18, 26 | 0.654 |
| BMI Underweight, n, % Normal, n, % Obese, n, % | 13 (50.00%) 12 (46.15%) 1 (3.85%) | 7 (38.89%) 6 (33.34%) 5 (27.77%) | 0.748 |
| Time from positive test to hospitalisation in the ICU [days] mean ± SD, median | 2.24 ± 3.73, 1 | 5.06 ± 10.65, 1 | 0.608 |
| Comorbidities 1. Neurological disorders, n, % 2. Immobilisation, n, % 3. Immunosuppressive drugs, n, % 4. Congenital heart diseases, n, % 5. Respiratory failure, n, % 6. None, n, % | 15 (55.55%) 10 (37.03%) 2 (7.4%) 3 (11.11%) 5 (18.5%) 5 (18.5%) | 5 (27.77%) 3 (16.66%) 2 (11.11%) 6 (33.33%) 7 (38.88%) - | 0.227 |
| From where the patient was admitted COVID-19 ward, n, % Emergency Room, n, % Home, n, % Paediatric Ward, n, % Other ICU, n, % Operating Room, n, % | 6 (22.22%) 5 (18.53%) 5 (18.52%) 9 (33.33%) 1 (3.70%) 1 (3.70%) | 6 (33.33%) 2 (11.11%) 4 (22.22) 3 (16.67%) 2 (11.11%) 1 (5.56%) | 0.691 * |
| Oxygen therapy, n, % | 7 (25.92%) | 18 (100%) | 0.018 * |
| Mechanical ventilation, n, % | 20 (7.07%) | 18 (100%) | |
| Circulatory failure | 15 (57.69%) | 12 (66.67%) | 0.54 |
| Hospitalisation time [days] mean ± SD, median | 16.56 ± 13.60, 12 | 10.82 ± 7.02, 11 | 0.330 |
| Mortality, n, % study group | 18 (40%) |
| Parameter (Mean ± SD; Median) | Survived | Died | P in Mann– Whitney U Test |
|---|---|---|---|
| First pH measurement | 7.33 ± 0.13, 7.36 | 7.34 ± 0.09, 7.31 | 0.763 |
| First pO2 measurement [mmHg] | 103.84 ± 53.61, 84.4 | 72.75 ± 49.49, 54.0 | 0.018 * |
| First pCO2 measurement [mmHg] | 52.91 ± 24.49, 47.0 | 51.68 ± 21.85, 45.5 | 0.990 |
| First arterial oxygen saturation measurement | 92.39 ± 7.91, 95.0 | 78.43 ± 20.92, 84.0 | 0.018 * |
| First Fi02 measurement | 0.65 ± 0.30, 0.5 | 0.89 ± 0.19, 1.0 | 0.020 * |
| First-day oxygen index [mmHg] | 207.73 ± 137.32, 172.5 | 121.82 ± 147.37, 57.6 | 0.003 * |
| Third-day oxygen index [mmHg] | 232.29 ± 179.71, 180.0 | 145.07 ± 142.96, 96.0 | 0.006 * |
| Fifth-day oxygen index [mmHg] | 229.86 ± 178.88, 158.5 | 143.87 ± 135.10, 84.5 | 0.124 |
| First-day lung injury score | 2.52 ± 1.81, 2.0 | 2.73 ± 1.35, 3.0 | 0.047 * |
| Third-day lung injury score | 2.62 ± 1.44, 2.3 | 2.47 ± 1.03, 3.0 | 0.939 |
| Fifth-day lung injury score | 2.23 ± 1.29, 2.15 | 2.59 ± 0.87, 3.0 | 0.873 |
| First-day pSOFA | 6.22 ± 2.88, 5.5 | 9.38 ± 3.12, 10.0 | 0.012 * |
| Third-day pSOFA | 6.19 ± 3.41, 5.0 | 10.50 ± 3.10, 10.5 | 0.007* |
| Fifth-day pSOFA | 5.78 ± 3.15, 5.0 | 11.50 ± 4.09, 12.0 | 0.001 * |
| WBC ** [×109 per L] | 10.86 ± 7.17, 9.69 | 10.81 ± 10.10, 6.45 | 0.614 |
