Brain Volume Loss Can Occur at the Rate of Normal Aging in Patients with Multiple Sclerosis Who Are Free from Disease Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Objective
2.2. MS Patients
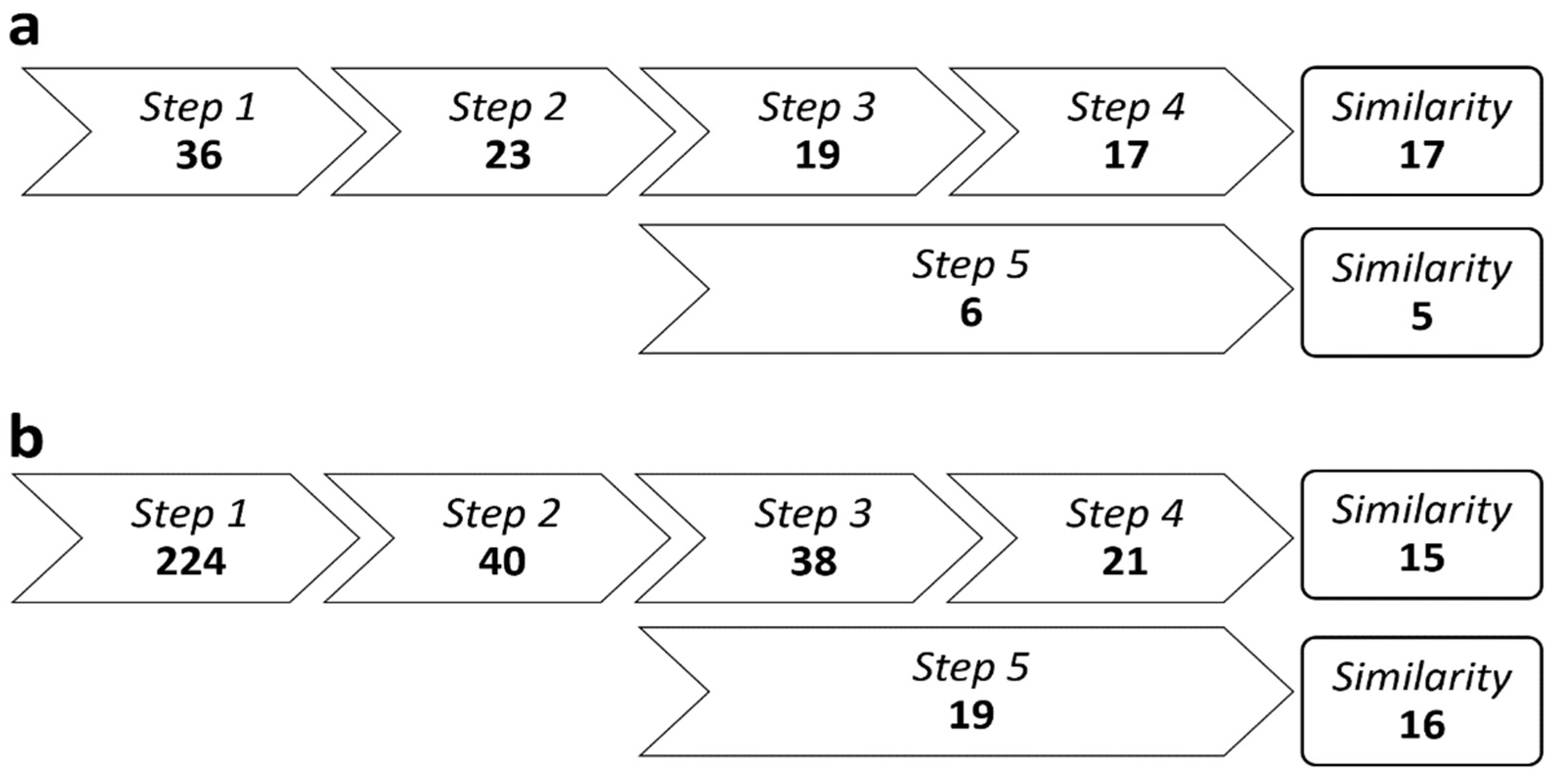
- Step 1
- A shortlist was generated on 24 August 2018 containing all unique patients with MS who were under treatment with natalizumab, fingolimod, or ocrelizumab in one of both centers at that time. Patients who had received treatment with alemtuzumab or cladribine in the 5 preceding years (and in whom no other subsequent DMT had been administered thereafter) were also added.
