Factors Associated with RANTES, EMMPIRIN, MMP2 and MMP9, and the Association of These Biomarkers with Cardiovascular Disease in a Multi-Ethnic Population
Abstract
Highlights
- This is the first multi-ethnic population-based study to use these serum biomarkers for preventive strategy.
- No association was found between RANTES, EMMPRIN, MMP2, and MMP9 with CVD.
- Our research improves the understanding of inflammatory biomarkers in the cardiovascular field. Currently, these biomarkers are ineffective for risk stratification or diagnosis when used as a single indicator.
- Prevention of CVD still requires a comprehensive evaluation of CVD risk factors.
Abstract
1. Introduction
2. Methods
2.1. Study Sample
2.2. Assessment of Biomarkers
2.3. Assessment of Covariates
2.4. Statistical Analysis
3. Results
3.1. Participant Characteristics
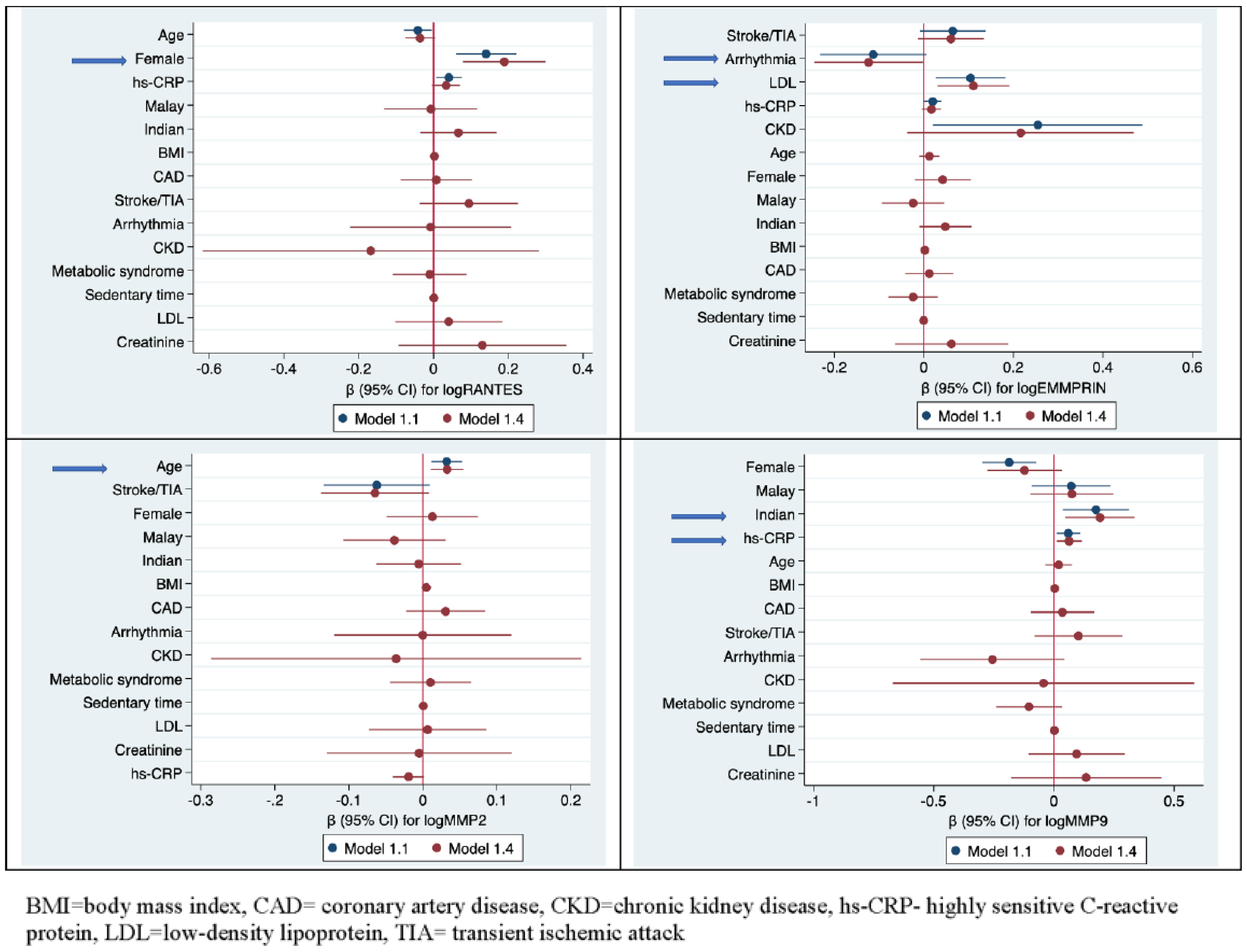
3.2. Factors Associated with the Biomarkers
3.2.1. RANTES
3.2.2. EMMPRIN
3.2.3. MMP-2
3.2.4. MMP-9
3.3. Association of Biomarkers with CVD
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations and Acronyms
| EMMPRIN: | extracellular matrix metalloproteinase inducer |
| FRS: | Framingham risk score |
| Hs-CRP: | highly sensitive C-reactive protein |
| MEC: | multi-ethnic cohort (MEC) |
| MMP: | matrix metalloproteinases |
| NLR: | neutrophil-to-lymphocyte ratio |
| PLR: | platelet-to-lymphocyte ratio |
| RANTES: | regulated on Activation, Normal T Cell Expressed and Secreted |
| SP2: | Singapore Prospective Study Program |
| SCCS2: | Singapore Cardiovascular Cohort Study |
| SII: | systemic inflammation index |
References
- WHO. Cardiovascular Diseases (CVDs). 2017. Available online: https://www.who.int/en/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds) (accessed on 1 September 2022).
