The Relevance in the Neutrophil to Lymphocyte Ratio and the SARC-F Score in Gastrointestinal Diseases
Abstract
:1. Introduction
2. Patients and Methods
2.1. Patients and Our Study
2.2. Statistical Procedure
3. Results
3.1. Patient Baseline Features
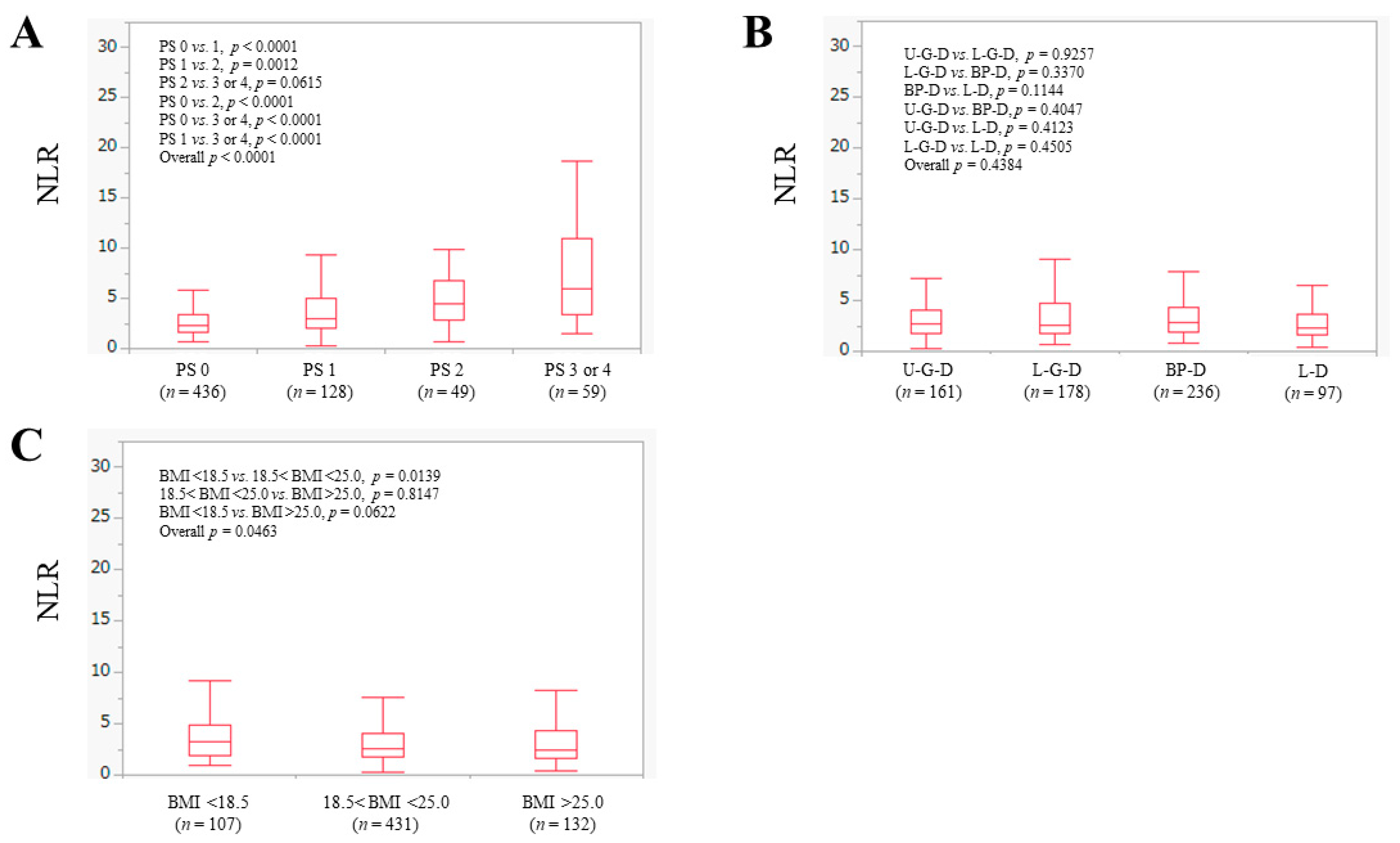
3.2. The NLR according to ECOG-PS, Anatomical Category of Disease and BMI
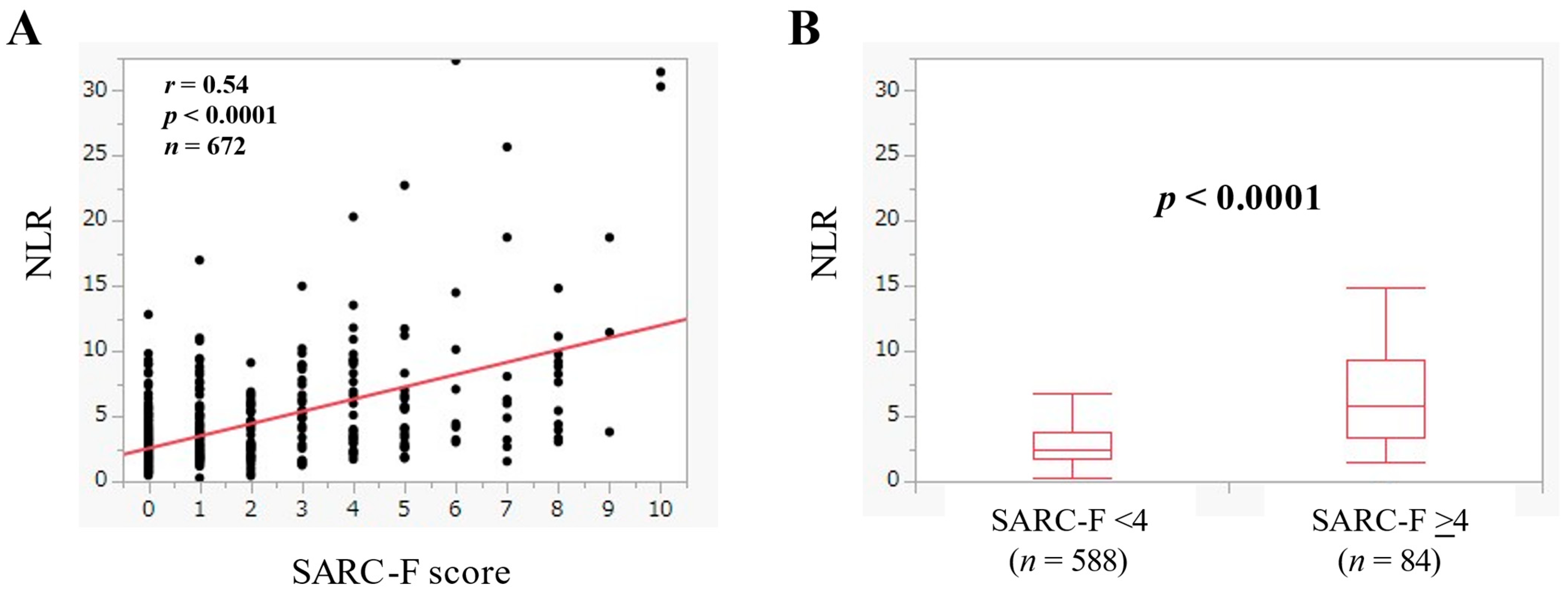
3.3. The Relevance in the NLR and the SARC-F Score
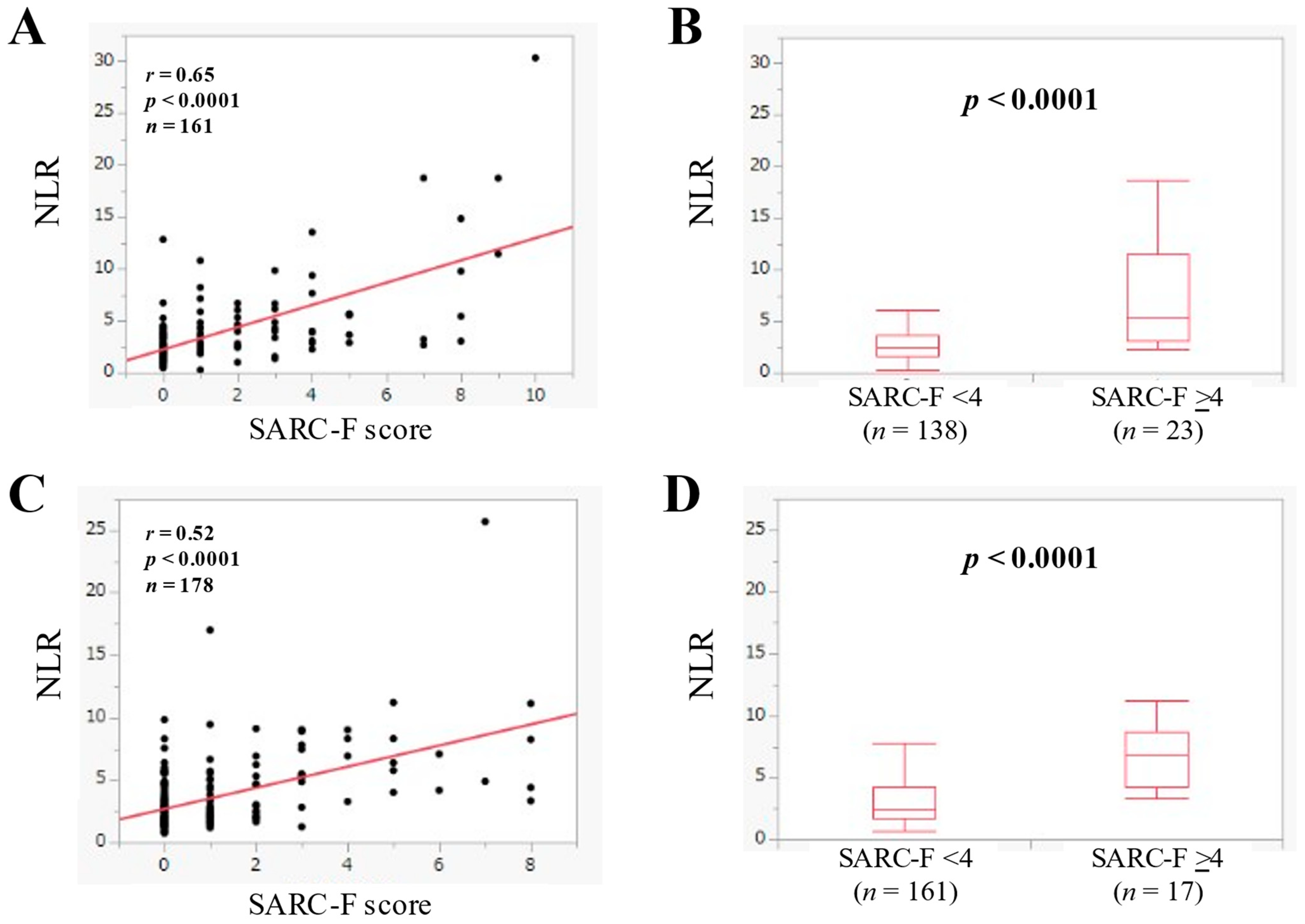
3.4. The Relevance in the NLR and the SARC-F Score according to the Anatomical Category
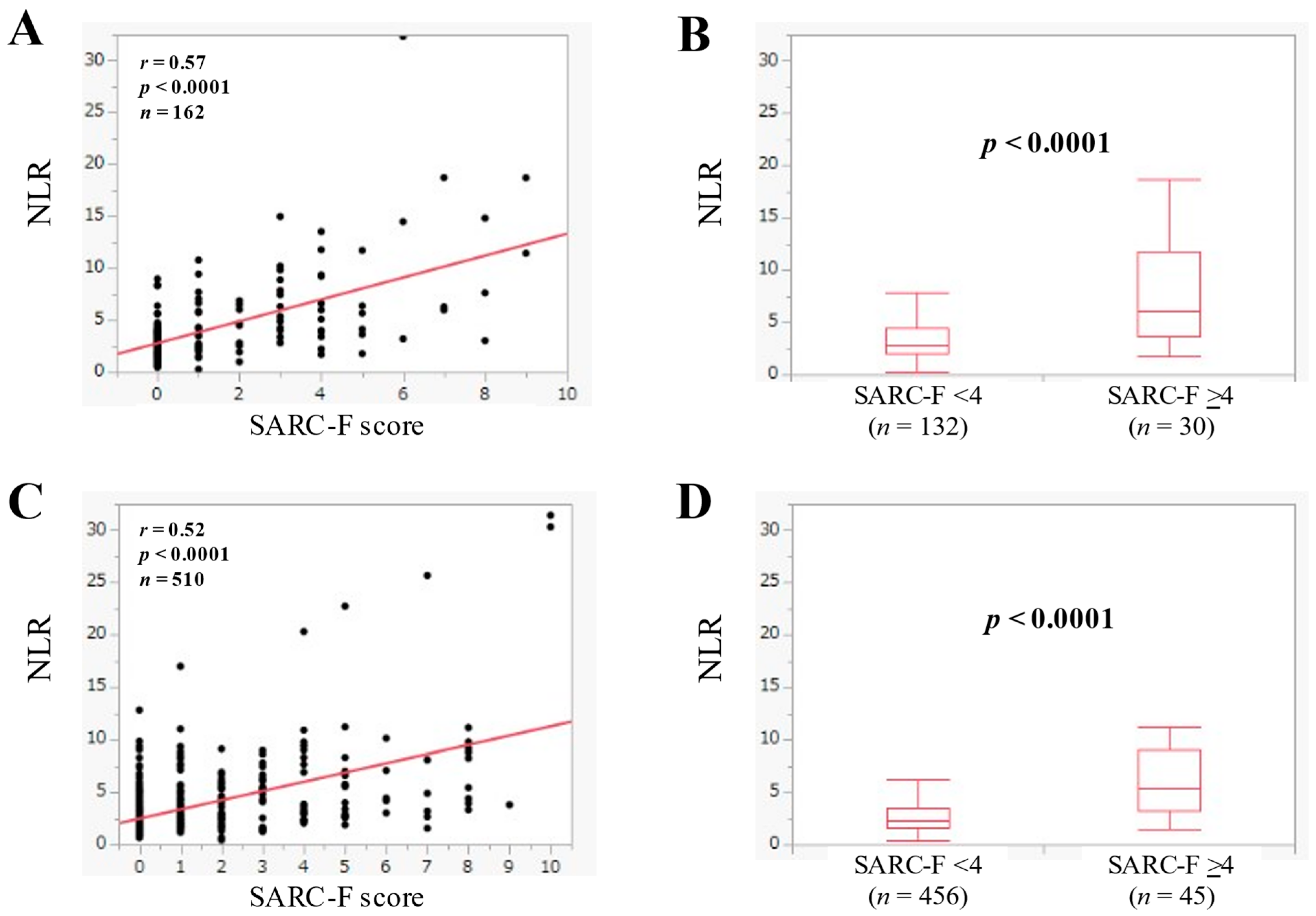
3.5. The Relevance in the NLR and the SARC-F Score in Patients with Advanced Cancer
3.6. The Relevance in the NLR and the SARC-F Score in Patients without Advanced Cancer
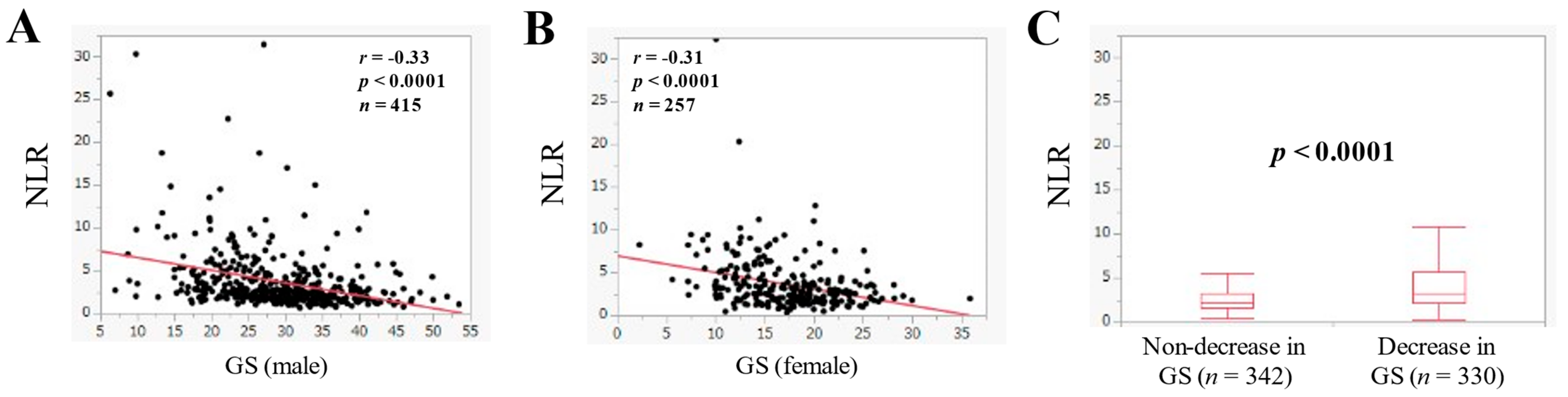
3.7. The Relevance in the NLR and GS
3.8. Univariate and Multivariate Analysis of Factors Linking to the SARC-F Score
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviation
References
- Dunne, R.F.; Loh, K.P.; Williams, G.R.; Jatoi, A.; Mustian, K.M.; Mohile, S.G. Cachexia and Sarcopenia in Older Adults with Cancer: A Comprehensive Review. Cancers 2019, 11, 1861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chhetri, J.K.; de Souto Barreto, P.; Fougère, B.; Rolland, Y.; Vellas, B.; Cesari, M. Chronic inflammation and sarcopenia: A regenerative cell therapy perspective. Exp. Gerontol. 2018, 103, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Ganapathy, A.; Nieves, J.W. Nutrition and Sarcopenia—What Do We Know? Nutrients 2020, 12, 1755. [Google Scholar] [CrossRef] [PubMed]
