Long-Term Outcomes of Extra-Anatomic Femoro-Tibial Bypass Reconstructions in Chronic Limb-Threating Ischemia
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Inclusion and Exclusion Criteria
2.3. Study Design and Clinical Parameters
2.4. Data Collection
2.5. Statistical Analysis
3. Results
3.1. Patients and Procedure Characteristics
3.2. Early Outcomes
3.3. Survival
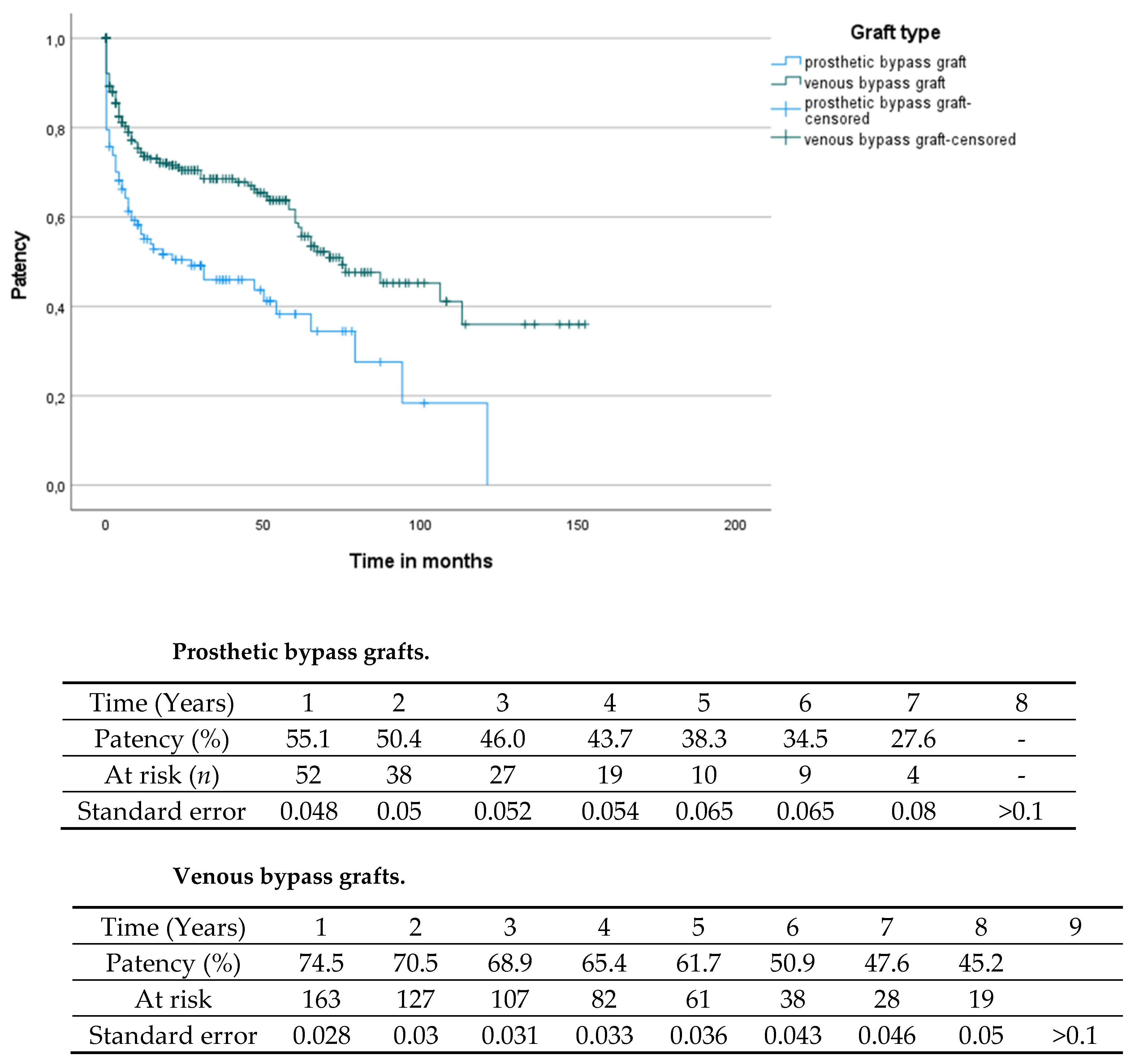
3.4. Comparison of Prosthetic and Venous Bypass Material
3.5. Analysis of Extra-Anatomic Reconstructions
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Van de Pavoordt, H.D.; Eikelboom, B.C.; De Geest, R.; Vermeulen, F.E. Results of prosthetic grafts in femoro-crural bypass operations as compared to autogenous saphenous vein grafts. Neth. J. Surg. 1986, 38, 177–179. [Google Scholar] [PubMed]
- Rostad, H.; Hall, K.V.; Rostad, B. Bypass To the Tibial and Peroneal Arteries Using the Great Saphenous Vein in Situ. Vasc. Surg. 1977, 11, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Almasri, J.; Adusumalli, J.; Asi, N.; Lakis, S.; Alsawas, M.; Prokop, L.J.; Bradbury, A.; Kolh, P.; Conte, M.S.; Murad, M.H. A systematic review and meta-analysis of revascularization outcomes of infrainguinal chronic limb-threatening ischemia. Eur. J. Vasc. Endovasc. Surg. 2019, 58, S110–S119. [Google Scholar] [CrossRef]
- Meyer, A.; Schilling, A.; Kott, M.; Rother, U.; Lang, W.; Regus, S. Open Versus Endovascular Revascularization of Below-Knee Arteries in Patients With End-Stage Renal Disease and Critical Limb Ischemia. Vasc. Endovasc. Surg. 2018, 52, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.; Schinz, K.; Lang, W.; Schmid, A.; Regus, S.; Rother, U. Outcomes and Influence of the Pedal Arch in Below-the-Knee Angioplasty in Patients with End-Stage Renal Disease and Critical Limb Ischemia. Ann. Vasc. Surg. 2016, 35, 121–129. [Google Scholar] [CrossRef]
- Kotov, A.; Peters, F.; Debus, E.S.; Zeller, T.; Heider, P.; Stavroulakis, K.; Remig, J.; Gussmann, A.; Hoffmann, J.; Friedrich, O.; et al. The prospective GermanVasc cohort study. Vasa 2021, 50, 446–452. [Google Scholar] [CrossRef]
- Aboyans, V.; Ricco, J.B.; Bartelink, M.L.; Björck, M.; Brodmann, M.; Cohner, T.; Desormais, I. 2017 ESC Guidelines on the Diagnosis and Treatment of Peripheral Arterial Diseases, in collaboration with the European Society for Vascular Surgery (ESVS): Document covering atherosclerotic disease of extracranial carotid and vertebral, mesenteric, renal, upper and lower extremity arteriesEndorsed by: The European Stroke Organization (ESO)The Task Force for the Diagnosis and Treatment of Peripheral Arterial Diseases of the European Society of Cardiology (ESC) and of the European Society for Vascular Surgery (ESVS). Eur. Heart J. 2018, 39, 763–816. [Google Scholar]
