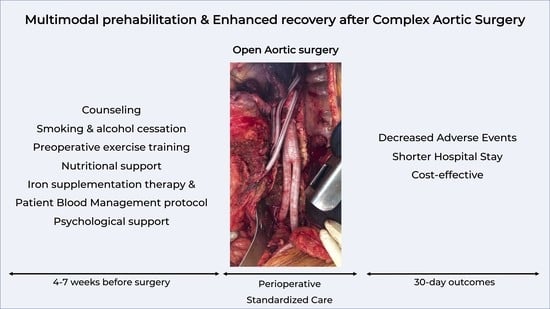
Benefits of Prehabilitation before Complex Aortic Surgery
Abstract
1. Introduction
2. Physiological Consequences of OAS for Complex Aortic Surgery
3. Preoperative Risk Assessment
4. Management of Postoperative Risk with an Active Preoperative Program
4.1. Prehabilitation
4.2. Enhanced Recovery Pathways
5. Limitations to Application
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Antoniou, G.A.; Antoniou, S.A.; Torella, F. Editor’s Choice-Endovascular vs. Open Repair for Abdominal Aortic Aneurysm: Systematic Review and Meta-analysis of Updated Peri-operative and Long Term Data of Randomised Controlled Trials. Eur. J. Vasc. Endovasc. Surg. Off. J. Eur. Soc. Vasc. Surg. 2020, 59, 385–397. [Google Scholar] [CrossRef] [PubMed]
- Dua, A.; Kuy, S.; Lee, C.J.; Upchurch, G.R.; Desai, S.S. Epidemiology of aortic aneurysm repair in the United States from 2000 to 2010. J. Vasc. Surg. 2014, 59, 1512–1517. [Google Scholar] [CrossRef] [PubMed]
- Farber, A.; Eberhardt, R.T. The Current State of Critical Limb Ischemia: A Systematic Review. JAMA Surg. 2016, 151, 1070–1077. [Google Scholar] [CrossRef]
- Upchurch, G.R.; Dimick, J.B.; Wainess, R.M.; Eliason, J.L.; Henke, P.K.; Cowan, J.A.; Eagleton, M.J.; Srivastava, S.D.; Stanley, J.C. Diffusion of new technology in health care: The case of aorto-iliac occlusive disease. Surgery 2004, 136, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Aboyans, V.; Ricco, J.B.; Bartelink, M.L.E.L.; Björck, M.; Brodmann, M.; Cohnert, T.; Collet, J.-P.; Czerny, M.; De Carlo, M.; Debus, S.; et al. 2017 ESC Guidelines on the Diagnosis and Treatment of Peripheral Arterial Diseases, in collaboration with the European Society for Vascular Surgery (ESVS): Document covering atherosclerotic disease of extracranial carotid and vertebral, mesenteric, renal, upper and lower extremity arteriesEndorsed by: The European Stroke Organization (ESO)The Task Force for the Diagnosis and Treatment of Peripheral Arterial Diseases of the European Society of Cardiology (ESC) and of the European Society for Vascular Surgery (ESVS). Eur. Heart J. 2018, 39, 763–816. [Google Scholar]
- Doumenc, B.; Mesnard, T.; Patterson, B.O.; Azzaoui, R.; De Préville, A.; Haulon, S.; Sobocinski, J. Management of Type IA Endoleak after EVAR by Explantation or Custom Made Fenestrated Endovascular Aortic Aneurysm Repair. Eur. J. Vasc. Endovasc. Surg. 2021, 61, 571–578. [Google Scholar] [CrossRef]
- Coselli, J.S.; LeMaire, S.A.; Preventza, O.; de la Cruz, K.I.; Cooley, D.A.; Price, M.D.; Stolz, A.P.; Green, S.Y.; Arredondo, C.N.; Rosengart, T.K. Outcomes of 3309 thoracoabdominal aortic aneurysm repairs. J. Thorac. Cardiovasc. Surg. 2016, 151, 1323–1337. [Google Scholar] [CrossRef]
- Deery, S.E.; Lancaster, R.T.; Baril, D.T.; Indes, J.E.; Bertges, D.J.; Conrad, M.F.; Cambria, R.P.; Patel, V.I. Contemporary outcomes of open complex abdominal aortic aneurysm repair. J. Vasc. Surg. 2016, 63, 1195–1200. [Google Scholar] [CrossRef]
- Latz, C.A.; Cambria, R.P.; Patel, V.I.; Mohebali, J.; Ergul, E.A.; Lancaster, R.T.; Conrad, M.F.; Clouse, W.D. Durability of open surgical repair of type I-III thoracoabdominal aortic aneurysm. J. Vasc. Surg. 2019, 70, 413–423. [Google Scholar] [CrossRef]
- D’oria, M.; Scali, S.; Mao, J.; Szeberin, Z.; Thomson, I.; Beiles, B.; Stone, D.; Sedrakyan, A.; Eldrup, N.; Venermo, M.; et al. Association Between Hospital Volume and Failure to Rescue after Open or Endovascular Repair of Intact Abdominal Aortic Aneurysms in the VASCUNET and International Consortium of Vascular Registries. Ann. Surg. 2021, 274, e452–e459. [Google Scholar] [CrossRef]
- Bredahl, K.; Jensen, L.P.; Schroeder, T.; Sillesen, H.; Nielsen, H.; Eiberg, J.P. Mortality and complications after aortic bifurcated bypass procedures for chronic aortoiliac occlusive disease. J. Vasc. Surg. 2015, 62, 75–82. [Google Scholar] [CrossRef]