- Sieber, C.C. Malnutrition and sarcopenia. Aging Clin. Exp. Res. 2019, 31, 793–798. [Google Scholar] [CrossRef]
- Nishikawa, H.; Fukunishi, S.; Asai, A.; Yokohama, K.; Nishiguchi, S.; Higuchi, K. Pathophysiology and mechanisms of primary sarcopenia (Review). Int. J. Mol. Med. 2021, 48, 156. [Google Scholar] [CrossRef]
- Nishikawa, H.; Shiraki, M.; Hiramatsu, A.; Moriya, K.; Hino, K.; Nishiguchi, S. Japan Society of Hepatology guidelines for sarcopenia in liver disease (1st edition): Recommendation from the working group for creation of sarcopenia assessment criteria. Hepatol. Res. 2016, 46, 951–963. [Google Scholar] [CrossRef]
- Balestrieri, P.; Ribolsi, M.; Guarino, M.P.L.; Emerenziani, S.; Altomare, A.; Cicala, M. Nutritional Aspects in Inflammatory Bowel Diseases. Nutrients 2020, 12, 372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shaw, C. Management of diet in gastrointestinal cancer. Proc. Nutr. Soc. 2021, 80, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yang, R.; Xu, J.; Fang, K.; Abdelrahim, M.; Chang, L. Sarcopenia as a predictor of postoperative risk of complications, mortality and length of stay following gastrointestinal oncological surgery. Ann. R. Coll. Surg. Engl. 2021, 103, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Meyer, F.; Valentini, L. Disease-Related Malnutrition and Sarcopenia as Determinants of Clinical Outcome. Visc. Med. 2019, 35, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Matsui, M.; Nishikawa, H.; Goto, M.; Asai, A.; Ushiro, K.; Ogura, T.; Takeuchi, T.; Nakamura, S.; Kakimoto, K.; Miyazaki, T.; et al. Prognostic Impact of the SARC-F Score in Gastrointestinal Advanced Cancers. Cancers 2021, 14, 10. [Google Scholar] [CrossRef] [PubMed]
