Evaluation of Quality and Bone Microstructure Alterations in Patients with Type 2 Diabetes: A Narrative Review
Abstract
:1. Introduction
2. Search Strategy and Limitations of the Review
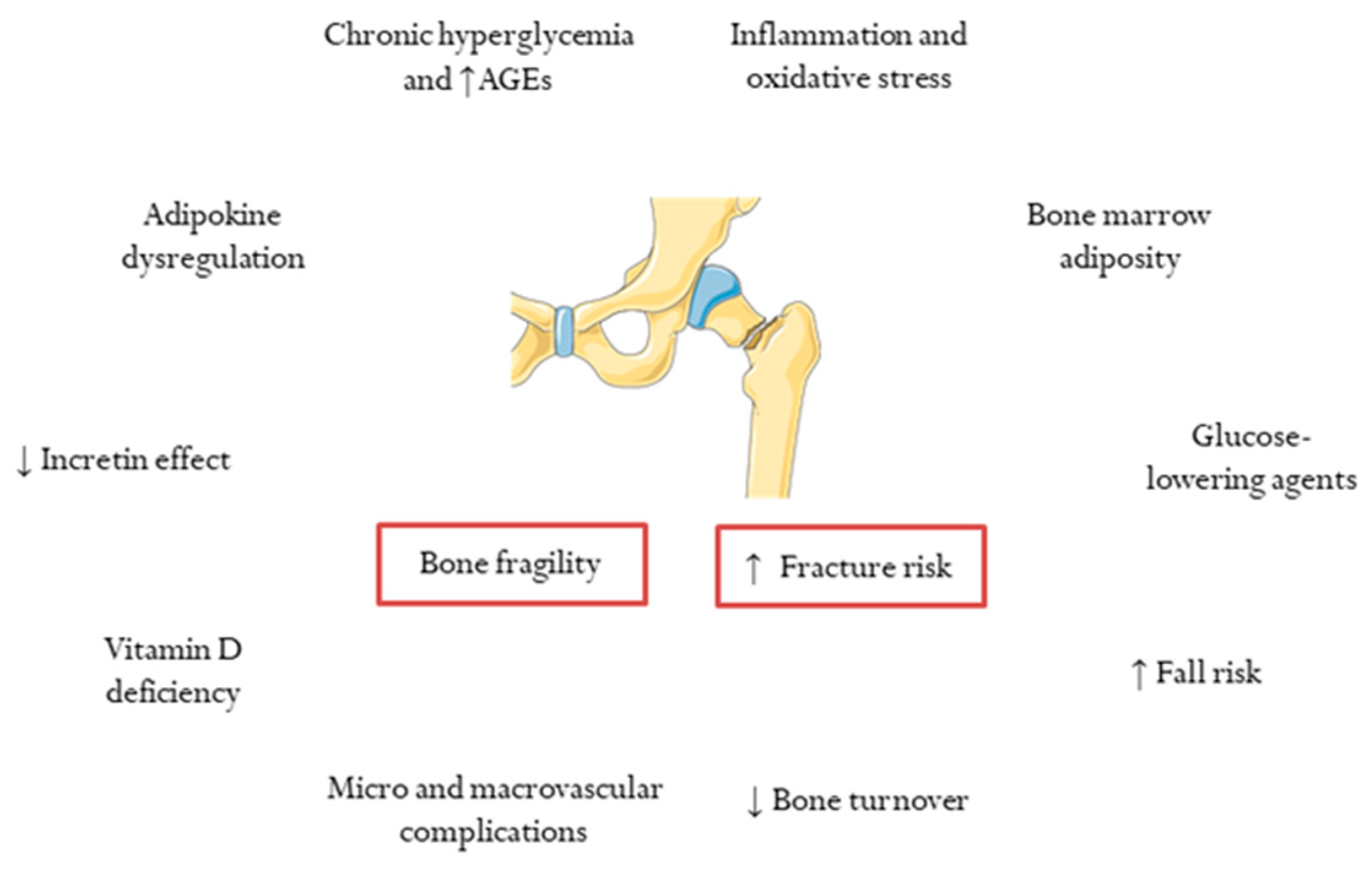
3. Determinants of Skeletal Fragility and Increased Risk of Fracture in T2DM
4. Bone Density and Fracture Risk Prediction in T2DM
5. Bone Microstructure in T2DM
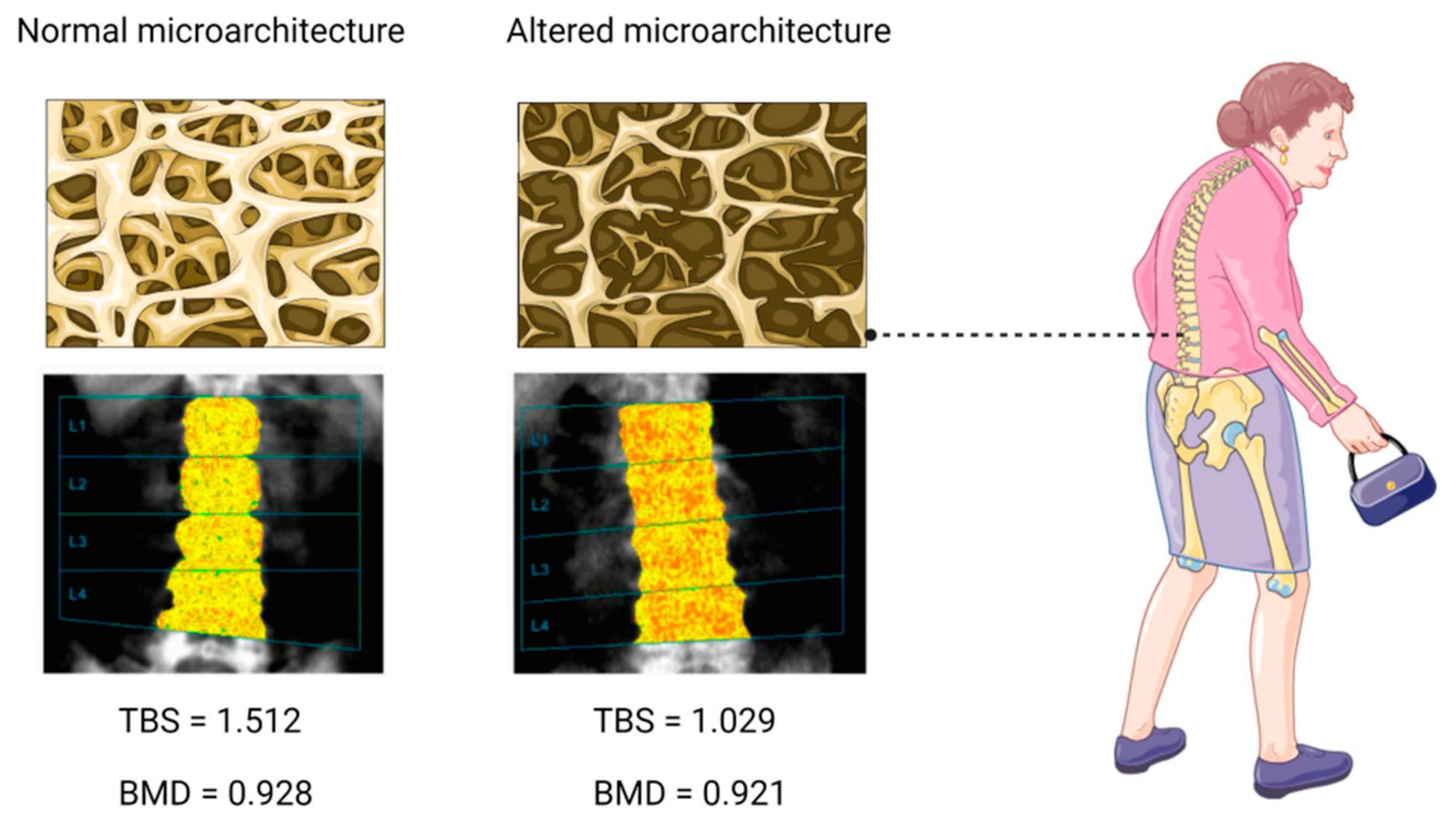
5.1. Trabecular Bone Score
5.2. High-Resolution, Peripheral, Quantitative Computed Tomography
5.3. Microindentation
6. Bone Quality in T2DM: The Role of Biomarkers of Bone Fragility
6.1. Pentosidine
6.2. N-carboxymethyl Lysine
6.3. Sclerostin
6.4. MicroRNAs
6.5. Other Biomarkers
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Dal Canto, E.; Ceriello, A.; Rydén, L.; Ferrini, M.; Hansen, T.B.; Schnell, O.; Standl, E.; Beulens, J.W. Diabetes as a cardiovascular risk factor: An overview of global trends of macro and micro vascular complications. Eur. J. Prev. Cardiol. 2019, 26 (Suppl. 2), 25–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alaofè, H.; Amoussa Hounkpatin, W.; Djrolo, F.; Ehiri, J.; Rosales, C. Factors Associated with Quality of Life in Patients with Type 2 Diabetes of South Benin: A Cross-Sectional Study. Int. J. Environ. Res. Public Health 2022, 19, 2360. [Google Scholar] [CrossRef] [PubMed]
- Fenwick, E.K.; Pesudovs, K.; Khadka, J.; Dirani, M.; Rees, G.; Wong, T.Y.; Lamoureux, E.L. The impact of diabetic retinopathy on quality of life: Qualitative findings from an item bank development project. Qual. Life Res. 2012, 21, 1771–1782. [Google Scholar] [CrossRef] [PubMed]
- Degu, H.; Wondimagegnehu, A.; Yifru, Y.M.; Belachew, A. Is health related quality of life influenced by diabetic neuropathic pain among type II diabetes mellitus patients in Ethiopia? PLoS ONE 2019, 14, e0211449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sinjari, B.; Feragalli, B.; Cornelli, U.; Belcaro, G.; Vitacolonna, E.; Santilli, M.; Rexhepi, I.; D’Addazio, G.; Zuccari, F.; Caputi, S. Artificial Saliva in Diabetic Xerostomia (ASDIX): Double Blind Trial of Aldiamed® Versus Placebo. J. Clin. Med. 2020, 9, 2196. [Google Scholar] [CrossRef]
- Khosla, S.; Samakkarnthai, P.; Monroe, D.G.; Farr, J.N. Update on the pathogenesis and treatment of skeletal fragility in type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2021, 17, 685–697. [Google Scholar] [CrossRef]
- Schousboe, J.T.; Morin, S.N.; Kline, G.A.; Lix, L.M.; Leslie, W.D. Differential risk of fracture attributable to type 2 diabetes mellitus according to skeletal site. Bone 2021, 154, 116220. [Google Scholar] [CrossRef]
- Wang, B.; Wang, Z.; Poundarik, A.A.; Zaki, M.J.; Bockman, R.S.; Glicksberg, B.S.; Nadkarni, G.N.; Vashishth, D. Unmasking Fracture Risk in Type 2 Diabetes: The Association of Longitudinal Glycemic Hemoglobin Level and Medications. J. Clin. Endocrinol. Metab. 2021, 107, e1390–e1401. [Google Scholar] [CrossRef]
- Schwartz, A.V. Epidemiology of fractures in type 2 diabetes. Bone 2016, 82, 2–8. [Google Scholar] [CrossRef]
- Koromani, F.; Ghatan, S.; van Hoek, M.; Zillikens, M.C.; Oei, E.H.G.; Rivadeneira, F.; Oei, L. Type 2 Diabetes Mellitus and Vertebral Fracture Risk. Curr. Osteoporos. Rep. 2021, 19, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Koromani, F.; Oei, L.; Shevroja, E.; Trajanoska, K.; Schoufour, J.; Muka, T.; Franco, O.H.; Ikram, M.A.; Zillikens, M.C.; Uitterlinden, A.G.; et al. Vertebral Fractures in Individuals with Type 2 Diabetes: More Than Skeletal Complications Alone. Diabetes Care 2020, 43, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Vilaca, T.; Schini, M.; Harnan, S.; Sutton, A.; Poku, E.; Allen, I.E.; Cummings, S.R.; Eastell, R. The risk of hip and non-vertebral fractures in type 1 and type 2 diabetes: A systematic review and meta-analysis update. Bone 2020, 137, 115457. [Google Scholar] [CrossRef] [PubMed]
- Janghorbani, M.; van Dam, R.M.; Willett, W.C.; Hu, F.B. Systematic Review of Type 1 and Type 2 Diabetes Mellitus and Risk of Fracture. Am. J. Epidemiol. 2007, 166, 495–505. [Google Scholar] [CrossRef]
- Ammarullah, M.I.; Afif, I.Y.; Maula, M.I.; Winarni, T.I.; Tauviqirrahman, M.; Akbar, I.; Basri, H.; van der Heide, E.; Jamari, J. Tresca Stress Simulation of Metal-on-Metal Total Hip Arthroplasty during Normal Walking Activity. Materials 2021, 14, 7554. [Google Scholar] [CrossRef]
- Jamari, J.; Ammarullah, M.; Saad, A.P.M.; Syahrom, A.; Uddin, M.; van der Heide, E.; Basri, H. The Effect of Bottom Profile Dimples on the Femoral Head on Wear in Metal-on-Metal Total Hip Arthroplasty. J. Funct. Biomater. 2021, 12, 38. [Google Scholar] [CrossRef]