- Pouwels, S.; Willigendael, E.M.; van Sambeek, M.R.H.M.; Nienhuijs, S.W.; Cuypers, P.W.M.; Teijink, J.a.W. Beneficial Effects of Pre-operative Exercise Therapy in Patients with an Abdominal Aortic Aneurysm: A Systematic Review. Eur. J. Vasc. Endovasc. Surg. 2015, 49, 66–76. [Google Scholar] [CrossRef]
- Kehlet, H.; Dahl, J.B. Anaesthesia, surgery, and challenges in postoperative recovery. Lancet Lond. Engl. 2003, 362, 1921–1928. [Google Scholar] [CrossRef]
- Fearon, K.C.H.; Ljungqvist, O.; Von Meyenfeldt, M.; Revhaug, A.; Dejong, C.H.C.; Lassen, K.; Nygren, J.; Hausel, J.; Soop, M.; Andersen, J.; et al. Enhanced recovery after surgery: A consensus review of clinical care for patients undergoing colonic resection. Clin. Nutr. 2005, 24, 466–477. [Google Scholar] [CrossRef]
- Feldheiser, A.; Aziz, O.; Baldini, G.; Cox, B.P.B.W.; Fearon, K.C.H.; Feldman, L.S.; Gan, T.J.; Kennedy, R.H.; Ljungqvist, O.; Lobo, D.N.; et al. Enhanced Recovery after Surgery (ERAS) for gastrointestinal surgery, part 2: Consensus statement for anaesthesia practice. Acta Anaesthesiol. Scand. 2016, 60, 289–334. [Google Scholar] [CrossRef]
- Batchelor, T.J.P.; Rasburn, N.J.; Abdelnour-Berchtold, E.; Brunelli, A.; Cerfolio, R.; Gonzalez, M.; Ljungqvist, O.; Petersen, R.H.; Popescu, W.M.; Slinger, P.D.; et al. Guidelines for enhanced recovery after lung surgery: Recommendations of the Enhanced Recovery after Surgery (ERAS®) Society and the European Society of Thoracic Surgeons (ESTS). Eur. J. Cardio-Thoracic Surg. 2019, 55, 91–115. [Google Scholar] [CrossRef]
- Melloul, E.; Hübner, M.; Scott, M.; Snowden, C.; Prentis, J.; Dejong, C.H.C.; Garden, O.J.; Farges, O.; Kokudo, N.; Vauthey, J.-N.; et al. Guidelines for Perioperative Care for Liver Surgery: Enhanced Recovery after Surgery (ERAS) Society Recommendations. World J. Surg. 2016, 40, 2425–2440. [Google Scholar] [CrossRef]
- Wainwright, T.W.; Gill, M.; McDonald, D.A.; Middleton, R.G.; Reed, M.; Sahota, O.; Yates, P.; Ljungqvist, O. Consensus statement for perioperative care in total hip replacement and total knee replacement surgery: Enhanced Recovery after Surgery (ERAS®) Society recommendations. Acta Orthop. 2020, 91, 3–19. [Google Scholar] [CrossRef]
- Debono, B.; Wainwright, T.W.; Wang, M.Y.; Sigmundsson, F.G.; Yang, M.M.; Smid-Nanninga, H.; Bonnal, A.; Le Huec, J.-C.; Fawcett, W.J.; Ljungqvist, O.; et al. Consensus statement for perioperative care in lumbar spinal fusion: Enhanced Recovery after Surgery (ERAS®) Society recommendations. Spine J. 2021, 21, 729–752. [Google Scholar] [CrossRef]
- McGinigle, K.L.; Spangler, E.L.; Pichel, A.C.; Ayyash, K.; Arya, S.; Settembrini, A.M.; Garg, J.; Thomas, M.M.; Dell, K.E.; Swiderski, I.J.; et al. Perioperative care in open aortic vascular surgery: A consensus statement by the Enhanced Recovery after Surgery (ERAS) Society and Society for Vascular Surgery. J. Vasc. Surg. 2022, 75, 1796–1820. [Google Scholar] [CrossRef]
- Cusack, B.; Buggy, D. Anaesthesia, analgesia, and the surgical stress response. BJA Educ. 2020, 20, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Soeters, P.B.; Grimble, R.F. Dangers, and benefits of the cytokine mediated response to injury and infection. Clin. Nutr. 2009, 28, 583–596. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.; Sabanathan, S. Prevention of respiratory complications after abdominal surgery. Thorax 1997, 52, S35–S40. [Google Scholar] [CrossRef] [PubMed]
- Twine, C.P.; Humphreys, A.K.; Williams, I.M. Systematic Review and Meta-analysis of the Retroperitoneal versus the Transperitoneal Approach to the Abdominal Aorta. Eur. J. Vasc. Endovasc. Surg. 2013, 46, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Bakhos, C.T.; Fabian, T.; Oyasiji, T.O.; Gautam, S.; Gangadharan, S.P.; Kent, M.S.; Martin, J.; Critchlow, J.F.; DeCamp, M.M. Impact of the Surgical Technique on Pulmonary Morbidity after Esophagectomy. Ann. Thorac. Surg. 2012, 93, 221–226; discussion 226–227. [Google Scholar] [CrossRef]
