Abstract
Diverticular disease (DD) is a common gastrointestinal condition. Patients with DD experience a huge variety of chronic nonspecific symptoms, including abdominal pain, bloating, and altered bowel habits. They are also at risk of complications such as acute diverticulitis, abscess formation, hemorrhage, and perforation. Intestinal dysbiosis and chronic inflammation have recently been recognized as potential key factors contributing to disease progression. Probiotics, due to their ability to modify colonic microbiota balance and to their immunomodulatory effects, could present a promising treatment option for patients with DD. Lactobacillus paracasei CNCM I 1572 (LCDG) is a probiotic strain with the capacity to rebalance gut microbiota and to decrease intestinal inflammation. This review summarizes the available clinical data on the use of LCDG in subjects with colonic DD.
1. Introduction
Diverticular disease (DD) is a spectrum of gastrointestinal conditions characterized by the presence of diverticula, defined as small, balloon-like sacs protruding through the layers of the colon [1]. DD is the fifth most important gastrointestinal disease in terms of healthcare costs in Western countries, with the highest rates occurring in the United States and Europe with 60–70% prevalence rates in those older than 60 [1,2]. The presence of one or more diverticula in the colon is called diverticulosis [2,3]. Diverticulosis is a common condition that generally occurs during middle age and remains asymptomatic [2,3]. Its finding is incidental [4]. Approximately 20% of patients with DD develop symptoms, including abdominal pain, bloating, and altered bowel habits, the condition is called symptomatic uncomplicated diverticular disease (SUDD) [1,2]. The major clinical complication, occurring in about 4% of individuals, is acute diverticulitis, that could be uncomplicated or complicated by abscess formation, haemorrhage, and perforation [1,5,6]. Moreover, due to persistent mucosal inflammation, patients with acute diverticulitis may be susceptible to the consecutive development of SUDD [7]. DD appear to share similar pathophysiological mechanisms with irritable bowel syndrome (IBS) [8,9]. In DD, alteration in bacterial microbiota occurs primarily because of faecal material stasis which predisposes to diverticular bacterial overgrowth [10,11,12,13]. This leads to impairment of the mucosal barrier function and up-regulates inflammatory cytokine release with low-grade microscopic inflammation; this inflammation has the potential to progress to microperforation and, ultimately, to acute diverticulitis [10,11,12,13]. In addition, dysbiosis and mucosal inflammation are associated with dysmotility; they alter nerve fibre activation leading to subsequent neuronal and muscular dysfunction, thus favouring the development of abdominal symptoms [14,15].
Given these observations, probiotics may be an appealing treatment option for this condition, due to their ability to modify colonic microbiota balance and to their immunomodulatory effects [16,17,18,19]. Although probiotics have already been proposed for use in inflammatory, infectious, neoplastic, and allergic disorders, the ideal probiotic strain for use in any of these indications has yet to be identified [20,21,22,23,24]. The interpretation of available data on probiotics is further confounded by variability in strain selection, dose, delivery vehicle, and evaluation of viability and efficacy. Lacticaseibacillus paracasei (formerly Lactibacillus paracasei) CNCM I1572 (LCDG; L. casei DG®; Enterolactis®, Sofar S.p.A., Trezzano Rosa, Milan, Italy, deposited at Institute Pasteur of Paris with number I1572) is a probiotic strain with the capacity to rebalance gut microbiota and to decrease intestinal inflammation. This review aims to summarize the available evidence on the use of L. casei DG® (Lactobacillus paracasei CNCM I 1572—LCDG; Enterolactis®) in the management of DD.
2. Probiotics and Their Mechanisms of Actions
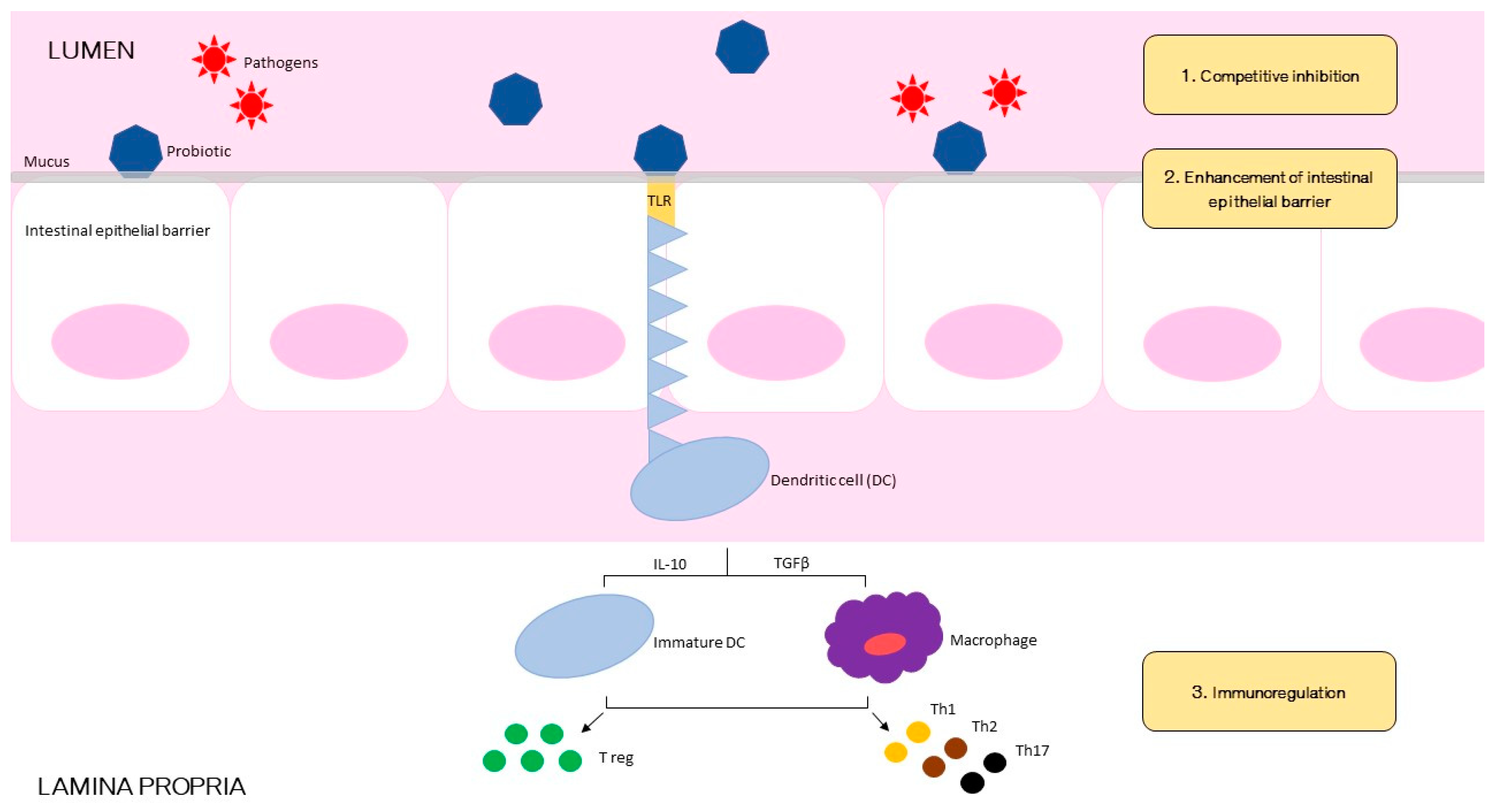
Probiotics are living organisms that are originally found in the intestine. They can also be synthesized in laboratories and therefore be available in commercial products [25,26,27]. Probiotics are defined as live microorganisms that, when administered in adequate amounts, confer a health benefit on the host [27]. The minimum quantity to obtain a temporary colonization of the intestine is generally at least 1 billion live cells per day [28]. Probiotics must also be resistant to gastric acid and bile to survive through the gastrointestinal tract [29,30,31]. Once present in the colon, probiotics must adhere to the colon’s epithelial cells to ensure adequate colonization [32]. Probiotic organisms have the ability to produce antimicrobial substances or antagonize pathogenic bacteria in the gut [33,34]. Finally, commercially manufactured probiotics must be stable for storage before being ingested and must be safe for use in humans’ large amounts [35,36]. The mechanism of action of probiotics is likely to be multifactorial. To simplify, we can resume 3 basic functional principles by which probiotics may confer health benefits: restoration of intestinal microbiota; regulation of immune function; and enhancement of barrier function of gut epithelium (Figure 1) [37,38]. Through competitive inhibition probiotics hinder the ability of pathogenic Gram-negative bacteria to adhere and colonize the intestinal mucosa [39,40,41]. Some probiotic strains secrete proteases that help to degrade toxins [42,43]. Also, by fermenting dietary fibers, probiotics can produce SCFAs (short-chain fatty acids), such as butyrate, propionate, and acetate, with protective properties against intestinal pathogens [44]. Moreover, probiotics have been associated to decreased secretion of inflammatory cytokines such as tumor necrosis factor-α, interferon-γ, and interleukin-1, and increased production of the anti-inflammatory cytokines such as interferon-α and interleukin-10 [45,46,47]. Some species displayed the capacity to stimulate immunoglobulin A secretion in Peyer patches [48]. Finally, probiotics exert a direct effect on the intestinal epithelial barrier function: by binding with toll-like receptors on the apical surface of the epithelial cells [49], they activate protein kinase C within the cell, which results in clenching of the tight junctions between epithelial cells and improving the barrier function of the gut mucosa, thus potentially limiting bacterial translocation [50,51,52].