- Sato, M.; Ye, W.; Sugihara, T.; Isaka, Y. Fracture risk and healthcare resource utilization and costs among osteoporosis patients with type 2 diabetes mellitus and without diabetes mellitus in Japan: Retrospective analysis of a hospital claims database. BMC Musculoskelet. Disord. 2016, 17, 489. [Google Scholar] [CrossRef] [Green Version]
- Shah, A.; Wu, F.; Jones, G.; Cicuttini, F.; Toh, L.S.; Laslett, L.L. The association between incident vertebral deformities, health-related quality of life and functional impairment: A 10.7-year cohort study. Osteoporos. Int. 2021, 32, 2247–2255. [Google Scholar] [CrossRef]
- Peeters, C.M.M.; Visser, E.; Van de Ree, C.L.P.; Gosens, T.; Den Oudsten, B.L.; De Vries, J. Quality of life after hip fracture in the elderly: A systematic literature review. Injury 2016, 47, 1369–1382. [Google Scholar] [CrossRef]
- Ferrari, S.L.; Abrahamsen, B.; Napoli, N.; Akesson, K.; Chandran, M.; Eastell, R.; El-Hajj Fuleihan, G.; Josse, R.; Kendler, D.L.; Kraenzlin, M.; et al. Diagnosis and management of bone fragility in diabetes: An emerging challenge. Osteoporos. Int. 2018, 29, 2585–2596. [Google Scholar] [CrossRef] [Green Version]
- de Waard, E.A.C.; van Geel, T.A.C.M.; Savelberg, H.H.C.M.; Koster, A.; Geusens, P.P.M.M.; van den Bergh, J.P.W. Increased fracture risk in patients with type 2 diabetes mellitus: An overview of the underlying mechanisms and the usefulness of imaging modalities and fracture risk assessment tools. Maturitas 2014, 79, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.S.; Vilaca, T. Obesity, Type 2 Diabetes and Bone in Adults. Calcif. Tissue Int. 2017, 100, 528–535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hofbauer, L.C.; Busse, B.; Eastell, R.; Ferrari, S.; Frost, M.; Müller, R.; Burden, A.M.; Rivadeneira, F.; Napoli, N.; Rauner, M. Bone fragility in diabetes: Novel concepts and clinical implications. Lancet Diabetes Endocrinol. 2022, 10, 207–220. [Google Scholar] [CrossRef]
- Majumdar, S.R.; Leslie, W.D.; Lix, L.M.; Morin, S.N.; Johansson, H.; Oden, A.; McCloskey, E.V.; Kanis, J.A. Longer Duration of Diabetes Strongly Impacts Fracture Risk Assessment: The Manitoba BMD Cohort. J. Clin. Endocrinol. Metab. 2016, 101, 4489–4496. [Google Scholar] [CrossRef] [PubMed]
- Dufour, A.B.; Kiel, D.P.; Williams, S.A.; Weiss, R.J.; Samelson, E.J. Risk Factors for Incident Fracture in Older Adults with Type 2 Diabetes: The Framingham Heart Study. Diabetes Care 2021, 44, 1547–1555. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-I.; Liu, C.-S.; Lin, W.-Y.; Meng, N.-H.; Chen, C.-C.; Yang, S.-Y.; Chen, H.-J.; Lin, C.-C.; Li, T.-C. Glycated Hemoglobin Level and Risk of Hip Fracture in Older People with Type 2 Diabetes: A Competing Risk Analysis of Taiwan Diabetes Cohort Study. J. Bone Miner. Res. 2015, 30, 1338–1346. [Google Scholar] [CrossRef] [Green Version]
- Oei, L.; Zillikens, M.C.; Dehghan, A.; Buitendijk, G.H.S.; Castaño-Betancourt, M.C.; Estrada, K.; Stolk, L.; Oei, E.H.G.; van Meurs, J.B.J.; Janssen, J.A.M.J.L.; et al. High Bone Mineral Density and Fracture Risk in Type 2 Diabetes as Skeletal Complications of Inadequate Glucose Control: The Rotterdam Study. Diabetes Care 2013, 36, 1619–1628. [Google Scholar] [CrossRef] [Green Version]
- Romero-Díaz, C.; Duarte-Montero, D.; Gutiérrez-Romero, S.A.; Mendivil, C.O. Diabetes and Bone Fragility. Diabetes Ther. 2020, 12, 71–86. [Google Scholar] [CrossRef]
- Starup-Linde, J.; Vestergaard, P. Biochemical bone turnover markers in diabetes mellitus—A systematic review. Bone 2016, 82, 69–78. [Google Scholar] [CrossRef]
- Tonks, K.T.; White, C.; Center, J.R.; Samocha-Bonet, D.; Greenfield, J. Bone Turnover Is Suppressed in Insulin Resistance, Independent of Adiposity. J. Clin. Endocrinol. Metab. 2017, 102, 1112–1121. [Google Scholar] [CrossRef] [Green Version]
- Napoli, N.; Chandran, M.; Pierroz, D.D.; Abrahamsen, B.; Schwartz, A.V.; Ferrari, S.L. Mechanisms of diabetes mellitus-induced bone fragility. Nat. Rev. Endocrinol. 2017, 13, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Nuche-Berenguer, B.; Portal-Núñez, S.; Moreno, P.; González, N.; Acitores, A.; López-Herradón, A.; Esbrit, P.; Valverde, I.; Villanueva-Peñacarrillo, M.L. Presence of a functional receptor for GLP-1 in osteoblastic cells, independent of the cAMP-linked GLP-1 receptor. J. Cell. Physiol. 2010, 225, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Torres, M.; Reyes-García, R.; García-Martin, A.; Jiménez-Moleón, J.J.; Gonzalez-Ramírez, A.R.; Lara-Villoslada, M.J.; Moreno, P.R. Ischemic heart disease is associated with vertebral fractures in patients with type 2 diabetes mellitus. J. Diabetes Investig. 2013, 4, 310–315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitri, J.; Pittas, A.G. Vitamin D and Diabetes. Endocrinol. Metab. Clin. N. Am. 2014, 43, 205–232. [Google Scholar] [CrossRef] [Green Version]
- Knudsen, J.K.; Leutscher, P.; Sørensen, S. Gut Microbiota in Bone Health and Diabetes. Curr. Osteoporos. Rep. 2021, 19, 462–479. [Google Scholar] [CrossRef]