- Older, P.; Smith, R. Experience with the Preoperative Invasive Measurement of Haemodynamic, Respiratory and Renal Function in 100 Elderly Patients Scheduled for Major Abdominal Surgery. Anaesth. Intensiv. Care 1988, 16, 389–395. [Google Scholar] [CrossRef]
- Zammert, M.; Gelman, S. The pathophysiology of aortic cross-clamping. Best Pract. Res. Clin. Anaesthesiol. 2016, 30, 257–269. [Google Scholar] [CrossRef]
- Norwood, M.; Bown, M.; Sayers, R. Ischaemia-Reperfusion Injury and Regional Inflammatory Responses in Abdominal Aortic Aneurysm Repair. Eur. J. Vasc. Endovasc. Surg. 2004, 28, 234–245. [Google Scholar] [CrossRef]
- Katseni, K.; Chalkias, A.; Kotsis, T.; Dafnios, N.; Arapoglou, V.; Kaparos, G.; Logothetis, E.; Iacovidou, N.; Karvouni, E. The Effect of Perioperative Ischemia and Reperfusion on Multiorgan Dysfunction following Abdominal Aortic Aneurysm Repair. BioMed. Res. Int. 2015, 2015, 98980. [Google Scholar] [CrossRef]
- Chee, Y.; Liu, S.; Irwin, M. Management of bleeding in vascular surgery. Br. J. Anaesth. 2016, 117, 85–94. [Google Scholar] [CrossRef]
- Pham, H.P.; Shaz, B.H. Update on massive transfusion. Br. J. Anaesth. 2013, 111, 71–82. [Google Scholar] [CrossRef]
- Engström, M.; Schött, U.; Romner, B.; Reinstrup, P. Acidosis Impairs the Coagulation: A Thromboelastographic Study. J. Trauma: Inj. Infect. Crit. Care 2006, 61, 624–628. [Google Scholar] [CrossRef]
- Vascular Events In Noncardiac Surgery Patients Cohort Evaluation (VISION) Study Investigators; Devereaux, P.J.; Chan, M.T.V.; Alonso-Coello, P.; Walsh, M.; Berwanger, O.; Villar, J.C.; Wang, C.Y.; Garutti, R.I.; Jacka, M.J.; et al. Association Between Postoperative Troponin Levels and 30-Day Mortality among Patients Undergoing Noncardiac Surgery. JAMA 2012, 307, 2295–2304. [Google Scholar]
- Wanhainen, A.; Verzini, F.; Van Herzeele, I.; Allaire, E.; Bown, M.; Cohnert, T.; Dick, F.; van Herwaarden, J.; Karkos, C.; Koelemay, M.; et al. Editor’s Choice–European Society for Vascular Surgery (ESVS) 2019 Clinical Practice Guidelines on the Management of Abdominal Aorto-iliac Artery Aneurysms. Eur. J. Vasc. Endovasc. Surg. 2019, 57, 8–93. [Google Scholar] [CrossRef]
- Qaseem, A.; Snow, V.; Fitterman, N.; Hornbake, E.R.; Lawrence, V.A.; Smetana, G.W.; Weiss, K.; Owens, D.K.; Clinical Efficacy Assessment Subcommittee of the American College of Physicians. Risk assessment for and strategies to reduce perioperative pulmonary complications for patients undergoing noncardiothoracic surgery: A guideline from the American College of Physicians. Ann. Intern. Med. 2006, 144, 575–580. [Google Scholar] [CrossRef]
- Fleisher, L.A.; Fleischmann, K.E.; Auerbach, A.D.; Barnason, S.A.; Beckman, J.A.; Bozkurt, B.; Davila-Roman, V.G.; Gerhard-Herman, M.D.; Holly, T.A.; Kane, G.C.; et al. 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery: A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines. J. Am. Coll. Cardiol. 2014, 64, e77–e137. [Google Scholar] [CrossRef]
- Morris, C.K.; Ueshima, K.; Kawaguchi, T.; Hideg, A.; Froelicher, V.F. The prognostic value of exercise capacity: A review of the literature. Am. Heart J. 1991, 122, 1423–1431. [Google Scholar] [CrossRef]
- Young, E.; Karthikesalingam, A.; Huddart, S.; Pearse, R.; Hinchliffe, R.; Loftus, I.M.; Thompson, M.; Holt, P. A Systematic Review of the Role of Cardiopulmonary Exercise Testing in Vascular Surgery. Eur. J. Vasc. Endovasc. Surg. Off. J. Eur. Soc. Vasc. Surg. 2012, 44, 64–71. [Google Scholar] [CrossRef]
- Ainsworth, B.E.; Haskell, W.L.; Herrmann, S.D.; Meckes, N.; Bassett, D.R.; Tudor-Locke, C.; Greer, J.L.; Vezina, J.; Whitt-Glover, M.C.; Leon, A.S. 2011 Compendium of Physical Activities: A second update of codes and MET values. Med. Sci. Sport. Exerc. 2011, 43, 1575–1581. [Google Scholar] [CrossRef]