Figure 1.
Schematic illustration of postulated mechanisms of probiotic bacterial actions against gastrointestinal pathogenic infection.
3. Lactobacillus paracasei CNCM I 1572
LCDG is a Gram-positive bacterial strain isolated from human faeces and normally present in healthy individuals’ intestinal microbiota. It survives the gastrointestinal transit in healthy children and adults when ingested with the probiotic drinkable formulation containing no less than 1 × 109 CFU, demonstrating resistance to digestive juices, hydrolytic enzymes, and bile acids [53,54,55]. LCDG’s human origin guarantees a long-lasting intestinal colonization, persisting in the gut of patients up to one week after the end of probiotic consumption [53,54]. LCDG produces lactic acid, providing a quick rebalancing action of faecal microbiota [48]. Moreover, LCDG does not induce antibiotics resistance, guaranteeing safe human consumption [54]. Consistently with these peculiarities, several in vitro/in vivo experiments demonstrated that the polysaccharides present on the surface of the bacteria, referred either as capsule or as exopolysaccharides (EPSs) can play a role both in the modulation of the intestinal microbial ecosystem and in the stimulation of the host’s immune responses; protecting LCDG, these polysaccharides also allow the probiotic strain to reach the intestine alive [48,56,57,58,59,60,61]. Finally, a genomic analysis of LCDG revealed that this strain produces a unique rhamnose-rich hetero-exopolysaccharide, named DG-EPS, with the ability to stimulate the production of proinflammatory cytokines by antigen-presenting cells (APCs) responsible for the detection of microorganisms and involved in their clearance through phagocytosis [57]. Acting as mild booster of the innate immunity, LCDG may contribute to a more efficient and faster immune response against potential infectious agents [61,62,63].
3.1. Lactobacillus paracasei CNCM I 1572 in Human Health
The efficacy of LCDG has been investigated in several clinical settings, including healthy humans or patients with urological diseases such as chronic bacterial prostatitis and gastrointestinal disorders such as IBS, small intestinal bacterial overgrowth (SIBO), and DD [48,58,59,60,64,65,66,67,68]. To determine the impact of LCDG on the intestinal microbial ecology of healthy patients, Ferrario et al. [48] conducted a randomized, double-blind, placebo-controlled crossover trial on 34 healthy human volunteers’ faecal microbiota. Participants were randomly assigned to 2 parallel groups receiving, once a day for 4 weeks, in addition to their habitual diet, either placebo or a probiotic capsule containing at least 24 billion viable cells of LCDG. Each volunteers’ faecal microbiota was evaluated before and after LCDGs’ consumption. Despite inter-individual variability in intestinal microbiota, this probiotic strain has been shown to positively modulate microbiota of healthy human, increasing the percentage of bacteria that—according to the literature—can potentially confer a health benefit to the host [69,70,71,72,73,74,75]. In fact, LCDG intake induced an increase in the gram-negative phylum Proteobacteria (p = 0.006), which is the most abundant phyla in the human gut microbiota, and in the gram-positive Clostridiales genus Coprococcus (p = 0.009), which play a crucial role in folate biosynthesis and in the colonic fermentation of dietary fiber leading to short chain fatty acids (SCFAs) production [48]. SCFAs (i.e., acetate, butyrate, and propionate) are crucial in preserving gut equilibrium, an increased level of SCFAs leads to the enhancement of barrier function of intestinal epithelium [76]. Butyrate in particular, is linked with a number of beneficial activities on the intestinal mucosa; drastic increase or reduction in its concentration is typical for several pathologies such as IBS and metabolic syndrome [77,78,79,80,81,82,83]. So, modifying the concentration of bacterial groups able to produce SCFAs in the gut, this probiotic strain can “rebalance” SCFAs and butyrate concentrations. After LCDG intervention, participants with butyrate > 100 mmol/kg of wet feces had a mean butyrate reduction of 49 ± 21%. In contrast, in participants with initial butyrate concentrations < 25 mmol/kg of wet feces, the probiotic contributed to a 329 ± 255% (p > 0.05) increment in butyrate. Finally, a declining trend was observed in genus Ruminococcus (p = 0.016) known for its role in the etiopathogenesis of IBS [73,84,85]. LCDG seems to work in the direction of a potential protective and healthy microbiota, rebalancing gut physiological conditions. Additional studies were then conducted to investigate its application in different colonic diseases. Rosania et al. [65] evaluated the effects of addition of LCDG to antibiotics in patients with SIBO during a 6-month follow-up. Twenty patients reporting abdominal compliant without gastrointestinal diseases/alarm symptoms were enrolled. SIBO was diagnosed by the agreement of lactulose and glucose breath tests. Patients received rifaximin 400 mg/day for 7 days/month followed by LCDG for 7 days more. All patients recorded a questionnaire for subjective symptom evaluation according to Rome III criteria and Bristol scale for stool characteristics before the study and after 6 months. A significant improvement was obtained in 5 (diffuse abdominal pain p < 0.001; pain in the left iliac area p < 0.002; meteorism p < 0.002; flatulence p < 0.001; nausea p < 0.01) out of 6 symptoms. The analysis for each single patient also showed an improvement in the number of bowel movements and stool characters in 16 out 20 patients (80%). Besides, in a recent multicenter randomized study, Cremon et al. [58] evaluated the effects of LCDG on gut microbiota-related factors in IBS patients’ faecal samples. The participants were randomly assigned to different groups: in one group they had to take LCDG two times a day for four weeks, in the other group they had to take the equivalent product without bacteria (placebo), this phase was followed by a washout period of four more weeks before crossing over to the alternate treatment (twice daily for four weeks). After 14 weeks, patients entered a four-week follow-up phase. In all cases, faecal samples were obtained before and after each treatment and follow-up period. The intestinal microbial ecosystem was then characterized. In IBS patients, at baseline, members of the gut microbiota attributed to the genus Ruminococcus were increased. Concentration of the proinflammatory cytokine IL-15 was also enhanced, whereas SCFAs levels were decreased. Interestingly, LCDG induced a significant trend of reduction in Ruminococcus (p = 0.042) and in IL-15 (mean change −173.4; p = 0.042). In contrast, faecal short chain fatty acids acetate (p = 0.021) and butyrate (p = 0.047) were increased, confirming once again the potential role of the probiotic strain in such disorders. Finally, to better understand the molecular mechanisms of action of LCDG, Compare et al. [59], by using human intestinal biopsy specimens in culture, analyzed the effect of the Lactobacillus on ileal and colonic mucosa of 10 post-infectious irritable bowel syndrome (PI-IBS) patients. At baseline, IL-1α, IL-6, IL-8 mRNA levels, and TLR-4 proteins expression were higher while anti-inflammatory IL-10 mRNA levels were lower in PI-IBS patients than in healthy controls. Treatment of colonic biopsies with LCDG significantly reduced the levels of all proinflammatory cytokines (Il-1 α p < 0.002, IL-6 p < 0.0001 and IL-8 p < 0.0001) in respect to baseline. In ileal mucosa, LCDG treatment was effective in reducing IL-1α and IL-8 mRNA levels (p < 0.0002 and p < 0.0001, respectively) but did not affect IL-6 levels. In contrast, IL-10 m-RNA levels significantly increased in both ileal and colonic mucosa (p < 0.0001 and p < 0.0001, respectively). Finally, the increase of TLR-4 protein expression was attenuated by LCDG (p < 0.0001).