- Shanbhogue, V.V.; Mitchell, D.M.; Rosen, C.J.; Bouxsein, M.L. Type 2 diabetes and the skeleton: New insights into sweet bones. Lancet Diabetes Endocrinol. 2016, 4, 159–173. [Google Scholar] [CrossRef]
- Rozas-Moreno, P.; Reyes-García, R.; Jódar-Gimeno, E.; Varsavsky, M.; Luque-Fernández, I.; Cortés-Berdonces, M.; Muñoz-Torres, M. Recomendaciones sobre el efecto de los fármacos antidiabéticos en el hueso. Endocrinol. Diabetes Nutr. 2017, 64, 1–6. [Google Scholar] [CrossRef]
- Molinuevo, M.S.; Schurman, L.; McCarthy, A.D.; Cortizo, A.M.; Tolosa, M.J.; Gangoiti, M.V.; Arnol, V.; Sedlinsky, C. Effect of metformin on bone marrow progenitor cell differentiation: In vivo and in vitro studies. J. Bone Miner. Res. 2010, 25, 211–221. [Google Scholar] [CrossRef] [Green Version]
- Monami, M.; Dicembrini, I.; Antenore, A.; Mannucci, E. Dipeptidyl Peptidase-4 Inhibitors and Bone Fractures: A meta-analysis of randomized clinical trials. Diabetes Care 2011, 34, 2474–2476. [Google Scholar] [CrossRef] [Green Version]
- Su, B.; Sheng, H.; Zhang, M.; Bu, L.; Yang, P.; Li, L.; Li, F.; Sheng, C.; Han, Y.; Qu, S.; et al. Risk of bone fractures associated with glucagon-like peptide-1 receptor agonists’ treatment: A meta-analysis of randomized controlled trials. Endocrine 2014, 48, 107–115. [Google Scholar] [CrossRef]
- Zhu, Z.-N.; Jiang, Y.-F.; Ding, T. Risk of fracture with thiazolidinediones: An updated meta-analysis of randomized clinical trials. Bone 2014, 68, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Kohan, D.E.; Fioretto, P.; Tang, W.; List, J.F. Long-term study of patients with type 2 diabetes and moderate renal impairment shows that dapagliflozin reduces weight and blood pressure but does not improve glycemic control. Kidney Int. 2014, 85, 962–971. [Google Scholar] [CrossRef] [Green Version]
- Neal, B.; Perkovic, V.; Mahaffey, K.W.; de Zeeuw, D.; Fulcher, G.; Erondu, N.; Shaw, W.; Law, G.; Desai, M.; Matthews, D.R.; et al. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N. Engl. J. Med. 2017, 377, 644–657. [Google Scholar] [CrossRef] [PubMed]
- Johnston, S.S.; Conner, C.; Aagren, M.; Ruiz, K.; Bouchard, J. Association between hypoglycaemic events and fall-related fractures in Medicare-covered patients with type 2 diabetes. Diabetes Obes. Metab. 2012, 14, 634–643. [Google Scholar] [CrossRef] [PubMed]
- Mayne, D.; Stout, N.R.; Aspray, T.J. Diabetes, falls and fractures. Age Ageing 2010, 39, 522–525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dennison, E.M.; Syddall, H.E.; Aihie Sayer, A.; Craighead, S.; Phillips, D.I.W.; Cooper, C. Type 2 diabetes mellitus is associated with increased axial bone density in men and women from the Hertfordshire Cohort Study: Evidence for an indirect effect of insulin resistance? Diabetologia 2004, 47, 1963–1968. [Google Scholar] [CrossRef]
- Bonds, D.E.; Larson, J.C.; Schwartz, A.V.; Strotmeyer, E.S.; Robbins, J.; Rodriguez, B.L.; Johnson, K.C.; Margolis, K. Risk of Fracture in Women with Type 2 Diabetes: The Women’s Health Initiative Observational Study. J. Clin. Endocrinol. Metab. 2006, 91, 3404–3410. [Google Scholar] [CrossRef] [Green Version]
- Mitchell, A.; Fall, T.; Melhus, H.; Wolk, A.; Michaëlsson, K.; Byberg, L. Type 2 Diabetes in Relation to Hip Bone Density, Area, and Bone Turnover in Swedish Men and Women: A Cross-Sectional Study. Calcif. Tissue Int. 2018, 103, 501–511. [Google Scholar] [CrossRef] [Green Version]
- Ma, L.; Oei, L.; Jiang, L.; Estrada, K.; Chen, H.; Wang, Z.; Yu, Q.; Zillikens, M.C.; Gao, X.; Rivadeneira, F. Association between bone mineral density and type 2 diabetes mellitus: A meta-analysis of observational studies. Eur. J. Epidemiol. 2012, 27, 319–332. [Google Scholar] [CrossRef] [Green Version]
- Pan, H.; Wu, N.; Yang, T.; He, W. Association between bone mineral density and type 1 diabetes mellitus: A meta-analysis of cross-sectional studies. Diabetes Metab. Res. Rev. 2014, 30, 531–542. [Google Scholar] [CrossRef]
- Srikanthan, P.; Crandall, C.J.; Miller-Martinez, D.; Seeman, T.E.; Greendale, G.A.; Binkley, N.; Karlamangla, A.S. Insulin Resistance and Bone Strength: Findings From the Study of Midlife in the United States. J. Bone Miner. Res. 2014, 29, 796–803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Upadhyay, J.; Farr, O.M.; Mantzoros, C.S. The role of leptin in regulating bone metabolism. Metabolism 2015, 64, 105–113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Botella Martínez, S.; Varo Cenarruzabeitia, N.; Escalada San Martin, J.; Calleja Canelas, A. The diabetic paradox: Bone mineral density and fracture in type 2 diabetes. Endocrinol. Nutr. 2016, 63, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Giangregorio, L.M.; Leslie, W.D.; Lix, L.M.; Johansson, H.; Oden, A.; McCloskey, E.; Kanis, J.A. FRAX underestimates fracture risk in patients with diabetes. J. Bone Miner. Res. 2012, 27, 301–308. [Google Scholar] [CrossRef] [PubMed]