- Tsao, C.W.; Aday, A.W.; Almarzooq, Z.I.; Alonso, A.; Beaton, A.Z.; Bittencourt, M.S.; Boehme, A.K.; Buxton, A.E.; Carson, A.P.; Commodore-Mensah, Y.; et al. Heart Disease and Stroke Statistics-2022 Update: A Report From the American Heart Association. Circulation 2022, 145, e153–e639. [Google Scholar] [CrossRef] [PubMed]
- Abete, P.; della Morte, D.; Gargiulo, G.; Basile, C.; Langellotto, A.; Galizia, G.; Testa, G.; Canonico, V.; Bonaduce, D.; Cacciatore, F. Cognitive impairment and cardiovascular diseases in the elderly. A heart-brain continuum hypothesis. Ageing Res. Rev. 2014, 18, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Weber, C.; Noels, H. Atherosclerosis: Current pathogenesis and therapeutic options. Nat. Med. 2011, 17, 1410–1422. [Google Scholar] [CrossRef] [PubMed]
- Tsimikas, S.; Willerson, J.T.; Ridker, P.M. C-reactive protein and other emerging blood biomarkers to optimize risk stratification of vulnerable patients. J. Am. Coll. Cardiol. 2006, 47, C19–C31. [Google Scholar] [CrossRef] [PubMed]
- Soeki, T.; Sata, M. Inflammatory Biomarkers and Atherosclerosis. Int. Heart J. 2016, 57, 134–139. [Google Scholar] [CrossRef]
- Lipkova, J.; Parenica, J.; Duris, K.; Helanova, K.; Tomandl, J.; Kubkova, L.; Vasku, A.; Goldbergova Pavkova, M. Association of circulating levels of RANTES and -403G/A promoter polymorphism to acute heart failure after STEMI and to cardiogenic shock. Clin. Exp. Med. 2015, 15, 405–414. [Google Scholar] [CrossRef]
- Appay, V.; Rowland-Jones, S.L. RANTES: A versatile and controversial chemokine. Trends Immunol. 2001, 22, 83–87. [Google Scholar] [CrossRef]
- Cavusoglu, E.; Eng, C.; Chopra, V.; Clark, L.T.; Pinsky, D.J.; Marmur, J.D. Low plasma RANTES levels are an independent predictor of cardiac mortality in patients referred for coronary angiography. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 929–935. [Google Scholar] [CrossRef]
- Kaushik, D.K.; Hahn, J.N.; Yong, V.W. EMMPRIN, an upstream regulator of MMPs, in CNS biology. Matrix Biol. 2015, 44-46, 138–146. [Google Scholar] [CrossRef]
- Agrawal, S.M.; Yong, V.W. The many faces of EMMPRIN—Roles in neuroinflammation. Biochim. Et Biophys. Acta 2011, 1812, 213–219. [Google Scholar] [CrossRef]
- Schmidt, R.; Bültmann, A.; Ungerer, M.; Joghetaei, N.; Bülbül, O.; Thieme, S.; Chavakis, T.; Toole, B.P.; Gawaz, M.; Schömig, A.; et al. Extracellular matrix metalloproteinase inducer regulates matrix metalloproteinase activity in cardiovascular cells: Implications in acute myocardial infarction. Circulation 2006, 113, 834–841. [Google Scholar] [CrossRef] [PubMed]
- Opstad, T.B.; Seljeflot, I.; Bohmer, E.; Arnesen, H.; Halvorsen, S. MMP-9 and Its Regulators TIMP-1 and EMMPRIN in Patients with Acute ST-Elevation Myocardial Infarction: A NORDISTEMI Substudy. Cardiology 2018, 139, 17–24. [Google Scholar] [CrossRef]
- Galis, Z.S.; Khatri, J.J. Matrix metalloproteinases in vascular remodeling and atherogenesis: The good, the bad, and the ugly. Circ. Res. 2002, 90, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Newby, A.C. Metalloproteinases promote plaque rupture and myocardial infarction: A persuasive concept waiting for clinical translation. Matrix Biol. 2015, 44–46, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Tan, G.J.; Han, L.N.; Bai, Y.Y.; He, M.; Liu, H.B. Novel biomarkers for cardiovascular risk prediction. J. Geriatr. Cardiol. 2017, 14, 135–150. [Google Scholar] [PubMed]
- Blankenberg, S.; Rupprecht, H.J.; Poirier, O.; Bickel, C.; Smieja, M.; Hafner, G.; Meyer, J.; Cambien, F.; Tiret, L.; AtheroGene Investigators. Plasma concentrations and genetic variation of matrix metalloproteinase 9 and prognosis of patients with cardiovascular disease. Circulation 2003, 107, 1579–1585. [Google Scholar] [CrossRef]
- Kelly, D.; Cockerill, G.; Ng, L.L.; Thompson, M.; Khan, S.; Samani, N.J.; Squire, I.B. Plasma matrix metalloproteinase-9 and left ventricular remodelling after acute myocardial infarction in man: A prospective cohort study. Eur. Heart J. 2007, 28, 711–718. [Google Scholar] [CrossRef]
