Effectiveness of Interventions to Improve the Anticholinergic Prescribing Practice in Older Adults: A Systematic Review
Abstract
:1. Introduction
2. Methods
2.1. Data Sources and Search Strategy
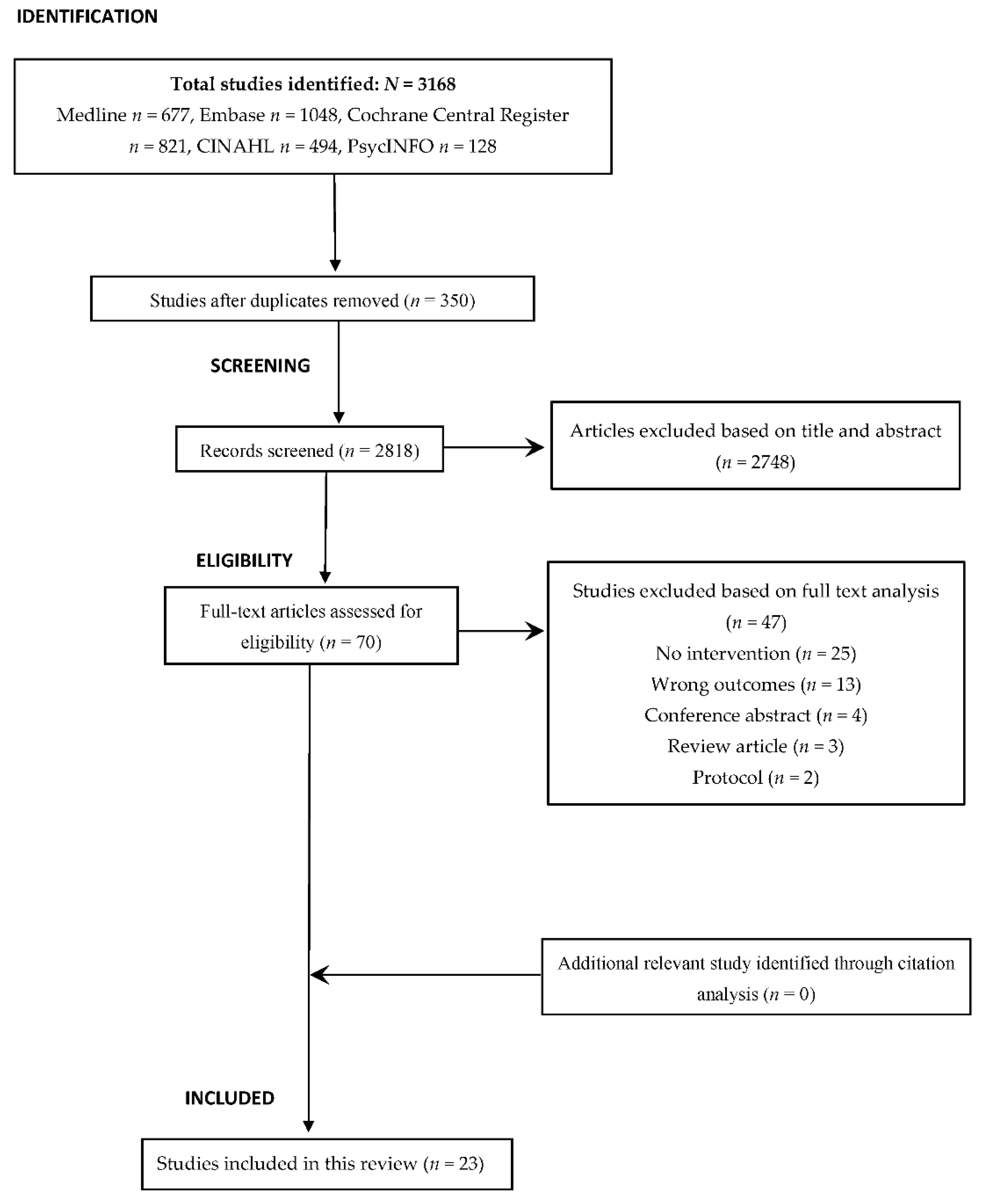
2.2. Study Screening and Selection
2.3. Inclusion Criteria
2.4. Exclusion Criteria
2.5. Data Extraction and Synthesis
2.6. Quality Assessment
3. Results
3.1. Overview of the Included Studies
| Author, Year, Country | Study Design | Intervention | Description of Intervention(s) | Effect on Outcome/Key Findings |
|---|---|---|---|---|
| Riordan et al., 2019, Ireland [47] | Convergent parallel mixed-methods design (before and after) | Academic Detailing (pharmacist-led) | Pharmacist conducted face-to-face education sessions and small focus group academic detailing sessions of 19–48 min with physicians. | Pharmacist-led academic detailing intervention was acceptable to GPs. Behavioural Change: awareness of non-pharmacological methods in treating urinary incontinence. Knowledge Gain: intervention served to refresh their knowledge |
| Ailabouni et al., 2019, New Zealand [48] | A single group (pre-and post-comparison) feasibility study | Medication review (deprescribing) | A collaborative pharmacist-led medication review with GPs was employed. New Zealand registered pharmacists used peer-reviewed deprescribing guidelines. The cumulative use of anticholinergic and sedative medicines for each participant was quantified using the DBI. | Deprescribing resulted in a significant reduction in falls, depression and frailty scores, and adverse drug reactions. No improvement in cognition and quality of life. Total regular medicines use reduced statistically, by a mean difference of 2.13 medicines per patient, among patients where deprescribing was initiated. |
| Toivo et al., 2019, Belgium [49] | Cluster RCT | Care coordination intervention (coordinated medication risk management) | Practical nurses were trained to make the preliminary medication risk assessment during home visits and report findings to the coordinating pharmacist. The coordinating pharmacist prepared the cases for the triage meeting with the physician and home care nurse to decide further actions. | No significant impact on the medication risks between the intervention and the control group. The per-protocol analysis indicated a tendency for effectiveness, particularly in optimising central nervous system medication use. |
| Hernandez et al., 2020, Spain [50] | Prospective pre-and post- interventional study | Medication review | Pharmacists reviewed the medications and detected drug-related problems using the Drug Burden Index (DBI) tool. Their recommendations were communicated to the physician via telephone, weekly meetings, and email. Further review was conducted at the weekly meeting between physician and pharmacist. | Statistically significant differences were found between pre- and post-intervention in NPI at admission, drug-related problems, MAI criteria (interactions, dosage and duplication), and mean (SD) DBI score. |
| Lenander et al., 2018, Sweden [51] | Cross-sectional | Medication Review | Clinical Pharmacist led medication review to assess the prevalence of DRPs and recommendations to discontinue, followed by team-based discussions with general practitioners (GPs) and nurses | It shows that the medication reviews decreased the use of potentially inappropriate medication. |
| Weichert et al., 2018, Finland [44] | Multicentre observational study | Medication Review | Medication review was conducted for ACB in patients at the time of admission and discharge | 21.1% of patients had their ACB reduced. There is considerable scope for improvement of prescribing practices in older people. |
| Lenander et al., 2017, Sweden [30] | Interventional pilot study | SÄKLÄK project, a developed intervention model | Multi-professional intervention model created to improve medication safety for elderly | Significant decrease in the prescription of anticholinergic drugs indicated the SÄKLÄK intervention is effective in reducing potential DRPs |
| Moga et al., 2017, USA [29] | Parallel arm Randomised Interventional study | Targeted medication therapy management intervention | Targeted patient-centred pharmacist–physician team medication therapy management intervention was used to reduce the use of inappropriate anticholinergic medications in older patients. | The targeted medication therapy management intervention resulted in improvement in anticholinergic medication appropriateness and reduced the use of inappropriate anticholinergic medications in older patients. |