- Step 2
- The most recent (before 24 August 2018) brain MRI including 3D T1-weighted and fluid-attenuated inversion recovery (FLAIR) images was tracked in all candidates obtained from step 1 and labeled as MRI-1. We subsequently searched for the brain MRI including 3D T1-weighted and FLAIR images which was performed the closest to the day 24 months prior to MRI-1; the latter was labeled as MRI-0. A minimum interval of 12 months between MRI-0 and MRI-1 was deemed mandatory to enable sufficient between-group BVL contrastation. Only patients who had been continuously treated with natalizumab, fingolimod, or ocrelizumab between MRI-0 and MRI-1, or those who had received alemtuzumab or cladribine prior to MRI-0 (and in whom no other DMT was administered between MRI-0 and MRI-1), were retained. The substantial disparity in retention between the two study sites (i.e., 64% versus 18%) after this step might be explained by fundamental differences with regard to the general working environment and/or facilities at both centers. The UZ Brussel is a classic academic hospital where the vast majority of patients followed at the MS unit are referred to the in-house radiology department for MRI evaluation, using a standardized protocol with 3D FLAIR and T1-weighted imaging in each subject. In contrast, the NMSC Melsbroek is a not a general hospital but a tertiary center specifically focusing on the neurological management, multidisciplinary care, and rehabilitation of individuals with MS. The latter has a larger database (which is also reflected by the higher number of patients resulting from step 1) but relies on referral to other hospitals for MRI monitoring, introducing radiological heterogeneity and thereby reducing the likelihood of meeting the inclusion criteria of step 2.
- Step 3
- MRI-0 was compared with MRI-1, as well as with each available intermediate brain MR. Patients were removed if, at any point throughout the follow-up period, there was (a) at least one new or increased hyperintense lesion on FLAIR imaging or (b) contrast enhancement on T1-weighted imaging.
- Step 4
- We identified the neurological report that chronologically preceded MRI-0 first, as well as the report that was the first to follow MRI-1 (respectively labeled as R-0 and R-1). All interim reports were screened for the occurrence of MS relapses; defined as subacute new or worsening neurological deficits, disabling and present for at least 24 h, and in the absence of fever/infection or a more appropriate alternative explanation. Expanded Disability Status Scale (EDSS) scores were recorded from R-0 and R-1 [12]. If not available in these reports, EDSS scores were reconstructed by the study team to the best of their ability based on the information in the patient records. Patients with evidence for one or more MS relapses, as well as those with significant EDSS progression, between R-0 and R-1, were not retained. The following criteria were used to define significant EDSS progression: at least 1.5 points if the R-0 score was 0, at least 1.0 points if the R-0 score was 1.0–5.0, or at least 0.5 points if the R-0 score was 5.5 or higher [13].
- Step 5
- In parallel, steps 1 to 4 also resulted in the identification of MS patients with evidence of disease activity (EDA) while being on high-efficacy DMT between MRI-0 and MRI-1, who served as an additional control group.