- El Miedany, Y. FRAX: Re-adjust or re-think. Arch. Osteoporos. 2020, 15, 150. [Google Scholar] [CrossRef]
- Valentini, A.; Cianfarani, M.A.; De Meo, L.; Morabito, P.; Romanello, D.; Tarantino, U.; Federici, M.; Bertoli, A. FRAX tool in type 2 diabetic subjects: The use of HbA1c in estimating fracture risk. Acta Diabetol. 2018, 55, 1043–1050. [Google Scholar] [CrossRef]
- Wen, Z.; Ding, N.; Chen, R.; Liu, S.; Wang, Q.; Sheng, Z.; Liu, H. Comparison of methods to improve fracture risk assessment in chinese diabetic postmenopausal women: A case-control study. Endocrine 2021, 73, 209–216. [Google Scholar] [CrossRef]
- Hu, L.; Li, T.; Zou, Y.; Yin, X.-L.; Gan, H. The Clinical Value of the RA-Adjusted Fracture Risk Assessment Tool in the Fracture Risk Prediction of Patients with Type 2 Diabetes Mellitus in China. Int. J. Gen. Med. 2021, 14, 327–333. [Google Scholar] [CrossRef]
- Leslie, W.D.; Johansson, H.; McCloskey, E.V.; Harvey, N.C.; Kanis, J.A.; Hans, D. Comparison of Methods for Improving Fracture Risk Assessment in Diabetes: The Manitoba BMD Registry. J. Bone Miner. Res. 2018, 33, 1923–1930. [Google Scholar] [CrossRef] [Green Version]
- Hunt, H.B.; Torres, A.M.; Palomino, P.M.; Marty, E.; Saiyed, R.; Cohn, M.; Jo, J.; Warner, S.; Sroga, G.E.; King, K.B.; et al. Altered Tissue Composition, Microarchitecture, and Mechanical Performance in Cancellous Bone From Men With Type 2 Diabetes Mellitus. J. Bone Miner. Res. 2019, 34, 1191–1206. [Google Scholar] [CrossRef]
- Silva, B.C.; Leslie, W.D.; Resch, H.; Lamy, O.; Lesnyak, O.; Binkley, N.; McCloskey, E.V.; Kanis, J.A.; Bilezikian, J.P. Trabecular Bone Score: A Noninvasive Analytical Method Based Upon the DXA Image. J. Bone Miner. Res. 2014, 29, 518–530. [Google Scholar] [CrossRef] [PubMed]
- McCloskey, E.V.; Oden, A.; Harvey, N.C.; Leslie, W.D.; Hans, D.; Johansson, H.; Barkmann, R.; Boutroy, S.; Brown, J.; Chapurlat, R.; et al. A meta-analysis of trabecular bone score in fracture risk prediction and its relationship to FRAX. J. Bone Miner. Res. 2016, 31, 940–948. [Google Scholar] [CrossRef] [PubMed]
- Hans, D.; Goertzen, A.L.; Krieg, M.-A.; Leslie, W.D. Bone microarchitecture assessed by TBS predicts osteoporotic fractures independent of bone density: The Manitoba study. J. Bone Miner. Res. 2011, 26, 2762–2769. [Google Scholar] [CrossRef] [PubMed]
- Leslie, W.D.; Aubry-Rozier, B.; Lix, L.M.; Morin, S.N.; Majumdar, S.R.; Hans, D. Spine bone texture assessed by trabecular bone score (TBS) predicts osteoporotic fractures in men: The Manitoba Bone Density Program. Bone 2014, 67, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Briot, K.; Paternotte, S.; Kolta, S.; Eastell, R.; Reid, D.M.; Felsenberg, D.; Glüer, C.C.; Roux, C. Added value of trabecular bone score to bone mineral density for prediction of osteoporotic fractures in postmenopausal women: The OPUS study. Bone 2013, 57, 232–236. [Google Scholar] [CrossRef]
- Boutroy, S.; Hans, D.; Sornay-Rendu, E.; Vilayphiou, N.; Winzenrieth, R.; Chapurlat, R. Trabecular bone score improves fracture risk prediction in non-osteoporotic women: The OFELY study. Osteoporos. Int. 2013, 24, 77–85. [Google Scholar] [CrossRef]
- Ho-Pham, L.T.; Nguyen, T.V. Association between trabecular bone score and type 2 diabetes: A quantitative update of evidence. Osteoporos. Int. 2019, 30, 2079–2085. [Google Scholar] [CrossRef]
- Ho-Pham, L.T.; Tran, B.; Do, A.T.; Nguyen, T.V. Association between pre-diabetes, type 2 diabetes and trabecular bone score: The Vietnam Osteoporosis Study. Diabetes Res. Clin. Pract. 2019, 155, 107790. [Google Scholar] [CrossRef]
- Hayón-Ponce, M.; García-Fontana, B.; Avilés-Pérez, M.D.; González-Salvatierra, S.; Andújar-Vera, F.; Moratalla-Aranda, E.; Muñoz-Torres, M. Lower trabecular bone score in type 2 diabetes mellitus: A role for fat mass and insulin resistance beyond hyperglycaemia. Diabetes Metab. 2021, 47, 101276. [Google Scholar] [CrossRef]
- Moon, H.U.; Lee, N.; Chung, Y.-S.; Choi, Y.J. Reduction of visceral fat could be related to the improvement of TBS in diabetes mellitus. J. Bone Miner. Metab. 2020, 38, 702–709. [Google Scholar] [CrossRef]
- Palomo, T.; Dreyer, P.; Muszkat, P.; Weiler, F.G.; Bonansea, T.C.P.; Domingues, F.C.; Vieira, J.G.H.; Silva, B.C.; Brandão, C.M.A. Effect of soft tissue noise on trabecular bone score in postmenopausal women with diabetes: A cross sectional study. Bone 2022, 157, 116339. [Google Scholar] [CrossRef] [PubMed]
- Depczynski, B.; Liew, P.Y.; White, C. Association of glycaemic variables with trabecular bone score in post-menopausal women with type 2 diabetes mellitus. Diabet. Med. 2020, 37, 1545–1552. [Google Scholar] [CrossRef] [PubMed]