| Lagrange et al., 2017, France [52] | Retrospective study | A context-aware pharmaceutical analysis tool | A context-aware computerised decision-support system designed to automatically compare prescriptions recorded in computerised patient files against the main consensual guidelines for medical management in older adults. | Prescription of anticholinergics was significantly decreased (28%). |
| Carnahan et al., 2017, USA [39] | Quasi-experimental study design | Educational program on medication use | IA-ADAPT/CMS Partnership is an evidence-based training program to improve dispensing drugs for elderly | Suggests that the IA-ADAPT and the CMS Partnership improved medication use with no adverse impact on BPSD. |
| Hanus et al., 2016, USA [40] | Observational Pilot study | Pharmacist-led EHR-based population health initiative and ARS Service | Physicians in the primary care settings could communicate with pharmacists employing a shared EHR. As part of a quality improvement project, a pharmacist-led EHR-based medication therapy recommendation service was implemented at 2 DHS medical clinics to reduce the anticholinergic burden | High recommendation acceptance rates were achieved using objective anticholinergic risk assessment and algorithm-driven medication therapy recommendations. |
| McLarin et al., 2016, Australia [43] | Retrospective study | RMMR | Impact of RMMRs on anticholinergic burden quantified by seven anticholinergic risk scales | Demonstrated that RMMRs are effective in reducing ACM prescribing in elderly |
| Kersten et al., 2015, Norway [46] | Retrospective study | Medication review | Investigated the clinical impact of PIMs in acutely hospitalised older adults. | Anticholinergic prescriptions were reduced from 39.2% to 37.9% |
| Juola et al., 2015, Finland [45] | Cluster RCT | Educational intervention | Nursing staff working in the intervention wards received two 4-h interactive training sessions based on constructive learning theory to recognise harmful medications and adverse drug events. | No significant differences in the change in prevalence of anticholinergic drugs. |
| Kersten et al., 2013, Norway [21] | RCT | Multidisciplinary drug review | Single Blind MDRD was conducted that recruited long-term nursing home residents with a total ADS score of greater than or equal to 3 | After 8 weeks, the median ADS score was significantly reduced from 4 to 2 in the intervention group. The largest improvement in immediate recall after 8 weeks was observed in the five patients in the intervention group who had their ADS score reduced to 0 |
| Ghibelli et al., 2013, Italy [53] | Pre, post-intervention study | INTERcheck CPSS | INTERcheck is a CPSS developed to optimise drug prescription for older people with multimorbidity and minimise the occurrence of adverse drug reactions. | The use of INTERCheck was associated with a significant reduction in PIMs and new-onset potentially severe DDIs. |
| Yeh et al., 2013, Taiwan [54] | Prospective case-control study | Educational program for primary care physicians | Educational program for primary care physicians serving in Veterans’ Homes, focusing on anticholinergic adverse reactions in geriatrics and the CR-ACHS | CR-ACHS was significantly reduced in the intervention group at 12-week follow-up. |
| Boustani et al., 2012, USA [41] | RCT | CDSS Alert (anticholinergic discontinuation) | CDSS alert system sends an interruptive alert if any of the 18 anticholinergics were prescribed, recommending stopping the drug, suggesting an alternative, or recommending dose modification. | Physicians receiving the CDSS issued more discontinuation orders of definite anticholinergics, but the results were not statistically significant. Results suggest that human interaction may play an important role in accepting recommendations aimed at improving the care of hospitalised older adults with CI. |
| Gnjidic et al., 2010, Australia [23] | Cluster RCT | Medication review | The study intervention included a letter and phone call to GPs, using DBI to prompt them to consider dose reduction or cessation of anticholinergic and sedative medications. | At follow-up, a DBI change was observed in 16 participants. DBI decreased in 12 participants, 6 (19%) in the control group, and 6 (32%) in the intervention group. |
| Castelino et al., 2010, Australia [22] | Retrospective study | Medication reviews by pharmacist | HMR by pharmacists for leads to an improvement in the use of medications | DBI and PIMs identified in 60.5% and 39.8% of the patients. Significant reduction in the cumulative DBI scores for all patients was observed following pharmacists’ recommendations |
| Starner et al., 2009, USA [38] | Retrospective study | Educational Intervention | Intervention letters were mailed to the physicians for patients having ≥1 DAE claim | Noticeable decrease was observed after a 6-month follow-up of the intervention in the reduction of DAE claims (48.8%) specifically reduction of anticholinergics (66.7%) was highest |