- Kashyap, V.; Ahn, S.S.; Quinones-Baldrich, W.J.; Choi, B.-U.; Dorey, F.; Reil, T.D.; Freischlag, J.A.; Moore, W.S. Infrapopliteal-Lower Extremity Revascularization with Prosthetic Conduit: A 20-Year Experience. Vasc. Endovasc. Surg. 2002, 36, 255–262. [Google Scholar] [CrossRef]
- Cavillon, A.; Melliere, D.; Allaire, E.; Blancas, E.A.; Berrahal, D.; Desgranges, P.; Becquemin, J.P. Are femoro-infrapopliteal bypasses worthwhile for limb salvage? J. Cardiovasc. Surg. 1998, 39, 267–272. [Google Scholar]
- Daenens, K.; Schepers, S.; Fourneau, I.; Houthoofd, S.; Nevelsteen, A. Heparin-bonded ePTFE grafts compared with vein grafts in femoropopliteal and femorocrural bypasses: 1- and 2-year results. J. Vasc. Surg. 2009, 49, 1210–1216. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Prev. Med. 2007, 45, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Panayiotopoulos, Y.P.; Tyrrell, M.R.; Owen, E.S.; Reidy, J.F.; Taylor, P.R. Outcome and cost analysis after femorocrural and femoropedal grafting for critical limb ischaemia. Br. J. Surg. 1997, 84, 207–212. [Google Scholar] [PubMed]
- Tyrrell, M.; Grigg, M.; Wolfe, J. Is arterial reconstruction to the ankle worthwhile in the absence of autologous vein? Eur. J. Vasc. Surg. 1989, 3, 429–434. [Google Scholar] [CrossRef]
- Tordoir, J.H.; Van Der Plas, J.P.; Jacobs, M.J.; Kitslaar, P.J. Factors determining the outcome of crural and pedal revascularisation for critical limb ischaemia. Eur. J. Vasc. Surg. 1993, 7, 82–86. [Google Scholar] [CrossRef]
- Lundgren, F. PTFE bypass to below-knee arteries: Distal vein collar or not? A prospective randomised multicentre study. Eur. J. Vasc. Endovasc. Surg. 2010, 39, 747–754. [Google Scholar]
- Branco, B.C.; Kougias, P.; Braun, J.D.; Mills, J.L.; Barshes, N.R. Distal vein patch use and limb events after infragenicular prosthetic bypasses. J. Vasc. Surg. 2018, 68, 145–152. [Google Scholar] [CrossRef]
- Kissin, M.; Kansal, N.; Pappas, P.J.; DeFouw, D.O.; Durán, W.N.; Hobson II, R.W. Vein interposition cuffs decrease the intimal hyperplastic response of polytetrafluoroethylene bypass grafts. J. Vasc. Surg. 2000, 31, 69–83. [Google Scholar] [CrossRef]
- Stonebridge, P.; Prescott, R.; Ruckley, C. Randomized trial comparing infrainguinal polytetrafluoroethylene bypass grafting with and without vein interposition cuff at the distal anastomosis. J. Vasc. Surg. 1997, 26, 543–550. [Google Scholar] [CrossRef]