- Ikegami, T.; Nishikawa, H.; Goto, M.; Matsui, M.; Asai, A.; Ushiro, K.; Ogura, T.; Takeuchi, T.; Nakamura, S.; Kakimoto, K.; et al. The Relationship between the SARC-F Score and the Controlling Nutritional Status Score in Gastrointestinal Diseases. J Clin Med. 2022, 11, 582. [Google Scholar] [CrossRef]
- Vellas, B.; Pahor, M.; Manini, T.; Rooks, D.; Guralnik, J.M.; Morley, J.; Studenski, S.; Evans, W.; Asbrand, C.; Fariello, R.; et al. Designing pharmaceutical trials for sarcopenia in frail older adults: EU/US Task Force recommendations. J. Nutr. Health Aging 2013, 17, 612–618. [Google Scholar] [CrossRef] [Green Version]
- Malmstrom, T.K.; Miller, D.K.; Simonsick, E.M.; Ferrucci, L.; Morley, J.E. SARC-F: A symptom score to predict persons with sarcopenia at risk for poor functional outcomes. J. Cachexia Sarcopenia Muscle 2016, 7, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Ida, S.; Kaneko, R.; Murata, K. SARC-F for Screening of Sarcopenia among Older Adults: A Meta-analysis of Screening Test Accuracy. J. Am. Med. Dir. Assoc. 2018, 19, 685–689. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, M.; Won, C.W. Validation of the Korean Version of the SARC-F Questionnaire to Assess Sarcopenia: Korean Frailty and Aging Cohort Study. J. Am. Med. Dir. Assoc. 2018, 19, 40–45.e41. [Google Scholar] [CrossRef]