- Iki, M.; Fujita, Y.; Kouda, K.; Yura, A.; Tachiki, T.; Tamaki, J.; Winzenrieth, R.; Sato, Y.; Moon, J.-S.; Okamoto, N.; et al. Hyperglycemia is associated with increased bone mineral density and decreased trabecular bone score in elderly Japanese men: The Fujiwara-kyo osteoporosis risk in men (FORMEN) study. Bone 2017, 105, 18–25. [Google Scholar] [CrossRef] [PubMed]
- El Asri, M.M.; Rodrigo, E.P.; de la Flor, S.D.-S.; Valdivieso, S.P.; Barrón, M.C.R.; Martínez, J.M.O.; Hernández, J.L.H. Índice trabecular óseo y niveles de 25-hidroxivitamina D en las complicaciones microvasculares de la diabetes mellitus tipo 2. Med. Clin. 2021, in press. [Google Scholar] [CrossRef]
- Leslie, W.D.; Aubry-Rozier, B.; Lamy, O.; Hans, D.; Manitoba Bone Density Program. TBS (Trabecular Bone Score) and Diabetes-Related Fracture Risk. J. Clin. Endocrinol. Metab. 2013, 98, 602–609. [Google Scholar] [CrossRef] [Green Version]
- Zhukouskaya, V.V.; Ellen-Vainicher, C.; Gaudio, A.; Privitera, F.; Cairoli, E.; Ulivieri, F.M.; Palmieri, S.; Morelli, V.; Grancini, V.; Orsi, E.; et al. The utility of lumbar spine trabecular bone score and femoral neck bone mineral density for identifying asymptomatic vertebral fractures in well-compensated type 2 diabetic patients. Osteoporos. Int. 2016, 27, 49–56. [Google Scholar] [CrossRef]
- Yamamoto, M.; Yamauchi, M.; Sugimoto, T. Prevalent vertebral fracture is dominantly associated with spinal microstructural deterioration rather than bone mineral density in patients with type 2 diabetes mellitus. PLoS ONE 2019, 14, e0222571. [Google Scholar] [CrossRef]
- Lin, Y.-C.; Wu, J.; Kuo, S.-F.; Cheung, Y.-C.; Sung, C.-M.; Fan, C.-M.; Chen, F.-P.; Mhuircheartaigh, J.N. Vertebral Fractures in Type 2 Diabetes Patients: Utility of Trabecular Bone Score and Relationship with Serum Bone Turnover Biomarkers. J. Clin. Densitom. 2020, 23, 37–43. [Google Scholar] [CrossRef]
- Nishiyama, K.K.; Shane, E. Clinical Imaging of Bone Microarchitecture with HR-pQCT. Curr. Osteoporos. Rep. 2013, 11, 147–155. [Google Scholar] [CrossRef]
- Mikolajewicz, N.; Bishop, N.; Burghardt, A.J.; Folkestad, L.; Hall, A.; Kozloff, K.M.; Lukey, P.T.; Molloy-Bland, M.; Morin, S.N.; Offiah, A.; et al. HR-pQCT Measures of Bone Microarchitecture Predict Fracture: Systematic Review and Meta-Analysis. J. Bone Miner. Res. 2019, 35, 446–459. [Google Scholar] [CrossRef]
- Cheung, W.; Hung, V.W.; Cheuk, K.; Chau, W.; Tsoi, K.K.; Wong, R.M.; Chow, S.K.; Lam, T.; Yung, P.S.; Law, S.; et al. Best Performance Parameters of HR-pQCT to Predict Fragility Fracture: Systematic Review and Meta-Analysis. J. Bone Miner. Res. 2021, 36, 2381–2398. [Google Scholar] [CrossRef] [PubMed]
- Burghardt, A.J.; Issever, A.S.; Schwartz, A.V.; Davis, K.A.; Masharani, U.; Majumdar, S.; Link, T.M. High-Resolution Peripheral Quantitative Computed Tomographic Imaging of Cortical and Trabecular Bone Microarchitecture in Patients with Type 2 Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2010, 95, 5045–5055. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patsch, J.M.; Burghardt, A.J.; Yap, S.P.; Baum, T.; Schwartz, A.V.; Joseph, G.B.; Link, T.M. Increased cortical porosity in type 2 diabetic postmenopausal women with fragility fractures. J. Bone Miner. Res. 2013, 28, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Yu, E.W.; Putman, M.S.; Derrico, N.; Abrishamanian-Garcia, G.; Finkelstein, J.S.; Bouxsein, M.L. Defects in cortical microarchitecture among African-American women with type 2 diabetes. Osteoporos. Int. 2015, 26, 673–679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Samelson, E.J.; Demissie, S.; Cupples, L.A.; Zhang, X.; Xu, H.; Liu, C.-T.; Boyd, S.K.; McLean, R.R.; Broe, K.E.; Kiel, D.P.; et al. Diabetes and Deficits in Cortical Bone Density, Microarchitecture, and Bone Size: Framingham HR-pQCT Study. J. Bone Miner. Res. 2018, 33, 54–62. [Google Scholar] [CrossRef]
- Heilmeier, U.; Joseph, G.B.; Pasco, C.; Dinh, N.; Torabi, S.; Darakananda, K.; Youm, J.; Carballido-Gamio, J.; Burghardt, A.J.; Link, T.M.; et al. Longitudinal Evolution of Bone Microarchitecture and Bone Strength in Type 2 Diabetic Postmenopausal Women with and without History of Fragility Fractures—A 5-Year Follow-Up Study Using High Resolution Peripheral Quantitative Computed Tomography. Front. Endocrinol. 2021, 12, 599316. [Google Scholar] [CrossRef]
- Shanbhogue, V.V.; Hansen, S.; Frost, M.; Jørgensen, N.R.; Hermann, A.P.; Henriksen, J.E.; Brixen, K. Compromised cortical bone compartment in type 2 diabetes mellitus patients with microvascular disease. Eur. J. Endocrinol. 2016, 174, 115–124. [Google Scholar] [CrossRef]