- Thompson, A.R.; Peters, N.; Lovegrove, R.E.; Ledwidge, S.; Kitching, A.; Magee, T.R.; Galland, R.B. Cardiopulmonary exercise testing provides a predictive tool for early and late outcomes in abdominal aortic aneurysm patients. Ind. Mark. Manag. 2011, 93, 474–481. [Google Scholar] [CrossRef]
- Wijeysundera, D.N.; Pearse, R.M.; Shulman, M.A.; Abbott, T.E.F.; Torres, E.; Ambosta, A.; Croal, B.L.; Granton, J.T.; Thorpe, K.E.; Grocott, M.P.W.; et al. Assessment of functional capacity before major non-cardiac surgery: An international, prospective cohort study. Lancet 2018, 391, 2631–2640. [Google Scholar] [CrossRef] [PubMed]
- Smetana, G.W.; Lawrence, V.A.; Cornell, J.E.; American College of Physicians. Preoperative Pulmonary Risk Stratification for Noncardiothoracic Surgery: Systematic Review for the American College of Physicians. Ann. Intern. Med. 2006, 144, 581–595. [Google Scholar] [CrossRef] [PubMed]
- Haridas, M.; Malangoni, M.A. Predictive factors for surgical site infection in general surgery. Surgery 2008, 144, 496–503; 501–503. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, E.; Farber, A.; Eslami, M.; Kalish, J.; Rybin, D.V.; Doros, G.; Peacock, M.R.; Siracuse, J.J. Preoperative hypoalbuminemia is associated with poor clinical outcomes after open and endovascular abdominal aortic aneurysm repair. J. Vasc. Surg. 2017, 66, 53–63.e1. [Google Scholar] [CrossRef] [PubMed]
- Weimann, A.; Braga, M.; Carli, F.; Higashiguchi, T.; Hübner, M.; Klek, S.; Laviano, A.; Ljungqvist, O.; Lobo, D.N.; Martindale, R.; et al. ESPEN guideline: Clinical nutrition in surgery. Clin. Nutr. Edinb. Scotl. 2017, 36, 623–650. [Google Scholar] [CrossRef]
- Beattie, W.S.; Karkouti, K.; Wijeysundera, D.N.; Tait, G. Risk associated with preoperative anemia in noncardiac surgery: A single-center cohort study. Anesthesiology 2009, 110, 574–581. [Google Scholar] [CrossRef]
- Desai, N.; Schofield, N.; Richards, T. Perioperative Patient Blood Management to Improve Outcomes. Obstet. Anesthesia Dig. 2018, 127, 1211–1220. [Google Scholar] [CrossRef]
- Kozek-Langenecker, S.A.; Ahmed, A.B.; Afshari, A.; Albaladejo, P.; Aldecoa, C.; Barauskas, G.; De Robertis, E.; Faraoni, D.; Filipescu, D.C.; Fries, D.; et al. Management of severe perioperative bleeding: Guidelines from the European Society of Anaesthesiology: First update 2016. Eur. J. Anaesthesiol. 2017, 34, 332–395. [Google Scholar] [CrossRef]
- Clegg, A.; Young, J.; Iliffe, S.; Rikkert, M.O.; Rockwood, K. Frailty in elderly people. Lancet 2013, 381, 752–762. [Google Scholar] [CrossRef]
- Chow, W.B.; Rosenthal, R.A.; Merkow, R.P.; Ko, C.Y.; Esnaola, N.F. Optimal Preoperative Assessment of the Geriatric Surgical Patient: A Best Practices Guideline from the American College of Surgeons National Surgical Quality Improvement Program and the American Geriatrics Society. J. Am. Coll. Surg. 2012, 215, 453–466. [Google Scholar] [CrossRef]
- McIsaac, D.I.; MacDonald, D.B.; Aucoin, S.D. Frailty for Perioperative Clinicians: A Narrative Review. Anesth. Analg. 2020, 130, 1450–1460. [Google Scholar] [CrossRef]
- Poh, A.W.Y.; Teo, S.P. Utility of Frailty Screening Tools in Older Surgical Patients. Ann. Geriatr. Med. Res. 2020, 24, 75–82. [Google Scholar] [CrossRef]
- Moran, J.; Wilson, F.; Guinan, E.; McCormick, P.; Hussey, J.; Moriarty, J. Role of cardiopulmonary exercise testing as a risk-assessment method in patients undergoing intra-abdominal surgery: A systematic review. Br. J. Anaesth. 2016, 116, 177–191. [Google Scholar] [CrossRef]
- West, M.A.; Jack, S.; Grocott, M.P.W. Prehabilitation before surgery: Is it for all patients? Best Pract. Res. Clin. Anaesthesiol. 2021, 35, 507–516. [Google Scholar] [CrossRef]