- Tanaka, S.; Kamiya, K.; Hamazaki, N.; Matsuzawa, R.; Nozaki, K.; Maekawa, E.; Noda, C.; Yamaoka-Tojo, M.; Matsunaga, A.; Masuda, T.; et al. Utility of SARC-F for Assessing Physical Function in Elderly Patients With Cardiovascular Disease. J. Am. Med. Dir. Assoc. 2017, 18, 176–181. [Google Scholar] [CrossRef]
- Chen, L.K.; Woo, J.; Assantachai, P.; Auyeung, T.W.; Chou, M.Y.; Iijima, K.; Jang, H.C.; Kang, L.; Kim, M.; Kim, S.; et al. Asian Working Group for Sarcopenia: 2019 Consensus Update on Sarcopenia Diagnosis and Treatment. J. Am. Med. Dir. Assoc. 2020, 21, 300–307.e2. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Writing Group for the European Working Group on Sarcopenia in Older People 2 (EWGSOP2), and the Extended Group for EWGSOP2. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef] [Green Version]
- Jackaman, C.; Tomay, F.; Duong, L.; Abdol Razak, N.B.; Pixley, F.J.; Metharom, P.; Nelson, D.J. Aging and cancer: The role of macrophages and neutrophils. Ageing Res. Rev. 2017, 36, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Templeton, A.J.; McNamara, M.G.; Šeruga, B.; Vera-Badillo, F.E.; Aneja, P.; Ocaña, A.; Leibowitz-Amit, R.; Sonpavde, G.; Knox, J.J.; Tran, B.; et al. Prognostic role of neutrophil-to-lymphocyte ratio in solid tumors: A systematic review and meta-analysis. J. Natl. Cancer Inst. 2014, 106, dju124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Terzić, J.; Grivennikov, S.; Karin, E.; Karin, M. Inflammation and colon cancer. Gastroenterology 2010, 138, 2101–2114. [Google Scholar] [CrossRef] [PubMed]