| Nishtala et al., 2009, Australia [42] | Retrospective study | RMMR | Clinical Pharmacist-led medication review decreased the DBI in older people | GP’s uptake of recommendations made by pharmacists resulted in a decrease in DBI score. Clinical pharmacist-conducted medication reviews can reduce prescribing of anticholinergic drugs and significantly decrease the DBI score of the study population. |
| van Eijk et al., 2001, Netherlands [55] | RCT | Educational visits as an individual and a group for general practitioners and pharmacists | Educational visits used academic detailing to discuss prescribing of highly anticholinergic antidepressants in elderly people. | The rate of starting anticholinergic antidepressants in the elderly reduced 26% (in the individual intervention) and 45% (in the group intervention) The use of less anticholinergic antidepressants increased by 40% and 29%, respectively |
| Author, Year, Country | Study Design | Setting | Sample Size | Mean Age (Years) | Gender (Female %) | Study Duration | Follow-Up | Relevant Outcome(s) | Outcome Measure | Significant Association (±) | Statistical Tests | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control/Pre | Intervention/Post | |||||||||||
| Riordan et al., 2019, Ireland [47] | Convergent parallel mixed-methods design (before and after) | General Practice | 154 | 75.0 | 72.1 | 5 months | 6 months | Effects on DBI and ACB scores | Patients having an ACB score of 0 (34%) | Patients having an ACB score of 0 (31%) 65% of patients did not show any change in DBI over time | − | SD, Range, IQR, Frequency, Percentages |
| Ailabouni et al., 2019, New Zealand [48] | A single group (pre- and post-comparison) feasibility study | Residential care facilities | 46 | 65.0 | 74.0 | 6 months | 2 weeks | Reduction in DBI score | ≥0.5 (median DBI) | 0.34 (median DBI) | + | Wilcox-signed Rank test (WSR) t-test Fisher’s exact test |
| Toivo et al., 2019, Belgium [49] | Cluster RCT | Primary care | 129 | 82.8 | 69.8 | 1 year | 1 year | Anticholinergic use | 18.8% (Anticholinergic use at baseline) 18.8% (Anticholinergic use at 12 months) | 29.6% (Anticholinergic use at baseline) 18.5% (Anticholinergic use at 12 months) | − | Binary logistic regression, two-sided statistical tests |
| Hernandez et al., 2020, Spain [50] | Prospective pre- and post-interventional study | Intermediate care hospital | 55 | 84.6 | 60.0 | 12 months | NA | Anticholinergic burden per Drug Burden Index (DBI) | 1.38 ± 0.7 (Mean DBI) | 1.08 ± 0.7 (Mean DBI) | + | Kolmogorov–Smirnov test Student’s t-test |
| Lenander et al., 2018, Sweden [51] | Cross-sectional | Primary care | 1720 | 87.5 | 74.5 | 1 year | 8 weeks | Discontinuation of DRPs | Pts with anticholinergics = 9.2% | Pts with anticholinergics = 4.2% | + | Student’s t-test, Chi-square |
| Weichert et al., 2018, Finland [44] | Observational study | Hospital | 549 | 79.6 | 58.3 | 1 year, 5 months | 30 days | Reduction in ACB Score during the hospital stay | Patients on DAPs on admission = 60.8% | Patients on DAPs on discharge = 57.7 | − | Shapiro–Wilk test, Wilcoxon signed-rank test,2 sample t-test, Yates and Pearson’s chi-square test multivariate binary logistic regression |
| Lenander et al., 2017 Sweden [30] | Interventional pilot study | Primary care | 2400 to 13,700 patients (estimated) | 65–79 (range) | 63 | 9 months | 6 months | Reduction in anticholinergic PIMs (before/after) | Anticholinergic prescriptions before intervention (4513) | Anticholinergic prescriptions after intervention (3824) | + | Chi-square test |
| Moga et al., 2017, USA [29] | Parallel arm Randomised Interventional study | Alzheimer’s Disease Center | 49 | 77.7 ± 6.6 | 70.0 | 1 year | 8 weeks | Significant reduction in anticholinergic drug scale (ADS) Score | 1.0 (0.3) | 0.2 (0.3) | + | Student’s t-tests (or Wilcoxon rank-sum tests for non-normally distributed variables), Chi-square or Fisher’s exact tests |
| Lagrange et al., 2017, France [52] | Retrospective study | Hospital | 187 | 73.9 | 63.1 | 10.5 months | 33 and 37 days | Change in number of prescriptions | 6538 doses (Anticholinergics) | 4696 doses (Anticholinergics) | + | Descriptive statistics |
| Carnahan et al., 2017, USA [39] | Quasi-experimental study design | Nursing home | 411 | 86.7 | 77.0 | 1 year 9 months | 276 days | Anticholinergic use | Mean (SD) 35.9% (12.0%) | Mean (SD) 36.1% (10.9%) | − | Generalised linear mixed logistic regression |
| Antipsychotic use | Mean (SD) 17.7% (10.4%) | Mean (SD) 20.7% (10.6%) | + | |||||||||
| Hanus et al., 2016, USA [40] | Observational Pilot study | Medical clinics | 59 | 77 ± 9.3 | 51.0 | 2 months | 2 weeks | Reduction in ACB Score, Increased medication acceptance rate | 1.08 50% | 0.89 95% | + | Generalised linear mixed-effects model, paired t-test |
