Thromboprophylaxis Reduced Venous Thromboembolism in Sickle Cell Patients with Central Venous Access Devices: A Retrospective Cohort Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
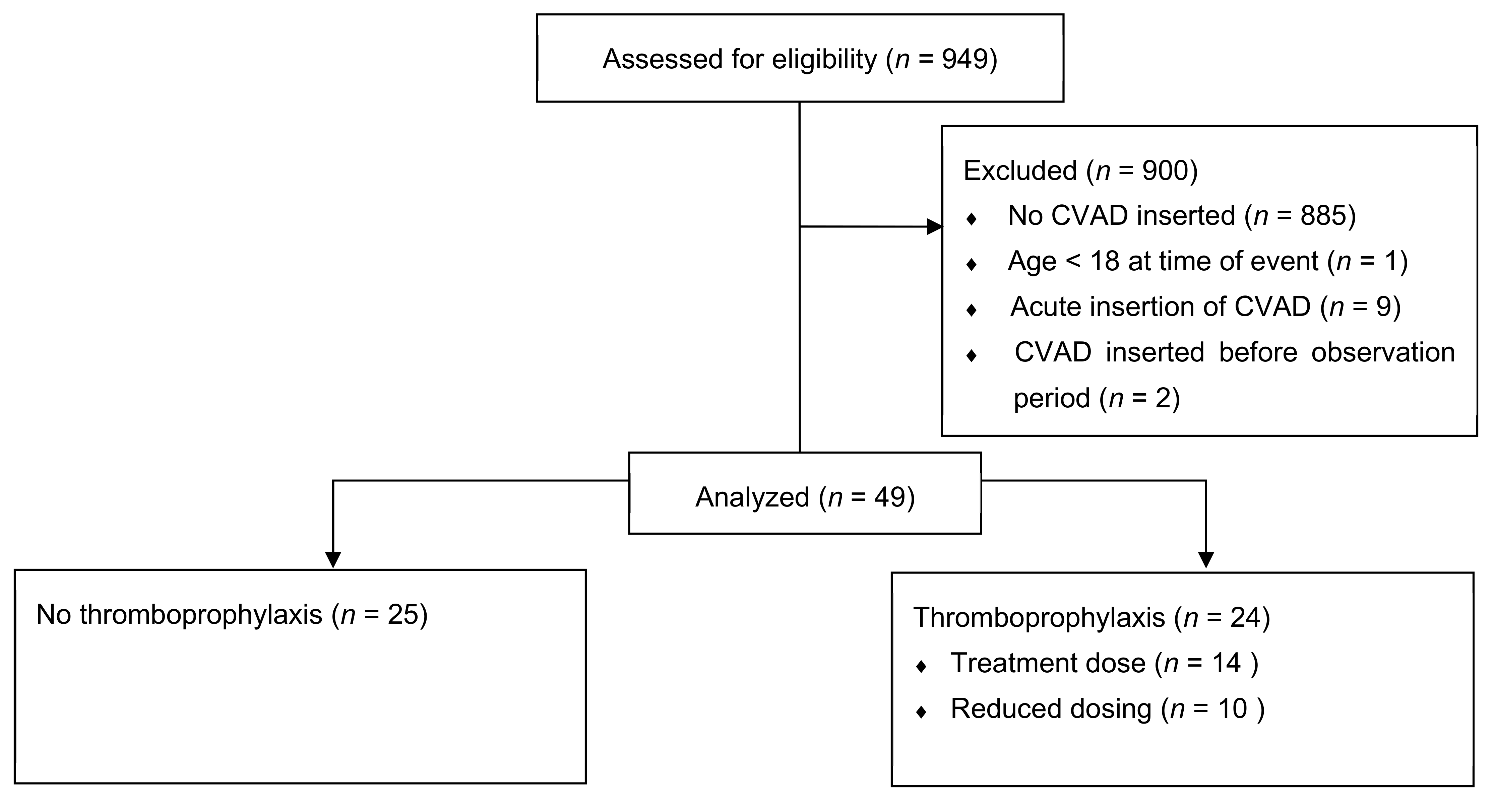
2.2. Inclusion and Exclusion Criteria
2.3. Patient Characteristics
2.4. Exposure
2.5. Outcome
2.6. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wun, T.; Brunson, A. Sickle cell disease: An inherited thrombophilia. Hematol. Am. Soc. Hematol. Educ. Program 2016, 2016, 640–647. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naik, R.P.; Streiff, M.B.; Lanzkron, S. Sickle cell disease and venous thromboembolism: What the anticoagulation expert needs to know. J. Thromb. Thrombolysis 2013, 35, 352–358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kato, G.J.; Piel, F.B.; Reid, C.D.; Gaston, M.H.; Ohene-Frempong, K.; Krishnamurti, L.; Smith, W.R.; Panepinto, J.A.; Weatherall, D.J.; Costa, F.F.; et al. Sickle cell disease. Nat. Rev. Dis. Primers 2018, 4, 1–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shet, A.S.; Lizarralde-Iragorri, M.A.; Naik, R.P. The molecular basis for the prothrombotic state in sickle cell disease. Haematologica 2020, 105, 2368–2379. [Google Scholar] [CrossRef] [PubMed]
- Naik, R.P.; Streiff, M.B.; Haywood, C., Jr.; Nelson, J.A.; Lanzkron, S. Venous thromboembolism in adults with sickle cell disease: A serious and under-recognized complication. Am. J. Med. 2013, 126, 443–449. [Google Scholar] [CrossRef] [Green Version]
- Brunson, A.; Keegan, T.; Mahajan, A.; White, R.; Wun, T. High incidence of venous thromboembolism recurrence in patients with sickle cell disease. Am. J. Hematol. 2019, 94, 862–870. [Google Scholar] [CrossRef]
- Naik, R.P.; Streiff, M.B.; Haywood, C., Jr.; Segal, J.B.; Lanzkron, S. Venous thromboembolism incidence in the Cooperative Study of Sickle Cell Disease. J. Thromb. Haemost. 2014, 12, 2010–2016. [Google Scholar] [CrossRef] [Green Version]
- Brewin, J.N.; Crowley, M.P.; Kesse-Adu, R.; Stuart-Smith, S.; Awogbade, M.; Howard, J. Catheter associated thromboses in patients with sickle cell anaemia and dual lumen Vortex apheresis ports are common and can be clinically asymptomatic. Br. J. Haematol. 2020, 189, e198–e200. [Google Scholar] [CrossRef] [Green Version]
- Howard, J.; Malfroy, M.; Llewelyn, C.; Choo, L.; Hodge, R.; Johnson, T.; Purohit, S.; Rees, D.C.; Tillyer, L.; Walker, I.; et al. The Transfusion Alternatives Preoperatively in Sickle Cell Disease (TAPS) study: A randomised, controlled, multicentre clinical trial. Lancet 2013, 381, 930–938. [Google Scholar] [CrossRef]