- De Waard, E.A.C.; De Jong, J.J.A.; Koster, A.; Savelberg, H.H.C.M.; Van Geel, T.A.; Houben, A.J.H.M.; Schram, M.T.; Dagnelie, P.C.; Van Der Kallen, C.J.; Sep, S.J.S.; et al. The association between diabetes status, HbA1c, diabetes duration, microvascular disease, and bone quality of the distal radius and tibia as measured with high-resolution peripheral quantitative computed tomography—The Maastricht Study. Osteoporos. Int. 2018, 29, 2725–2738. [Google Scholar] [CrossRef] [Green Version]
- Farr, J.N.; Drake, M.T.; Amin, S.; Melton, L.J., 3rd; McCready, L.K.; Khosla, S. In Vivo Assessment of Bone Quality in Postmenopausal Women with Type 2 Diabetes. J. Bone Miner. Res. 2014, 29, 787–795. [Google Scholar] [CrossRef]
- Nilsson, A.G.; Sundh, D.; Johansson, L.; Nilsson, M.; Mellström, D.; Rudäng, R.; Zoulakis, M.; Wallander, M.; Darelid, A.; Lorentzon, M. Type 2 Diabetes Mellitus Is Associated with Better Bone Microarchitecture But Lower Bone Material Strength and Poorer Physical Function in Elderly Women: A Population-Based Study. J. Bone Miner. Res. 2017, 32, 1062–1071. [Google Scholar] [CrossRef] [Green Version]
- Randall, C.; Bridges, D.; Guerri, R.; Nogues, X.; Puig, L.; Torres, E.; Mellibovsky, L.; Hoffseth, K.; Stalbaum, T.; Srikanth, A.; et al. Applications of a New Handheld Reference Point Indentation Instrument Measuring Bone Material Strength. J. Med. Devices 2013, 7, 041005. [Google Scholar] [CrossRef] [PubMed]
- Bridges, D.; Randall, C.; Hansma, P.K. A new device for performing reference point indentation without a reference probe. Rev. Sci. Instrum. 2012, 83, 044301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herrera, S.; Diez-Perez, A. Clinical experience with microindentation in vivo in humans. Bone 2017, 95, 175–182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Furst, J.R.; Bandeira, L.C.; Fan, W.-W.; Agarwal, S.; Nishiyama, K.K.; McMahon, D.J.; Dworakowski, E.; Jiang, H.; Silverberg, S.J.; Rubin, M.R. Advanced Glycation Endproducts and Bone Material Strength in Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2016, 101, 2502–2510. [Google Scholar] [CrossRef] [Green Version]
- Holloway-Kew, K.L.; Betson, A.; Rufus-Membere, P.G.; Gaston, J.; Diez-Perez, A.; Kotowicz, M.A.; Pasco, J.A. Impact microindentation in men with impaired fasting glucose and type 2 diabetes. Bone 2021, 142, 115685. [Google Scholar] [CrossRef]
- Starup-Linde, J.; Lykkeboe, S.; Handberg, A.; Vestergaard, P.; Høyem, P.; Fleischer, J.; Hansen, T.K.; Poulsen, P.L.; Laugesen, E. Glucose variability and low bone turnover in people with type 2 diabetes. Bone 2021, 153, 116159. [Google Scholar] [CrossRef]
- Starup-Linde, J.; Eriksen, S.A.; Lykkeboe, S.; Handberg, A.; Vestergaard, P. Biochemical markers of bone turnover in diabetes patients—A meta-analysis, and a methodological study on the effects of glucose on bone markers. Osteoporos. Int. 2014, 25, 1697–1708. [Google Scholar] [CrossRef]
- Reyes-Garcia, R.; Rozas-Moreno, P.; López-Gallardo, G.; Garcia-Martin, A.; Varsavsky, M.; Avilés-Pérez, M.D.; Muñoz-Torres, M. Serum levels of bone resorption markers are decreased in patients with type 2 diabetes. Acta Diabetol. 2013, 50, 47–52. [Google Scholar] [CrossRef]
- Yamamoto, M.; Yamaguchi, T.; Nawata, K.; Yamauchi, M.; Sugimoto, T. Decreased PTH Levels Accompanied by Low Bone Formation Are Associated with Vertebral Fractures in Postmenopausal Women with Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2012, 97, 1277–1284. [Google Scholar] [CrossRef]
- Napoli, N.; Conte, C.; Eastell, R.; Ewing, S.K.; Bauer, D.C.; Strotmeyer, E.S.; Black, D.M.; Samelson, E.J.; Vittinghoff, E.; Schwartz, A.V. Bone Turnover Markers Do Not Predict Fracture Risk in Type 2 Diabetes. J. Bone Miner. Res. 2020, 35, 2363–2371. [Google Scholar] [CrossRef]
- Karim, L.; Moulton, J.; Van Vliet, M.; Velie, K.; Robbins, A.; Malekipour, F.; Abdeen, A.; Ayres, D.; Bouxsein, M.L. Bone microarchitecture, biomechanical properties, and advanced glycation end-products in the proximal femur of adults with type 2 diabetes. Bone 2018, 114, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Wölfel, E.M.; Jähn-Rickert, K.; Schmidt, F.N.; Wulff, B.; Mushumba, H.; Sroga, G.E.; Püschel, K.; Milovanovic, P.; Amling, M.; Campbell, G.M.; et al. Individuals with type 2 diabetes mellitus show dimorphic and heterogeneous patterns of loss in femoral bone quality. Bone 2020, 140, 115556. [Google Scholar] [CrossRef] [PubMed]
- Sell, D.R.; Monnier, V.M. Structure elucidation of a senescence cross-link from human extracellular matrix. Implication of pen-toses in the aging process. J. Biol. Chem. 1989, 264, 21597–21602. [Google Scholar] [CrossRef]