3.2. Lactobacillus paracasei CNCM I 1572 and Diverticular Disease
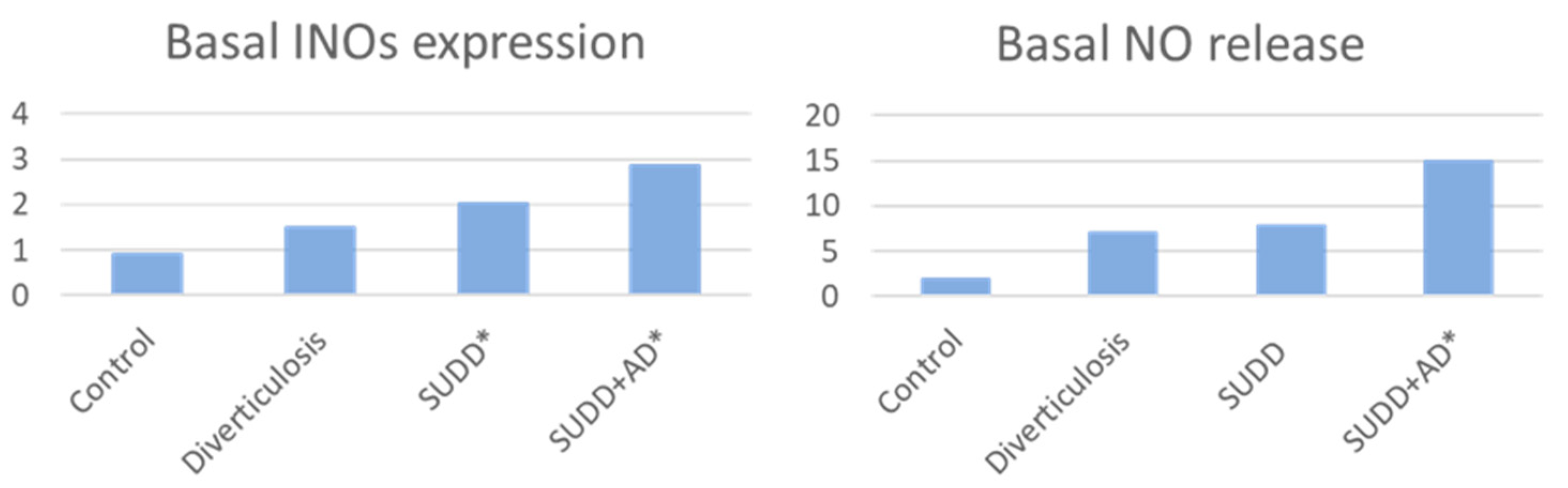
Some new data on the role of LCDG in the management of diverticular disease have emerged in the last years. Anti-inflammatory action of LCDG on patients with DD was investigated in an in vitro study conducted by Turco et al. [60] Intestinal biopsies were collected during endoscopy in 40 consecutive individuals, divided as follow: 10 patients with diverticulosis, 10 patients with SUDD, 10 patients with SUDD with previous acute diverticulitis (SUDD+AD), and a control group of 10 people without gastrointestinal diseases. Biopsies were then stimulated with the probiotic LCDG and/or the pathogen enteroinvasive Escherichia coli (EIEC). As previous studies demonstrated an increase in nitric oxide (NO)-mediated responses in patients with DD; the rationale was to evaluate NO release and inducible nitric oxide synthase (iNOS) expression before and after biopsies stimulation [86,87,88,89]. Basal iNOS expression was significantly increased in SUDD and SUDD+AD patients (+2.04- and +2.86-fold increase vs. CTRLs, respectively; p < 0.05). Basal NO expression was significantly increased in SUDD+AD (+7.77-fold increase vs. CTRLs; p < 0.05) (Figure 2).
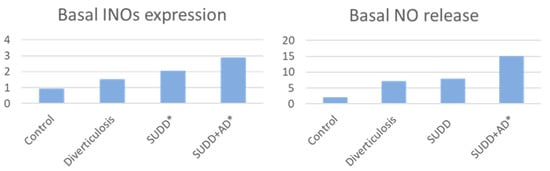
Figure 2.
Basal INOs expression and NO release in patients vs. controls * p < 0.05 vs. controls INOs = inducible nitric oxide synthase; NO = nitric oxide; SUDD = symptomatic uncomplicated diverticular disease; AD = acute diverticulitis.
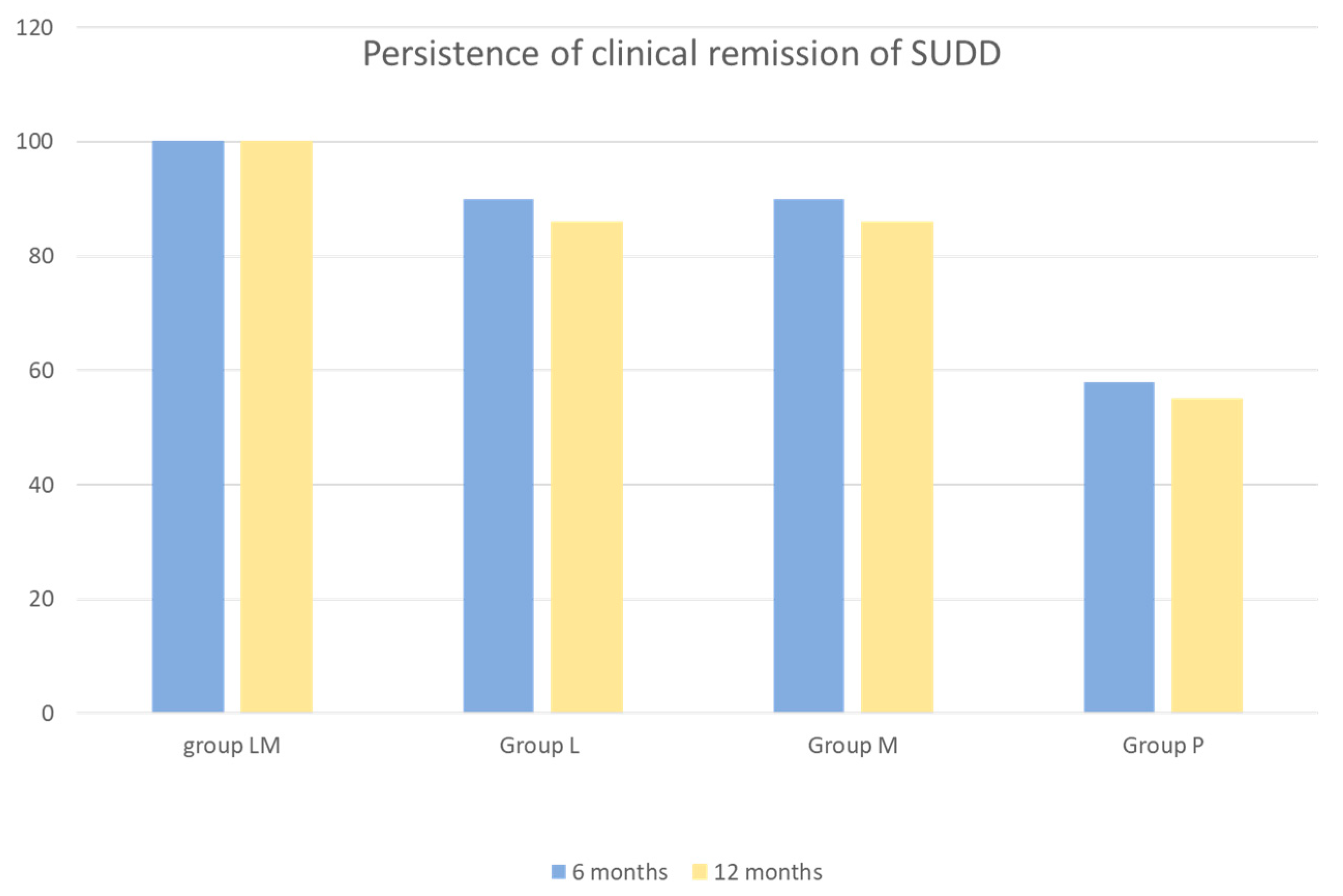
In all groups, iNOS expression was significantly increased by EIEC and reduced by LCDG (p < 0.05 and p < 0.05, respectively). In all groups, except for SUDD+AD, EIEC significantly increased NO release and LCDG significantly reduced NO release (p < 0.05 and p < 0.05 respectively). Data confirmed an activation of NO-dependent inflammation related to iNOS expression and NO release that appeared progressively increased from diverticulosis to SUDD with previous diverticulitis. At baseline, a significantly increased release of the anti-inflammatory cytokine IL-10 in patients with SUDD+AD (−11.25+ fold increase vs. controls was also observed; p < 0.05), clear evidence of the body’s attempt to control inflammation after acute diverticulitis. Finally, this study demonstrated that colonic mucosa of patients with DD is characterized by a different reactivity towards pathogenic stimuli. LCDGs’ role in counteracting the pro-inflammatory effects exerted by EIEC was confirmed, suggesting a beneficial role of this probiotic in DD. Three in vivo studies (Table 1) have then investigated the efficacy of LCDG administered in combination with mesalazine (5-ASA), which is commonly used in the treatment of inflammatory bowel disease. The rationale for this approach was to control peri-diverticular inflammation while simultaneously restore local microbiota [90,91,92]. Tursi et al. [66] conducted a multicenter, prospective, randomized controlled study comparing 5-ASA, LCDG, and their combination in 90 patients with recurrent SUDD. Subjects periodically self-rated their symptoms using a tool that generated an “overall symptom score”, including constipation, diarrhea, abdominal pain, rectal bleeding, and mucus with the stools, that could range from 0 to 50. After 12 months, 76.7% of subjects (23/30) in each monotherapy arm were symptom-free, compared with 96.7% of those on combination therapy (29/30; one subject lost to follow up). In general, when symptoms occurred, they were rated as mild, but the overall symptom score was lower in the group receiving the combination of 5-ASA plus LCDG compared with the other two groups (p < 0.001). A related randomized study was reported by Tursi et al. [67] in 75 subjects with additional study arms to assess 5-ASA doses of 0.8 g versus 1.6 