| PLT ** [×109 per L] | 202.82 ± 113.68, 197.0 | 207.64 ± 171.74, 142.0 | 0.832 |
| HGB ** [g/dL] | 10.91 ± 3.75, 9.8 | 10.94 ± 1.51, 10.5 | 0.178 |
| Procalcitonin ** [ng/mL] | 7.21 ± 8.96, 2.34 | 6.64 ± 17.33, 0.37 | 0.118 |
| C-reactive protein ** [mg/L] | 58.85 ± 106.87, 12.48 | 57.75 ± 77.61, 17.4 | 0.365 |
| D-dimer ** [ng FEU/mL] | 3573.71 ± 4953.15, 1900 | 2813.75 ± 3343.10, 1485.5 | 0.561 |
| Creatynine ** [mg/dL] | 0.59 ± 0.87,0.26 | 0.47 ± 0.32, 0.37 | 0.456 |
| Urea ** [mg/dL] | 48.70 ± 54.35, 32.0 | 27.23 ± 18.06, 24.0 | 0.217 |
| Ferritine [µg/L] | 2307.18 ± 6288.94, 193.0 | 1112.53 ± 1431.25, 430.75 | 0.433 |
| LDH [U/L] | 687.78 ± 575.99, 434.5 | 817.23 ± 678.26,505.0 | 0.232 |
| Troponin [pg/mL] | 49.90 ± 62.55, 24.6 | 950.94 ± 2293.97, 204.0 | 0.089 |
| IL-6 [pg/mL] | 74.22 ± 149.74,18.085 | 95.12 ± 124.83, 51.27 | 0.798 |
| Lactate ** [mmol/L] | 3.03 ± 4.23, 1.1 | 17.76 ± 48.76, 3.26 | 0.070 |
| AST ** [IU/L] | 83.04 ± 132.24, 41.0 | 112.89 ± 139.33, 66.5 | 0.028 * |
| ALT ** [IU/L] | 75.87 ± 176.89; 25.0 | 118.22 ± 281.05, 38.5 | 0.351 |
| Number (%) | Survived (n = 27) | Died (n = 18) | p in Chi-Square Test |
|---|---|---|---|
| COVID-19 convalescent plasma | 7 (26.92%) | 4 (23.53%) | 0.803 |
| Tocilizumab | 2 (8%) | 1 (5.88%) | 0.793 |
| Remdesivir | 5 (19.23%) | 6 (33.33%) | 0.288 |
| Steroid therapy | 27 (100%) | 16 (88.88%) | 0.076 |
| Anticoagulants | 24 (92.30%) | 13 (72.22%) | 0.073 |
| Immunoglobulins | 7 (25.92%) | 8 (44.44%) | 0.196 |
| Muscle relaxants | 11 (40.74%) | 10 (55.55%) | 0.329 |
| Parameter [Mean ± SD; Median] | Ventilated Patients Who Survived (n = 21) | Ventilated Patients Who Died (n = 17) | p Value |
|---|---|---|---|
| Time between positive test and hospitalisation in the ICU [days] | 1.63 ± 2.63, 2.13 | 5.24 ± 10.59, 1 | 0.247 |
| First-day oxygen index [mmHg] | 206.72 ± 147.89, 153.0 | 122.70 ± 151.86, 56.0 | 0.007 * |
| Third-day oxygen index [mmHg] | 232.29 ± 179.71, 180.0 | 145.07 ± 142.96, 96.0 | 0.086 |
| Fifth-day oxygen index [mmHg] | 229.86 ± 178.88, 158.5 | 143.87 ± 135.10, 84.5 | 0.124 |
| First-day lung injury score | 2.52 ± 1.81, 2.0 | 3.0 ± 1.35, 3.0 | 0.106 |
| Third-day lung injury score | 2.62 ± 1.44, 2.3 | 2.47 ± 1.03, 3.0 | 0.909 |
| Fifth-day lung injury score | 2.213 ± 1.34,2.1 | 2.59 ± 0.87, 3.0 | 0.48 |
| First-day pSOFA | 6.62 ± 2.78, 6.0 | 9.17 ± 3.16, 9.0 | 0.043 * |
| Third-day pSOFA | 6.47 ± 3.33, 5.0 | 10.33 ± 3.24, 10.0 | 0.020 * |