- Lenti, M.; Falcinelli, E.; Pompili, M.; de Rango, P.; Conti, V.; Guglielmini, G.; Momi, S.; Corazzi, T.; Giordano, G.; Gresele, P. Matrix metalloproteinase-2 of human carotid atherosclerotic plaques promotes platelet activation. Correlation with ischaemic events. Thromb. Haemost. 2014, 111, 1089–1101. [Google Scholar] [CrossRef]
- Dhillon, O.S.; Khan, S.Q.; Narayan, H.K.; Ng, K.H.; Mohammed, N.; Quinn, P.A.; Squire, I.B.; Davies, J.E.; Ng, L.L. Matrix metalloproteinase-2 predicts mortality in patients with acute coronary syndrome. Clin. Sci. 2009, 118, 249–257. [Google Scholar] [CrossRef]
- Tan, K.H.X.; Tan, L.W.L.; Sim, X.; Tai, E.S.; Lee, J.J.; Chia, K.S.; van Dam, R.M. Cohort Profile: The Singapore Multi-Ethnic Cohort (MEC) study. Int. J. Epidemiol. 2018, 47, 699. [Google Scholar] [CrossRef]
- D’Agostino, R.B., Sr.; Vasan, R.S.; Pencina, M.J.; Wolf, P.A.; Cobain, M.; Massaro, J.M.; Kannel, W.B. General cardiovascular risk profile for use in primary care: The Framingham Heart Study. Circulation 2008, 117, 743–753. [Google Scholar] [CrossRef] [PubMed]
- Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002, 106, 3143–3421. [CrossRef]
- Yao, L.; Herlea-Pana, O.; Heuser-Baker, J.; Chen, Y.; Barlic-Dicen, J. Roles of the Chemokine System in Development of Obesity, Insulin Resistance, and Cardiovascular Disease. J. Immunol. Res. 2014, 2014, 181450. [Google Scholar] [CrossRef]
- Rothenbacher, D.; Müller-Scholze, S.; Herder, C.; Koenig, W.; Kolb, H. Differential Expression of Chemokines, Risk of Stable Coronary Heart Disease, and Correlation with Established Cardiovascular Risk Markers. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Mikolajczyk, T.P.; Szczepaniak, P.; Vidler, F.; Maffia, P.; Graham, G.J.; Guzik, T.J. Role of inflammatory chemokines in hypertension. Pharmacol. Ther. 2021, 223, 107799. [Google Scholar] [CrossRef]
- Wang, C.-H.; Dai, J.-Y.; Wang, L.; Jia, J.-F.; Zheng, Z.-H.; Ding, J.; Chen, Z.-N.; Zhu, P. Expression of CD147 (EMMPRIN) on neutrophils in rheumatoid arthritis enhances chemotaxis, matrix metalloproteinase production and invasiveness of synoviocytes. J. Cell. Mol. Med. 2011, 15, 850–860. [Google Scholar] [CrossRef]
- von Ungern-Sternberg, S.N.I.; Zernecke, A.; Seizer, P. Extracellular Matrix Metalloproteinase Inducer EMMPRIN (CD147) in Cardiovascular Disease. Int. J. Mol. Sci. 2018, 19, 507. [Google Scholar] [CrossRef]
- Elahirad, S.; Elieh Ali Komi, D.; Kiani, A.; Mohammadi-Noori, E.; Vaisi-Raygani, A.; Mozafari, H.; Bahrehmand, F.; Saidi, M.; Toupchi-Khosroshahi, V.; Salehi, N. Association of Matrix Metalloproteinase-2 (MMP-2) and MMP-9 Promoter Polymorphisms, Their Serum Levels, and Activities with Coronary Artery Calcification (CAC) in an Iranian Population. Cardiovasc. Toxicol. 2022, 22, 118–129. [Google Scholar] [CrossRef]
- Derosa, G.; Maffioli, P.; D’Angelo, A.; Salvadeo, S.A.; Ferrari, I.; Fogari, E.; Gravina, A.; Mereu, R.; Palumbo, I.; Randazzo, S.; et al. Evaluation of metalloproteinase 2 and 9 levels and their inhibitors in combined dyslipidemia. Clin. Investig. Med. 2009, 32, E124–E132. [Google Scholar] [CrossRef]
- Boumiza, S.; Chahed, K.; Tabka, Z.; Jacob, M.-P.; Norel, X.; Ozen, G. MMPs and TIMPs levels are correlated with anthropometric parameters, blood pressure, and endothelial function in obesity. Sci. Rep. 2021, 11, 20052. [Google Scholar] [CrossRef]
- Salamonsen, L.A.; Woolley, D.E. Matrix metalloproteinases in normal menstruation. Hum. Reprod. 1996, 11 (Suppl. S2), 124–133. [Google Scholar] [CrossRef] [PubMed]
- Ueba, T.; Nomura, S.; Inami, N.; Yokoi, T.; Inoue, T. Elevated RANTES level is associated with metabolic syndrome and correlated with activated platelets associated markers in healthy younger men. Clin. Appl. Thromb. Hemost. 2014, 20, 813–818. [Google Scholar] [CrossRef]
- Herder, C.; Peeters, W.; Illig, T.; Baumert, J.; de Kleijn, D.P.; Moll, F.L.; Poschen, U.; Klopp, N.; Müller-Nurasyid, M.; Roden, M.; et al. RANTES/CCL5 and risk for coronary events: Results from the MONICA/KORA Augsburg case-cohort, Athero-Express and CARDIoGRAM studies. PLoS ONE 2011, 6, e25734. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Yang, L.X.; Guo, R.W.; Zhu, G.F.; Shi, Y.K.; Wang, X.M.; Qi, F.; Guo, C.M.; Ye, J.S.; Yang, Z.H.; et al. Functional blockage of EMMPRIN ameliorates atherosclerosis in apolipoprotein E-deficient mice. Int. J. Cardiol. 2013, 168, 3248–3253. [Google Scholar] [CrossRef] [PubMed]