- Boden, I.; Skinner, E.H.; Browning, L.; Reeve, J.; Anderson, L.; Hill, C.; Robertson, I.K.; Story, D.; Denehy, L. Preoperative physiotherapy for the prevention of respiratory complications after upper abdominal surgery: Pragmatic, double blinded, multicentre randomised controlled trial. BMJ 2018, 360, j5916. [Google Scholar] [CrossRef]
- Theadom, A.; Cropley, M. Effects of preoperative smoking cessation on the incidence and risk of intraoperative and postoperative complications in adult smokers: A systematic review. Tob. Control 2006, 15, 352–358. [Google Scholar] [CrossRef]
- Wong, J.; Lam, D.P.; Abrishami, A.; Chan, M.T.V.; Chung, F. Short-term preoperative smoking cessation and postoperative complications: A systematic review and meta-analysis. Can. J. Anaesth. 2012, 59, 268–279. [Google Scholar] [CrossRef]
- Sørensen, L.T. Wound healing and infection in surgery. The clinical impact of smoking and smoking cessation: A systematic review and meta-analysis. Arch. Surg. 2012, 147, 373–383. [Google Scholar] [CrossRef]
- Herdy, A.H.; Marcchi, P.L.B.; Vila, A.; Tavares, C.; Collaço, J.; Niebauer, J.; Ribeiro, J.P. Pre- and postoperative cardiopulmonary rehabilitation in hospitalized patients undergoing coronary artery bypass surgery: A randomized controlled trial. Am. J. Phys. Med. Rehabil. 2008, 87, 714–719. [Google Scholar] [CrossRef]
- Benzo, R.; Wigle, D.; Novotny, P.; Wetzstein, M.; Nichols, F.; Shen, R.K.; Cassivi, S.; Deschamps, C. Preoperative pulmonary rehabilitation before lung cancer resection: Results from two randomized studies. Lung Cancer 2011, 74, 441–445. [Google Scholar] [CrossRef]
- Sewell, L.; Singh, S.J.; Williams, J.E.A.; Collier, R.; Morgan, M.D.L. How long should outpatient pulmonary rehabilitation be? A randomised controlled trial of 4 weeks versus 7 weeks. Thorax 2006, 61, 767–771. [Google Scholar] [CrossRef] [PubMed]
- Barakat, H.M.; Shahin, Y.; Khan, J.A.; McCollum, P.T.; Chetter, I.C. Preoperative Supervised Exercise Improves Outcomes after Elective Abdominal Aortic Aneurysm Repair: A Randomized Controlled Trial. Ann. Surg. 2016, 264, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Dronkers, J.; Veldman, A.; Hoberg, E.; van der Waal, C.; van Meeteren, N. Prevention of pulmonary complications after upper abdominal surgery by preoperative intensive inspiratory muscle training: A randomized controlled pilot study. Clin. Rehabil. 2008, 22, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Weitz, J.I.; Byrne, J.; Clagett, G.P.; Farkouh, M.E.; Porter, J.M.; Sackett, D.L.; Strandness, D.E., Jr.; Taylor, L.M. Diagnosis and Treatment of Chronic Arterial Insufficiency of the Lower Extremities: A Critical Review. Circulation 1996, 94, 3026–3049. [Google Scholar] [CrossRef]
- Thomas, J.; Delaney, C.; Suen, J.; Miller, M. Nutritional status of patients admitted to a metropolitan tertiary care vascular surgery unit. Asia Pac. J. Clin. Nutr. 2019, 28, 64–71. [Google Scholar]
- Mizobuchi, K.; Jujo, K.; Minami, Y.; Ishida, I.; Nakao, M.; Hagiwara, N. The Baseline Nutritional Status Predicts Long-Term Mortality in Patients Undergoing Endovascular Therapy. Nutrients 2019, 11, 1745. [Google Scholar] [CrossRef]
- Carvalho, J.; Correia, M.A.; Kanegusuku, H.; Longano, P.; Wolosker, N.; Ritti-Dias, R.M.; Cucato, G.G. Association between the risk of malnutrition and functional capacity in patients with peripheral arterial disease: A cross-sectional study. PLoS ONE 2022, 17, e0273051. [Google Scholar] [CrossRef]
- Schechter, D.C. “Coral reef” atherosclerosis of the suprarenal aorta. J. Vasc. Surg. 1986, 3, 679–680. [Google Scholar] [CrossRef][Green Version]
- Björck, M.; Koelemay, M.; Acosta, S.; Bastos Goncalves, F.; Kölbel, T.; Kolkman, J.J.; Lees, T.; Lefevre, J.H.; Menyhei, G.; Oderich, G.; et al. Editor’s Choice-Management of the Diseases of Mesenteric Arteries and Veins: Clinical Practice Guidelines of the European Society of Vascular Surgery (ESVS). Eur. J. Vasc. Endovasc. Surg. 2017, 53, 460–510. [Google Scholar] [CrossRef]
