Impact of Amiodarone Therapy on the Ablation Outcome of Ventricular Tachycardia in Arrhythmogenic Right Ventricular Cardiomyopathy
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Electrophysiological Study, Mapping, and Ablation
2.3. Follow-Up and Recurrences of Ventricular Arrhythmia (VA)
2.4. Statistical Analysis
3. Results
3.1. Study Population
3.1.1. Patient Selection and Baseline Characteristics of ARVC Patients
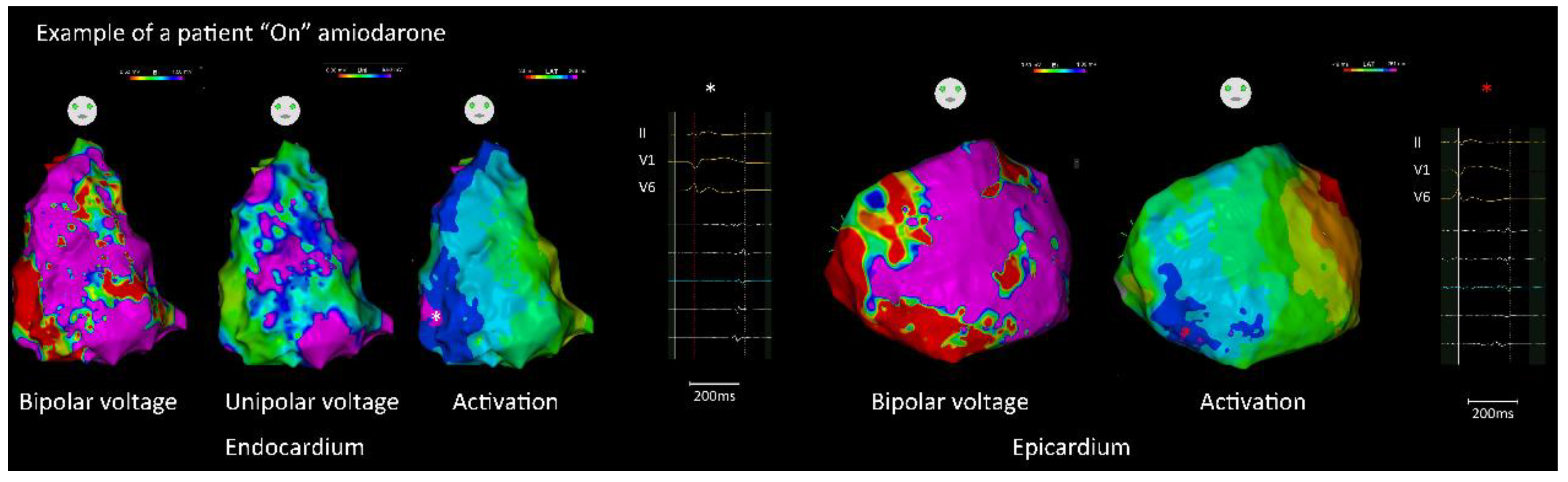
3.1.2. Endocardial and Epicardial Substrate Characteristics
3.1.3. Mapping of Ventricular Tachycardia and Catheter Ablation
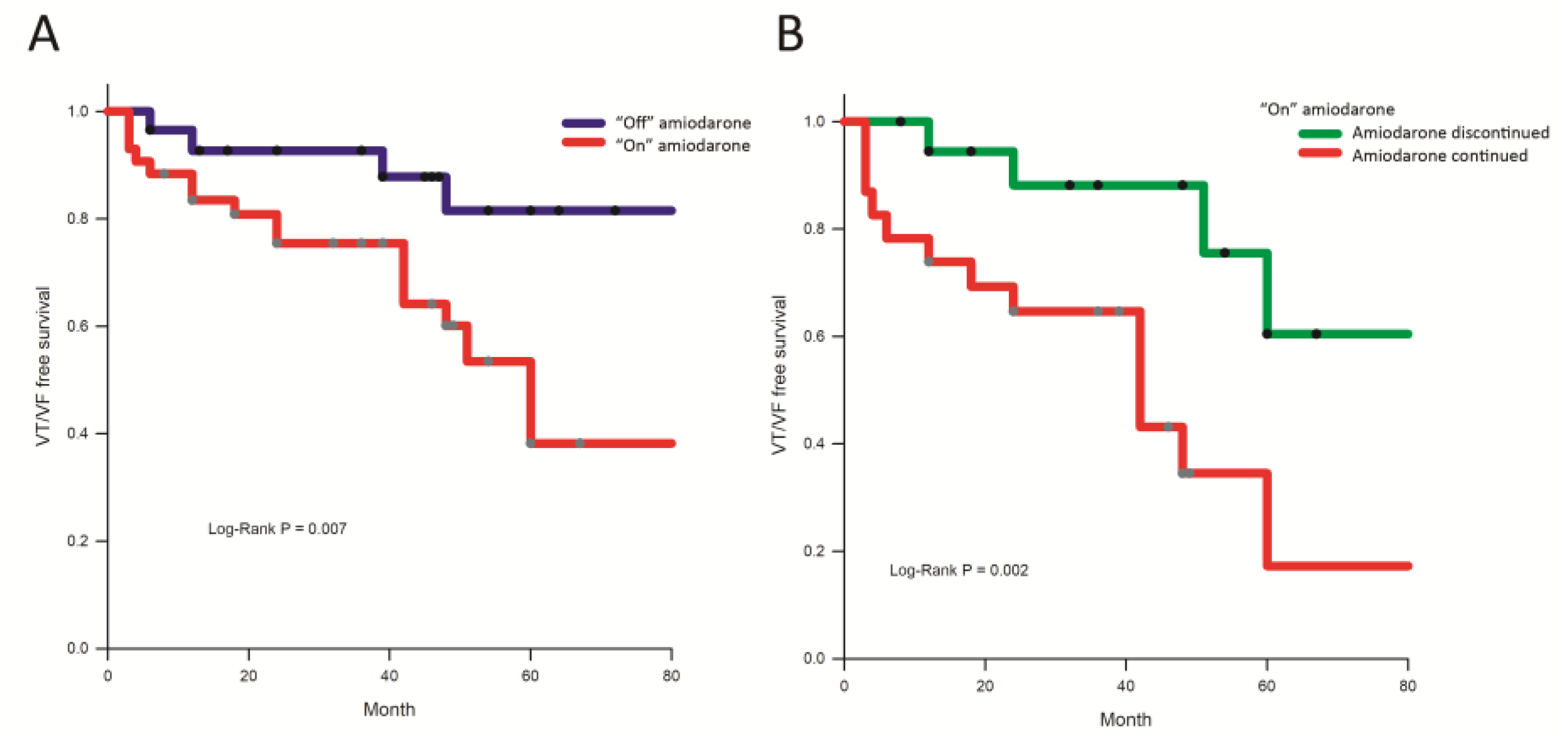
3.1.4. Follow-Up
4. Discussion
4.1. Main Findings
4.2. Amiodarone and the Abnormal Substrate
4.3. Amiodarone Use and Ablation Outcomes
4.4. Clinical Implications
4.5. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Corrado, D.; Link, M.S.; Calkins, H. Arrhythmogenic Right Ventricular Cardiomyopathy. N. Engl. J. Med. 2017, 376, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Dalal, D.; Nasir, K.; Bomma, C.; Prakasa, K.; Tandri, H.; Piccini, J.; Roguin, A.; Tichnell, C.; James, C.; Russell, S.D.; et al. Arrhythmogenic right ventricular dysplasia: A United States experience. Circulation 2005, 112, 3823–3832. [Google Scholar] [CrossRef] [PubMed]
- Towbin, J.A.; McKenna, W.J.; Abrams, D.J.; Ackerman, M.J.; Calkins, H.; Darrieux, F.C.C.; Daubert, J.P.; de Chillou, C.; DePasquale, E.C.; Desai, M.Y.; et al. 2019 HRS expert consensus statement on evaluation, risk stratification, and management of arrhythmogenic cardiomyopathy. Heart Rhythm 2019, 16, e301–e372. [Google Scholar] [CrossRef] [PubMed]
- Vassallo, P.; Trohman, R.G. Prescribing amiodarone: An evidence-based review of clinical indications. JAMA 2007, 298, 1312–1322. [Google Scholar] [CrossRef] [PubMed]
- Colunga Biancatelli, R.M.; Congedo, V.; Calvosa, L.; Ciacciarelli, M.; Polidoro, A.; Iuliano, L. Adverse reactions of Amiodarone. J. Geriatr. Cardiol. 2019, 16, 552–566. [Google Scholar] [CrossRef]