- Sugiyama, S.; Miyata, T.; Ueda, Y.; Tanaka, H.; Maeda, K.; Kawashima, S.; Strihou, C.V.Y.D.; Kurokawa, K. Plasma levels of pentosidine in diabetic patients: An advanced glycation end product. J. Am. Soc. Nephrol. 1998, 9, 1681–1688. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, N.; Okumura, K.-I.; Aso, Y. High serum pentosidine concentrations are associated with increased arterial stiffness and thickness in patients with type 2 diabetes. Metabolism 2005, 54, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Kerkeni, M.; Saïdi, A.; Bouzidi, H.; Letaief, A.; Ben Yahia, S.; Hammami, M. Pentosidine as a biomarker for microvascular complications in type 2 diabetic patients. Diabetes Vasc. Dis. Res. 2013, 10, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Sugimoto, T. Advanced Glycation End Products, Diabetes, and Bone Strength. Curr. Osteoporos. Rep. 2016, 14, 320–326. [Google Scholar] [CrossRef] [Green Version]
- Viguet-Carrin, S.; Roux, J.P.; Arlot, M.E.; Merabet, Z.; Leeming, D.; Byrjalsen, I.; Delmas, P.D.; Bouxsein, M.L. Contribution of the advanced glycation end product pentosidine and of maturation of type I collagen to compressive biomechanical properties of human lumbar vertebrae. Bone 2006, 39, 1073–1079. [Google Scholar] [CrossRef]
- Valcourt, U.; Merle, B.; Gineyts, E.; Viguet-Carrin, S.; Delmas, P.D.; Garnero, P. Non-enzymatic Glycation of Bone Collagen Modifies Osteoclastic Activity and Differentiation. J. Biol. Chem. 2007, 282, 5691–5703. [Google Scholar] [CrossRef] [Green Version]
- Yamamoto, M.; Yamaguchi, T.; Yamauchi, M.; Yano, S.; Sugimoto, T. Serum Pentosidine Levels Are Positively Associated with the Presence of Vertebral Fractures in Postmenopausal Women with Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2008, 93, 1013–1019. [Google Scholar] [CrossRef] [Green Version]
- Choi, Y.J.; Ock, S.Y.; Jin, Y.; Lee, J.S.; Kim, S.H.; Chung, Y.-S. Urinary Pentosidine levels negatively associates with trabecular bone scores in patients with type 2 diabetes mellitus. Osteoporos. Int. 2018, 29, 907–915. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, A.V.; Garnero, P.; Hillier, T.A.; Sellmeyer, D.E.; Strotmeyer, E.S.; Feingold, K.R.; Resnick, H.E.; Tylavsky, F.A.; Black, D.M.; Cummings, S.R.; et al. Pentosidine and Increased Fracture Risk in Older Adults with Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2009, 94, 2380–2386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barzilay, J.I.; Bůžková, P.; Zieman, S.J.; Kizer, J.R.; Djoussé, L.; Ix, J.H.; Tracy, R.P.; Siscovick, D.S.; Cauley, J.A.; Mukamal, K.J. Circulating Levels of Carboxy-Methyl-Lysine (CML) Are Associated with Hip Fracture Risk: The Cardiovascular Health Study. J. Bone Miner. Res. 2014, 29, 1061–1066. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dhaliwal, R.; Ewing, S.K.; Vashishth, D.; Semba, R.D.; Schwartz, A.V. Greater Carboxy-Methyl-Lysine Is Associated with Increased Fracture Risk in Type 2 Diabetes. J. Bone Miner. Res. 2022, 37, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Calle, J.; Sato, A.Y.; Bellido, T. Role and mechanism of action of sclerostin in bone. Bone 2017, 96, 29–37. [Google Scholar] [CrossRef] [Green Version]
- Ardawi, M.-S.M.; Rouzi, A.A.; Al-Sibiani, S.A.; Al-Senani, N.S.; Qari, M.H.; Mousa, S.A. High serum sclerostin predicts the occurrence of osteoporotic fractures in postmenopausal women: The center of excellence for osteoporosis research study. J. Bone Miner. Res. 2012, 27, 2592–2602. [Google Scholar] [CrossRef]
- Arasu, A.; Cawthon, P.M.; Lui, L.-Y.; Do, T.P.; Arora, P.S.; Cauley, J.A.; Ensrud, K.E.; Cummings, S.R. The Study of Osteoporotic Fractures Research Group Serum Sclerostin and Risk of Hip Fracture in Older Caucasian Women. J. Clin. Endocrinol. Metab. 2012, 97, 2027–2032. [Google Scholar] [CrossRef] [Green Version]
- García-Martín, A.; Rozas-Moreno, P.; Reyes-Garcia, R.; Morales-Santana, S.; García-Fontana, B.; Garcia-Salcedo, J.A.; Muñoz-Torres, M. Circulating Levels of Sclerostin Are Increased in Patients with Type 2 Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2012, 97, 234–241. [Google Scholar] [CrossRef] [Green Version]
- Heilmeier, U.; Carpenter, D.R.; Patsch, J.M.; Harnish, R.; Joseph, G.B.; Burghardt, A.J.; Baum, T.; Schwartz, A.V.; Lang, T.F.; Link, T.M. Volumetric femoral BMD, bone geometry, and serum sclerostin levels differ between type 2 diabetic postmenopausal women with and without fragility fractures. Osteoporos. Int. 2015, 26, 1283–1293. [Google Scholar] [CrossRef] [Green Version]
- Ardawi, M.-S.M.; Akhbar, D.H.; AlShaikh, A.; Ahmed, M.M.; Qari, M.H.; Rouzi, A.A.; Ali, A.Y.; Abdulrafee, A.A.; Saeda, M.Y. Increased serum sclerostin and decreased serum IGF-1 are associated with vertebral fractures among postmenopausal women with type-2 diabetes. Bone 2013, 56, 355–362. [Google Scholar] [CrossRef]
- Yamamoto, M.; Yamauchi, M.; Sugimoto, T. Elevated Sclerostin Levels Are Associated with Vertebral Fractures in Patients with Type 2 Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2013, 98, 4030–4037. [Google Scholar] [CrossRef] [PubMed]
- Hensley, A.P.; McAlinden, A. The role of microRNAs in bone development. Bone 2021, 143, 115760. [Google Scholar] [CrossRef] [PubMed]
