Annexin A6 Polymorphism Is Associated with Pro-atherogenic Lipid Profiles and with the Downregulation of Methotrexate on Anti-Atherogenic Lipid Profiles in Psoriasis
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Study Design
2.2. Treatment
2.3. Assessment of Lipid Levels and Disease Characteristics
2.4. DNA Extraction and Genotyping
2.5. Western Blot Assay
2.6. Statistical Analysis
3. Results
3.1. The CC Genotype of AnxA6, rs11960458, Is Associated with Lower Levels of Pro-Atherogenic Lipids in Psoriatic Patients
3.2. The Reduction of MTX on the Levels of Anti-Atherogenic Lipids and TGs Was Associated with the rs11960458 Genotype
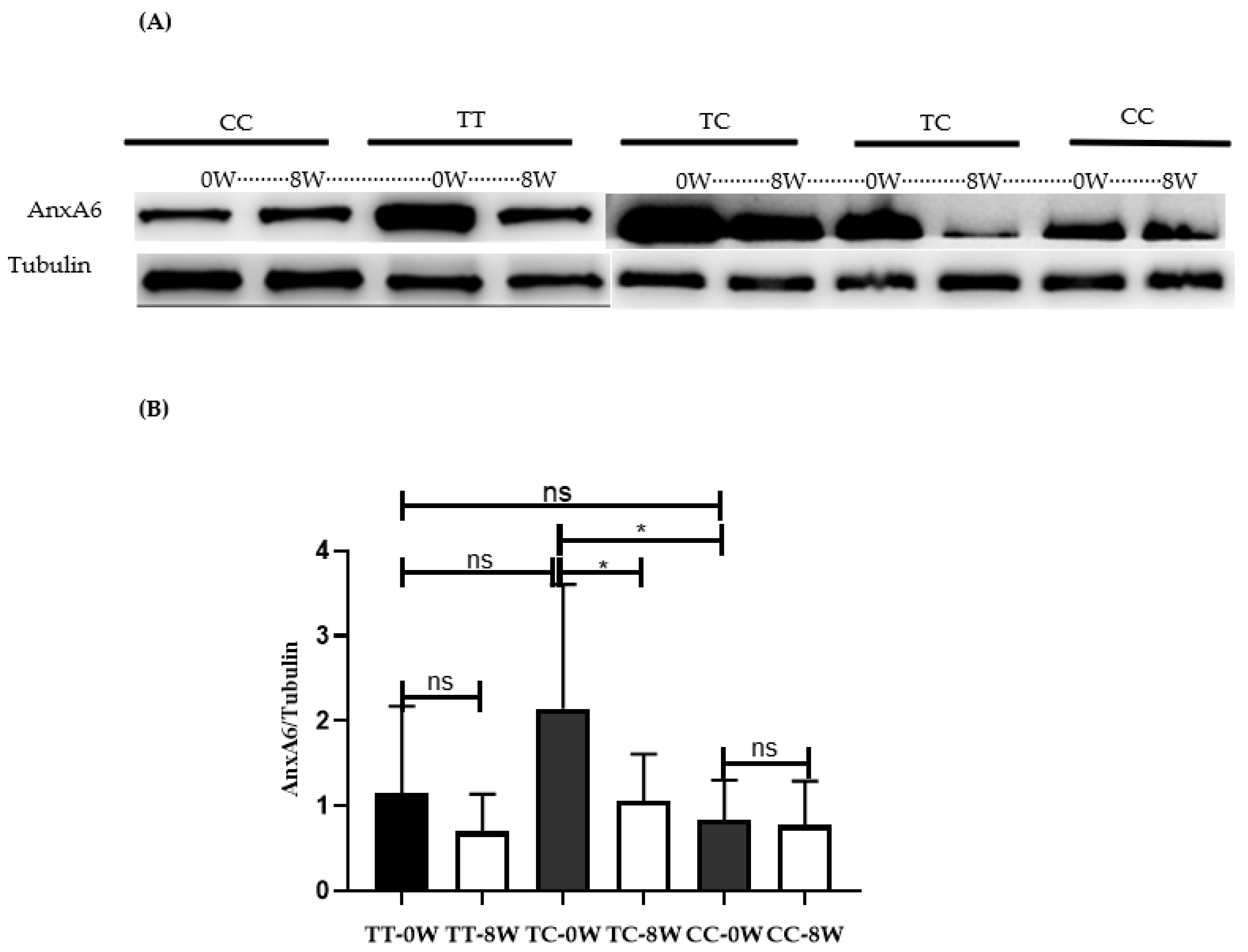
3.3. The Level of AnxA6 Protein in PBMCs Was Significantly Increased in TC Genotype Carriers and Was Significantly Downregulated after MTX Treatment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AnxA6 | AnnexinA6 |
| ApoA1 | Apolipoprotein A1 |
| ApoB | Apolipoprotein B |
| BMI | Body mass index |
| BSA | Body surface area |
| HDL-C | High-density lipoprotein–cholesterol |
| LDL | Low-density lipoprotein |
| LP(a) | Lipoprotein (a) |
| MTX | Methotrexate |
| PASI | Psoriasis area severity index |
| PBMCs | Peripheral blood mononuclear cells |
| SNPs | Single-nucleotide polymorphisms |
| TC | Total cholesterol |
| TG | Triglyceride |
References
- Michalek, I.M.; Loring, B.; John, S.M. A systematic review of worldwide epidemiology of psoriasis. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Dobrzyńska, I.; Szachowicz-Petelska, B.; Wroński, A.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Changes in the Physicochemical Properties of Blood and Skin Cell Membranes as a Result of Psoriasis Vulgaris and Psoriatic Arthritis Development. Int. J. Mol. Sci. 2020, 21, 9129. [Google Scholar] [CrossRef] [PubMed]
- Oliveira Mde, F.; Rocha Bde, O.; Duarte, G.V. Psoriasis: Classical and emerging comorbidities. An. Bras. Dermatol. 2015, 90, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Khalil, S.; Bardawil, T.; Stephan, C.; Darwiche, N.; Abbas, O.; Kibbi, A.G.; Nemer, G.; Kurban, M. Retinoids: A journey from the molecular structures and mechanisms of action to clinical uses in dermatology and adverse effects. J. Dermatol. Treat. 2017, 28, 684–696. [Google Scholar] [CrossRef]
- Oh, E.H.; Ro, Y.S.; Kim, J.E. Epidemiology and cardiovascular comorbidities in patients with psoriasis: A Korean nationwide population-based cohort study. J. Dermatol. 2017, 44, 621–629. [Google Scholar] [CrossRef]
- Edson-Heredia, E.; Zhu, B.; Lefevre, C.; Wang, M.; Barrett, A.; Bushe, C.J.; Cox, A.; Wu, J.J.; Maeda-Chubachi, T. Prevalence and incidence rates of cardiovascular, autoimmune, and other diseases in patients with psoriatic or psoriatic arthritis: A retrospective study using Clinical Practice Research Datalink. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 955–963. [Google Scholar] [CrossRef]