2.3. Healthy Controls
2.4. Brain Volume Analysis
2.5. Statistics
3. Results
3.1. Study Groups
3.2. Brain Volume Change
3.3. Correlations
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Walton, C.; King, R.; Rechtman, L.; Kaye, W.; Leray, E.; Marrie, R.A.; Robertson, N.; La Rocca, N.; Uitdehaag, B.; Van Der Mei, I.; et al. Rising prevalence of multiple sclerosis worldwide: Insights from the Atlas of MS, third edition. Mult. Scler. J. 2020, 26, 1816–1821. [Google Scholar] [CrossRef]
- Dobson, R.; Giovannoni, G. Multiple sclerosis—A review. Eur. J. Neurol. 2019, 26, 27–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sastre-Garriga, J.; Pareto, D.; Rovira, A. Brain Atrophy in Multiple Sclerosis: Clinical Relevance and Technical Aspects. Neuroimaging Clin. N. Am. 2017, 27, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Sastre-Garriga, J.; on behalf of the MAGNIMS Study Group; Pareto, D.; Battaglini, M.; Rocca, M.A.; Ciccarelli, O.; Enzinger, C.; Wuerfel, J.; Sormani, M.P.; Barkhof, F.; et al. MAGNIMS consensus recommendations on the use of brain and spinal cord atrophy measures in clinical practice. Nat. Rev. Neurol. 2020, 16, 171–182. [Google Scholar] [CrossRef] [Green Version]
- Battaglini, M.; Gentile, G.; Luchetti, L.; Giorgio, A.; Vrenken, H.; Barkhof, F.; Cover, K.S.; Bakshi, R.; Chu, R.; Sormani, M.P.; et al. Lifespan normative data on rates of brain volume changes. Neurobiol. Aging 2019, 81, 30–37. [Google Scholar] [CrossRef]
- De Stefano, N.; Giorgio, A.; Battaglini, M.; Rovaris, M.; Sormani, M.P.; Barkhof, F.; Korteweg, T.; Enzinger, C.; Fazekas, F.; Calabrese, M.; et al. Assessing brain atrophy rates in a large population of untreated multiple sclerosis subtypes. Neurology 2010, 74, 1868–1876. [Google Scholar] [CrossRef] [PubMed]
- Rotstein, D.L.; Healy, B.C.; Malik, M.T.; Chitnis, T.; Weiner, H.L. Evaluation of No Evidence of Disease Activity in a 7-Year Longitudinal Multiple Sclerosis Cohort. JAMA Neurol. 2015, 72, 152–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pantano, P.; Mainero, C.; Caramia, F. Functional Brain Reorganization in Multiple Sclerosis: Evidence from fMRI Studies. J. Neuroimaging 2006, 16, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, J.; Audoin, B.; Reuter, F.; Ranjeva, J.-P. Plasticity in MS: From Functional Imaging to Rehabilitation. Int. MS J. 2009, 16, 26–31. [Google Scholar] [PubMed]
- Colorado, R.A.; Shukla, K.; Zhou, Y.; Wolinsky, J.S.; Narayana, P.A. Multi-task functional MRI in multiple sclerosis patients without clinical disability. NeuroImage 2012, 59, 573–581. [Google Scholar] [CrossRef] [Green Version]
- Mayssam, E.N.; Eid, C.; Khoury, S.J.; Hannoun, S. “No evidence of disease activity”: Is it an aspirational therapeutic goal in multiple sclerosis? Mult. Scler. Relat. Disord. 2020, 40, 101935. [Google Scholar]
- Kurtzke, J.F. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 1983, 33, 1444–1452. [Google Scholar] [CrossRef] [Green Version]
- Rio, J.; Nos, C.; Tintore, M.; Tellez, N.; Galan, I.; Pelayo, R.; Comabella, M.; Montalban, X. Defining the response to interferon-beta in relapsing-remitting multiple sclerosis patients. Ann. Neurol. 2006, 59, 344–352. [Google Scholar] [CrossRef]