- Heilmeier, U.; Hackl, M.; Schroeder, F.; Torabi, S.; Kapoor, P.; Vierlinger, K.; Eiriksdottir, G.; Gudmundsson, E.F.; Harris, T.B.; Gudnason, V.; et al. Circulating serum microRNAs including senescent miR-31-5p are associated with incident fragility fractures in older postmenopausal women with type 2 diabetes mellitus. Bone 2022, 158, 116308. [Google Scholar] [CrossRef] [PubMed]
- Heilmeier, U.; Hackl, M.; Skalicky, S.; Weilner, S.; Schroeder, F.; Vierlinger, K.; Patsch, J.M.; Baum, T.; Oberbauer, E.; Lobach, I.; et al. Serum miRNA Signatures Are Indicative of Skeletal Fractures in Postmenopausal Women with and without Type 2 Diabetes and Influence Osteogenic and Adipogenic Differentiation of Adipose Tissue-Derived Mesenchymal Stem Cells In Vitro. J. Bone Miner. Res. 2016, 31, 2173–2192. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.-S.; Kang, X.-R.; Zhou, Z.-H.; Yang, J.; Xin, Q.; Ying, C.-T.; Zhang, Y.-P.; Tao, J. MiR-1908/EXO1 and MiR-203a/FOS, regulated by scd1, are associated with fracture risk and bone health in postmenopausal diabetic women. Aging 2020, 12, 9549–9584. [Google Scholar] [CrossRef]
- Mulder, D.J.; Van De Water, T.; Lutgers, H.L.; Graaff, R.; Gans, R.O.; Zijlstra, F.; Smit, A.J. Skin Autofluorescence, a Novel Marker for Glycemic and Oxidative Stress-Derived Advanced Glycation Endproducts: An Overview of Current Clinical Studies, Evidence, and Limitations. Diabetes Technol. Ther. 2006, 8, 523–535. [Google Scholar] [CrossRef] [Green Version]
- Samakkarnthai, P.; Sfeir, J.G.; Atkinson, E.J.; Achenbach, S.J.; Wennberg, P.W.; Dyck, P.J.; Tweed, A.J.; Volkman, T.L.; Amin, S.; Farr, J.N.; et al. Determinants of Bone Material Strength and Cortical Porosity in Patients with Type 2 Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2020, 105, e3718–e3729. [Google Scholar] [CrossRef]
- Waqas, K.; Chen, J.; Koromani, F.; Trajanoska, K.; Van Der Eerden, B.C.J.; Uitterlinden, A.G.; Rivadeneira, F.; Zillikens, M.C. Skin Autofluorescence, a Noninvasive Biomarker for Advanced Glycation End-Products, Is Associated with Prevalent Vertebral and Major Osteoporotic Fractures: The Rotterdam Study. J. Bone Miner. Res. 2020, 35, 1904–1913. [Google Scholar] [CrossRef]
- Sihota, P.; Pal, R.; Yadav, R.N.; Neradi, D.; Karn, S.; Goni, V.G.; Sharma, S.; Mehandia, V.; Bhadada, S.K.; Kumar, N.; et al. Can fingernail quality predict bone damage in Type 2 diabetes mellitus? A pilot study. PLoS ONE 2021, 16, e0257955. [Google Scholar] [CrossRef]
- Sihota, P.; Yadav, R.N.; Dhiman, V.; Bhadada, S.K.; Mehandia, V.; Kumar, N. Investigation of diabetic patient’s fingernail quality to monitor type 2 diabetes induced tissue damage. Sci. Rep. 2019, 9, 3193. [Google Scholar] [CrossRef]
| Study | Design | Study Population | Results |
|---|---|---|---|
| Leslie et al., 2013 [75] | Retrospective cohort (mean follow-up 4.7 years) | 29,407 women ≥ 50 years (2356 with diagnosed T2DM) | TBS predicted major osteoporotic fractures (hip, spine, forearm and humerus) in T2DM (HR 1.27, CI 1.10–1.46) |
| Zhukouskaya et al., 2015 [76] | Cross-sectional | 99 postmenopausal women with T2DM/107 healthy controls | TBS was associated with VF (AUC 0.69, cut-off value 1.130 in ROC curve analysis) |
| Yamamoto et al., 2019 [77] | Cross-sectional | 584 patients with T2DM (257 postmenopausal women and 291 men > 50 years) | TBS correlated with prevalent VF in multivariate logistic regression analysis |
| Lin et al., 2019 [78] | Cross-sectional | 285 postmenopausal women with T2DM | TBS had the strongest association with VF (AUC 0.775) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Montoro, J.I.; García-Fontana, B.; García-Fontana, C.; Muñoz-Torres, M. Evaluation of Quality and Bone Microstructure Alterations in Patients with Type 2 Diabetes: A Narrative Review. J. Clin. Med. 2022, 11, 2206. https://doi.org/10.3390/jcm11082206
Martínez-Montoro JI, García-Fontana B, García-Fontana C, Muñoz-Torres M. Evaluation of Quality and Bone Microstructure Alterations in Patients with Type 2 Diabetes: A Narrative Review. Journal of Clinical Medicine. 2022; 11(8):2206. https://doi.org/10.3390/jcm11082206
Chicago/Turabian StyleMartínez-Montoro, José Ignacio, Beatriz García-Fontana, Cristina García-Fontana, and Manuel Muñoz-Torres. 2022. "Evaluation of Quality and Bone Microstructure Alterations in Patients with Type 2 Diabetes: A Narrative Review" Journal of Clinical Medicine 11, no. 8: 2206. https://doi.org/10.3390/jcm11082206
APA StyleMartínez-Montoro, J. I., García-Fontana, B., García-Fontana, C., & Muñoz-Torres, M. (2022). Evaluation of Quality and Bone Microstructure Alterations in Patients with Type 2 Diabetes: A Narrative Review. Journal of Clinical Medicine, 11(8), 2206. https://doi.org/10.3390/jcm11082206