- Lin, E.Y.; Pollard, J.W. Role of infiltrated leucocytes in tumour growth and spread. Br. J. Cancer 2004, 90, 2053–2058. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nishikawa, H.; Goto, M.; Fukunishi, S.; Asai, A.; Nishiguchi, S.; Higuchi, K. Cancer Cachexia: Its Mechanism and Clinical Significance. Int. J. Mol. Sci. 2021, 22, 8491. [Google Scholar] [CrossRef]
- An, X.; Ding, P.R.; Li, Y.H.; Wang, F.H.; Shi, Y.X.; Wang, Z.Q.; He, Y.J.; Xu, R.H.; Jiang, W.Q. Elevated neutrophil to lymphocyte ratio predicts survival in advanced pancreatic cancer. Biomarkers 2010, 15, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Shaul, M.E.; Fridlender, Z.G. Tumour-associated neutrophils in patients with cancer. Nat. Rev. Clin. Oncol. 2019, 16, 601–620. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Chen, M.H.; Li, S.; Guo, Y.L.; Zhu, C.G.; Xu, R.X.; Zhang, Y.; Sun, J.; Qing, P.; Liu, G.; et al. Usefulness of the neutrophil-to-lymphocyte ratio in predicting the severity of coronary artery disease: A Gensini score assessment. Atheroscler. Thromb. 2014, 21, 1271–1282. [Google Scholar] [CrossRef] [Green Version]
- Afsar, B. The relationship between neutrophil lymphocyte ratio with urinary protein and albumin excretion in newly diagnosed patients with type 2 diabetes. Am. J. Med. Sci. 2014, 347, 217–220. [Google Scholar] [CrossRef]
- Bekdas, M.; Goksugur, S.B.; Sarac, E.G.; Erkocoglu, M.; Demircioglu, F. Neutrophil/lymphocyte and C-reactive protein/mean platelet volume ratios in differentiating between viral and bacterial pneumonias and diagnosing early complications in children. Saudi Med. J. 2014, 35, 442–447. [Google Scholar]