- Sana, A.; Van Noord, D.; Mensink, P.B.; Kooij, S.; Van Dijk, K.; Bravenboer, B.; Lieverse, A.G.; Sijbrands, E.J.; Langendonk, J.G.; Kuipers, E.J. Patients with chronic gastrointestinal ischemia have a higher cardiovascular disease risk and mortality. Atherosclerosis 2012, 224, 235–241. [Google Scholar] [CrossRef][Green Version]
- Levett, D.Z.H.; Grimmett, C. Psychological factors, prehabilitation and surgical outcomes: Evidence and future directions. Anaesthesia 2019, 74, 36–42. [Google Scholar] [CrossRef]
- Tsimopoulou, I.; Pasquali, S.; Howard, R.; Desai, A.; Gourevitch, D.; Tolosa, I.; Vohra, R. Psychological Prehabilitation before Cancer Surgery: A Systematic Review. Ann. Surg. Oncol. 2015, 22, 4117–4123. [Google Scholar] [CrossRef]
- Dao, T.K.; Youssef, N.A.; Armsworth, M.; Wear, E.; Papathopoulos, K.N.; Gopaldas, R. Randomized controlled trial of brief cognitive behavioral intervention for depression and anxiety symptoms preoperatively in patients undergoing coronary artery bypass graft surgery. J. Thorac. Cardiovasc. Surg. 2011, 142, e109–e115. [Google Scholar] [CrossRef]
- Liberzon, I.; Abelson, J.L.; Amdur, R.L.; King, A.P.; Cardneau, J.D.; Henke, P.; Graham, L.M. Increased psychiatric morbidity after abdominal aortic surgery: Risk factors for stress-related disorders. J. Vasc. Surg. 2006, 43, 929–934. [Google Scholar] [CrossRef]
- King, A.P.; Abelson, J.L.; Gholami, B.; Upchurch, G.R.; Henke, P.; Graham, L.; Liberzon, I. Presurgical Psychological and Neuroendocrine Predictors of Psychiatric Morbidity after Major Vascular Surgery: A Prospective Longitudinal Study. Psychosom. Med. 2015, 77, 993–1005. [Google Scholar] [CrossRef]
- Khoo, C.K.; Vickery, C.J.; Forsyth, N.; Vinall, N.S.; Eyre-Brook, I.A. A prospective randomized controlled trial of multimodal perioperative management protocol in patients undergoing elective colorectal resection for cancer. Ann. Surg. 2007, 245, 867–872. [Google Scholar] [CrossRef]
- Muehling, B.; Schelzig, H.; Steffen, P.; Meierhenrich, R.; Sunder-Plassmann, L.; Orend, K.H. A prospective randomized trial comparing traditional and fast-track patient care in elective open infrarenal aneurysm repair. World J. Surg. 2009, 33, 577–585. [Google Scholar] [CrossRef]
- Docherty, J.; Morgan-Bates, K.; Stather, P. A Systematic Review and Meta-Analysis of Enhanced Recovery for Open Abdominal Aortic Aneurysm Surgery. Vasc. Endovasc. Surg. 2022, 56, 15385744221098810. [Google Scholar] [CrossRef]
- Kehlet, H.; Wilmore, D.W. Evidence-based surgical care and the evolution of fast-track surgery. Ann. Surg. 2008, 248, 189–198. [Google Scholar] [CrossRef]
- Baron, D.M.; Hochrieser, H.; Posch, M.; Metnitz, B.; Rhodes, A.; Moreno, R.P.; Pearse, R.M.; Metnitz, P. Preoperative anaemia is associated with poor clinical outcome in non-cardiac surgery patients. Br. J. Anaesth. 2014, 113, 416–423. [Google Scholar] [CrossRef]
- Dunkelgrun, M.; Hoeks, S.E.; Welten, G.M.J.M.; Vidakovic, R.; Winkel, T.A.; Schouten, O.; van Domburg, R.T.; Bax, J.J.; Kuijper, R.; Chonchol, M.; et al. Anemia as an independent predictor of perioperative and long-term cardiovascular outcome in patients scheduled for elective vascular surgery. Am. J. Cardiol. 2008, 101, 1196–1200. [Google Scholar] [CrossRef] [PubMed]
- Fowler, A.J.; Ahmad, T.; Phull, M.K.; Allard, S.; Gillies, M.A.; Pearse, R.M. Meta-analysis of the association between preoperative anaemia and mortality after surgery. Br. J. Surg. 2015, 102, 1314–1324. [Google Scholar] [CrossRef] [PubMed]
- Obi, A.T.; Park, Y.J.; Bove, P.; Cuff, R.; Kazmers, A.; Gurm, H.S.; Grossman, P.M.; Henke, P.K. The association of perioperative transfusion with 30-day morbidity and mortality in patients undergoing major vascular surgery. J. Vasc. Surg. 2015, 61, 1000–1009.e1. [Google Scholar] [CrossRef] [PubMed]
- Shander, A.; Javidroozi, M.; Ozawa, S.; Hare, G.M.T. What is really dangerous: Anaemia or transfusion? Br. J. Anaesth. 2011, 107, i41–i59. [Google Scholar] [CrossRef]