- Kjekshus, H.; Skrebelyte-Strom, L.; Bakkelund, V.; Arnesen, H.; Ronning, O.M.; Steine, K.; Seljeflot, I. Biomarkers in patients with cryptogenic stroke/TIA and subclinical atrial fibrillation. Eur. Heart J. 2020, 41, ehaa946-2439. [Google Scholar] [CrossRef]
- Yasmin; Wallace, S.; McEniery, C.M.; Dakham, Z.; Pusalkar, P.; Maki-Petaja, K.; Ashby, M.J.; Cockcroft, J.R.; Wilkinson, I.B. Matrix Metalloproteinase-9 (MMP-9), MMP-2, and Serum Elastase Activity Are Associated With Systolic Hypertension and Arterial Stiffness. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Porhcisaliyan, V.D.; Wang, Y.; Tan, N.C.; Jafar, T.H. Socioeconomic status and ethnic variation associated with type 2 diabetes mellitus in patients with uncontrolled hypertension in Singapore. BMJ Open Diabetes Res. Care 2021, 9, e002064. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Heng, D.; Chia, K.S.; Chew, S.K.; Tan, B.Y.; Hughes, K. Risk factors and incident coronary heart disease in Chinese, Malay and Asian Indian males: The Singapore Cardiovascular Cohort Study. Int. J. Epidemiol. 2001, 30, 983–988. [Google Scholar] [CrossRef]
- Gibson, A.O.; Blaha, M.J.; Arnan, M.K.; Sacco, R.L.; Szklo, M.; Herrington, D.M.; Yeboah, J. Coronary artery calcium and incident cerebrovascular events in an asymptomatic cohort. The MESA Study. JACC Cardiovasc. Imaging 2014, 7, 1108–1115. [Google Scholar] [CrossRef]
- Ridker, P.M.; Everett, B.M.; Pradhan, A.; MacFadyen, J.G.; Solomon, D.H.; Zaharris, E.; Mam, V.; Hasan, A.; Rosenberg, Y.; Iturriaga, E.; et al. Low-Dose Methotrexate for the Prevention of Atherosclerotic Events. N. Engl. J. Med. 2019, 380, 752–762. [Google Scholar] [CrossRef]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N. Engl. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef] [PubMed]
- Pasqui, E.; de Donato, G.; Brancaccio, B.; Casilli, G.; Ferrante, G.; Cappelli, A.; Palasciano, G. The Predictive Role of Inflammatory Biochemical Markers in Post-Operative Delirium After Vascular Surgery Procedures. Vasc. Health Risk Manag. 2022, 18, 747–756. [Google Scholar] [CrossRef] [PubMed]
- Tamhane, U.U.; Aneja, S.; Montgomery, D.; Rogers, E.K.; Eagle, K.A.; Gurm, H.S. Association between admission neutrophil to lymphocyte ratio and outcomes in patients with acute coronary syndrome. Am. J. Cardiol. 2008, 102, 653–657. [Google Scholar] [CrossRef]
- Taurino, M.; Aloisi, F.; Del Porto, F.; Nespola, M.; Dezi, T.; Pranteda, C.; Rizzo, L.; Sirignano, P. Neutrophil-to-Lymphocyte Ratio Could Predict Outcome in Patients Presenting with Acute Limb Ischemia. J. Clin. Med. 2021, 10, 4343. [Google Scholar] [CrossRef]
- Pasqui, E.; de Donato, G.; Lazzeri, E.; Molino, C.; Galzerano, G.; Giubbolini, M.; Palasciano, G. High Neutrophil-to-Lymphocyte and Platelet-to-Lymphocyte Ratios Are Associated with a Higher Risk of Hemodialysis Vascular Access Failure. Biomedicines 2022, 10, 2218. [Google Scholar] [CrossRef] [PubMed]
- Iyer, R.P.; Patterson, N.L.; Fields, G.B.; Lindsey, M.L. The history of matrix metalloproteinases: Milestones, myths, and misperceptions. Am. J. Physiol. Circ. Physiol. 2012, 303, H919–H930. [Google Scholar] [CrossRef]
- Halade, G.V.; Jin, Y.F.; Lindsey, M.L. Matrix metalloproteinase (MMP)-9: A proximal biomarker for cardiac remodeling and a distal biomarker for inflammation. Pharmacol. Ther. 2013, 139, 32–40. [Google Scholar] [CrossRef]
- Virani, S.S.; Nambi, V.; Hoogeveen, R.; Wasserman, B.A.; Coresh, J.; Gonzalez, F., 2nd; Chambless, L.E.; Mosley, T.H.; Boerwinkle, E.; Ballantyne, C.M. Relationship between circulating levels of RANTES (regulated on activation, normal T-cell expressed, and secreted) and carotid plaque characteristics: The Atherosclerosis Risk in Communities (ARIC) Carotid MRI Study. Eur. Heart J. 2011, 32, 459–468. [Google Scholar] [CrossRef]
- Tokami, H.; Ago, T.; Sugimori, H.; Kuroda, J.; Awano, H.; Suzuki, K.; Kiyohara, Y.; Kamouchi, M.; Kitazono, T. RANTES has a potential to play a neuroprotective role in an autocrine/paracrine manner after ischemic stroke. Brain Res. 2013, 1517, 122–132. [Google Scholar] [CrossRef]