- Lagunas-Rangel, F.A. Neutrophil-to-lymphocyte ratio and lymphocyte-to-C-reactive protein ratio in patients with severe coronavirus disease 2019 (COVID-19): A meta-analysis. J Med. Virol. 2020, 92, 1733–1734. [Google Scholar] [CrossRef] [Green Version]
- Ponti, G.; Maccaferri, M.; Ruini, C.; Tomasi, A.; Ozben, T. Biomarkers associated with COVID-19 disease progression. Crit. Rev. Clin. Lab. Sci. 2020, 57, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Li, H.; Zhang, C.; Chen, Z.; Liu, H.; Lei, F.; Qin, J.J.; Liu, Y.M.; Zhou, F.; Song, X.; et al. The Neutrophil-to-Lymphocyte Ratio Determines Clinical Efficacy of Corticosteroid Therapy in Patients with COVID-19. Cell Metab. 2021, 33, 258–269.e3. [Google Scholar] [CrossRef] [PubMed]
- Sayah, W.; Berkane, I.; Guermache, I.; Sabri, M.; Lakhal, F.Z.; Yasmine Rahali, S.; Djidjeli, A.; Lamara Mahammed, L.; Merah, F.; Belaid, B.; et al. Interleukin-6, procalcitonin and neutrophil-to-lymphocyte ratio: Potential immune-inflammatory parameters to identify severe and fatal forms of COVID-19. Cytokine 2021, 141, 155428. [Google Scholar] [CrossRef]
- Forget, P.; Khalifa, C.; Defour, J.P.; Latinne, D.; Van Pel, M.C.; De Kock, M. What is the normal value of the neutrophil-to-lymphocyte ratio? BMC Res. Notes 2017, 10, 12. [Google Scholar] [CrossRef] [Green Version]
- Chandrashekara, S.; Mukhtar Ahmad, M.; Renuka, P.; Anupama, K.R.; Renuka, K. Characterization of neutrophil-to-lymphocyte ratio as a measure of inflammation in rheumatoid arthritis. Int. J. Rheum. Dis. 2017, 20, 1457–1467. [Google Scholar] [CrossRef] [PubMed]