g daily; follow up was extended to 24 months. In the present case, 80% and 87% of patients on monotherapy remained symptom-free versus 92% and 94% of those on combination therapy, the differences were not statistically significant. No 5-ASA dose-response was reported. Of note, all subjects who stopped treatment developed symptoms or a diverticular complication. Finally, Tursi et al. [68] conducted another randomized, controlled double-blind, double-dummy trial to evaluate the effectiveness of 5-ASA and/or LCDG in maintaining remission in SUDD. The study was conducted on 210 patients with SUDD. Participants were randomly enrolled in four groups: Group M (active 5-ASA 1.6 g/day plus LCDG placebo), Group L (LCDG 24 billion/day plus 5-ASA placebo), Group LM (active LCDG 24 billion/day plus active 5-ASA 1.6 g/day), Group P (LCDG placebo plus 5-ASA placebo). Patients received treatment during 12 months for 10 days/month. Recurrence of SUDD was defined as the reoccurrence, for at least 24 consecutive hours, of abdominal pain during follow-up, scored as ≥5 (0: best; 10: worst). Reappearance of SUDD happened in no (0%) patient in group LM, in 7 (13.7%) patients in group M, in 8 (14.5%) patients in group L, and in 23 (46.0%) patients in group P (LM group vs. M group, p = 0.015; LM group vs. L group, p = 0.011; LM group vs. P group, p = 0.000; M group vs. P group, p = 0.000; L group vs. P group, p = 0.000). Compared with all other groups, in group P there was a significant number of recurrences (p = 0.003). Both cyclic 5-ASA and LCDG emerged to be better than placebo for maintaining remission in SUDD, particularly when used together (Figure 3). Moreover, both treatments, alone or in combination, were significantly better than placebo in preventing occurrence of acute diverticulitis in SUDD patients. No adverse effects related to the probiotic treatment were observed in the overall studies [66,67,68].

Table 1.
Main characteristics of the 3 in vivo selected studies on LCDG treatment in DD.
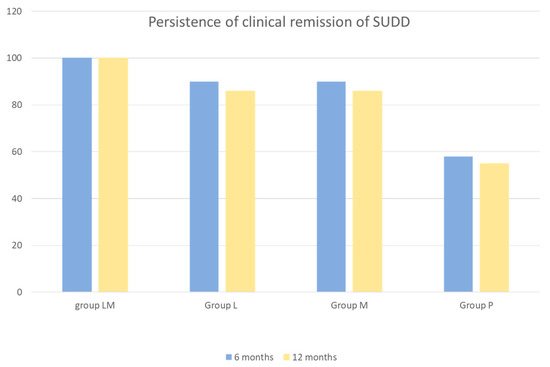
Figure 3.
Persistence of clinical remission of SUDD at 6 and 12 months by study group. Absence of recurring abdominal pain (scored ≥ 5 for at least 24 consecutive hours and recorded at any time during the follow up) was defined as clinical remission. SUDD = symptomatic uncomplicated diverticular disease; group LM = active mesalazine + active Lactobacillus casei; group L = active Lactobacillus casei + mesalazine placebo; group M = active mesalazine + Lactobacillus casei placebo; group P = mesalazine placebo + Lactobacillus casei placebo.
4. Discussion
Dysbiosis has been described in patients with symptomatic diverticular disease. Thus, changing gut bacterial composition and switching off inflammatory patterns through probiotics seems an interesting therapeutic strategy, breaking the vicious circle in which dysbiosis and inflammation promote each other. These observations have encouraged to investigate the potential role of LCDG as a new therapy for DD [93,94,95,96].
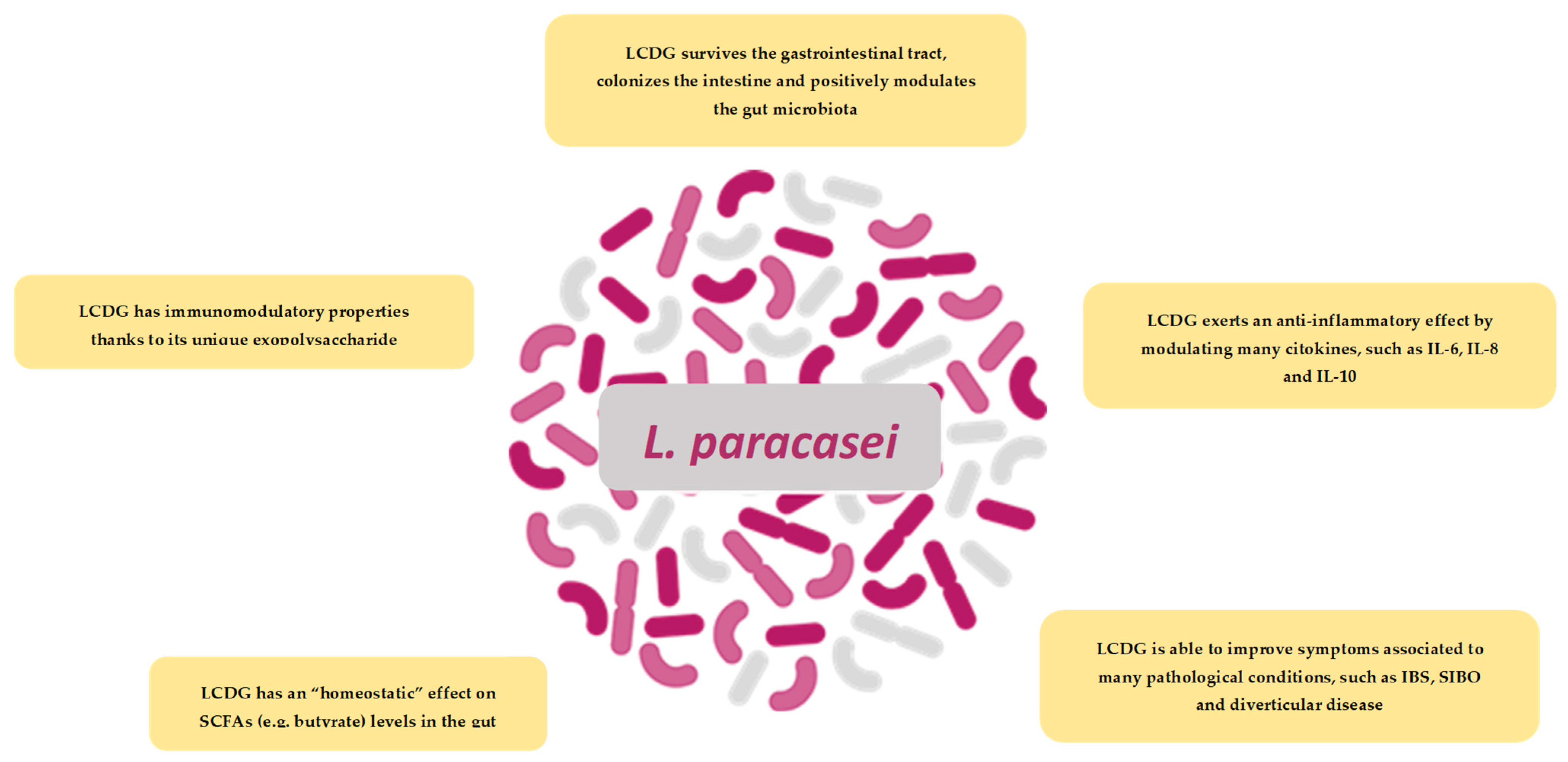
LCDG has multiple modes of action (Figure 4), including rebalancing of the intestinal microbiotas’ ecology and regulation of the immune system activity. This probiotic strain has a clear impact on faecal microbiota, modifying specific microbial groups at the phylum and genus levels, inhibiting colonic bacterial overgrowth and metabolism of pathogens, and increasing levels of SCFAs playing an important role in maintaining intestinal homeostasis [48,58]. Moreover, data coming from in vitro and in vivo studies, demonstrated LCDGs’ capacity to regulate the immune system activity in IBS and DD, by controlling pro- and anti-inflammatory cytokines levels [58,59,60]. Acting on inflammation, LCDG may also act on symptom development in individuals affected by such intestinal diseases [58,65].