- Schulman, S.; Kearon, C.; Subcommittee on Control of Anticoagulation of the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non-surgical patients. J. Thromb. Haemost. 2005, 3, 692–694. [Google Scholar] [CrossRef]
- Matusiak, K.; Forté, S.; Abdulrehman, J.; Verhovsek, M.; Kuo, K.H.M. Use of Thromboprophylaxis for Central Venous Access Devices in Patients with Sickle Cell Disease: A Survey of Canadian Providers. Blood 2021, 138, 4173. [Google Scholar] [CrossRef]
- Yeral, M.; Boga, C.; Oguzkurt, L.; Asma, S.; Kasar, M.; Kozanoglu, I. Short-term central venous catheter complications in patients with sickle cell disease who undergo apheresis. J. Thromb. Thrombolysis 2014, 37, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Phillips, G.; Slingluff, C.; Hartman, J.; Thomas, P.; Akwari, O. Totally implantable intravenous catheters in the management of sickle cell anemia. Am. J. Hematol. 1988, 29, 134–138. [Google Scholar] [CrossRef]
- Jeng, M.R.; Feusner, J.; Skibola, C.; Vichinsky, E. Central venous catheter complications in sickle cell disease. Am. J. Hematol. 2002, 69, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Alkindi, S.; Matwani, S.; Al-Maawali, A.; Al-Maskari, B.; Pathare, A. Complications of PORT-A-CATH(R) in patients with sickle cell disease. J. Infect. Public Health 2012, 5, 57–62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raj, A.; Bertolone, S.; Bond, S.; Burnett, D.; Denker, A. Cathlink 20: A subcutaneous implanted central venous access device used in children with sickle cell disease on long-term erythrocytapheresis—A report of low complication rates. Pediatr. Blood Cancer 2005, 44, 669–672. [Google Scholar] [CrossRef]
- Abdul-Rauf, A.; Gauderer, M.; Chiarucci, K.; Berman, B. Long-term central venous access in patients with sickle cell disease. Incidence of thrombotic and infectious complications. J. Pediatr. Hematol. Oncol. 1995, 17, 342–345. [Google Scholar] [CrossRef]
- Shah, N.; Landi, D.; Shah, R.; Rothman, J.; De Castro, L.M.; Thornburg, C.D. Complications of implantable venous access devices in patients with sickle cell disease. Am. J. Hematol. 2011, 87, 224–226. [Google Scholar] [CrossRef]
- Srisuwananukorn, A.; Raslan, R.; Zhang, X.; Shah, B.N.; Han, J.; Gowhari, M.; Molokie, R.E.; Gordeuk, V.R.; Saraf, S.L. Clinical, laboratory, and genetic risk factors for thrombosis in sickle cell disease. Blood Adv. 2020, 4, 1978–1986. [Google Scholar] [CrossRef]
- Colella, M.P.; De Paula, E.V.; Conran, N.; Machado-Neto, J.A.; Annicchino-Bizzacchi, J.M.; Costa, F.F.; Saad, S.T.O.; Traina, F. Hydroxyurea is associated with reductions in hypercoagulability markers in sickle cell anemia. J. Thromb. Haemost. 2012, 10, 1967–1970. [Google Scholar] [CrossRef]