- Van Schependom, J.; Vidaurre, D.; Costers, L.; Sjogard, M.; D’Hooghe, M.B.; D’Haeseleer, M.; Wens, V.; De Tiège, X.; Goldman, S.; Woolrich, M.; et al. Altered transient brain dynamics in multiple sclerosis: Treatment or pathology? Hum. Brain Mapp. 2019, 40, 4789–4800. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, S.; Ribbens, A.; Sima, D.M.; Cambron, M.; De Keyser, J.; Wang, C.; Barnett, M.H.; Van Huffel, S.; Maes, F.; Smeets, D. Two Time Point MS Lesion Segmentation in Brain MRI: An Expectation-Maximization Framework. Front. Neurosci. 2016, 10, 576. [Google Scholar] [CrossRef] [Green Version]
- Smeets, D.; Ribbens, A.; Sima, D.M.; Cambron, M.; Horáková, D.; Jain, S.; Maertens, A.; Van Vlierberghe, E.; Terzopoulos, V.; Vanbinst, A.-M.; et al. Reliable measurements of brain atrophy in individual patients with multiple sclerosis. Brain Behav. 2016, 6, e00518. [Google Scholar] [CrossRef]
- Rojas, J.; Sanchez, F.; Caro, F.; Miguez, J.; Patrucco, L.; Funes, J.; Cristiano, E. Brain volume loss and no evidence of disease activity over 3 years in multiple sclerosis patients under interferon beta 1a subcutaneous treatment. J. Clin. Neurosci. 2019, 59, 175–178. [Google Scholar] [CrossRef]
- Håkansson, I.; Tisell, A.; Cassel, P.; Blennow, K.; Zetterberg, H.; Lundberg, P.; Dahle, C.; Vrethem, M.; Ernerudh, J. Neurofilament levels, disease activity and brain volume during follow-up in multiple sclerosis. J. Neuroinflammation 2018, 15, 209. [Google Scholar] [CrossRef] [Green Version]
- Nygaard, G.O.; Celius, E.G.; Benavent, S.A.D.R.; Sowa, P.; Gustavsen, M.W.; Fjell, A.M.; Landrø, N.I.; Walhovd, K.B.; Harbo, H.F. A Longitudinal Study of Disability, Cognition and Gray Matter Atrophy in Early Multiple Sclerosis Patients According to Evidence of Disease Activity. PLoS ONE 2015, 10, e0135974. [Google Scholar] [CrossRef] [PubMed]
- Ontaneda, D.; Raza, P.C.; Mahajan, K.R.; Arnold, D.L.; Dwyer, M.G.; Gauthier, S.A.; Greve, D.N.; Harrison, D.M.; Henry, R.G.; Li, D.K.B.; et al. Deep grey matter injury in multiple sclerosis: A NAIMS consensus statement. Brain 2021, 144, 1974–1984. [Google Scholar] [CrossRef] [PubMed]
- Steenwijk, M.D.; Amiri, H.; Schoonheim, M.M.; de Sitter, A.; Barkhof, F.; Pouwels, P.J.; Vrenken, H. Agreement of MSmetrix with established methods for measuring cross-sectional and longitudinal brain atrophy. NeuroImage Clin. 2017, 15, 843–853. [Google Scholar] [CrossRef]
- Guevara, C.; Garrido, C.; Martinez, M.; Farías, G.; Orellana, P.; Soruco, W.; Alarcón, P.; Diaz, V.; Silva, C.; Kempton, M.J.; et al. Prospective Assessment of No Evidence of Disease Activity-4 Status in Early Disease Stages of Multiple Sclerosis in Routine Clinical Practice. Front. Neurol. 2019, 10, 788. [Google Scholar] [CrossRef] [Green Version]
- Yokote, H.; Kamata, T.; Toru, S.; Sanjo, N.; Yokota, T. Brain volume loss is present in Japanese multiple sclerosis patients with no evidence of disease activity. Neurol. Sci. 2018, 39, 1713–1716. [Google Scholar] [CrossRef]