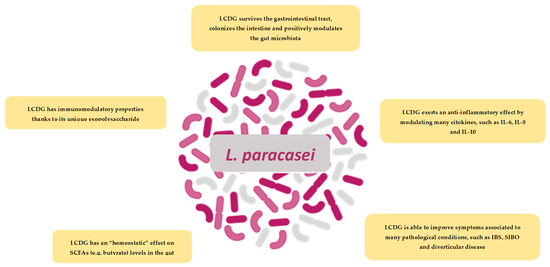
Figure 4.
Schematic illustration of the most important finding on LCDG.
In three clinical trials, this probiotic strain seemed to show an apparent trend to significantly obtain remission and reduction of the recurrence of SUDD, especially when used in combination with 5-ASA [66,67,68] (Table 1). However, the poor number of overall studies, the heterogeneous nature, and the relatively poor quality of the available studies on the use of LCDG make it difficult to evaluate the cumulative efficacy of this probiotic strain. Treatment protocols about timing, dosage, or combination with other drugs and the follow-up periods in the different studies were very variable. Only 1 study was a double-blinded randomized controlled trial. Furthermore, the type of DD was heterogeneous between the mentioned studies because there is a lack of a globally recognized clinical classification for diagnosing and defining SUDD and its recurrence.
Two main considerations can be extrapolated from this work. First, even if the amount of data present is not sufficient to draw robust conclusions, LCDG appears a promising option to promote the health of the individual, equilibrating gut physiological conditions in colonic pathologies such as SIBO, IBS, and DD. Especially in the latter pathology, the efficacy and safety of LCDG in improving symptoms of SUDD have already been mentioned in the WGO Global Guidelines of the use of Probiotics and Prebiotics (2017) [97]. It could also be interesting to evaluate its use in inflammatory bowel disease (IBD) and microscopic colitis, two more multifactorial gastrointestinal diseases in which we find the same pathogenetic pattern of chronic inflammation and protracted dysbiosis. Secondly, data emerging from this review suggest that large, randomized placebo-controlled studies are needed to establish efficacy, dose-responses, optimal timing for introduction, and duration of LCDG therapy in the spectrum of colonic diverticular disease.
5. Conclusions
Lactibacillus paracasei is a promising candidate in the management of diverticular disease, ensuring multiple benefic effects on the intestinal homeostasis.
Specific studies to evaluate LCDGs’ efficacy in each clinical setting of DD are warranted, including treatment of acute diverticulitis and SUDD, prevention of recurrent diverticulitis, and management of chronic symptoms.
Author Contributions
E.B. wrote the article and created tables and figures. F.D., W.F., A.T. and S.D. critically reviewed the content of the paper and supervised the project. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Conflicts of Interest
E.B. declares no conflict of interest. F.D. has served has a speaker and declares no conflict of interest. W.F. has served as a speaker, is an employee of Sofar S.p.A. A.T. has served has a speaker and declares no conflict of interest. S.D. has served as a speaker, consultant, and advisory board member for Schering-Plough, AbbVie, Actelion, Alphawasserman, AstraZeneca, Cellerix, Cosmo Pharmaceuticals, Ferring, Genentech, Grunenthal, Johnson and Johnson, Millenium Takeda, MSD, Nikkiso Europe GmbH, Novo Nordisk, Nycomed, Pfizer, Pharmacosmos, UCB Pharma, and Vifor.
References
- Talutis, S.D.; Kuhnen, F.A.H. Pathophysiology and Epidemiology of Diverticular Disease. Clin. Colon Rectal Surg. 2021, 34, 081–085. [Google Scholar] [CrossRef] [PubMed]
- Strate, L.L.; Morris, A.M. Epidemiology, Pathophysiology, and Treatment of Diverticulitis. Gastroenterology 2019, 156, 1282–1298.e1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McSweeney, W.; Srinath, H. Diverticular disease practice points. Aust. Fam. Physician 2017, 46, 829–832. [Google Scholar] [PubMed]
- Rezapour, M.; Ali, S.; Stollman, N. Diverticular Disease: An Update on Pathogenesis and Management. Gut Liver 2018, 12, 125–132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.L.; Teng, G.G.; Wu, T.; Chen, G.W.; Wang, P.Y.; Jiang, Y.; Wu, Y.C.; Sun, L.; Liu, T.; Zuo, S.; et al. Clinical analysis of 554 patients with colorectal diverticulosis. Zhonghua Wei Chang Wai Ke Za Zhi Chin. J. Gastrointest. Surg. 2021, 24, 1008–1014. [Google Scholar]
- Swanson, S.M.; Strate, L.L. Acute Colonic Diverticulitis. Ann. Intern. Med. 2018, 168, ITC65–ITC80. [Google Scholar] [CrossRef]
- Lahat, A.; Necula, D.; Yavzori, M.; Picard, O.; Halperin, S.; Eliakim, R.; Ben-Horin, S. Prolonged Recurrent Abdominal Pain is Associated with Ongoing Underlying Mucosal Inflammation in Patients who had an Episode of Acute Complicated Diverticulitis. J. Clin. Gastroenterol. 2019, 53, e178–e185. [Google Scholar] [CrossRef] [PubMed]
- Spiller, R. Editorial: New Thoughts on the Association Between Diverticulosis and Irritable Bowel Syndrome. Am. J. Gastroenterol. 2014, 109, 1906–1908. [Google Scholar] [CrossRef]
- Alamo, R.Z.; Quigley, E.M.M. Irritable bowel syndrome and colonic diverticular disease: Overlapping symptoms and overlapping therapeutic approaches. Curr. Opin. Gastroenterol. 2019, 35, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Piscopo, N.; Ellul, P. Diverticular Disease: A Review on Pathophysiology and Recent Evidence. Ulst. Med. J. 2020, 89, 83–88. [Google Scholar]
- Tursi, A.; Scarpignato, C.; Strate, L.L.; Lanas, A.; Kruis, W.; Lahat, A.; Danese, S. Colonic diverticular disease. Nat. Rev. Dis. Primers 2020, 6, 20. [Google Scholar] [CrossRef] [PubMed]
- Severi, C.; Carabotti, M.; Cicenia, A.; Pallotta, L.; Annibale, B. Recent advances in understanding and managing diverticulitis. F1000Research 2018, 7, 971. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreno, A.M.; Wille-Jørgensen, P. Long-term outcome in 445 patients after diagnosis of diverticular disease. Colorectal Dis. 2007, 9, 464–468. [Google Scholar] [CrossRef] [PubMed]
- Quigley, E.M.M. Gut microbiota, inflammation and symptomatic diverticular disease. New insights into an old and neglected disorder. J. Gastrointest. Liver Dis. 2010, 19, 127–129. [Google Scholar]
- Humes, D.J.; Simpson, J.; Smith, J.; Sutton, P.; Zaitoun, A.; Bush, D.; Bennett, A.; Scholefield, J.H.; Spiller, R.C. Visceral hypersensitivity in symptomatic diverticular disease and the role of neuropeptides and low grade inflammation. Neurogastroenterol. Motil. Off. J. Eur. Gastrointest. Motil. Soc. 2012, 24, 318-e163. [Google Scholar] [CrossRef] [PubMed]
- Langenfeld, S.J. Evaluation and Medical Management of Uncomplicated Diverticulitis. Clin. Colon Rectal Surg. 2021, 34, 086–090. [Google Scholar] [CrossRef] [PubMed]
- Ticinesi, A.; Nouvenne, A.; Corrente, V.; Tana, C.; Di Mario, F.; Meschi, T. Diverticular Disease: A Gut Microbiota Perspective. J. Gastrointest. Liver Dis. 2019, 28, 327–337. [Google Scholar] [CrossRef]
- Ojetti, V.; Petruzziello, C.; Cardone, S.; Saviano, L.; Migneco, A.; Santarelli, L.; Gabrielli, M.; Zaccaria, R.; Lopetuso, L.; Covino, M.; et al. The Use of Probiotics in Different Phases of Diverticular Disease. Rev. Recent Clin. Trials 2018, 13, 89–96. [Google Scholar] [CrossRef]
- Piccioni, A.; Franza, L.; Brigida, M.; Zanza, C.; Torelli, E.; Petrucci, M.; Nicolò, R.; Covino, M.; Candelli, M.; Saviano, A.; et al. Gut Microbiota and Acute Diverticulitis: Role of Probiotics in Management of This Delicate Pathophysiological Balance. J. Pers. Med. 2021, 11, 298. [Google Scholar] [CrossRef]
- Shahrokhi, M.; Nagalli, S. Probiotics. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. Available online: http://www.ncbi.nlm.nih.gov/books/NBK553134/ (accessed on 7 January 2022).