- Brunetta, D.M.; De Santis, G.C.; Silva-Pinto, A.C.; de Oliveira, L.C.O.; Covas, D.T. Hydroxyurea increases plasma concentrations of microparticles and reduces coagulation activation and fibrinolysis in patients with sickle cell anemia. Acta Haematol. 2015, 133, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Gollamudi, J.; Sarvepalli, S.; Vadaparti Binf, A.; Alin, T.; Little, J.A.; Nayak, L. Venous Thromboembolism in Sickle Cell Disease is Associated with Neutrophilia. Hemoglobin 2021, 45, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Sathya, B.; Rios, R.N.; Arai, A.E.; Brofferio, A.; Thein, S.L.; Ruhl, A.P. Catheter-related right atrial thrombus in sickle cell disease. Clin. Case Rep. 2017, 5, 1898–1900. [Google Scholar] [CrossRef] [PubMed]
- Gilon, D.; Schechter, D.; Rein, A.J.; Gimmon, Z.; Or, R.; Rozenman, Y.; Slavin, S.; Gotsman, M.S.; Nagler, A. Right atrial thrombi are related to indwelling central venous catheter position: Insights into time course and possible mechanism of formation. Am. Heart J. 1998, 135, 457–462. [Google Scholar] [CrossRef]
- Stavroulopoulos, A.; Aresti, V.; Zounis, C. Right atrial thrombi complicating haemodialysis catheters. A meta-analysis of reported cases and a proposal of a management algorithm. Nephrol. Dial. Transpl. 2012, 27, 2936–2944. [Google Scholar] [CrossRef] [Green Version]
- Hariharan, N.; Brunson, A.; Mahajan, A.; Keegan, T.H.; Wun, T. Bleeding in patients with sickle cell disease: A population-based study. Blood Adv. 2020, 4, 793–802. [Google Scholar] [CrossRef]
| Thromboprophylaxis | ||||
|---|---|---|---|---|
| Variable | No (n = 25) | Yes (n = 24) | ||
| Age, years (mean (SD)) | 33.8 (9.1) | 31.9 (9.1) | ||
| <30 years | 10 (40) | 13 (54) | ||
| 30 to <40 years | 8 (32) | 7 (29) | ||
| ≥40 years | 7 (28) | 4 (17) | ||
| Sex, n (%) | Male | 9 (36) | 7 (29) | |
| Female | 16 (64) | 17 (71) | ||
| BMI, kg/m2 (mean (SD)) | 22.4 (4.2) | 24.4 (3.9) | ||
| BMI < 25 kg/m2, n (%) | 21 (84) | 14 (64) | ||
| BMI ≥ 25 kg/m2, n (%) | 4 (16) | 8 (36) | ||
| Genotype, n (%) | SS | 24 (96) | 20 (83) | |
| SC | 1 (4) | 3 (13) | ||
| Sβ0 | 0 | 1(4) | ||
| Main indication for CVAD insertion, n (%) | Transfusion | |||
| Secondary stroke prevention | 0 | 7 (29) | ||
| Primary stroke prevention | 2 (8) | 1 (4) | ||
| Vasculopathy, moyamoya-like | 5 (20) | 3 (13) | ||
| Priapism | 1 (4) | 1 (4) | ||
| Pain | 5 (20) | 4 (17) | ||
| Secondary prevention of acute chest syndrome | 2 (8) | 0 | ||
| Solid organ allograft protection | 1 (4) | 1 (4) | ||
| Pregnancy | 2 (8) | 1 (4) | ||
| Leg ulcer | 1 (4) | 1 (4) | ||
| Pulmonary hypertension | 2 (8) | 4 (17) | ||
| Other | 3 (12) | 0 | ||
| Venous access | 0 | 1 (4) | ||