| n or Median (IQR) | |
|---|---|
| Age (years) | 73 (63–79) |
| Gender, male/female | 415/257 |
| ECOG-PS, 0/1/2/3/4 | 436/128/49/41/18 |
| Anatomical disease type | |
| Upper gastrointestinal disease | 161 |
| Lower gastrointestinal disease | 178 |
| Biliary and pancreatic disease | 236 |
| Liver disease | 97 |
| Advanced cancer, yes/no | 162/510 |
| Body mass index (kg/m2) | 22.0 (19.6–24.4) |
| Alanine aminotransferase (IU/L) | 19 (12–32) |
| C reactive protein (mg/dL) | 0.18 (0.06–0.94) |
| eGFR (ml/min/1.73 m2) | 67 (55–81) |
| Serum albumin (g/dL) | 3.8 (3.4–4.2) |
| Hemoglobin (g/dL) | 12.5 (11.1–13.8) |
| Platelet count (×104/μL) | 22.0 (16.7–27.9) |
| White blood cell (/μL) | 5910 (4768–7693) |
| Neutrophil count (/μL) | 3706 (2809–5408) |
| Total lymphocyte count (/μL) | 1397 (1069–1842) |
| Neutrophil to lymphocyte ratio | 2.65 (1.79–4.31) |
| SARC-F score | 0 (0–2) |
| Grip strength (male, kg) | 28.9 (23.6–34.2) |
| Grip strength (female, kg) | 17.0 (13.2–20.4) |
| r | p Value | |
|---|---|---|
| Age | 0.25 | <0.0001 |
| BMI | −0.03 | 0.3967 |
| ECOG-PS | 0.79 | <0.0001 |
| ALT | −0.009 | 0.8207 |
| Hemoglobin | −0.27 | <0.0001 |
| Platelet count | 0.03 | 0.4707 |
| Serum albumin | −0.30 | <0.0001 |
| NLR | 0.54 | <0.0001 |
| CRP | 0.21 | <0.0001 |
| eGFR | −0.10 | 0.0074 |
| Estimates | Standard Error | p Value | |
|---|---|---|---|
| Age | 0.0066012 | 0.003619 | 0.0686 |
| ECOG-PS | 1.2992242 | 0.050599 | <0.0001 |
| Hemoglobin | −0.017435 | 0.025668 | 0.4972 |
| Serum albumin | 0.042363 | 0.095874 | 0.6587 |