- Nadal, I.; Sanchez, E. Probiotics as Drugs Against Human Gastrointestinal Infections. Recent Patents Anti-Infect. Drug Discov. 2007, 2, 148–156. [Google Scholar] [CrossRef]
- Butler, C.C.; Owen-Jones, E.; Lau, M.; Gillespie, D.; Lown, M.; Calder, P.C.; Stanton, H.; Wootton, M.; Herrera, V.C.; Bayer, A.; et al. Probiotics to Reduce Antibiotic Administration in Care Home Residents Aged 65 Years and Older: The PRINCESS RCT; NIHR Journals Library: Southampton, UK, 2021. Available online: http://www.ncbi.nlm.nih.gov/books/NBK570117/ (accessed on 7 January 2022).
- Daliri, E.B.-M.; Ofosu, F.K.; Xiuqin, C.; Chelliah, R.; Oh, D.-H. Probiotic Effector Compounds: Current Knowledge and Future Perspectives. Front. Microbiol. 2021, 12, 655705. [Google Scholar] [CrossRef] [PubMed]
- Badgeley, A.; Anwar, H.; Modi, K.; Murphy, P.; Lakshmikuttyamma, A. Effect of probiotics and gut microbiota on anti-cancer drugs: Mechanistic perspectives. Biochim. Biophys. Acta 2021, 1875, 188494. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.E. Probiotics: Definition, Sources, Selection, and Uses. Clin. Infect. Dis. 2008, 46 (Suppl. 2), S58–S61. [Google Scholar] [CrossRef] [Green Version]
- Reid, G. Probiotics: Definition, scope and mechanisms of action. Best Pract. Res. Clin. Gastroenterol. 2016, 30, 17–25. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weitzel, M.L.J.; Vegge, C.S.; Pane, M.; Goldman, V.S.; Koshy, B.; Porsby, C.H.; Burguière, P.; Schoeni, J.L. Improving and Comparing Probiotic Plate Count Methods by Analytical Procedure Lifecycle Management. Front. Microbiol. 2021, 12, 693066. [Google Scholar] [CrossRef] [PubMed]
- Morelli, L.; Pellegrino, P. A critical evaluation of the factors affecting the survival and persistence of beneficial bacteria in healthy adults. Benef. Microbes 2021, 12, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, R.; Liaqat, A.; Chughtai, M.F.J.; Tanweer, S.; Tehseen, S.; Ahsan, S.; Nadeem, M.; Mehmood, T.; Rehman, S.J.U.; Saeed, K.; et al. Microencapsulation: A pragmatic approach towards delivery of probiotics in gut. J. Microencapsul. 2021, 38, 437–458. [Google Scholar] [CrossRef] [PubMed]
- Centurion, F.; Basit, A.W.; Liu, J.; Gaisford, S.; Rahim, A.; Kalantar-Zadeh, K. Nanoencapsulation for Probiotic Delivery. ACS Nano 2021, 15, 18653–18660. [Google Scholar] [CrossRef] [PubMed]
- Forssten, S.; Ouwehand, A.C. Dose-Response Recovery of Probiotic Strains in Simulated Gastro-Intestinal Passage. Microorganisms 2020, 8, 112. [Google Scholar] [CrossRef] [Green Version]
- Varankovich, N.; Nickerson, M.T.; Korber, D.R. Probiotic-based strategies for therapeutic and prophylactic use against multiple gastrointestinal diseases. Front. Microbiol. 2015, 6, 685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mishra, C.; Lambert, J. Production of anti-microbial substances by probiotics. Asia Pac. J. Clin. Nutr. 1996, 5, 20–24. [Google Scholar] [PubMed]
- Toscano, M.; De Grandi, R.; Pastorelli, L.; Vecchi, M.; Drago, L. A consumer’s guide for probiotics: 10 golden rules for a correct use. Dig. Liver Dis. Off. J. Ital. Soc. Gastroenterol. Ital. Assoc. Study Liver 2017, 49, 1177–1184. [Google Scholar] [CrossRef] [PubMed]
- Dicks, L.M.T.; Botes, M. Probiotic lactic acid bacteria in the gastro-intestinal tract: Health benefits, safety and mode of action. Benef. Microbes 2010, 1, 11–29. [Google Scholar] [CrossRef] [PubMed]
- Halloran, K.; Underwood, M.A. Probiotic mechanisms of action. Early Hum. Dev. 2019, 135, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Plaza-Diaz, J.; Ruiz-Ojeda, F.J.; Gil-Campos, M.; Gil, A. Mechanisms of Action of Probiotics. Adv. Nutr. 2019, 10, S49–S66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pedicord, V.A.; Lockhart, A.A.K.; Rangan, K.J.; Craig, J.W.; Loschko, J.; Rogoz, A.; Hang, H.C.; Mucida, D. Exploiting a host-commensal interaction to promote intestinal barrier function and enteric pathogen tolerance. Sci. Immunol. 2016, 1, eaai7732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hecht, A.L.; Casterline, B.; Earley, Z.M.; Goo, Y.A.; Goodlett, D.R.; Wardenburg, J.B. Strain competition restricts colonization of an enteric pathogen and prevents colitis. EMBO Rep. 2016, 17, 1281–1291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Zyl, W.F.; Deane, S.M.; Dicks, L.M. Molecular insights into probiotic mechanisms of action employed against intestinal pathogenic bacteria. Gut Microbes 2020, 12, 1831339. [Google Scholar] [CrossRef] [PubMed]
- El-Saadony, M.T.; Alagawany, M.; Patra, A.K.; Kar, I.; Tiwari, R.; Dawood, M.A.; Dhama, K.; Abdel-Latif, H.M. The functionality of probiotics in aquaculture: An overview. Fish Shellfish Immunol. 2021, 117, 36–52. [Google Scholar] [CrossRef]
- Zhao, W.; Yuan, T.; Piva, C.; Spinard, E.J.; Schuttert, C.W.; Rowley, D.C.; Nelson, D.R. The Probiotic Bacterium Phaeobacter inhibens Downregulates Virulence Factor Transcription in the Shellfish Pathogen Vibrio coralliilyticus by N-Acyl Homoserine Lactone Production. Appl. Environ. Microbiol. 2019, 85, e01545-18. [Google Scholar] [CrossRef] [Green Version]
- Markowiak-Kopeć, P.; Śliżewska, K. The Effect of Probiotics on the Production of Short-Chain Fatty Acids by Human Intestinal Microbiome. Nutrients 2020, 12, 1107. [Google Scholar] [CrossRef] [PubMed]
- Cortes-Perez, N.G.; de Moreno de LeBlanc, A.; Gomez-Gutierrez, J.G.; LeBlanc, J.G.; Bermúdez-Humarán, L.G. Probiotics and Trained Immunity. Biomolecules 2021, 11, 1402. [Google Scholar] [CrossRef]
- Vincenzi, A.; Goettert, M.I.; de Souza, C.F.V. An evaluation of the effects of probiotics on tumoral necrosis factor (TNF-α) signaling and gene expression. Cytokine Growth Factor Rev. 2021, 57, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Ashaolu, T.J. Immune boosting functional foods and their mechanisms: A critical evaluation of probiotics and prebiotics. Biomed. Pharmacother. 2020, 130, 110625. [Google Scholar] [CrossRef] [PubMed]
- Ferrario, C.; Taverniti, V.; Milani, C.; Fiore, W.; Laureati, M.; De Noni, I.; Stuknyte, M.; Chouaia, B.; Riso, P.; Guglielmetti, S. Modulation of Fecal Clostridiales Bacteria and Butyrate by Probiotic Intervention with Lactobacillus paracasei DG Varies among Healthy Adults. J. Nutr. 2014, 144, 1787–1796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raheem, A.; Liang, L.; Zhang, G.; Cui, S. Modulatory Effects of Probiotics During Pathogenic Infections with Emphasis on Immune Regulation. Front. Immunol. 2021, 12, 616713. [Google Scholar] [CrossRef]