| Intravenous iron chelation | 1 (4) | 0 | ||
| Type of transfusion, n (%) | Partial manual | 3 (12) | 9 (38) | |
| Automated | 19 (76) | 14 (58) | ||
| Simple transfusions | 1 (4) | 1 (4) | ||
| Other indication for CVAD | 2 (8) | 0 | ||
| CVAD type and subtype, n (%) | PICC line | 2 (8) | 4 (17) | |
| Subcutaneous port | 23 (92) | 20 (83) | ||
| Port-a-Cath | 7 (28) | 12 (50) | ||
| Vortex® | 15 (60) | 7 (29) | ||
| Xcela Power® | 1 (4) | 1 (4) | ||
| CVAD site, n (%) | Jugular | 23 (92) | 20 (83) | |
| Brachial | 2 (8) | 4 (17) | ||
| CVAD duration, years median (range) | 2.3 (0.2–8.9) | 3.5 (0.3–8.5) | ||
| Use of VTE prophylaxis, n (%) | Prophylaxis at reduced dosing | - | 10 (42) | |
| Prophylaxis at treatment dose | - | 14 (58) | ||
| Type of VTE prophylaxis, n (%) | VKA | - | 13 (54) | |
| LMWH | - | 2 (8) | ||
| ASA | - | 4 (17) | ||
| Rivaroxaban | - | 1 (4) | ||
| VKA and ASA | - | 3 (13) | ||
| Rivaroxaban and ASA | - | 1 (4) | ||
| Additional VTE risk factors, n (%) | None | 16 (64) | 14 (58) | |
| Sepsis | 1 (4) | 1 (4) | ||
| Pregnancy | 3 (12) | 1 (4) | ||
| Prior VTE | 3 (12) | 9 (38) | ||
| Medium-/high-risk surgery | 1 (4) | 1 (4) | ||
| Line infection | 2 (8) | 5 (21) | ||
| Hydroxyurea as disease-modifying therapy, n (%) | 17 (68) | 12 (50) | ||
| Ferritin, mcg/L (median (range)) | 2608.0 (16–7908) | 2114.5 (17–13065) | ||
| Ferritin < 1000 mcg/L, n (%) | 9 (36) | 10 (42) | ||
| Ferritin ≥ 1000 mcg/L, n (%) | 16 (64) | 14 (58) | ||
| Thromboprophylaxis | |||||
|---|---|---|---|---|---|
| No (n = 25) | Yes (n = 24) | ||||
| Observation period, patient-years | 62.2 | 86.2 | |||
| Events, type | Number of events | Incidence rate per 1000 implantation days (95% CI) | Number of events | Incidence rate per 1000 implantation days (95% CI) | |
| Any VTE * | 10 | 0.44 (0.28–0.70) | 4 | 0.13 (0.05–0.32) | |
| CRT | 3 | 0.13 (0.05–0.38) | 3 | 0.10 (0.03–0.28) | |
| PE | 1 | 0.04 (0.01–0.30) | 0 | 0 | |
| Atrial thrombi | 4 | 0.18 (0.07–0.43) | 0 | 0 | |
| Other & | 3 | 0.13 (0.05–0.38) | 1 | 0.03 (0.005–0.22) | |
| Univariate (n = 49) | Multivariable (n = 49) | ||||||
|---|---|---|---|---|---|---|---|
| Variable | VTE Rate Ratio | 95% CI | Significance (p-Value) | VTE Rate Ratio | 95% CI | Significance (p-Value) | |
| Thromboprophylaxis | |||||||
| Present | 1 | 1 | |||||
| Absent | 4.0 | (1.2–12.6) | 0.02 | 14.9 | (2.0–108.7) | 0.01 | |
| Age categories | |||||||
| ≥40 years | 1 | 1 | |||||
| <30 years | 0.3 | (0.1–1.5) | 0.15 | 13.4 | (0.8–230.6) | 0.07 | |
| 30 to <40 years | 0.4 | (0.1–1.8) | 0.22 | 2.7 | (0.2–38.1) | 0.47 | |
| Sex | |||||||
| Male | 1 | 1 | |||||
| Female | 0.4 | (0.1–1.2) | 0.11 | 0.8 | (0.1–4.9) | 0.81 | |