- Chihara, J.; Yasuba, H.; Tsuda, A.; Urayama, O.; Saito, N.; Honda, K.; Kayaba, H.; Yamashita, T.; Kurimoto, F.; Yamada, H. Elevation of the plasma level of RANTES during asthma attacks. J. Allergy Clin. Immunol. 1997, 100, S52–S55. [Google Scholar] [CrossRef]
- Birger, M.; Kaldjian, A.S.; Roth, G.A.; Moran, A.E.; Dieleman, J.L.; Bellows, B.K. Spending on Cardiovascular Disease and Cardiovascular Risk Factors in the United States: 1996 to 2016. Circulation 2021, 144, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Public Health England. Available online: https://www.gov.uk/government/publications/health-matters-preventing-cardiovascular-disease/health-matters-preventing-cardiovascular-disease (accessed on 14 February 2019).
- Principal Causes of Death: Ministry of Health. 2020. Available online: https://www.moh.gov.sg/resources-statistics/singapore-health-facts/principal-causes-of-death (accessed on 1 September 2022).
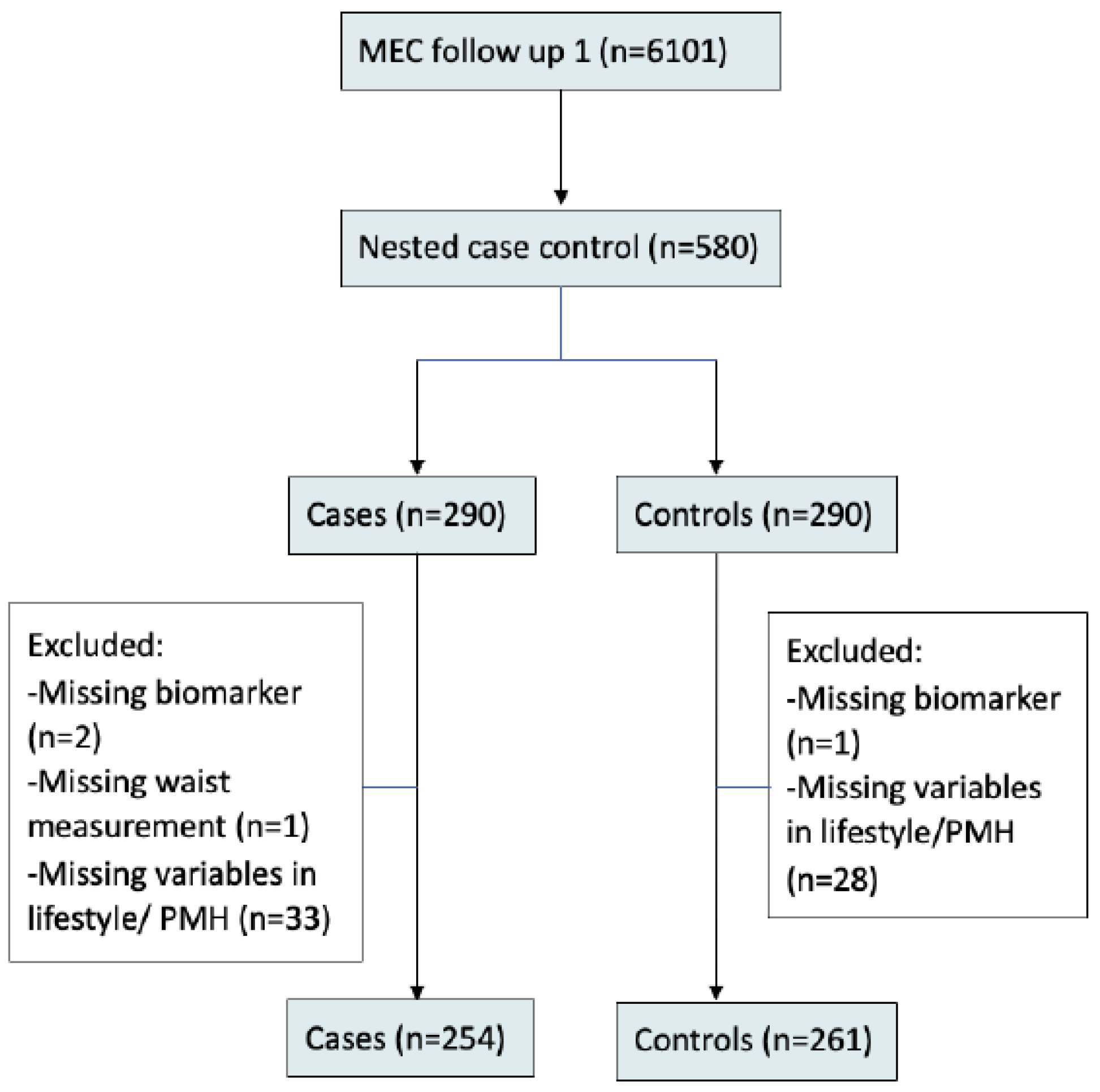
| Total | Case (with CVD) | Control (without CVD) | p-Value * | |
|---|---|---|---|---|
| N = 515 | N = 254 | N = 261 | ||
| Females, n (%) | 178 (34.6%) | 89 (35.0%) | 89 (34.1%) | 0.820 |
| Age | 55.65 (10.31) | 55.5827 (10.36) | 55.7126 (10.27) | 0.890 |
| Ethnicity, n (%) | 0.960 | |||
| Chinese | 331 (64.3%) | 163 (64.2%) | 168 (64.4%) | |
| Malay | 73 (14.2%) | 37 (14.6%) | 36 (13.8%) | |
| Indian | 111 (21.6%) | 54 (21.3%) | 57 (21.8%) | |
| logRANTES | 3.75 (0.43) | 3.76 (0.43) | 3.74 (0.43) | 0.730 |
| logEMMPRIN | 1.55 (0.24) | 1.56 (0.23) | 1.54 (0.25) | 0.340 |
| logMMP-2 | 5.39 (0.24) | 5.38 (0.25) | 5.39 (0.23) | 0.710 |
| logMMP-9 | 5.79 (0.59) | 5.80 (0.58) | 5.79 (0.60) | 0.770 |
| Current/previous smoker, n (%) | 140 (27.2%) | 78 (30.7%) | 62 (23.8%) | 0.076 |
| Hypertension, n (%) | 191 (37.1%) | 119 (46.9%) | 72 (27.6%) | <0.001 |
| Hyperlipidemia, n (%) | 197 (38.3%) | 112 (44.1%) | 85 (32.6%) | 0.007 |
| Diabetes Mellitus, n (%) | 84 (16.3%) | 55 (21.7%) | 29 (11.1%) | 0.001 |
| Metabolic Syndrome, n (%) | 158 (30.7%) | 87(34.3%) | 71(27.2%) | 0.083 |
| Framingham score | 0.18 (0.15) | 0.20 (0.16) | 0.16 (0.14) | 0.004 |
| Gout, n (%) | 38 (7.4%) | 21 (8.3%) | 17 (6.5%) | 0.450 |
| Chronic kidney disease, n (%) | 6 (1.2%) | 5 (2.0%) | 1 (0.4%) | 0.150 |
| Sedentary time total hrs/week | 38.58 (18.58) | 39.64 (18.67) | 37.56 (18.47) | 0.200 |
| Family history, n (%) | 137 (26.6%) | 76 (29.9%) | 61 (23.4%) | 0.093 |
| Waist, cm | 87.86 (11.48) | 89.30 (11.34) | 86.46 (11.46) | 0.005 |
| BMI, kg/m² | 25.45 (4.26) | 26.06 (4.41) | 24.86 (4.03) | 0.001 |
| SBP, mmHg | 129.66 (18.38) | 132.13 (18.87) | 127.26 (17.59) | 0.003 |
| DBP, mmHg | 77.04 (11.56) | 77.82 (12.22) | 76.28 (10.86 | 0.130 |
| LVH by ECG, n (%) | 17(3.3%) | 11(4.3%) | 6 (2.3%) | 0.200 |
| TC, mmol/L | 5.11 (0.94) | 5.10 (0.97) | 5.12 (0.91) | 0.790 |
| HDL, mmol/L | 1.27 (0.31) | 1.25 (0.27) | 1.30 (0.34) | 0.066 |
| LDL, mmol/L | 3.16 (0.82) | 3.16 (0.86) | 3.16 (0.77) | 1 |
| Cholesterol Ratio | 4.18 (1.00) | 4.24 (1.03) | 4.12 (0.99) | 0.18 |
| Triglycerides, mmol/L | 1.56 (0.85) | 1.61 (0.91) | 1.52 (0.80) | 0.22 |
| Creatinine, μmol/L | 75.11 (22.28) | 77.39 (26.80) | 72.89 (16.52) | 0.022 |
| eGFR, mL/min/1.73m2, | 91.96 (27.35) | 92.93 (29.66) | 91.01 (24.93) | 0.430 |
| Hs-CRP, mg/L | 2.81 (6.64) | 3.51 (8.97) | 2.14 (2.81) | 0.019 |
| HbA1c % | 6.10 (1.29) | 6.31(1.46) | 5.90 (1.07) | <0.001 |
| Lipid lowering medications, n (%) | 146(28.3) | 91(35.8) | 55(21.1) | <0.001 |
| Hypertensive medications, n (%) | 151(29.3) | 96(37.8) | 55(21.1) | <0.001 |
| Variable | (a) logRANTES | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Model 1.0 | Model 1.1 | Model 1.2 | Model 1.3 | Model 1.4 | |||||||||||
| β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | |
| CVD composite | 0.013 | (−0.061, 0.087) | 0.731 | ||||||||||||
| CAD | 0.078 | (−0.089,0.082) | 0.195 | 0.008 | (−0.088, 0.103) | 0.875 | |||||||||
| Arrhythmia | 0.048 | (−0.262, 0.165) | 0.656 | −0.008 | (−0.223, 0.207) | 0.943 | |||||||||