- Kappos, L.; de Stefano, N.; Freedman, M.S.; Cree, B.; Radue, E.-W.; Sprenger, T.; Sormani, M.P.; Smith, T.; Häring, D.A.; Meier, D.P.; et al. Inclusion of brain volume loss in a revised measure of ‘no evidence of disease activity’ (NEDA-4) in relapsing–remitting multiple sclerosis. Mult. Scler. J. 2016, 22, 1297–1305. [Google Scholar] [CrossRef] [Green Version]
- Khalil, M.; Teunissen, C.E.; Otto, M.; Piehl, F.; Sormani, M.P.; Gattringer, T.; Barro, C.; Kappos, L.; Comabella, M.; Fazekas, F.; et al. Neurofilaments as biomarkers in neurological disorders. Nat. Rev. Neurol. 2018, 14, 577–589. [Google Scholar] [CrossRef]
- Thebault, S.; Abdoli, M.; Fereshtehnejad, S.-M.; Tessier, D.; Tabard-Cossa, V.; Freedman, M.S. Serum neurofilament light chain predicts long term clinical outcomes in multiple sclerosis. Sci. Rep. 2020, 10, 10381. [Google Scholar] [CrossRef] [PubMed]
- Barro, C.; Benkert, P.; Disanto, G.; Tsagkas, C.; Amann, M.; Naegelin, Y.; Leppert, D.; Gobbi, C.; Granziera, C.; Yaldizli, Ö.; et al. Serum neurofilament as a predictor of disease worsening and brain and spinal cord atrophy in multiple sclerosis. Brain 2018, 141, 2382–2391. [Google Scholar] [CrossRef]
- Petzold, A.; Steenwijk, M.D.; Eikelenboom, J.M.; Wattjes, M.P.; Uitdehaag, B.M. Elevated CSF neurofilament proteins predict brain atrophy: A 15-year follow-up study. Mult. Scler. J. 2016, 22, 1154–1162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hyun, J.-W.; Kim, Y.; Kim, G.; Kim, S.-H.; Kim, H.J. Longitudinal analysis of serum neurofilament light chain: A potential therapeutic monitoring biomarker for multiple sclerosis. Mult. Scler. J. 2019, 26, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Havrdova, E.; Arnold, D.L.; Cohen, J.A.; Hartung, H.P.; Fox, E.J.; Giovannoni, G.; Schippling, S.; Selmaj, K.W.; Traboulsee, A.; Compston, D.A.S.; et al. Alemtuzumab CARE-MS I 5-year follow-up: Durable efficacy in the absence of continuous MS therapy. Neurology 2017, 89, 1107–1116. [Google Scholar] [CrossRef] [Green Version]
- Coles, A.J.; Cohen, J.A.; Fox, E.J.; Giovannoni, G.; Hartung, H.P.; Havrdova, E.; Schippling, S.; Selmaj, K.W.; Traboulsee, A.; Compston, D.A.S.; et al. Alemtuzumab CARE-MS II 5-year follow-up: Efficacy and safety findings. Neurology 2017, 89, 1117–1126. [Google Scholar] [CrossRef] [Green Version]
- Green, A.J.; McQuaid, S.; Hauser, S.L.; Allen, I.V.; Lyness, R. Ocular pathology in multiple sclerosis: Retinal atrophy and inflammation irrespective of disease duration. Brain 2010, 133, 1591–1601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petzold, A.; Balcer, L.J.; Calabresi, P.A.; Costello, F.; Frohman, T.C.; Frohman, E.M.; Martinez-Lapiscina, E.H.; Green, A.J.; Kardon, R.; Outteryck, O.; et al. Retinal layer segmentation in multiple sclerosis: A systematic review and meta-analysis. Lancet Neurol. 2017, 16, 797–812. [Google Scholar] [CrossRef] [Green Version]
- Costello, F.; Coupland, S.; Hodge, W.; Lorello, G.; Koroluk, J.; Pan, Y.I.; Freedman, M.S.; Zackon, D.H.; Kardon, R.H. Quantifying axonal loss after optic neuritis with optical coherence tomography. Ann. Neurol. 2006, 59, 963–969. [Google Scholar] [CrossRef]