| CVAD type | |||||||
| Subcutaneous port | 1 | - | - | - | |||
| PICC line | 1.8 | (0.6–5.8) | 0.32 | - | - | - | |
| CVAD location | |||||||
| Brachial | 1 | - | - | - | |||
| Jugular | 0.6 | (0.2–1.8) | 0.32 | - | - | - | |
| CVAD subtype | |||||||
| Conventional Port-a-Cath | 1 | 1 | |||||
| PICC line | 5.8 | (1.3–25.9) | 0.02 | 34.7 | (3.5–343.6) | 0.002 | |
| Vortex and Xcela Power | 58.2 | (15.0–225.0) | <0.001 | 20.3 | (3.8–107.9) | <0.001 | |
| BMI, kg/m2 | |||||||
| BMI ≥ 25 kg/m2 | 1 | 1 | |||||
| BMI < 25 kg/m2 | 3.1 | (1.0–9.8) | 0.06 | 1.3 | (0.1–18.6) | 0.85 | |
| Ferritin (per additional mcg/L) | |||||||
| Ferritin ≥ 1000 mcg/L | 1 | - | - | - | |||
| Ferritin < 1000 mcg/L | 1.3 | (0.4–4.8) | 0.66 | - | - | - | |
| Additional VTE risk factors | |||||||
| Present | 1 | 1 | |||||
| Absent | 2.6 | (0.9–7.6) | 0.09 | 1.1 | (0.2–4.9) | 0.93 | |
| Hydroxyurea | |||||||
| Present | 1 | 1 | |||||
| Absent | 20.5 | (6.4–65.3) | <0.001 | 47.1 | (8.0–276.0) | <0.001 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forté, S.; De Luna, G.; Abdulrehman, J.; Fadiga, N.; Pestrin, O.; Pham Hung d’Alexandry d’Orengiani, A.-L.; Aneke, J.C.; Guillet, H.; Budhram, D.; Habibi, A.; et al. Thromboprophylaxis Reduced Venous Thromboembolism in Sickle Cell Patients with Central Venous Access Devices: A Retrospective Cohort Study. J. Clin. Med. 2022, 11, 1193. https://doi.org/10.3390/jcm11051193
Forté S, De Luna G, Abdulrehman J, Fadiga N, Pestrin O, Pham Hung d’Alexandry d’Orengiani A-L, Aneke JC, Guillet H, Budhram D, Habibi A, et al. Thromboprophylaxis Reduced Venous Thromboembolism in Sickle Cell Patients with Central Venous Access Devices: A Retrospective Cohort Study. Journal of Clinical Medicine. 2022; 11(5):1193. https://doi.org/10.3390/jcm11051193
Chicago/Turabian StyleForté, Stéphanie, Gonzalo De Luna, Jameel Abdulrehman, Nafanta Fadiga, Olivia Pestrin, Anne-Laure Pham Hung d’Alexandry d’Orengiani, John Chinawaeze Aneke, Henri Guillet, Dalton Budhram, Anoosha Habibi, and et al. 2022. "Thromboprophylaxis Reduced Venous Thromboembolism in Sickle Cell Patients with Central Venous Access Devices: A Retrospective Cohort Study" Journal of Clinical Medicine 11, no. 5: 1193. https://doi.org/10.3390/jcm11051193
APA StyleForté, S., De Luna, G., Abdulrehman, J., Fadiga, N., Pestrin, O., Pham Hung d’Alexandry d’Orengiani, A.-L., Aneke, J. C., Guillet, H., Budhram, D., Habibi, A., Ward, R., Bartolucci, P., & Kuo, K. H. M. (2022). Thromboprophylaxis Reduced Venous Thromboembolism in Sickle Cell Patients with Central Venous Access Devices: A Retrospective Cohort Study. Journal of Clinical Medicine, 11(5), 1193. https://doi.org/10.3390/jcm11051193