- Meyer, A.; Fiessler, C.; Stavroulakis, K.; Torsello, G.; Bisdas, T.; Lang, W.; Adili, F.; Balzer, K.; Billing, A.; Böckler, D.; et al. Outcomes of dialysis patients with critical limb ischemia after revascularization compared with patients with normal renal function. J. Vasc. Surg. 2018, 68, 822–829.e1. [Google Scholar] [CrossRef]
- Samson, R.H.; Morales, R.; Showalter, D.P.; Lepore, M.R.; Nair, D.G. Heparin-bonded expanded polytetrafluoroethylene femoropopliteal bypass grafts outperform expanded polytetrafluoroethylene grafts without heparin in a long-term comparison. J. Vasc. Surg. 2016, 64, 638–647. [Google Scholar] [CrossRef]
- Stockmann, U. Extra-anatomic crural reconstruction. In Langenbecks Archiv fur Chirurgie. Supplement II, Verhandlungen der Deutschen Gesellschaft fur Chirurgie. Deutsche Gesellschaft fur Chirurgie. Kongress; Springer: Berlin, Germany, 1989; pp. 613–615. [Google Scholar]
- Kapfer, X.; Meichelboeck, W.; Groegler, F.M. Comparison of carbon-impregnated and standard ePTFE prostheses in extra-anatomical anterior tibial artery bypass: A prospective randomized multicenter study. Eur. J. Vasc. Endovasc. Surg. 2006, 32, 155–168. [Google Scholar] [CrossRef][Green Version]
- Pollock, A.V. Axillary-femoral by-pass grafts in the treatment of aorto-iliac occlusive disease. Br. J. Surg. 2005, 59, 704–707. [Google Scholar] [CrossRef] [PubMed]
- Koivunen, V.; Juonala, M.; Mikkola, K.; Hakovirta, H.H. Chronic limb threatening ischemia and diabetes mellitus: The severity of tibial atherosclerosis and outcome after infrapopliteal revascularization. Scand. J. Surg. 2020, 110, 472–482. [Google Scholar] [CrossRef] [PubMed]
- Dayama, A.; Tsilimparis, N.; Kolakowski, S.; Matolo, N.M.; Humphries, M.D. Clinical outcomes of bypass-first versus endovascular-first strategy in patients with chronic limb-threatening ischemia due to infrageniculate arterial disease. J. Vasc. Surg. 2019, 69, 156–163.e1. [Google Scholar] [CrossRef] [PubMed]

| Patients Characteristics | Total N (%) | Anatomic Bypass N (%) | Extra-Anatomic Bypass N (%) | p-Value |
|---|---|---|---|---|
| Arterial hypertension | 412 (90.5) | 365 (80.2) | 47 (10.3) | 0.804 |
| Diabetes | 229 (50.3) | 208 (45.7) | 21 (4.6) | 0.183 |
| Coronary artery disease | 244 (53.6) | 218 (47.9) | 26 (5.7) | 0.766 |
| Hyperlipidemia | 223 (49.0) | 195 (42.9) | 28 (6.2) | 0.616 |
| Smoking (currently and formal) | 255 (56.0) | 227 (49.9) | 28 (6.2) | 0.774 |