- Talman, L.S.; Bisker, E.R.; Sackel, D.J.; Long, D.A., Jr.; Galetta, K.M.; Ratchford, J.N.; Lile, D.J.; Farrell, S.K.; Loguidice, M.J.; Remington, G.; et al. Longitudinal study of vision and retinal nerve fiber layer thickness in multiple sclerosis. Ann. Neurol. 2010, 67, 749–760. [Google Scholar] [PubMed] [Green Version]
- Fisher, J.B.; Jacobs, D.A.; Markowitz, C.E.; Galetta, S.L.; Volpe, N.J.; Nano-Schiavi, M.L.; Baier, M.L.; Frohman, E.M.; Winslow, H.; Frohman, T.C. Relation of Visual Function to Retinal Nerve Fiber Layer Thickness in Multiple Sclerosis. Ophthalmology 2006, 113, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Martin, E.; Rodriguez-Mena, D.; Herrero, R.; Almarcegui, C.; Dolz, I.; Martin, J.; Ara, J.R.; Larrosa, J.M.; Polo, V.; Fernández, J.; et al. Neuro-ophthalmologic evaluation, quality of life, and functional disability in patients with MS. Neurology 2013, 81, 76–83. [Google Scholar] [CrossRef]
- Toledo, J.; Sepulcre, J.; Salinas-Alaman, A.; García-Layana, A.; Murie-Fernandez, M.; Bejarano, B.; Villoslada, P. Retinal nerve fiber layer atrophy is associated with physical and cognitive disability in multiple sclerosis. Mult. Scler. J. 2008, 14, 906–912. [Google Scholar] [CrossRef]
- Saidha, S.; Al-Louzi, O.; Ratchford, J.N.; Bhargava, P.; Oh, J.; Newsome, S.D.; Prince, J.L.; Pham, D.; Roy, S.; Van Zijl, P.; et al. Optical coherence tomography reflects brain atrophy in multiple sclerosis: A four-year study. Ann. Neurol. 2015, 78, 801–813. [Google Scholar] [CrossRef]
- Lambe, J.; Fitzgerald, K.C.; Murphy, O.C.; Filippatou, A.G.; Sotirchos, E.S.; Kalaitzidis, G.; Vasileiou, E.; Pellegrini, N.; Ogbuokiri, E.; Toliver, B.; et al. Association of Spectral-Domain OCT With Long-term Disability Worsening in Multiple Sclerosis. Neurology 2021, 96, e2058–e2069. [Google Scholar] [CrossRef]
- Pisa, M.; Guerrieri, S.; Di Maggio, G.; Medaglini, S.; Moiola, L.; Martinelli, V.; Comi, G.; Leocani, L. No evidence of disease activity is associated with reduced rate of axonal retinal atrophy in MS. Neurology 2017, 89, 2469–2475. [Google Scholar] [CrossRef]
- Pisa, M.; Ratti, F.; Vabanesi, M.; Radaelli, M.; Guerrieri, S.; Moiola, L.; Martinelli, V.; Comi, G.; Leocani, L. Subclinical neurodegeneration in multiple sclerosis and neuromyelitis optica spectrum disorder revealed by optical coherence tomography. Mult. Scler. J. 2019, 26, 1197–1206. [Google Scholar] [CrossRef] [PubMed]
- Compston, A.; Coles, A. Multiple sclerosis. Lancet 2008, 372, 1502–1517. [Google Scholar] [CrossRef]
- Sen, M.K.; Almuslehi, M.S.M.; Shortland, P.J.; Coorssen, J.R.; Mahns, D.A. Revisiting the Pathoetiology of Multiple Sclerosis: Has the Tail Been Wagging the Mouse? Front. Immunol. 2020, 11, 572186. [Google Scholar] [CrossRef] [PubMed]
- Hauser, S.L.; Cree, B.A. Treatment of Multiple Sclerosis: A Review. Am. J. Med. 2020, 133, 1380–1390.e2. [Google Scholar] [CrossRef]
- Tintore, M.; Vidal-Jordana, A.; Sastre-Garriga, J. Treatment of multiple sclerosis—Success from bench to bedside. Nat. Rev. Neurol. 2019, 15, 53–58. [Google Scholar] [CrossRef]