| McLarin et al., 2016, Australia [43] | Retrospective study. | Aged care facilities | 814 | 85.6 | 69.6 | NA | NA | Reduction in anticholinergic medications after a medication review | Mean (SD) 3.73 (1.46) | Mean (SD) 3.32 (1.7) | + | Wilcoxon signed-rank test, ANOVA |
| Kersten et al., 2015, Norway [46] | Retrospective study | Hospital | 232 | 86.1 | 59.1 | 8 months | 1 year | Reduction in anticholinergic prescriptions | Prevalence of anticholinergic drugs was significantly reduced (p < 0.02) | + | Paired samples Student’s t-test, McNamar’s test, Mann–Whitney U tests, ANOVA, linear regression | |
| Juola et al., 2015, Finland [45] | Cluster RCT | Assisted living facilities | 227 | 83.0 | 70.9 | 1 year | 1 year | Mean Anticholinergic drugs | 1.0 (Mean Anticholinergic drugs) | 1.2 (Mean Anticholinergic drugs) | − | t-tests, Mann–Whitney U tests, or Chi-square tests, GEE models, Poisson regression models |
| Kersten et al., 2013, Norway [21] | RCT | Nursing home | 87 | 85.0 | 39.0 | 8 weeks | 8 weeks | Marked reduction in ADS score | Median = 4 | Median = 2 | + | ANCOVA, Poisson regression analysis |
| Ghibelli et al., 2013, Italy [53] | Pre- and post-intervention study | Hospital | 75 for Pre 75 for Post | 81 | 58.3 | 4 months | NA | Reduction in ACB score | 1.3 | 1.1 | − | Pearson Chi-square test, Student’s t-test |
| Yeh et al., 2013, Taiwan [54] | Prospective case-control | Veteran Home | 67 | 83.4 | NA | 12 weeks | 12 weeks | Anticholinergic Burden (CR-ACHS) | 1.0 ± 1.1 (Mean CR-ACHS) | −0.5 ± 1.1 (Mean CR-ACHS) | + | Wilcoxon signed ranks test |
| Boustani et al.,2012, USA [41] | RCT | Hospital | 424 | 74.8 | 68.0 | 21 months | At the time of discharge | Discontinuation of AC prescriptions | anticholinergic discontinued = 31.2% | anticholinergic discontinued = 48.9% | _ | Fisher’s exact test, t-test, logistic regression, multiple regression |
| Gnjidic et al., 2010, Australia [23] | Cluster RCT | Self-care retirement village | 115 | 84.3 | 73.0 | 13 months | 3 months | Drug Burden Index (DBI) | 0.26 ± 0.34 (mean DBI) | 0.22 ± 0.42 (mean DBI) | − | Kolmogorov–Smirnov test Mann–Whitney nonparametric test X2 test |
| Castelino et al., 2010, Australia [22] | A retrospective analysis of medication reviews | Community-dwelling | 372 | 76.1 | 55.0 | NA | NA | Impact of pharmacist’s on DBI scores | Sum of DBI scores = 206.86 | Sum of DBI scores = 157.26 | + | Wilcoxon signed-rank test |
| Starner et al., 2009, USA [38] | Retrospective study | Pharmacy claims data | 10,364 | 65.0 | NA | 8 months | 6 months | Rate of discontinued anticholinergics | NA | 66.7% | + | NA |
| Nishtala et al., 2009, Australia [42] | Retrospective study | Aged care homes | 500 | 84.0 | 75.0 | 6 months | 2 months | Significant decrease in DBI score | NA | 12% decrease in DBI | + | 2-tailed Wilcoxon signed-rank test |
| van Eijk et al., 2001, Netherlands [55] | RCT | Primary care | 46,078 | 71 | 58.0 | 1 year | NA | Reduction in the prescribing of anticholinergics | 30% reduction in the rate of starting highly anticholinergic antidepressant in the individual intervention arms compared with the control arm | 40% reduction in the rate of starting highly anticholinergic antidepressants in the group intervention arms compared with the control arm | + | Poisson regression model |
3.2. Methodological Quality of Studies
3.3. Intervention Characteristics
3.4. Effectiveness of Interventions at Improving Anticholinergic Prescribing Practice
4. Discussion
4.1. Implications for Clinical Practice and Future Research
4.2. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Buxton, I.L.O. Principles of Prescription Order Writing and Patient Compliance. In Goodman &, Gilman’s: The Pharmacological Basis of Therapeutics, 12th ed.; Brunton, L.L., Chabner, B.A., Knollmann, B.C., Eds.; McGraw-Hill Education: New York, NY, USA, 2015. [Google Scholar]
- Likic, R.; Maxwell, S.R. Prevention of medication errors: Teaching and training. Br. J. Clin. Pharmacol. 2009, 67, 656–661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drag, L.L.; Wright, S.L.; Bieliauskas, L.A. Prescribing Practices of Anticholinergic Medications and Their Association with Cognition in an Extended Care Setting. J. Appl. Gerontol. 2012, 31, 239–259. [Google Scholar] [CrossRef]
- Barton, C.; Sklenicka, J.; Sayegh, P.; Yaffe, K. Contraindicated medication use among patients in a memory disorders clinic. Am. J. Geriatr. Pharmacother. 2008, 6, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Buck, M.D.; Atreja, A.; Brunker, C.P.; Jain, A.; Suh, T.T.; Palmer, R.M.; Dorr, D.A.; Harris, C.M.; Wilcox, A.B. Potentially inappropriate medication prescribing in outpatient practices: Prevalence and patient characteristics based on electronic health records. Am. J. Geriatr. Pharmacother. 2009, 7, 84–92. [Google Scholar] [CrossRef]