- Kuy, S.; Dua, A.; Desai, S.; Dua, A.; Patel, B.; Tondravi, N.; Seabrook, G.R.; Brown, K.R.; Lewis, B.D.; Lee, C.J.; et al. Surgical site infections after lower extremity revascularization procedures involving groin incisions. Ann. Vasc. Surg. 2014, 28, 53–58. [Google Scholar] [CrossRef]
- Tan, T.W.; Farber, A.; Hamburg, N.M.; Eberhardt, R.T.; Rybin, D.; Doros, G.; Eldrup-Jorgensen, J.; Goodney, P.P.; Cronenwett, J.L.; Kalish, J.A.; et al. Blood transfusion for lower extremity bypass is associated with increased wound infection and graft thrombosis. J. Am. Coll. Surg. 2013, 216, 1005–1014.e2. [Google Scholar] [CrossRef]
- Rohde, J.M.; Dimcheff, D.E.; Blumberg, N.; Saint, S.; Langa, K.M.; Kuhn, L.; Hickner, A.; Rogers, M.A. Health care-associated infection after red blood cell transfusion: A systematic review and meta-analysis. JAMA 2014, 311, 1317–1326. [Google Scholar] [CrossRef]
- Amin, M.; Fergusson, D.; Aziz, A.; Wilson, K.; Coyle, D.; Hébert, P. The cost of allogeneic red blood cells—A systematic review. Transfus. Med. Oxf. Engl. 2003, 13, 275–285. [Google Scholar] [CrossRef]
- Franchini, M.; Marano, G.; Veropalumbo, E.; Masiello, F.; Pati, I.; Candura, F.; Profili, S.; Catalano, L.; Piccinini, V.; Pupella, S.; et al. Patient Blood Management: A revolutionary approach to transfusion medicine. Blood Transfus. 2019, 17, 191–195. [Google Scholar]
- Kassebaum, N.J.; Jasrasaria, R.; Naghavi, M.; Wulf, S.K.; Johns, N.; Lozano, R.; Regan, M.; Weatherall, D.; Chou, D.P.; Eisele, T.P.; et al. A systematic analysis of global anemia burden from 1990 to 2010. Blood 2014, 123, 615–624. [Google Scholar] [CrossRef]
- Tolkien, Z.; Stecher, L.; Mander, A.P.; Pereira, D.I.A.; Powell, J.J. Ferrous sulfate supplementation causes significant gastrointestinal side-effects in adults: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0117383. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.; Gómez-Ramírez, S.; Cuenca, J.; García-Erce, J.A.; Iglesias-Aparicio, D.; Haman-Alcober, S.; Ariza, D.; Naveira, E. Very-short-term perioperative intravenous iron administration and postoperative outcome in major orthopedic surgery: A pooled analysis of observational data from 2547 patients. Transfusion 2014, 54, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Froessler, B.; Palm, P.; Weber, I.; Hodyl, N.A.; Singh, R.; Murphy, E.M. The Important Role for Intravenous Iron in Perioperative Patient Blood Management in Major Abdominal Surgery: A Randomized Controlled Trial. Ann. Surg. 2016, 264, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Winearls, C.G.; Oliver, D.O.; Pippard, M.J.; Reid, C.; Downing, M.R.; Cotes, P.M. Effect of human erythropoietin derived from recombinant DNA on the anaemia of patients ntained by chronic haemodialysis. Lancet 1986, 2, 1175–1178. [Google Scholar] [CrossRef]
- Carson, J.L.; Grossman, B.J.; Kleinman, S.; Tinmouth, A.T.; Marques, M.B.; Fung, M.K.; Holcomb, J.B.; Illoh, O.; Kaplan, L.J.; Katz, L.M.; et al. Red blood cell transfusion: A clinical practice guideline from the AABB. Ann. Intern. Med. 2012, 157, 49–58. [Google Scholar] [CrossRef]
- Kiser, K.A.; Tanaka, A.; Sandhu, H.K.; Miller, C.C.; Leonard, S.D.; Safi, H.J.; Estrera, A.L. Extensive cell salvage and postoperative outcomes following thoracoabdominal and descending aortic repair. J. Thorac. Cardiovasc. Surg. 2022, 163, 914–921.e1. [Google Scholar] [CrossRef]
- Pasternak, J.; Nikolic, D.; Milosevic, D.; Popovic, V.; Markovic, V. An analysis of the influence of intra-operative blood salvage and autologous transfusion on reducing the need for allogeneic transfusion in elective infrarenal abdominal aortic aneurysm repair. Blood Transfus. 2014, 12, s182–s186. [Google Scholar]