- Di Biase, L.; Romero, J.; Du, X.; Mohanty, S.; Trivedi, C.; Della Rocca, D.G.; Patel, K.; Sanchez, J.; Yang, R.; Alviz, I.; et al. Catheter ablation of ventricular tachycardia in ischemic cardiomyopathy: Impact of concomitant amiodarone therapy on short- and long-term clinical outcomes. Heart Rhythm 2021, 18, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Marcus, F.I.; McKenna, W.J.; Sherrill, D.; Basso, C.; Bauce, B.; Bluemke, D.A.; Calkins, H.; Corrado, D.; Cox, M.G.; Daubert, J.P.; et al. Diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia: Proposed modification of the Task Force Criteria. Eur. Heart J. 2010, 31, 806–814. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Chung, F.P.; Kuo, L.; Lin, Y.J.; Chang, S.L.; Lo, L.W.; Hu, Y.F.; Tuan, T.C.; Chao, T.F.; Liao, J.N.; et al. Characteristics of recurrent ventricular tachyarrhythmia after catheter ablation in patients with arrhythmogenic right ventricular cardiomyopathy. J. Cardiovasc. Electrophysiol. 2019, 30, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Chung, F.P.; Lin, Y.J.; Chang, S.L.; Lo, L.W.; Hu, Y.F.; Tuan, T.C.; Chao, T.F.; Liao, J.N.; Chang, T.Y.; et al. Clinical significance of J waves with respect to substrate characteristics and ablation outcomes in patients with arrhythmogenic right ventricular cardiomyopathy. Europace 2021, 23, 1418–1427. [Google Scholar] [CrossRef]
- Marchlinski, F.E.; Callans, D.J.; Gottlieb, C.D.; Zado, E. Linear ablation lesions for control of unmappable ventricular tachycardia in patients with ischemic and nonischemic cardiomyopathy. Circulation 2000, 101, 1288–1296. [Google Scholar] [CrossRef] [PubMed]
- Aliot, E.M.; Stevenson, W.G.; Almendral-Garrote, J.M.; Bogun, F.; Calkins, C.H.; Delacretaz, E.; Della Bella, P.; Hindricks, G.; Jais, P.; Josephson, M.E.; et al. EHRA/HRS Expert Consensus on Catheter Ablation of Ventricular Arrhythmias: Developed in a partnership with the European Heart Rhythm Association (EHRA), a Registered Branch of the European Society of Cardiology (ESC), and the Heart Rhythm Society (HRS); in collaboration with the American College of Cardiology (ACC) and the American Heart Association (AHA). Heart Rhythm 2009, 6, 886–933. [Google Scholar] [CrossRef] [PubMed]