| Stroke | 0.078 | (−0.40, 0.196) | 0.195 | 0.095 | (−0.037, 0.226) | 0.159 | |||||||||
| Age | −0.04 | (−0.076, −0.004) | 0.029 | −0.042 | (−0.079, −0.004) | 0.029 | −0.042 | (−0.078, −0.006) | 0.021 | −0.043 | (−0.080, −0.007) | 0.021 | −0.036 | (−0.076, 0.004) | 0.08 |
| Male | ref | ref | |||||||||||||
| Female | 0.153 | (0.077, 0.230) | <0.001 | 0.141 | (0.06, 0.221) | 0.001 | 0.167 | (0.090, 0.243) | <0.001 | 0.212 | (0.109, 0.314) | <0.001 | 0.189 | (0.078, 0.300) | 0.001 |
| Ethnicity | 0.055 * | 0.213 * | 0.268 * | 0.392* | |||||||||||
| Chinese | ref | ref | ref | ref | |||||||||||
| Malay | 0.059 | (−0.049, 0.167) | 0.282 | 0.013 | (−0.095, 0.121) | 0.814 | 0.002 | (−0.109, 0.113) | 0.976 | −0.007 | (−0.131, 0.117) | 0.911 | |||
| Indian | 0.109 | (0.017, 0.201) | 0.02 | 0.083 | (−0.010,0.176) | 0.081 | 0.076 | (−0.019, 0.172) | 0.118 | 0.067 | (−0.036, 0.169) | 0.204 | |||
| Gout | −0.105 | (−0.246, 0.036) | 0.143 | ||||||||||||
| CKD | −0.113 | (−0.458, 0.232) | 0.519 | −0.109 | (−0.466, 0.248) | 0.548 | −0.168 | (−0.617, 0.281) | 0.463 | ||||||
| Family History | −0.045 | (−0.129, 0.038) | 0.288 | ||||||||||||
| Sedentary time, | 0.001 | (−0.001, 0.003) | 0.279 | 0.001 | (−0.001, 0.004) | 0.209 | |||||||||
| Hrs/week | |||||||||||||||
| BMI kg/m² | 0.01 | (0.001, 0.018) | 0.03 | 0.004 | (−0.006, 0.014) | 0.444 | 0.003 | (−0.009, 0.014) | 0.652 | ||||||
| Metabolic Syndrome | 0.025 | (−0.055, 0.105) | 0.542 | −0.022 | (−0.111, 0.066) | 0.621 | −0.01 | (−0.108, 0.088) | 0.84 | ||||||
| LVH ECG | −0.161 | (−0.368, 0.046) | 0.127 | ||||||||||||
| Framingham score | −0.405 | (−0.645, −0.165) | 0.001 | ||||||||||||
| LDL mmol/L | 0.017 | (−0.119, 0.153) | 0.804 | 0.041 | (−0.102, 0.184) | 0.575 | |||||||||
| Creatinine mg/L | −0.133 | (−0.285, 0.183) | 0.085 | 0.136 | (−0.071, 0.343) | 0.198 | 0.131 | (−0.094, 0.355) | 0.254 | ||||||
| Hs−CRP mg/L | 0.044 | (0.012, 0.077) | 0.008 | 0.041 | (0.007, 0.075) | 0.019 | 0.035 | (0.001, 0.068) | 0.041 | 0.031 | (−0.004, 0.067) | 0.083 | 0.034 | (−0.004, 0.071) | 0.081 |
| Hba1c % | 0.096 | (−0.112, 0.304) | 0.366 | ||||||||||||
| Lipid lowering medications | 0.018 | (−0.064, 0.100) | 0.669 | ||||||||||||
| Hypertensive medications | 0.022 | (−0.060, 0.103) | 0.6 | ||||||||||||
| Variable | (b) logEMMPRIN | ||||||||||||||
| Model1.0 | Model1.1 | Model 1.2 | Model 1,3 | Model 1.4 | |||||||||||
| β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | |
| CVD composite | 0.02 | (−0.021, 0.062) | 0.342 | ||||||||||||
| CAD | 0.011 | (−0.037, 0.059) | 0.641 | 0.012 | (−0.042, 0.066) | 0.66 | |||||||||
| Arrhythmia | −0.115 | (−0.236, 0.005) | 0.061 | −0.113 | (−0.232, 0.005) | 0.061 | −0.11 | (−0.230, 0.009) | 0.07 | −0.11 | (−0.230,0.009) | 0.07 | −0.124 | (−0.245. −0.003 | 0.045 |
| Stroke | 0.075 | (0.009, 0.141) | 0.027 | 0.064 | (−0.009, 0.137) | 0.084 | 0.065 | (−0.008, 0.139) | 0.081 | 0.065 | (−0.008,0.139) | 0.081 | 0.06 | (−0.014, 0.135) | 0.11 |
| Age | 0.012 | (−0.008, 0.032) | 0.244 | 0.013 | (−.008, 0.035) | 0.232 | 0.012 | (−0.010,0.034) | 0.289 | 0.012 | (−0.010, 0.035) | 0.284 | |||
| Male | ref | ref | ref | ref | |||||||||||
| Female | 0.006 | (−0.037, 0.050) | 0.774 | 0.021 | (−0.025, 0.067) | 0.359 | 0.02 | (−0.026, 0.066) | 0.402 | 0.042 | (−0.020, 0.105) | 0.185 | |||
| Ethnicity | 0.018 * | 0.127 * | 0.130 * | 0.123 * | |||||||||||
| Chinese | ref | ref | ref | ref | |||||||||||
| Malay | −0.004 | (−0.065, 0.056) | 0.887 | −0.018 | (−0.086, 0.050) | 0.601 | −0.019 | (−0.087, 0.049) | 0.578 | −0.024 | (−0.094, 0.046) | 0.499 | |||
| Indian | 0.072 | (0.021, 0.123) | 0.006 | 0.051 | ( −0.006, 0.107) | 0.078 | 0.05 | (−0.007, 0.106) | 0.084 | 0.048 | (−0.010, 0.106) | 0.105 | |||
| Gout | 0.055 | (−0.024, 0.135) | 0.172 | ||||||||||||
| CKD | 0.163 | (−0.030, 0.357) | 0.098 | 0.255 | (0.021, 0.489) | 0.033 | 0.258 | (−0.006, 0.107) | 0.031 | 0.257 | (0.022,0.491) | 0.032 | 0.216 | (−0.037.0.469) | 0.094 |
| Family History | 0.028 | (−0.019, 0.075) | 0.241 | ||||||||||||
| Sedentary time, | −0.001 | (−0.002, 0.001) | 0.301 | 0 | (−0.002, 0.001) | 0.556 | 0 | (−0.002, 0.001) | 0.537 | ||||||
| Hrs/week | |||||||||||||||