- Amato, M.P.; Fonderico, M.; Portaccio, E.; Pastò, L.; Razzolini, L.; Prestipino, E.; Bellinvia, A.; Tudisco, L.; Fratangelo, R.; Comi, G.; et al. Disease-modifying drugs can reduce disability progression in relapsing multiple sclerosis. Brain 2020, 143, 3013–3024. [Google Scholar] [CrossRef]
- Kalincik, T.; Diouf, I.; Sharmin, S.; Malpas, C.; Spelman, T.; Horakova, D.; Havrdova, E.K.; Trojano, M.; Izquierdo, G.; Lugaresi, A.; et al. Effect of Disease-Modifying Therapy on Disability in Relapsing-Remitting Multiple Sclerosis over 15 Years. Neurology 2020, 96, e783–e797. [Google Scholar] [CrossRef] [PubMed]
- Chalmer, T.A.; Baggesen, L.M.; Nørgaard, M.; Koch-Henriksen, N.; Magyari, M.; Sorensen, P.S.; The Danish Multiple Sclerosis Group. Early versus later treatment start in multiple sclerosis: A register-based cohort study. Eur. J. Neurol. 2018, 25, 1262-e110. [Google Scholar] [CrossRef] [PubMed]
- Buron, M.D.; Chalmer, T.A.; Sellebjerg, F.; Barzinji, I.; Christensen, J.R.; Christensen, M.K.; Hansen, V.; Illes, Z.; Jensen, H.B.; Kant, M.; et al. Initial high-efficacy disease-modifying therapy in multiple sclerosis: A nationwide cohort study. Neurology 2020, 95, e1041–e1051. [Google Scholar] [CrossRef]
- He, A.; Merkel, B.; Brown, W.; Ryerson, L.Z.; Kister, I.; Malpas, C.B.; Sharmin, S.; Horakova, D.; Havrdova, E.K.; Spelman, T.; et al. Timing of high-efficacy therapy for multiple sclerosis: A retrospective observational cohort study. Lancet Neurol. 2020, 19, 307–316. [Google Scholar] [CrossRef]
- Prosperini, L.; Mancinelli, C.R.; Solaro, C.M.; Nociti, V.; Haggiag, S.; Cordioli, C.; De Giglio, L.; De Rossi, N.; Galgani, S.; Rasia, S.; et al. Induction Versus Escalation in Multiple Sclerosis: A 10-Year Real World Study. Neurotherapeutics 2020, 17, 994–1004. [Google Scholar] [CrossRef]
- Di Filippo, M.; Anderson, V.M.; Altmann, D.R.; Swanton, J.K.; Plant, G.T.; Thompson, A.J.; Miller, D.H. Brain atrophy and lesion load measures over 1 year relate to clinical status after 6 years in patients with clinically isolated syndromes. J. Neurol. Neurosurg. Psychiatry 2010, 81, 204–208. [Google Scholar] [CrossRef]
- Minneboo, A.; Jasperse, B.; Barkhof, F.; Uitdehaag, B.M.J.; Knol, D.L.; de Groot, V.; Polman, C.H.; Castelijns, J.A. Predicting short-term disability progression in early multiple sclerosis: Added value of MRI parameters. J. Neurol. Neurosurg. Psychiatry 2008, 79, 917–923. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Branger, P.; Parienti, J.-J.; Sormani, M.P.; Defer, G. The Effect of Disease-Modifying Drugs on Brain Atrophy in Relapsing-Remitting Multiple Sclerosis: A Meta-Analysis. PLoS ONE 2016, 11, e0149685. [Google Scholar] [CrossRef] [Green Version]
- Sormani, M.P.; Arnold, D.L.; de Stefano, N. Treatment effect on brain atrophy correlates with treatment effect on disability in multiple sclerosis. Ann. Neurol. 2013, 75, 43–49. [Google Scholar] [CrossRef]
- De Stefano, N.; Stromillo, M.L.; Giorgio, A.; Bartolozzi, M.L.; Battaglini, M.; Baldini, M.; Portaccio, E.; Amato, M.P.; Sormani, M.P. Establishing pathological cut-offs of brain atrophy rates in multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 2016, 87, 93–99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sprenger, T.; Kappos, L.; Radue, E.-W.; Gaetano, L.; Mueller-Lenke, N.; Wuerfel, J.; Poole, E.M.; Cavalier, S. Association of brain volume loss and long-term disability outcomes in patients with multiple sclerosis treated with teriflunomide. Mult. Scler. J. 2019, 26, 1207–1216. [Google Scholar] [CrossRef] [Green Version]