| Fifth-day pSOFA | 6.0 ± 3.10, 5.0 | 11.22 ± 4.24, 11.0 | 0.005 * |
| WBC ** [×109 per L] | 10.86 ± 7.17; 9.69 | 18.22 ± 10.10, 6.45 | 0.868 |
| PLT ** [×109 per L] | 202.82 ± 113.68, 197.0 | 162.0 ± 171.74, 142.0 | 0.985 |
| HGB ** [g/dL] | 10.91 ± 3.75, 9.8 | 10.2 ± 1.51, 10.5 | 0.126 |
| Procalcitonin ** [ng/mL] | 7.21 ± 8.96, 2.34 | 0.6 ± 17.33, 0.37 | 0.118 |
| C-reactive protein ** [mg/L] | 58.85 ± 106.87, 12.48 | 13.73 ± 77.61, 17.4 | 0.235 |
| D-dimer ** [ng FEU/mL] | 3573.71 ± 4953.15, 1900.0 | 429.0 ± 3343.09, 1485.5 | 0.897 |
| Creatynine ** [mg/dL] | 0.59 ± 0.87, 0.26 | 0.27 ± 0.32, 0.37 | 0.231 |
| Urea ** [mg/dL] | 48.70 ± 54.35, 32.0 | 8.0 ± 18.06, 24.0 | 0.362 |
| Ferritine [µg/L] | 1837.64 ± 5594.13, 152.65 | 1112.53 ± 1431.25, 430.75 | 0.650 |
| LDH [U/L] | 652.88 ± 526.68, 489.0 | 817.23 ± 678.26, 505.0 | 0.257 |
| Troponin [pg/mL] | 41.65 ± 59.86, 16.95 | 384.0 ± 2293.97, 205 | 0.130 |
| NT proBNP [pg/mL] | 10565.47 ± 15675.56, 2100.0 | 918.5 ± 441.94,918.5 | 0.932 |
| IL-6 [pg/mL] | 74.22 ± 149.74, 18.08 | 7.54 ± 124.83, 51.27 | 0.798 |
| Lactate [mmol/L] | 2.95 ± 3.80, 1.7 | 17.76 ± 48.76, 3.26 | 0.092 |
| AST on the 1st day [IU/L] | 81.90± 135.85, 41.0 | 117.23 ± 142.36, 77.0 | 0.039 * |
| ALT on the 1st day [IU/L] | 72.0 ± 166.48, 25.5 | 39.0 ± 281.05, 38.5 | 0.217 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Damps, M.; Byrska-Maciejasz, E.; Kowalska, M.; Rosada-Kurasińska, J.; Rybojad, B.; Sordyl, J.; Zielińska, M.; Bartkowska-Śniatkowska, A.; on behalf of the PAPITCO-19 Group. COVID-19 in Pediatric Intensive Care Units in Poland, PAPITCO-19 Study (Polish Analysis of PICU Trends during COVID-19). J. Clin. Med. 2023, 12, 3983. https://doi.org/10.3390/jcm12123983
Damps M, Byrska-Maciejasz E, Kowalska M, Rosada-Kurasińska J, Rybojad B, Sordyl J, Zielińska M, Bartkowska-Śniatkowska A, on behalf of the PAPITCO-19 Group. COVID-19 in Pediatric Intensive Care Units in Poland, PAPITCO-19 Study (Polish Analysis of PICU Trends during COVID-19). Journal of Clinical Medicine. 2023; 12(12):3983. https://doi.org/10.3390/jcm12123983
Chicago/Turabian StyleDamps, Maria, Elżbieta Byrska-Maciejasz, Małgorzata Kowalska, Jowita Rosada-Kurasińska, Beata Rybojad, Joanna Sordyl, Marzena Zielińska, Alicja Bartkowska-Śniatkowska, and on behalf of the PAPITCO-19 Group. 2023. "COVID-19 in Pediatric Intensive Care Units in Poland, PAPITCO-19 Study (Polish Analysis of PICU Trends during COVID-19)" Journal of Clinical Medicine 12, no. 12: 3983. https://doi.org/10.3390/jcm12123983