| BMI kg/m² | 0.003 | (−0.002, 0.008) | 0.222 | 0.002 | (−0.004. 0.009) | 0.459 | |||||||||
| Metabolic Syndrome | −0.017 | (−0.062, 0.028) | 0.453 | −0.024 | (−0.079, 0.031) | 0.392 | |||||||||
| LVH ECG | −0.02 | (−0.137, 0.096) | 0.731 | ||||||||||||
| Framingham score | 0.048 | (−0.089, 0.184) | 0.491 | ||||||||||||
| LDL mmol/L | 0.109 | (0.033, 0.184) | 0.005 | 0.105 | (0.027, 0.183) | 0.009 | 0.112 | (0.033, 0.191) | 0.005 | 0.112 | (0.034, 0.191) | 0.005 | 0.111 | (0.030, 0.191) | 0.007 |
| Creatinine mg/L | 0.066 | (−0.019, 0.151) | 0.129 | 0.062 | (−0.065, 0.188) | 0.339 | |||||||||
| Hs−CRP mg/L | 0.019 | (0.001, 0.038) | 0.04 | 0.02 | (0.000, 0.039) | 0.045 | 0.018 | (−0.002, 0.038) | 0.086 | 0.018 | (−0.002, 0.038) | 0.084 | 0.017 | (−0.004, 0.038) | 0.121 |
| Hba1c % | 0.013 | (−0.104, 0.130) | 0.827 | ||||||||||||
| Lipid lowering medications | −0.022 | (−0.069, 0.024) | 0.342 | ||||||||||||
| Hypertensive medications | −0.012 | (−0.058, 0.033) | 0.597 | ||||||||||||
| Variable | (c) logMMP2 | ||||||||||||||
| Model 1.0 | Model 1.1 | Model 1.2 | Model 1.3 | Model 1.4 | |||||||||||
| β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | |
| CVD composite | −0.008 | (−0.049, 0.034) | 0.713 | ||||||||||||
| CAD | 0.025 | (−0.023,0.072) | 0.311 | 0.027 | (−0.021, 0.075) | 0.269 | 0.031 | (−0.022, 0.084) | 0.256 | ||||||
| Arrhythmia | 0.004 | (−0.114, 0.122) | 0.953 | 0 | (−0.120, 0.120) | 0.998 | |||||||||
| Stroke | −0.031 | (−0.096, 0.035) | 0.359 | −0.062 | (−0.134, 0.010) | 0.089 | −0.038 | (−0.103, 0.028) | 0.26 | −0.041 | (−0.107, 0.025) | 0.221 | −0.065 | (−0.138, 0.009) | 0.084 |
| Age | 0.033 | (0.013, 0.053) | 0.001 | 0.032 | (0.011, 0.053) | 0.003 | 0.034 | (0.014, 0.054) | 0.001 | 0.036 | (0.015, 0.056) | 0.001 | 0.033 | (0.010, 0.055) | 0.004 |
| Male | ref | ref | ref | ref | |||||||||||
| Female | 0.005 | (−0.038, 0.049) | 0.809 | −0.001 | (−0.044, 0.042) | 0.963 | 0.001 | (−0.043, 0.044) | 0.982 | 0.013 | (−0.049, 0.075) | 0.684 | |||
| Ethnicity | 0.736 * | 0.815 * | 0.700 * | 0.545 * | |||||||||||
| Chinese | ref | ref | ref | ref | |||||||||||
| Malay | −0.023 | (−0.084, 0.037) | 0.454 | −0.011 | (−0.072, 0.050) | 0.72 | −0.026 | (−0.090, 0.037) | 0.418 | −0.039 | (−0.108, 0.031) | 0.274 | |||
| Indian | 0.002 | (−0.050, 0.053) | 0.946 | 0.011 | (−0.040, 0.063) | 0.664 | 0 | (−0.053, 0.053) | 0.999 | −0.006 | (−0.063, 0.052) | 0.849 | |||
| Gout | 0.011 | (−0.068, 0.090) | 0.782 | ||||||||||||
| CKD | −0.011 | (−0.203, 0.181) | 0.909 | −0.036 | (−0.286, 0.214) | 0.777 | |||||||||
| Family History | −0.025 | (−0.072, 0.022) | 0.294 | ||||||||||||
| Sedentary time, | 0.001 | (−0.001, 0.002) | 0.327 | 0.001 | (−0.001, 0.002) | 0.371 | |||||||||
| Hrs/week | |||||||||||||||
| BMI kg/m² | 0.002 | (−0.003, 0.007) | 0.361 | 0.004 | (−0.001, 0.009) | 0.164 | 0.005 | (−0.002, 0.011) | 0.144 | ||||||
| Metabolic Syndrome | 0.022 | (−0.023, 0.067) | 0.336 | 0.01 | (−0.044,0.65) | 0.715 | |||||||||
| LVH ECG | 0.013 | (−0.103, 0.128) | 0.829 | ||||||||||||
| Framingham score | 0.157 | (0.023, 0.292) | 0.022 | ||||||||||||
| LDL mmol/L | −0.013 | (−0.088, 0.063) | 0.739 | 0.006 | (−0.070, 0.082) | 0.873 | 0.006 | (−0.073, 0.086) | 0.873 | ||||||
| Creatinine mg/L | 0.002 | (−0.082, 0.087) | 0.956 | −0.005 | (−0.130, 0.120) | 0.938 | |||||||||
| Hs−CRP mg/L | −0.015 | (−0.034, 0.003) | 0.101 | −0.019 | (−0.040, 0.002) | 0.072 | |||||||||
| Hba1c % | 0.021 | (−0.095, 0.137) | 0.725 | ||||||||||||
| Lipid lowering medications | 0.012 | (−0.033, 0.058) | 0.598 | ||||||||||||
| Hypertensive medications | 0.024 | (−0.021, 0.070) | 0.29 | ||||||||||||
| Variable | (d) logMMP9 | ||||||||||||||
| Model1.0 | Model 1.1 | Model 1.2 | Model 1.3 | Model 1.4 | |||||||||||
| β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | |
| CVD composite | 0.013 | (−0.088, 0.118) | 0.731 | ||||||||||||
| CAD | 0.033 | (−0.085, 0.152) | 0.583 | −0.002 | (−0.120, 0.115) | 0.972 | 0.035 | (−0.097, 0.168) | 0.6 | ||||||
| Arrhythmia | −0.279 | (−0.579, 0.020) | 0.068 | −0.256 | (−0.082, 0.286) | 0.094 | |||||||||
| Stroke | 0.108 | (−0.055, 0.272) | 0.194 | 0.102 | (−0.556, 0.044) | 0.275 | |||||||||