- Chung, F.P.; Li, H.R.; Chong, E.; Pan, C.H.; Lin, Y.J.; Chang, S.L.; Lo, L.W.; Hu, Y.F.; Tuan, T.C.; Chao, T.F.; et al. Seasonal variation in the frequency of sudden cardiac death and ventricular tachyarrhythmia in patients with arrhythmogenic right ventricular dysplasia/cardiomyopathy: The effect of meteorological factors. Heart Rhythm 2013, 10, 1859–1866. [Google Scholar] [CrossRef] [PubMed]
- Brembilla-Perrot, B.; Claudon, O.; Houriez, P.; Beurrier, D.; Suty-Selton, C. Absence of change of signal-averaged electrocardiogram identifies patients with ventricular arrhythmias who are non-responders to amiodarone. Int. J. Cardiol. 2002, 83, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Van Herendael, H.; Dorian, P. Amiodarone for the treatment and prevention of ventricular fibrillation and ventricular tachycardia. Vasc. Health Risk Manag. 2010, 6, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Kawase, A.; Ikeda, T.; Nakazawa, K.; Ashihara, T.; Namba, T.; Kubota, T.; Sugi, K.; Hirai, H. Widening of the excitable gap and enlargement of the core of reentry during atrial fibrillation with a pure sodium channel blocker in canine atria. Circulation 2003, 107, 905–910. [Google Scholar] [CrossRef] [PubMed]
- Kirchhof, P.; Degen, H.; Franz, M.R.; Eckardt, L.; Fabritz, L.; Milberg, P.; Laer, S.; Neumann, J.; Breithardt, G.; Haverkamp, W. Amiodarone-induced postrepolarization refractoriness suppresses induction of ventricular fibrillation. J. Pharmacol. Exp. Ther. 2003, 305, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Corrado, D.; Wichter, T.; Link, M.S.; Hauer, R.N.; Marchlinski, F.E.; Anastasakis, A.; Bauce, B.; Basso, C.; Brunckhorst, C.; Tsatsopoulou, A.; et al. Treatment of Arrhythmogenic Right Ventricular Cardiomyopathy/Dysplasia: An International Task Force Consensus Statement. Circulation 2015, 132, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Siddoway, L.A. Amiodarone: Guidelines for use and monitoring. Am. Fam. Physician 2003, 68, 2189–2196. [Google Scholar] [PubMed]