| Dialysis dependency | 25 (5.5) | 22 (4.8) | 3 (0.6) | 0.904 |
| Procedure characteristics | ||||
| Acute limb ischemia (as indication for bypass surgery) | 53 (11.6) | 45 (9.8) | 8 (1.7) | 0.353 |
| Rutherford Stage | 0.06 | |||
| 3 | 29 (6.3) | 24 (5.3) | 4 (0.09) | |
| 4 | 57 (12.5) | 47 (10.3) | 10 (2.2) | |
| 5/6 | 316 (69.5) | 285 (62.6) | 31 (6.8) | |
| Prosthetic bypass grafts | 131 (28.8) | 80 (17.6) | 51 (11.2) | <0.001 |
| Bypass anatomy | 455 (100.0) | 404 (88.7) | 51 (11.2) | |
| WIFI Score amputation risk | 0.451 | |||
| Very low | 9 (2.0) | 9 (2.0) | 0 (0.0) | |
| Low | 76 (16.7) | 63 (13.8) | 13 (2.9) | |
| Moderate | 90 (19.8) | 81 (17.7) | 9 (1.9) | |
| High | 127 (27.9) | 113 (24.8) | 14 (3.1) | |
| Not assessable | 153 (33.6) | 135 (29.7) | 26 (5.7) | |
| No antithrombotic therapy | 12 (2.6) | 9 (1.9) | 3 (0.7) | 0.091 |
| Single antiplatelet therapy | 218 (47.9) | 194 (42.6) | 24 (5.3) | |
| Dual antiplatelet therapy | 46 (10.1) | 41 (9.0) | 5 (1.1) | |
| Vitamin K antagonist | 119 (26.2) | 106 (23.3) | 13 (2.9) | |
| Direct oral anticoagulation | 39 (8.6) | 36 (7.9) | 3 (0.7) | |
| Aspirin and direct oral anticoagulation | 5 (1.1) | 3 (0.7) | 2 (0.4) | |
| Aspirin and Vitamin K antagonist | 14 (3.1) | 13 (2.9) | 1 (0.2) | |
| Triple Therapy | 2 (0.4) | 2 (0.4) | 0 (0.0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meyer, A.; Boxberger, E.; Behrendt, C.-A.; Yagshyyev, S.; Welk, I.; Lang, W.; Rother, U. Long-Term Outcomes of Extra-Anatomic Femoro-Tibial Bypass Reconstructions in Chronic Limb-Threating Ischemia. J. Clin. Med. 2022, 11, 1237. https://doi.org/10.3390/jcm11051237
Meyer A, Boxberger E, Behrendt C-A, Yagshyyev S, Welk I, Lang W, Rother U. Long-Term Outcomes of Extra-Anatomic Femoro-Tibial Bypass Reconstructions in Chronic Limb-Threating Ischemia. Journal of Clinical Medicine. 2022; 11(5):1237. https://doi.org/10.3390/jcm11051237
Chicago/Turabian StyleMeyer, Alexander, Evgenia Boxberger, Christian-Alexander Behrendt, Shatlyk Yagshyyev, Irina Welk, Werner Lang, and Ulrich Rother. 2022. "Long-Term Outcomes of Extra-Anatomic Femoro-Tibial Bypass Reconstructions in Chronic Limb-Threating Ischemia" Journal of Clinical Medicine 11, no. 5: 1237. https://doi.org/10.3390/jcm11051237
APA StyleMeyer, A., Boxberger, E., Behrendt, C.-A., Yagshyyev, S., Welk, I., Lang, W., & Rother, U. (2022). Long-Term Outcomes of Extra-Anatomic Femoro-Tibial Bypass Reconstructions in Chronic Limb-Threating Ischemia. Journal of Clinical Medicine, 11(5), 1237. https://doi.org/10.3390/jcm11051237