| NLR | 0.1257596 | 0.016222 | <0.0001 |
| CRP | −0.009757 | 0.012562 | 0.4376 |
| eGFR | −0.001313 | 0.002261 | 0.5615 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamasaki, E.; Nishikawa, H.; Goto, M.; Matsui, M.; Asai, A.; Ushiro, K.; Ogura, T.; Takeuchi, T.; Nakamura, S.; Kakimoto, K.; et al. The Relevance in the Neutrophil to Lymphocyte Ratio and the SARC-F Score in Gastrointestinal Diseases. J. Clin. Med. 2022, 11, 2012. https://doi.org/10.3390/jcm11072012
Yamasaki E, Nishikawa H, Goto M, Matsui M, Asai A, Ushiro K, Ogura T, Takeuchi T, Nakamura S, Kakimoto K, et al. The Relevance in the Neutrophil to Lymphocyte Ratio and the SARC-F Score in Gastrointestinal Diseases. Journal of Clinical Medicine. 2022; 11(7):2012. https://doi.org/10.3390/jcm11072012
Chicago/Turabian StyleYamasaki, Eiki, Hiroki Nishikawa, Masahiro Goto, Masahiro Matsui, Akira Asai, Kosuke Ushiro, Takeshi Ogura, Toshihisa Takeuchi, Shiro Nakamura, Kazuki Kakimoto, and et al. 2022. "The Relevance in the Neutrophil to Lymphocyte Ratio and the SARC-F Score in Gastrointestinal Diseases" Journal of Clinical Medicine 11, no. 7: 2012. https://doi.org/10.3390/jcm11072012
APA StyleYamasaki, E., Nishikawa, H., Goto, M., Matsui, M., Asai, A., Ushiro, K., Ogura, T., Takeuchi, T., Nakamura, S., Kakimoto, K., Miyazaki, T., Fukunishi, S., Ohama, H., Yokohama, K., Yasuoka, H., & Higuchi, K. (2022). The Relevance in the Neutrophil to Lymphocyte Ratio and the SARC-F Score in Gastrointestinal Diseases. Journal of Clinical Medicine, 11(7), 2012. https://doi.org/10.3390/jcm11072012