| Age | −0.015 | (−0.065, 0.035) | 0.549 | 0.005 | (−0.044, 0.055) | 0.829 | 0.008 | (−0.043, 0.058) | 0.767 | 0.02 | (−0.036, 0.075) | 0.49 | |||
| Male | ref | ref | ref | ref | ref | ||||||||||
| Female | −0.199 | (−0.306, −0.092) | <0.001 | −0.186 | (−0.298, −0.074) | 0.001 | −0.196 | (−0.303, −0.090) | <0.001 | −0.196 | (−0.303, −0.089) | <0.001 | −0.122 | (−0.277, 0.033) | 0.122 |
| Ethnicity | 0.029 * | 0.043 * | 0.184 * | 0.208 * | 0.032* | ||||||||||
| Chinese | ref | ref | 0.208 | ref | ref | ref | |||||||||
| Malay | 0.05 | (−0.100, 0.199) | 0.515 | 0.072 | (−0.092, 0.236) | 0.388 | 0.03 | (−0.121, 0.181) | 0.698 | 0.018 | (−0.139, 0.174) | 0.823 | 0.075 | (−0.099, 0.248) | 0.397 |
| Indian | 0.173 | (0.046, 0.300) | 0.008 | 0.175 | (0.037, 0.313) | 0.013 | 0.122 | (−0.008, 0.251) | 0.066 | 0.118 | (−0.014, 0.250) | 0.08 | 0.192 | (0.048, 0.335) | 0.009 |
| Gout | 0.004 | (−0.192, 0.201) | 0.966 | ||||||||||||
| CKD | 0.249 | (−0.229, 0.728) | 0.306 | −0.044 | (−0.670, 0.583) | 0.892 | |||||||||
| Family History | −0.06 | (−0.176, 0.056) | 0.313 | ||||||||||||
| Sedentary time, | 0.002 | (−0.001, 0.005) | 0.115 | 0.002 | (−0.001, 0.005) | 0.258 | |||||||||
| Hrs/week | |||||||||||||||
| BMI kg/m² | 0.009 | (−0.003, 0.021) | 0.139 | 0.001 | (−0.012, 0.014) | 0.899 | 0.003 | (−0.013, 0.019) | 0.681 | ||||||
| Metabolic Syndrome | −0.026 | (−0.137, 0.086) | 0.649 | −0.104 | (−0.241, 0.033) | 0.136 | |||||||||
| LVH ECG | −0.062 | (−0.350, 0.225) | 0.67 | ||||||||||||
| Framingham score | 0.227 | (−0.109, 0.562) | 0.185 | ||||||||||||
| LDL mmol/L | 0.104 | (−0.084, 0.292) | 0.278 | 0.103 | (−0.085, 0.290) | 0.283 | 0.094 | (−0.105, 0.294) | 0.353 | ||||||
| Creatine mg/L | 0.316 | (0.107, 0.525) | 0.003 | 0.134 | (−0.179, 0.447) | 0.401 | |||||||||
| Hs−CRP mg/L | 0.072 | (0.027, 0.118) | 0.002 | 0.06 | (0.010, 0.110) | 0.019 | 0.063 | (0.017, 0.110) | 0.008 | 0.063 | (0.014, 0.112) | 0.012 | 0.063 | (0.011, 0.116) | 0.019 |
| Hba1c % | 0.079 | (−0.210, 0.367) | 0.591 | ||||||||||||
| Lipid lowering medications | −0.097 | (−0.210, 0.017) | 0.095 | ||||||||||||
| Hypertensive medications | −0.031 | (−0.144, 0.082) | 0.589 | ||||||||||||
| log RANTES | log EMMPRIN | log MMP-2 | log MMP-9 | |
|---|---|---|---|---|
| OR, 95% CI | OR, 95% CI | OR, 95% CI | OR, 95% CI | |
| Univariate | 1.074 | 1.421 | 0.872 | 1.044 |
| (0.716, 1.700) | (0.688, 2.936) | (0.422, 1.803) | (0.780–1.397) | |
| Model 2.1 | 1.064 | 1.441 | 0.876 | 1.009 |
| (0.703, 1.609) | (0.480, 1.720) | (0.421, 1.826) | (0.749, 1.358) | |
| Model 2.2 | 1.067 | 1.471 | 0.879 | 1.013 |
| (0.704, 1.617) | (0.702, 3.084) | (0.422, 1.832) | (0.750, 1.369) | |
| Model 2.3 | 0.937 | 1.348 | 0.834 | 1.046 |
| (0.602, 1.457) | (0.610, 2.980) | (0.384, 1.810) | (0.762, 1.436) | |
| Model 2.4 | 0.934 | 1.348 | 0.836 | 1.044 |
| (0.601, 1.452) | (0.610, 2.980) | (0.385, 1.817) | (0.760, 1.433) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.Y.-T.; Tan, C.S.; Lai, M.K.P.; Hilal, S. Factors Associated with RANTES, EMMPIRIN, MMP2 and MMP9, and the Association of These Biomarkers with Cardiovascular Disease in a Multi-Ethnic Population. J. Clin. Med. 2022, 11, 7281. https://doi.org/10.3390/jcm11247281
Wang LY-T, Tan CS, Lai MKP, Hilal S. Factors Associated with RANTES, EMMPIRIN, MMP2 and MMP9, and the Association of These Biomarkers with Cardiovascular Disease in a Multi-Ethnic Population. Journal of Clinical Medicine. 2022; 11(24):7281. https://doi.org/10.3390/jcm11247281
Chicago/Turabian StyleWang, Laureen Yi-Ting, Chuen Seng Tan, Mitchell K. P. Lai, and Saima Hilal. 2022. "Factors Associated with RANTES, EMMPIRIN, MMP2 and MMP9, and the Association of These Biomarkers with Cardiovascular Disease in a Multi-Ethnic Population" Journal of Clinical Medicine 11, no. 24: 7281. https://doi.org/10.3390/jcm11247281
APA StyleWang, L. Y.-T., Tan, C. S., Lai, M. K. P., & Hilal, S. (2022). Factors Associated with RANTES, EMMPIRIN, MMP2 and MMP9, and the Association of These Biomarkers with Cardiovascular Disease in a Multi-Ethnic Population. Journal of Clinical Medicine, 11(24), 7281. https://doi.org/10.3390/jcm11247281