- Corsonello, A.; Pedone, C.; Lattanzio, F.; Lucchetti, M.; Garasto, S.; Di Muzio, M.; Giunta, S.; Onder, G.; Di Iorio, A.; Volpato, S.; et al. Potentially Inappropriate Medications and Functional Decline in Elderly Hospitalized Patients. J. Am. Geriatr. Soc. 2009, 57, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Hale, L.; Griffin, A.; Cartwright, O.; Moulin, J.; Alford, S.; Fleming, R. Potentially inappropriate medication use in hospitalized older adults: A DUE using the full Beers criteria. Formulary 2008, 43, 326–336. [Google Scholar]
- Rajska-Neumann, A.; Wieczorowska-Tobis, K. Polypharmacy and potential inappropriateness of pharmaco-logical treatment among community-dwelling elderly patients. Arch. Gerontol. Geriatr. 2007, 44 (Suppl. S1), 303–309. [Google Scholar] [CrossRef]
- Gallagher, P.F.; Barry, P.J.; Ryan, C.; Hartigan, I.; O’Mahony, D. Inappropriate prescribing in an acutely ill population of elderly patients as determined by Beers’ Criteria. Age Ageing 2008, 37, 96–101. [Google Scholar] [CrossRef] [Green Version]
- Ruxton, K.; Woodman, R.J.; Mangoni, A.A. Drugs with anticholinergic effects and cognitive impairment, falls and all-cause mortality in older adults: A systematic review and meta-analysis. Br. J. Clin. Pharmacol. 2015, 80, 209–220. [Google Scholar] [CrossRef]
- Van Eijk, M.E.; Bahri, P.; Dekker, G.; Herings, R.M.; Porsius, A.; Avorn, J.; De Boer, A. Use of prevalence and incidence measures to describe age-related prescribing of antidepressants with and without anticholinergic effects. J. Clin. Epidemiol. 2000, 53, 645–651. [Google Scholar] [CrossRef]
- Campbell, N.; Boustani, M.; Limbil, T.; Ott, C.; Fox, C.; Maidment, I.; Schubert, C.C.; Munger, S.; Fick, D.; Miller, D.; et al. The cognitive impact of anticholinergics: A clinical review. Clin. Interv. Aging 2009, 4, 225–233. [Google Scholar] [PubMed] [Green Version]
- Rudolph, J.L.; Salow, M.J.; Angelini, M.C.; McGlinchey, R.E. The anticholinergic risk scale and anticholinergic adverse effects in older persons. Arch. Intern. Med. 2008, 168, 508–513. [Google Scholar] [CrossRef] [Green Version]
- Kersten, H.; Wyller, T.B. Anticholinergic drug burden in older people’s brain—How well is it measured? Basic Clin. Pharmacol. Toxicol. 2014, 114, 151–159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nishtala, P.S.; Salahudeen, M.S.; Hilmer, S.N. Anticholinergics: Theoretical and clinical overview. Expert Opin. Drug. Saf. 2016, 15, 753–768. [Google Scholar] [CrossRef] [PubMed]
- Salahudeen, M.S.; Nishtala, P.S. Examination and Estimation of Anticholinergic Burden: Current Trends and Implications for Future Research. Drugs Aging 2016, 33, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Peterson, G.M.; Bereznicki, L.R.; Salahudeen, M.S. Association between anticholinergic drug burden and mortality in older people: A systematic review. Eur. J. Clin. Pharmacol. 2020, 76, 319–335. [Google Scholar] [CrossRef] [PubMed]
- Tune, L.E. Anticholinergic effects of medication in elderly patients. J. Clin. Psychiatry 2001, 62 (Suppl. S21), 11–14. [Google Scholar]
- Alharafsheh, A.; Alsheikh, M.; Ali, S.; Baraiki, A.A.; Alharbi, G.; Alhabshi, T.; Aboutaleb, A. A retrospective cross-sectional study of antibiotics prescribing patterns in admitted patients at a tertiary care setting in the KSA. Int. J. Health Sci. 2018, 12, 67–71. [Google Scholar]
- Gorup, E.; Rifel, J.; Petek Šter, M. Anticholinergic Burden and Most Common Anticholinergic-acting Medicines in Older General Practice Patients. Zdr Varst. 2018, 57, 140–147. [Google Scholar] [CrossRef] [Green Version]
- Kersten, H.; Molden, E.; Tolo, I.K.; Skovlund, E.; Engedal, K.; Wyller, T.B. Cognitive effects of reducing anticholinergic drug burden in a frail elderly population: A randomized controlled trial. J. Gerontol. A. Biol. Sci. Med. Sci. 2013, 68, 271–278. [Google Scholar] [CrossRef] [Green Version]
- Castelino, R.L.; Hilmer, S.N.; Bajorek, B.V.; Nishtala, P.; Chen, T.F. Drug Burden Index and potentially inappropriate medications in community-dwelling older people: The impact of Home Medicines Review. Drugs Aging 2010, 27, 135–148. [Google Scholar] [CrossRef] [PubMed]
- Gnjidic, D.; Le Couteur, D.G.; Abernethy, D.R.; Hilmer, S.N. A pilot randomized clinical trial utilizing the drug burden index to reduce exposure to anticholinergic and sedative medications in older people. Ann. Pharmacother. 2010, 44, 1725–1732. [Google Scholar] [CrossRef] [PubMed]
- Tay, H.S.; Soiza, R.L.; Mangoni, A.A. Minimizing anticholinergic drug prescribing in older hospitalized patients: A full audit cycle. Ther. Adv. Drug Saf. 2014, 5, 121–128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Meer, H.G.; Wouters, H.; Pont, L.G.; Taxis, K. Reducing the anticholinergic and sedative load in older patients on polypharmacy by pharmacist-led medication review: A randomised controlled trial. BMJ Open 2018, 8, e019042. [Google Scholar] [CrossRef] [Green Version]