- Quigley, E.M.; Gajula, P. Recent advances in modulating the microbiome. F1000Research 2020, 9, 46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biswasroy, P.; Pradhan, D.; Sahu, D.K.; Sahu, A.; Ghosh, G.; Rath, G. Recent Advances in Clinical Utility of Probiotics in Gastrointestinal Tract Disorders. Curr. Pharm. Biotechnol. 2021, 22, 1559–1573. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Zuo, Z.; Mao, A.-P. Effect of Probiotics on Inducing Remission and Maintaining Therapy in Ulcerative Colitis, Crohnʼs Disease, and Pouchitis. Inflamm. Bowel Dis. 2014, 20, 21–35. [Google Scholar] [CrossRef] [Green Version]
- Radicioni, M.; Koirala, R.; Fiore, W.; Leuratti, C.; Guglielmetti, S.; Arioli, S. Survival of L. casei DG® (Lactobacillus paracasei CNCMI1572) in the gastrointestinal tract of a healthy paediatric population. Eur. J. Nutr. 2019, 58, 3161–3170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arioli, S.; Koirala, R.; Taverniti, V.; Fiore, W.; Guglielmetti, S. Quantitative Recovery of Viable Lactobacillus paracasei CNCM I-1572 (L. casei DG®) After Gastrointestinal Passage in Healthy Adults. Front. Microbiol. 2018, 9, 1720. [Google Scholar] [CrossRef] [PubMed]
- Anon. Detection Probiotic’s DNA of Lactobacillus paracasei in Healthy Human Faeces|Journal of Pharmaceutical Research International. Available online: https://www.journaljpri.com/index.php/JPRI/article/view/17790 (accessed on 8 January 2022).
- Balzaretti, S.; Taverniti, V.; Rondini, G.; Marcolegio, G.; Minuzzo, M.; Remagni, M.C.; Fiore, W.; Arioli, S.; Guglielmetti, S. The vaginal isolate Lactobacillus paracasei LPC-S01 (DSM 26760) is suitable for oral administration. Front. Microbiol. 2015, 6, 952. [Google Scholar] [CrossRef]
- Balzaretti, S.; Taverniti, V.; Guglielmetti, S.; Fiore, W.; Minuzzo, M.; Ngo, H.N.; Ngere, J.B.; Sadiq, S.; Humphreys, P.; Laws, A.P. A Novel Rhamnose-Rich Hetero-exopolysaccharide Isolated from Lactobacillus paracasei DG Activates THP-1 Human Monocytic Cells. Appl. Environ. Microbiol. 2017, 83, e02702-16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cremon, C.; Guglielmetti, S.; Gargari, G.; Taverniti, V.; Castellazzi, A.M.; Valsecchi, C.; Tagliacarne, C.; Fiore, W.; Bellini, M.; Bertani, L.; et al. Effect of Lactobacillus paracasei CNCM I-1572 on symptoms, gut microbiota, short chain fatty acids, and immune activation in patients with irritable bowel syndrome: A pilot randomized clinical trial. United Eur. Gastroenterol. J. 2018, 6, 604–613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Compare, D.; Rocco, A.; Coccoli, P.; Angrisani, D.; Sgamato, C.; Iovine, B.; Salvatore, U.; Nardone, G. Lactobacillus casei DG and its postbiotic reduce the inflammatory mucosal response: An ex-vivo organ culture model of post-infectious irritable bowel syndrome. BMC Gastroenterol. 2017, 17, 53. [Google Scholar] [CrossRef] [Green Version]
- Turco, F.; Andreozzi, P.; Palumbo, I.; Zito, F.P.; Cargiolli, M.; Fiore, W.; Gennarelli, N.; De Palma, G.D.; Sarnelli, G.; Cuomo, R. Bacterial stimuli activate nitric oxide colonic mucosal production in diverticular disease. Protective effects of L. casei DG® (Lactobacillus paracasei CNCM I-1572). United Eur. Gastroenterol. J. 2017, 5, 715–724. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.-F.; Tseng, K.-C.; Chiang, S.-S.; Lee, B.-H.; Hsu, W.-H.; Pan, T.-M. Immunomodulatory and antioxidant potential of Lactobacillus exopolysaccharides. J. Sci. Food Agric. 2011, 91, 2284–2291. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo-Cantabrana, C.; Lopez-Suarez, P.; Gueimonde, M.; Reyes-Gavilan, C.D.L.; Suarez-Diaz, A.M.; Margolles, A.; Ruas-Madiedo, P. Immune Modulation Capability of Exopolysaccharides Synthesised by Lactic Acid Bacteria and Bifidobacteria. Probiotics Antimicrob. Proteins 2012, 4, 227–237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patten, D.; Laws, A. Lactobacillus-produced exopolysaccharides and their potential health benefits: A review. Benef. Microbes 2015, 6, 457–471. [Google Scholar] [CrossRef] [PubMed]
- Cai, T.; Gallelli, L.; Cione, E.; Perletti, G.; Ciarleglio, F.; Malossini, G.; De Pretis, G.; Palmieri, A.; Mirone, V.; Bartoletti, R.; et al. The use of Lactobacillus casei DG® prevents symptomatic episodes and reduces the antibiotic use in patients affected by chronic bacterial prostatitis: Results from a phase IV study. World J. Urol. 2021, 39, 3433–3440. [Google Scholar] [CrossRef] [PubMed]
- Rosania, R.; Giorgio, F.; Principi, M.; Amoruso, A.; Monno, R.; Di Leo, A.; Ierardi, E. Effect of probiotic or prebiotic supplementation on antibiotic therapy in the small intestinal bacterial overgrowth: A comparative evaluation. Curr. Clin. Pharmacol. 2013, 8, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Tursi, A.; Brandimarte, G.; Giorgetti, G.M.; Elisei, W. Mesalazine and/or Lactobacillus casei in preventing recurrence of symptomatic uncomplicated diverticular disease of the colon: A prospective, randomized, open-label study. J. Clin. Gastroenterol. 2006, 40, 312–316. [Google Scholar] [CrossRef] [PubMed]
- Tursi, A.; Brandimarte, G.; Giorgetti, G.M.; Elisei, W. Mesalazine and/or Lactobacillus casei in maintaining long-term remission of symptomatic uncomplicated diverticular disease of the colon. Hepatogastroenterology 2008, 55, 916–920. [Google Scholar] [PubMed]
- Tursi, A.; Brandimarte, G.; Elisei, W.; Picchio, M.; Forti, G.; Pianese, G.; Rodino, S.; D’Amico, T.; Sacca, N.; Portincasa, P.; et al. Randomised clinical trial: Mesalazine and/or probiotics in maintaining remission of symptomatic uncomplicated diverticular disease-a double-blind, randomised, placebo-controlled study. Aliment. Pharmacol. Ther. 2013, 38, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Dai, D.; Zhu, J.; Sun, C.; Li, M.; Liu, J.; Wu, S.; Ning, K.; He, L.-J.; Zhao, X.-M.; Chen, W.-H. GMrepo v2: A curated human gut microbiome database with special focus on disease markers and cross-dataset comparison. Nucleic Acids Res. 2022, 50, D777–D784. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Hu, G.; Sun, C.; Duan, Y.; Zhang, Z.; Liu, Z.; Zhao, X.-M.; Chen, W.-H. mBodyMap: A curated database for microbes across human body and their associations with health and diseases. Nucleic Acids Res. 2022, 50, D808–D816. [Google Scholar] [CrossRef]
- Gosalbes, M.J.; Durbán, A.; Pignatelli, M.; Abellan-Andres, J.J.; Hernández, N.J.; Pérez-Cobas, A.E.; Latorre, A.; Moya, A. Metatranscriptomic Approach to Analyze the Functional Human Gut Microbiota. PLoS ONE 2011, 6, e17447. [Google Scholar] [CrossRef]