- Sikorski, R.A.; Rizkalla, N.A.; Yang, W.W.; Frank, S.M. Autologous blood salvage in the era of patient blood management. Vox. Sang. 2017, 112, 499–510. [Google Scholar] [CrossRef]
- Muñoz, M.; Campos, A.; Muñoz, E.; Carrero, A.; Cuenca, J.; GarcÍA-Erce, J.A. Red cell salvage in orthopedic surgery. Transfus. Altern. Transfus. Med. 2006, 8, 41–51. [Google Scholar] [CrossRef]
- Allen, S.J.; McBride, W.T.; McMurray, T.J.; Phillips, A.S.; Penugonda, S.P.; Campalani, G.; Young, I.S.; Armstrong, M.A. Cell salvage alters the systemic inflammatory response after off-pump coronary artery bypass grafting surgery. Ann. Thorac. Surg. 2007, 83, 578–585. [Google Scholar] [CrossRef][Green Version]
- Gäbel, J.; Westerberg, M.; Bengtsson, A.; Jeppsson, A. Cell salvage of cardiotomy suction blood improves the balance between pro- and anti-inflammatory cytokines after cardiac surgery. Eur. J. Cardio-Thorac. Surg. 2013, 44, 506–511. [Google Scholar] [CrossRef]
- McCarron, E.P.; Bailey, M.; Leonard, B.; McManus, T.E. Improving the uptake: Barriers and facilitators to pulmonary rehabilitation. Clin. Respir. J. 2019, 13, 624–629. [Google Scholar] [CrossRef]
- Petersen, J.; Kloth, B.; Konertz, J.; Kubitz, J.; Schulte-Uentrop, L.; Ketels, G.; Reichenspurner, H.; Girdauskas, E. Economic impact of enhanced recovery after surgery protocol in minimally invasive cardiac surgery. BMC Health Serv. Res. 2021, 21, 254. [Google Scholar] [CrossRef]
- Noba, L.; Rodgers, S.; Chandler, C.; Balfour, A.; Hariharan, D.; Yip, V.S. Enhanced Recovery after Surgery (ERAS) Reduces Hospital Costs and Improve Clinical Outcomes in Liver Surgery: A Systematic Review and Meta-Analysis. J. Gastrointest Surg. Off. J. Soc. Surg. Aliment. Tract. 2020, 24, 918–932. [Google Scholar] [CrossRef]
| Study (Design) | Study Population | Intervention | Main Outcomes | Results |
|---|---|---|---|---|
| Prehabilitation | Interv. vs. Control Group | |||
| Dronkers et al., 2008 [63] (RCT) | N = 20 Mean age 70 ± 6 years (intervention) vs. 59 ± 6 years (control) | Inspiratory muscle training program (≥2 weeks before surgery) | Postoperative pulmonary atelectasis | 3/10 vs. 8/10; p = 0.07 |
| Barakat et al., 2016 [62] (RCT) | N= 120 (including 46 EVAR) Male sex = 93% Mean age 73 ± 7 years | 6 weeks of preoperative supervised exercise | Composite endpoint including cardiac, pulmonary, and renal complications LOS | 23% vs. 42%; p = 0.021 7 (5, 9) vs. 8 (6, 12.3) days; p= 0.025 |
| ERAS | ||||
| Muehling et al., 2009 [77] (RCT) | N = 99 Median age 67 (40–81) | ERAS protocol | Surgical complications and reoperations Medical complications | 10% vs. 8%; p = 0.741 16% vs. 36%; p = 0.039 |
| Docherty et al., 2022 [78] (Meta-analysis) | Control group: N = 930 Intervention group: N = 709 10 studies included | Various ERAS programs | Postoperative complications LOS 30-day mortality | Ref = Interv. group OR = 0.38 (0.22, 0.65); p = < 0.001 −3.18 days (−5.01, −1.35); p < 0.001 No difference (p = 0.92) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mesnard, T.; Dubosq, M.; Pruvot, L.; Azzaoui, R.; Patterson, B.O.; Sobocinski, J. Benefits of Prehabilitation before Complex Aortic Surgery. J. Clin. Med. 2023, 12, 3691. https://doi.org/10.3390/jcm12113691
Mesnard T, Dubosq M, Pruvot L, Azzaoui R, Patterson BO, Sobocinski J. Benefits of Prehabilitation before Complex Aortic Surgery. Journal of Clinical Medicine. 2023; 12(11):3691. https://doi.org/10.3390/jcm12113691
Chicago/Turabian StyleMesnard, Thomas, Maxime Dubosq, Louis Pruvot, Richard Azzaoui, Benjamin O. Patterson, and Jonathan Sobocinski. 2023. "Benefits of Prehabilitation before Complex Aortic Surgery" Journal of Clinical Medicine 12, no. 11: 3691. https://doi.org/10.3390/jcm12113691
APA StyleMesnard, T., Dubosq, M., Pruvot, L., Azzaoui, R., Patterson, B. O., & Sobocinski, J. (2023). Benefits of Prehabilitation before Complex Aortic Surgery. Journal of Clinical Medicine, 12(11), 3691. https://doi.org/10.3390/jcm12113691