- Christensen, M.; Lundh, A. Medication review in hospitalised patients to reduce morbidity and mortality. Cochrane Database Syst. Rev. 2016, 2, Cd008986. [Google Scholar] [CrossRef]
- Holland, R.; Desborough, J.; Goodyer, L.; Hall, S.; Wright, D.; Loke, Y.K. Does pharmacist-led medication review help to reduce hospital admissions and deaths in older people? A systematic review and meta-analysis. Br. J. Clin. Pharmacol. 2008, 65, 303–316. [Google Scholar] [CrossRef] [Green Version]
- Wallerstedt, S.M.; Kindblom, J.M.; Nylén, K.; Samuelsson, O.; Strandell, A. Medication reviews for nursing home residents to reduce mortality and hospitalization: Systematic review and meta-analysis. Br. J. Clin. Pharmacol. 2014, 78, 488–497. [Google Scholar] [CrossRef]
- Moga, D.C.; Abner, E.L.; Rigsby, D.N.; Eckmann, L.; Huffmyer, M.; Murphy, R.R.; Coy, B.B.; Jicha, G.A. Optimizing medication appropriateness in older adults: A randomized clinical interventional trial to decrease anticholinergic burden. Alzheimers Res. Ther. 2017, 9, 36. [Google Scholar] [CrossRef]
- Lenander, C.; Bondesson, Å.; Viberg, N.; Jakobsson, U.; Beckman, A.; Midlöv, P. Effects of an intervention (SÄKLÄK) on prescription of potentially inappropriate medication in elderly patients. Fam. Pract. 2017, 34, 213–218. [Google Scholar] [CrossRef]
- Aronson, J.K. Medication errors: Definitions and classification. Br. J. Clin. Pharmacol. 2009, 67, 599–604. [Google Scholar] [CrossRef] [Green Version]
- Velo, G.P.; Minuz, P. Medication errors: Prescribing faults and prescription errors. Br. J. Clin. Pharmacol. 2009, 67, 624–628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ali, S.; Salahudeen, M.S.; Bereznicki, L.R.E.; Curtain, C.M. Pharmacist-led interventions to reduce adverse drug events in older people living in residential aged care facilities: A systematic review. Br. J. Clin. Pharmacol. 2021, 87, 3672–3689. [Google Scholar] [CrossRef] [PubMed]
- Guise, J.M.; Chang, C.; Viswanathan, M.; Glick, S.; Treadwell, J.; Umscheid, C.A.; Whitlock, E.; Fu, R.; Berliner, E.; Paynter, R.; et al. Agency for Healthcare Research and Quality Evidence-based Practice Center methods for systematically reviewing complex multicomponent health care interventions. J. Clin. Epidemiol. 2014, 67, 1181–1191. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G. Assessing risk of bias in included studies. In Cochrane Handbook for Systematic Reviews of Interventions; Higgins, J.P.T., Green, S., Eds.; Wiley-Blackwell: Chichester, UK, 2011; pp. 187–242. [Google Scholar]
- Wells, G.A.; O’Connell, B.S.D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomized Studies in Meta-Analysis. Available online: www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 25 December 2021).
- Starner, C.I.; Norman, S.A.; Reynolds, R.G.; Gleason, P.P. Effect of a retrospective drug utilization review on potentially inappropriate prescribing in the elderly. Am. J. Geriatr. Pharmacother. 2009, 7, 11–19. [Google Scholar] [CrossRef]
- Carnahan, R.M.; Brown, G.D.; Letuchy, E.M.; Rubenstein, L.M.; Gryzlak, B.M.; Smith, M.; Reist, J.C.; Kelly, M.W.; Schultz, S.K.; Weckmann, M.T.; et al. Impact of programs to reduce antipsychotic and anticholinergic use in nursing homes. Alzheimers Dement. 2017, 3, 553–561. [Google Scholar] [CrossRef]
- Hanus, R.J.; Lisowe, K.S.; Eickhoff, J.C.; Kieser, M.A.; Statz-Paynter, J.L.; Zorek, J.A. Evaluation of a pharmacist-led pilot service based on the anticholinergic risk scale. J. Am. Pharm. Assoc. 2016, 56, 555–561. [Google Scholar] [CrossRef]
- Boustani, M.A.; Campbell, N.L.; Khan, B.A.; Abernathy, G.; Zawahiri, M.; Campbell, T.; Tricker, J.; Hui, S.L.; Buckley, J.D.; Perkins, A.J.; et al. Enhancing care for hospitalized older adults with cognitive impairment: A randomized controlled trial. J. Gen. Intern. Med. 2012, 27, 561–567. [Google Scholar] [CrossRef] [Green Version]
- Nishtala, P.S.; Hilmer, S.N.; McLachlan, A.J.; Hannan, P.J.; Chen, T.F. Impact of residential medication management reviews on drug burden index in aged-care homes: A retrospective analysis. Drugs Aging 2009, 26, 677–686. [Google Scholar] [CrossRef]
- McLarin, P.E.; Peterson, G.M.; Curtain, C.M.; Nishtala, P.S.; Hannan, P.J.; Castelino, R.L. Impact of residential medication management reviews on anticholinergic burden in aged care residents. Curr. Med. Res. Opin. 2016, 32, 123–131. [Google Scholar] [CrossRef]
- Weichert, I.; Romero-Ortuno, R.; Tolonen, J.; Soe, T.; Lebus, C.; Choudhury, S.; Nadarajah, C.V.; Nanayakkara, P.; Orrù, M.; Di Somma, S.; et al. Anticholinergic medications in patients admitted with cognitive impairment or falls (AMiCI). The impact of hospital admission on anticholinergic cognitive medication burden. Results of a multicentre observational study. J. Clin. Pharm. Ther. 2018, 43, 682–694. [Google Scholar] [CrossRef] [PubMed]