- Kang, D.-W.; Park, J.G.; Ilhan, Z.E.; Wallstrom, G.; LaBaer, J.; Adams, J.B.; Krajmalnik-Brown, R. Reduced Incidence of Prevotella and Other Fermenters in Intestinal Microflora of Autistic Children. PLoS ONE 2013, 8, e68322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malinen, E.; Rinttilä, T.; Kajander, K.; Mättö, J.; Kassinen, A.; Krogius, L.; Saarela, M.; Korpela, R.; Palva, A. Analysis of the fecal microbiota of irritable bowel syndrome patients and healthy controls with real-time PCR. Am. J. Gastroenterol. 2005, 100, 373–382. [Google Scholar] [CrossRef]
- Rajilic-Stojanovic, M.; Biagi, E.; Heilig, H.G.; Kajander, K.; Kekkonen, R.A.; Tims, S.; de Vos, W.M. Global and Deep Molecular Analysis of Microbiota Signatures in Fecal Samples From Patients with Irritable Bowel Syndrome. Gastroenterology 2011, 141, 1792–1801. [Google Scholar] [CrossRef]
- Huttenhower, C.; Gevers, D.; Knight, R.; Abubucker, S.; Badger, J.H.; Chinwalla, A.T.; Creasy, H.H.; Earl, A.M.; FitzGerald, M.G.; Fulton, R.S.; et al. Structure, function and diversity of the healthy human microbiome. Nature 2012, 486, 207–214. [Google Scholar] [CrossRef] [Green Version]
- Ríos-Covián, D.; Ruas-Madiedo, P.; Margolles, A.; Gueimonde, M.; De Los Reyes-Gavilán, C.G.; Salazar, N. Intestinal Short Chain Fatty Acids and their Link with Diet and Human Health. Front. Microbiol. 2016, 7, 185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leonel, A.J.; Alvarez-Leite, J.I. Butyrate: Implications for intestinal function. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 474–479. [Google Scholar] [CrossRef] [PubMed]
- Bourdu, S.; Dapoigny, M.; Chapuy, E.; Artigue, F.; Vasson, M.-P.; Dechelotte, P.; Bommelaer, G.; Eschalier, A.; Ardid, D. Rectal Instillation of Butyrate Provides a Novel Clinically Relevant Model of Noninflammatory Colonic Hypersensitivity in Rats. Gastroenterology 2005, 128, 1996–2008. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, I.; O’Toole, P.; Öhman, L.; Claesson, M.; Deane, J.; Quigley, E.M.M.; Simrén, M. An irritable bowel syndrome subtype defined by species-specific alterations in faecal microbiota. Gut 2012, 61, 997–1006. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, T.F.S.; Grześkowiak, Ł.; Franceschini, S.C.C.; Bressan, J.; Ferreira, C.L.L.F.; Peluzio, M.C.G. Higher level of faecal SCFA in women correlates with metabolic syndrome risk factors. Br. J. Nutr. 2013, 109, 914–919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Payne, A.N.; Chassard, C.; Zimmermann, M.; Müller, P.; Stinca, S.; Lacroix, C. The metabolic activity of gut microbiota in obese children is increased compared with normal-weight children and exhibits more exhaustive substrate utilization. Nutr. Diabetes 2011, 1, e12. [Google Scholar] [CrossRef] [Green Version]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.H.; Belenguer, A.; Holtrop, G.; Johnstone, A.M.; Flint, H.J.; Lobley, G.E. Reduced Dietary Intake of Carbohydrates by Obese Subjects Results in Decreased Concentrations of Butyrate and Butyrate-Producing Bacteria in Feces. Appl. Environ. Microbiol. 2007, 73, 1073–1078. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parkes, G.C.; Rayment, N.B.; Hudspith, B.N.; Petrovska, L.; Lomer, M.C.; Brostoff, J.; Whelan, K.; Sanderson, J.D. Distinct microbial populations exist in the mucosa-associated microbiota of sub-groups of irritable bowel syndrome. Neurogastroenterol. Motil. Off. J. Eur. Gastrointest. Motil. Soc. 2012, 24, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Swidsinski, A.; Weber, J.; Loening-Baucke, V.; Hale, L.P.; Lochs, H. Spatial Organization and Composition of the Mucosal Flora in Patients with Inflammatory Bowel Disease. J. Clin. Microbiol. 2005, 43, 3380–3389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Espin, F.; Rofes, L.; Ortega, O.; Clavé, P.; Gallego, D. Nitrergic neuro-muscular transmission is up-regulated in patients with diverticulosis. Neurogastroenterol. Motil. Off. J. Eur. Gastrointest. Motil. Soc. 2014, 26, 1458–1468. [Google Scholar] [CrossRef] [PubMed]
- Golder, M.; Burleigh, D.E.; Ghali, L.; Feakins, R.M.; Lunniss, P.J.; Williams, N.S.; Navsaria, H.A. Longitudinal muscle shows abnormal relaxation responses to nitric oxide and contains altered levels of NOS1 and elastin in uncomplicated diverticular disease. Colorectal Dis. 2007, 9, 218–228. [Google Scholar] [CrossRef] [PubMed]
- Jeyarajah, S.; Papagrigoriadis, S. Review article: The pathogenesis of diverticular disease-current perspectives on motility and neurotransmitters. Aliment. Pharmacol. Ther. 2011, 33, 789–800. [Google Scholar] [CrossRef] [Green Version]
- Zamora, R.; Vodovotz, Y.; Billiar, T.R. Inducible Nitric Oxide Synthase and Inflammatory Diseases. Mol. Med. 2000, 6, 347–373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Mario, F.; Miraglia, C.; Cambiè, G.; Violi, A.; Nouvenne, A.; Franceschi, M.; Brandimarte, G.; Elisei, W.; Picchio, M.; Tursi, A. Long-term efficacy of rifaximin to manage the symptomatic uncomplicated diverticular disease of the colon. J. Investig. Med. Off. Publ. Am. Fed. Clin. Res. 2019, 67, 767–770. [Google Scholar] [CrossRef] [PubMed]
- Latella, G.; Pimpo, M.; Sottili, S.; Zippi, M.; Viscido, A.; Chiaramonte, M.; Frieri, G. Rifaximin improves symptoms of acquired uncomplicated diverticular disease of the colon. Int. J. Colorectal Dis. 2003, 18, 55–62. [Google Scholar] [CrossRef] [PubMed]
- De Bastiani, R.; Sanna, G.; Bertolusso, L.; Casella, G.; De Polo, M.; Zamparella, M.; Cottone, C.; Tosetti, C.; Mancuso, M.; Pirrotta, E. General practitioners’ management of symptomatic uncomplicated diverticular disease of the colon by using rifaximin, a non-adsorbable antibiotic. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 423–430. [Google Scholar] [PubMed]
- Scaioli, E.; Colecchia, A.; Marasco, G.; Schiumerini, R.; Festi, D. Pathophysiology and Therapeutic Strategies for Symptomatic Uncomplicated Diverticular Disease of the Colon. Am. J. Dig. Dis. 2016, 61, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Tursi, A. New physiopathological and therapeutic approaches to diverticular disease: An update. Expert Opin. Pharmacother. 2014, 15, 1005–1017. [Google Scholar] [CrossRef] [PubMed]
- Tursi, A. New physiopathological and therapeutic approaches to diverticular disease of the colon. Expert Opin. Pharmacother. 2007, 8, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Tursi, A.; Papa, A.; Danese, S. Review article: The pathophysiology and medical management of diverticulosis and diverticular disease of the colon. Aliment. Pharmacol. Ther. 2015, 42, 664–684. [Google Scholar] [CrossRef] [PubMed]
- Anon; World Gastroenterology Organisation (WGO). World Gastroenterol Organ WGO. Available online: https://www.worldgastroenterology.org (accessed on 28 January 2022).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).