- De Stefano, N.; Tomic, D.; Radue, E.W.; Sprenger, T.; Meier, D.P.; Haring, D.; Sormani, M.P. Effect of fingolimod on diffuse brain tissue damage in relapsing-remitting multiple sclerosis patients. Mult. Scler. Relat. Disord. 2016, 7, 98–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| NEDA-3 | HC | EDA | |
|---|---|---|---|
| Number of subjects | 32 | 27 | 21 |
| Age (years) 1 | 43 (10) *,^ | 48 (13) * | 39 (10) ^ |
| Gender (female/male–female/total) | 23/9–0.72 | 16/11–0.59 | 14/7–0.67 |
| EDSS score 1 | 3.6 (2.0) ° | NA | 4.0 (1.0) ° |
| Disease duration (years) 2 | 12 (27) § | NA | 10 (35) § |
| Current DMT duration (years) 2 | 3 (9) # | NA | 3 (9) # |
| Interval between MRI exams (months) 2 | 22 (18) ~ | 32 (28) | 20 (30) ~ |
| NEDA-3 | HC | |
|---|---|---|
| Number of subjects | 29 | 24 |
| Age (years) 1,* | 44 (10) ~ | 46 (12) ~ |
| Gender (female/male–female/total) | 20/9–0.69 | 16/8–0.67 |
| EDSS score 1 | 3.5 (1.9) | NA |
| Disease duration (years) 2,^ | 12 (6) | NA |
| Current DMT duration (years) 2 | 2 (9) | NA |
| Interval between MRI exams (months) 2 | 21 (18) ° | 33 (28) ° |
| Annualized WBV change (%) 1 | −0.25 (0.49) § | −0.24 (0.20) § |
| Annualized TGMV change (%) 1 | −0.29 (0.60) # | −0.29 (0.23) # |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Temmerman, J.; Van Der Veken, F.; Engelborghs, S.; Guldolf, K.; Nagels, G.; Smeets, D.; Allemeersch, G.-J.; Costers, L.; D’hooghe, M.B.; Vanbinst, A.-M.; et al. Brain Volume Loss Can Occur at the Rate of Normal Aging in Patients with Multiple Sclerosis Who Are Free from Disease Activity. J. Clin. Med. 2022, 11, 523. https://doi.org/10.3390/jcm11030523
Temmerman J, Van Der Veken F, Engelborghs S, Guldolf K, Nagels G, Smeets D, Allemeersch G-J, Costers L, D’hooghe MB, Vanbinst A-M, et al. Brain Volume Loss Can Occur at the Rate of Normal Aging in Patients with Multiple Sclerosis Who Are Free from Disease Activity. Journal of Clinical Medicine. 2022; 11(3):523. https://doi.org/10.3390/jcm11030523
Chicago/Turabian StyleTemmerman, Joke, Floris Van Der Veken, Sebastiaan Engelborghs, Kaat Guldolf, Guy Nagels, Dirk Smeets, Gert-Jan Allemeersch, Lars Costers, Marie B. D’hooghe, Anne-Marie Vanbinst, and et al. 2022. "Brain Volume Loss Can Occur at the Rate of Normal Aging in Patients with Multiple Sclerosis Who Are Free from Disease Activity" Journal of Clinical Medicine 11, no. 3: 523. https://doi.org/10.3390/jcm11030523
APA StyleTemmerman, J., Van Der Veken, F., Engelborghs, S., Guldolf, K., Nagels, G., Smeets, D., Allemeersch, G.-J., Costers, L., D’hooghe, M. B., Vanbinst, A.-M., Van Schependom, J., Bjerke, M., & D’haeseleer, M. (2022). Brain Volume Loss Can Occur at the Rate of Normal Aging in Patients with Multiple Sclerosis Who Are Free from Disease Activity. Journal of Clinical Medicine, 11(3), 523. https://doi.org/10.3390/jcm11030523