- Juola, A.L.; Bjorkman, M.P.; Pylkkanen, S.; Finne-Soveri, H.; Soini, H.; Kautiainen, H.; Bell, J.S.; Pitkala, K.H. Nurse Education to Reduce Harmful Medication Use in Assisted Living Facilities: Effects of a Randomized Controlled Trial on Falls and Cognition. Drugs Aging 2015, 32, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Kersten, H.; Hvidsten, L.T.; Gløersen, G.; Wyller, T.B.; Wang-Hansen, M.S. Clinical impact of potentially inappropriate medications during hospitalization of acutely ill older patients with multimorbidity. Scand. J. Prim. Health Care 2015, 33, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Riordan, D.O.; Hurley, E.; Sinnott, C.; Galvin, R.; Dalton, K.; Kearney, P.M.; Halpin, J.D.; Byrne, S. Pharmacist-led academic detailing intervention in primary care: A mixed methods feasibility study. Int. J. Clin. Pharm. 2019, 41, 574–582. [Google Scholar] [CrossRef]
- Ailabouni, N.; Mangin, D.; Nishtala, P.S. DEFEAT-polypharmacy: Deprescribing anticholinergic and sedative medicines feasibility trial in residential aged care facilities. Int. J. Clin. Pharm. 2019, 41, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Toivo, T.; Airaksinen, M.; Dimitrow, M.; Savela, E.; Pelkonen, K.; Kiuru, V.; Suominen, T.; Uunimäki, M.; Kivelä, S.L.; Leikola, S.; et al. Enhanced coordination of care to reduce medication risks in older home care clients in primary care: A randomized controlled trial. BMC Geriatr. 2019, 19, 332. [Google Scholar] [CrossRef]
- Hernandez, M.; Mestres, C.; Junyent, J.; Costa-Tutusaus, L.; Modamio, P.; Lastra, C.F.; Mariño, E.L. Effects of a multifaceted intervention in psychogeriatric patients: One-year prospective study. Eur. J. Hosp. Pharm. 2020, 27, 226–231. [Google Scholar] [CrossRef]
- Lenander, C.; Bondesson, Å.; Viberg, N.; Beckman, A.; Midlöv, P. Effects of medication reviews on use of potentially inappropriate medications in elderly patients, a cross-sectional study in Swedish primary care. BMC Health Serv. Res. 2018, 18, 616. [Google Scholar] [CrossRef]
- Lagrange, F.; Lagrange, J.; Bennaga, C.; Taloub, F.; Keddi, M.; Dumoulin, B. A context-aware decision-support system in clinical pharmacy: Drug monitoring in the elderly. Le Pharmacien Hospitalier et Clinicien 2017, 52, 100–110. [Google Scholar] [CrossRef]
- Ghibelli, S.; Marengoni, A.; Djade, C.D.; Nobili, A.; Tettamanti, M.; Franchi, C.; Caccia, S.; Giovarruscio, F.; Remuzzi, A.; Pasina, L. Prevention of inappropriate prescribing in hospitalized older patients using a computerized prescription support system (INTERcheck(®)). Drugs Aging 2013, 30, 821–828. [Google Scholar] [CrossRef]
- Yeh, Y.C.; Liu, C.L.; Peng, L.N.; Lin, M.H.; Chen, L.K. Potential benefits of reducing medication-related anticholinergic burden for demented older adults: A prospective cohort study. Geriatr. Gerontol. Int. 2013, 13, 694–700. [Google Scholar] [CrossRef] [PubMed]
- van Eijk, M.E.; Avorn, J.; Porsius, A.J.; de Boer, A. Reducing prescribing of highly anticholinergic antidepressants for elderly people: Randomised trial of group versus individual academic detailing. BMJ 2001, 322, 654–657. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fox, C.; Smith, T.; Maidment, I.; Chan, W.Y.; Bua, N.; Myint, P.K.; Boustani, M.; Kwok, C.S.; Glover, M.; Koopmans, I.; et al. Effect of medications with anti-cholinergic properties on cognitive function, delirium, physical function and mortality: A systematic review. Age Ageing 2014, 43, 604–615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riordan, D.O.; Walsh, K.A.; Galvin, R.; Sinnott, C.; Kearney, P.M.; Byrne, S. The effect of pharmacist-led interventions in optimising prescribing in older adults in primary care: A systematic review. SAGE Open Med. 2016, 4, 2050312116652568. [Google Scholar] [CrossRef] [Green Version]
- Øyane, N.M.F.; Finckenhagen, M.; Ruths, S.; Thue, G.; Lindahl, A.K. Improving drug prescription in general practice using a novel quality improvement model. Scand. J. Prim. Health Care. 2021, 39, 174–183. [Google Scholar] [CrossRef]
- van der Meer, H.G.; Wouters, H.; van Hulten, R.; Pras, N.; Taxis, K. Decreasing the load? Is a Multidisciplinary Multistep Medication Review in older people an effective intervention to reduce a patient’s Drug Burden Index? Protocol of a randomised controlled trial. BMJ Open 2015, 5, e009213. [Google Scholar] [CrossRef]
- Gurwitz, J.H.; Field, T.S.; Judge, J.; Rochon, P.; Harrold, L.R.; Cadoret, C.; Lee, M.; White, K.; LaPrino, J.; Erramuspe-Mainard, J.; et al. The incidence of adverse drug events in two large academic long-term care facilities. Am. J. Med. 2005, 118, 251–258. [Google Scholar] [CrossRef]