- Connolly, S.J. Evidence-based analysis of amiodarone efficacy and safety. Circulation 1999, 100, 2025–2034. [Google Scholar] [CrossRef] [PubMed]
- Doyle, J.F.; Ho, K.M. Benefits and risks of long-term amiodarone therapy for persistent atrial fibrillation: A meta-analysis. Mayo Clin. Proc. 2009, 84, 234–242. [Google Scholar] [CrossRef] [PubMed]


| “Off” Amiodarone | “On” Amiodarone | p-Value | |
|---|---|---|---|
| (N = 29) | (N = 43) | ||
| Baseline characteristics | |||
| Age | 43.0 ± 13.1 | 46.9 ± 15.4 | 0.260 |
| Sex (Men, %) | 13 (44.8%) | 31 (72.1%) | 0.027 |
| Hypertension | 7 (24.1%) | 14 (32.6%) | 0.598 |
| Diabetes mellitus | 2 (6.9%) | 2 (4.7%) | 0.999 |
| LVEF (%) | 57.2 ± 8.1 | 53.5 ± 8.7 | 0.081 |
| RVEF (%) | 41.0 ± 9.7 | 38.0 ± 11.5 | 0.240 |
| Medication | |||
| Amiodarone dose before CA (mg) | 0 | 232.6 ± 97.9 | |
| Beta blocker before CA | 21 (72.4%) | 29 (67.4%) | 0.795 |
| Class Ic AAD before CA | 10 (34.5%) | 3 (7.0%) | 0.004 |
| Class Ib AAD before CA | 7 (24.1%) | 4 (9.3%) | 0.105 |
| “Continued” amiodarone before discharge | 0 | 38 (88.4%) | |
| “Continued” amiodarone 3 months after CA | 0 (0.0%) | 23 (53.5%) | |
| Amiodarone dose after CA (mg) | 0 | 223.9 ± 106.5 | |
| Task Force criteria * | |||
| Structural abnormalities | |||
| Major | 9 (31.0%) | 25 (58.1%) | 0.076 |
| Minor | 15 (51.7%) | 14 (32.6%) | |
| Fibrofatty replacement | |||
| Major | 7 (24.1%) | 9 (20.9%) | 0.483 |
| Minor | 2 (6.9%) | 13 (30.2%) | |
| Depolarization abnormalities | |||
| Major | 2 (6.9%) | 9 (20.9%) | 0.138 |
| Minor | 26 (89.7%) | 30 (69.8%) | |
| Repolarization abnormalities | |||
| Major | 9 (27.3%) | 7 (15.2%) | 0.421 |
| Minor | 13 (39.4%) | 21 (45.7%) | |
| Ventricular arrhythmias | |||
| Major | 10 (34.5%) | 19 (44.2%) | 0.469 |
| Minor | 19 (65.5%) | 24 (55.8%) | |
| Family history | |||
| Major | 5 (17.2%) | 10 (23.3%) | 0.568 |
| Minor | 0 (0.0%) | 1 (2.3%) | |
| “Off” Amiodarone | “On” Amiodarone | p-Value | |
|---|---|---|---|
| (N = 29) | (N = 43) | ||
| VT morphologies | 1.3 ± 0.6 | 1.7 ± 0.9 | 0.014 |
| VT cycle length | 309.2 ± 54.7 | 304.5 ± 72.5 | 0.774 |
| Procedure time (minutes) | 184.5 ± 51.1 | 220.3 ± 43.9 | 0.019 |
| Ablation time (minutes) | 28.2 ± 24.4 | 55.4 ± 37.7 | 0.001 |
| RV endocardium | |||
| Mapping points | 600.7 ± 528.5 | 850.0 ± 666.8 | 0.096 |
| Bipolar voltage * | 2.1 ± 0.6 | 1.9 ± 0.7 | 0.142 |
| Unipolar voltage * | 5.3 ± 0.7 | 4.9 ± 1.4 | 0.253 |
| Total activation time (ms) | 144.0 ± 32.7 | 158.2 ± 31.8 | <0.001 |
| Bipolar low voltage zone (cm2) | 28.4 ±17.1 | 34.7± 27.4 | 0.275 |
| Bipolar low voltage zone, % | 13.5 ± 7.6 | 15.4 ± 10.7 | 0.415 |
| Bipolar scar (cm2) | 14.2 ± 12.5 | 17.4 ± 13.6 | 0.317 |
| Bipolar scar, % | 6.8 ± 5.3 | 8.0 ± 5.5 | 0.341 |
| Unipolar low voltage zone (cm2) | 55.1 ± 36.3 | 65.3 ± 27.9 | 0.183 |
| Unipolar low voltage zone, % | 22.6 ± 11.7 | 25.8 ±10.2 | 0.215 |
| Area with abnormal electrograms | 6.8 ± 5.0 | 16.8 ± 15.4 | 0.001 |
| RV epicardium | (N = 12) | (N = 35) | |
| Mapping points | 1495.8 ± 950.9 | 2076.8 ± 1776.9 | 0.270 |
| Averaged bipolar voltage (mV) * | 1.3 ± 0.5 | 1.3 ± 0.8 | 0.919 |
| Total activation time (ms) | 183.6 ± 24.2 | 211.2 ± 26.0 | 0.002 |
| Bipolar low voltage zone (cm2) | 95.0 ± 54.8 | 111.5 ± 55.8 | 0.378 |
| Bipolar low voltage zone, % | 36.0 ± 22.2 | 36.3 ± 20.4 | 0.968 |
| Bipolar scar (cm2) | 53.4 ± 43.7 | 56.6 ± 34.2 | 0.797 |
| Bipolar scar, % | 19.0 ± 14.9 | 19.1 ± 11.4 | 0.842 |
| Area with abnormal electrograms | 13.7 ± 21.4 | 28.2 ± 29.0 | 0.024 |
| Univariate Analysis | Multivariate Analysis | |||
|---|---|---|---|---|
| p-Value | HR (95% CI) | p-Value | HR (95% CI) | |
| Age (year) | 0.995 | 1.00 (0.97–1.03) | - | - |
| Sex (male) | 0.051 | 2.56 (0.99–6.56) | 0.605 | 1.38 (0.41–4.68) |
| Hypertension | 0.062 | 2.21 (0.96–5.10) | 0.665 | 1.24 (0.47–3.22) |
| Diabetes mellitus | 0.758 | 1.38 (0.18–10.46) | - | - |