- Martin, R.M.; Lunec, S.G.; Rink, E. UK postal survey of pharmacists working with general practices on prescribing issues: Characteristics, roles and working arrangements. Int. J. Pharm. Pract. 1998, 6, 133–139. [Google Scholar] [CrossRef]
- Veggeland, T.; Dyb, S. The contribution of a clinical pharmacist to the improvement of medication at a geriatric hospital unit in Norway. Pharm. Pract. 2008, 6, 20–24. [Google Scholar] [CrossRef] [Green Version]
- Romskaug, R.; Skovlund, E.; Straand, J.; Molden, E.; Kersten, H.; Pitkala, K.H.; Lundqvist, C.; Wyller, T.B. Effect of Clinical Geriatric Assessments and Collaborative Medication Reviews by Geriatrician and Family Physician for Improving Health-Related Quality of Life in Home-Dwelling Older Patients Receiving Polypharmacy: A Cluster Randomized Clinical Trial. JAMA Intern. Med. 2020, 180, 181–189. [Google Scholar] [CrossRef]
- Gallagher, P.F.; O’Connor, M.N.; O’Mahony, D. Prevention of potentially inappropriate prescribing for elderly patients: A randomized controlled trial using STOPP/START criteria. Clin. Pharmacol. Ther. 2011, 89, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Clyne, B.; Smith, S.M.; Hughes, C.M.; Boland, F.; Bradley, M.C.; Cooper, J.A.; Fahey, T. Effectiveness of a Multifaceted Intervention for Potentially Inappropriate Prescribing in Older Patients in Primary Care: A Cluster-Randomized Controlled Trial (OPTI-SCRIPT Study). Ann. Fam. Med. 2015, 13, 545–553. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pasina, L.; Marengoni, A.; Ghibelli, S.; Suardi, F.; Djade, C.D.; Nobili, A.; Franchi, C.; Guerrini, G. A Multicomponent Intervention to Optimize Psychotropic Drug Prescription in Elderly Nursing Home Residents: An Italian Multicenter, Prospective, Pilot Study. Drugs Aging 2016, 33, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Jaam, M.; Naseralallah, L.M.; Hussain, T.A.; Pawluk, S.A. Pharmacist-led educational interventions provided to healthcare providers to reduce medication errors: A systematic review and meta-analysis. PLoS ONE 2021, 16, e0253588. [Google Scholar] [CrossRef]
- García-Gollarte, F.; Baleriola-Júlvez, J.; Ferrero-López, I.; Cuenllas-Díaz, Á.; Cruz-Jentoft, A.J. An educational intervention on drug use in nursing homes improves health outcomes resource utilization and reduces inappropriate drug prescription. J. Am. Med. Dir. Assoc. 2014, 15, 885–891. [Google Scholar] [CrossRef]
- Trivalle, C.; Cartier, T.; Verny, C.; Mathieu, A.M.; Davrinche, P.; Agostini, H.; Becquemont, L.; Demolis, P. Identifying and preventing adverse drug events in elderly hospitalised patients: A randomised trial of a program to reduce adverse drug effects. J. Nutr. Health Aging 2010, 14, 57–61. [Google Scholar] [CrossRef]
- López-Álvarez, J.; Sevilla-Llewellyn-Jones, J.; Agüera-Ortiz, L. Anticholinergic Drugs in Geriatric Psychopharmacology. Front Neurosci. 2019, 13, 309. [Google Scholar] [CrossRef] [Green Version]
- Kamarudin, G.; Penm, J.; Chaar, B.; Moles, R. Educational interventions to improve prescribing competency: A systematic review. BMJ Open 2013, 3, e003291. [Google Scholar] [CrossRef] [Green Version]
- Picton, C.; Loughrey, C.; Webb, A. The need for a prescribing competency framework to address the burden of complex polypharmacy among multiple long-term conditions. Clin. Med. 2016, 16, 470–474. [Google Scholar] [CrossRef] [Green Version]
- Salahudeen, M.S.; Duffull, S.B.; Nishtala, P.S. Anticholinergic burden quantified by anticholinergic risk scales and adverse outcomes in older people: A systematic review. BMC Geriatr. 2015, 15, 31. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salahudeen, M.S.; Alfahmi, A.; Farooq, A.; Akhtar, M.; Ajaz, S.; Alotaibi, S.; Faiz, M.; Ali, S. Effectiveness of Interventions to Improve the Anticholinergic Prescribing Practice in Older Adults: A Systematic Review. J. Clin. Med. 2022, 11, 714. https://doi.org/10.3390/jcm11030714
Salahudeen MS, Alfahmi A, Farooq A, Akhtar M, Ajaz S, Alotaibi S, Faiz M, Ali S. Effectiveness of Interventions to Improve the Anticholinergic Prescribing Practice in Older Adults: A Systematic Review. Journal of Clinical Medicine. 2022; 11(3):714. https://doi.org/10.3390/jcm11030714
Chicago/Turabian StyleSalahudeen, Mohammed S., Adel Alfahmi, Anam Farooq, Mehnaz Akhtar, Sana Ajaz, Saud Alotaibi, Manal Faiz, and Sheraz Ali. 2022. "Effectiveness of Interventions to Improve the Anticholinergic Prescribing Practice in Older Adults: A Systematic Review" Journal of Clinical Medicine 11, no. 3: 714. https://doi.org/10.3390/jcm11030714
APA StyleSalahudeen, M. S., Alfahmi, A., Farooq, A., Akhtar, M., Ajaz, S., Alotaibi, S., Faiz, M., & Ali, S. (2022). Effectiveness of Interventions to Improve the Anticholinergic Prescribing Practice in Older Adults: A Systematic Review. Journal of Clinical Medicine, 11(3), 714. https://doi.org/10.3390/jcm11030714