| LVEF (%) | 0.642 | 0.99 (0.95–1.04) | - | - |
| RVEF (%) | 0.049 | 0.96 (0.93–1.00) | 0.494 | 0.98 (0.94–1.03) |
| Amiodarone before CA | 0.013 | 3.54 (1.31–9.60) | 0.038 | 3.04 (1.07–8.66) |
| Amiodarone > 6 months | 0.283 | 1.56 (0.69–3.54) | ||
| Task Force score | 0.550 | 1.08 (0.84–1.38) | - | - |
| Total activation time (RV endo) | 0.009 | 1.02 (1.00–1.03) | 0.385 | 1.03 (0.96–1.12) |
| Bipolar scar (%, RV endo) | 0.283 | 1.04 (0.97–1.12) | - | - |
| Epicardial approach | 0.413 | 1.44 (0.60–3.46) | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, C.-Y.; Chung, F.-P.; Nwe, N.; Hsieh, Y.-C.; Li, C.-H.; Lin, Y.-J.; Chang, S.-L.; Lo, L.-W.; Hu, Y.-F.; Tuan, T.-C.; et al. Impact of Amiodarone Therapy on the Ablation Outcome of Ventricular Tachycardia in Arrhythmogenic Right Ventricular Cardiomyopathy. J. Clin. Med. 2022, 11, 7265. https://doi.org/10.3390/jcm11247265
Lin C-Y, Chung F-P, Nwe N, Hsieh Y-C, Li C-H, Lin Y-J, Chang S-L, Lo L-W, Hu Y-F, Tuan T-C, et al. Impact of Amiodarone Therapy on the Ablation Outcome of Ventricular Tachycardia in Arrhythmogenic Right Ventricular Cardiomyopathy. Journal of Clinical Medicine. 2022; 11(24):7265. https://doi.org/10.3390/jcm11247265
Chicago/Turabian StyleLin, Chin-Yu, Fa-Po Chung, Nwe Nwe, Yu-Cheng Hsieh, Cheng-Hung Li, Yenn-Jiang Lin, Shih-Lin Chang, Li-Wei Lo, Yu-Feng Hu, Ta-Chuan Tuan, and et al. 2022. "Impact of Amiodarone Therapy on the Ablation Outcome of Ventricular Tachycardia in Arrhythmogenic Right Ventricular Cardiomyopathy" Journal of Clinical Medicine 11, no. 24: 7265. https://doi.org/10.3390/jcm11247265
APA StyleLin, C.-Y., Chung, F.-P., Nwe, N., Hsieh, Y.-C., Li, C.-H., Lin, Y.-J., Chang, S.-L., Lo, L.-W., Hu, Y.-F., Tuan, T.-C., Chao, T.-F., Liao, J.-N., Chang, T.-Y., Kuo, L., Wu, C.-I., Liu, C.-M., Liu, S.-H., Cheng, W.-H., & Chen, S.-A. (2022). Impact of Amiodarone Therapy on the Ablation Outcome of Ventricular Tachycardia in Arrhythmogenic Right Ventricular Cardiomyopathy. Journal of Clinical Medicine, 11(24), 7265. https://doi.org/10.3390/jcm11247265