- Saurat, J.H.; Langley, R.G.; Reich, K.; Unnebrink, K.; Sasso, E.H.; Kampman, W. Relationship between methotrexate dosing and clinical response in patients with moderate to severe psoriasis: Subanalysis of the CHAMPION study. Br. J. Dermatol. 2011, 165, 399–406. [Google Scholar] [CrossRef]
- Ronda, N.; Greco, D.; Adorni, M.P.; Zimetti, F.; Favari, E.; Hjeltnes, G.; Mikkelsen, K.; Borghi, M.O.; Favalli, E.G.; Gatti, R.; et al. Newly identified antiatherosclerotic activity of methotrexate and adalimumab: Complementary effects on lipoprotein function and macrophage cholesterol metabolism. Arthritis Rheumatol. 2015, 67, 1155–1164. [Google Scholar] [CrossRef]
- Reiss, A.B.; Carsons, S.E.; Anwar, K.; Rao, S.; Edelman, S.D.; Zhang, H.; Fernandez, P.; Cronstein, B.N.; Chan, E.S. Atheroprotective effects of methotrexate on reverse cholesterol transport proteins and foam cell transformation in human THP-1 monocyte/macrophages. Arthritis Rheum. 2008, 58, 3675–3683. [Google Scholar] [CrossRef]
- Westlake, S.L.; Colebatch, A.N.; Baird, J.; Kiely, P.; Quinn, M.; Choy, E.; Ostor, A.J.; Edwards, C.J. The effect of methotrexate on cardiovascular disease in patients with rheumatoid arthritis: A systematic literature review. Rheumatology 2010, 49, 295–307. [Google Scholar] [CrossRef]
- Bordy, R.; Verhoeven, F.; Tournier-Nappey, M.; Wendling, D.; Demougeot, C.; Totoson, P. Methotrexate did not improve endothelial function in rheumatoid arthritis: A study in rats with adjuvant-induced arthritis. Clin. Exp. Rheumatol. 2019, 37, 81–88. [Google Scholar] [PubMed]
- Grewal, T.; Hoque, M.; Conway, J.R.W.; Reverter, M.; Wahba, M.; Beevi, S.S.; Timpson, P.; Enrich, C.; Rentero, C. Annexin A6-A multifunctional scaffold in cell motility. Cell Adhes. Migr. 2017, 11, 288–304. [Google Scholar] [CrossRef] [PubMed]
- Enrich, C.; Rentero, C.; de Muga, S.V.; Reverter, M.; Mulay, V.; Wood, P.; Koese, M.; Grewal, T. Annexin A6-Linking Ca2+ signaling with cholesterol transport. Biochim. Biophys. Acta 2011, 1813, 935–947. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.D.; Cheng, H.; Wang, Z.X.; Zhang, A.P.; Wang, P.G.; Xu, J.H.; Zhu, Q.X.; Zhou, H.S.; Ellinghaus, E.; Zhang, F.R.; et al. Association analyses identify six new psoriasis susceptibility loci in the Chinese population. Nat. Genet. 2010, 42, 1005–1009. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.Y.; Cheng, H.; Wang, W.J.; Wang, W.J.; Fu, H.Y.; Liu, L.H.; Zhang, F.Y.; Yang, S.; Zhang, X.J. TNIP1/ANXA6 and CSMD1 variants interacting with cigarette smoking, alcohol intake affect risk of psoriasis. J. Dermatol. Sci. 2013, 70, 94–98. [Google Scholar] [CrossRef]
- Villarreal-Martinez, A.; Gallardo-Blanco, H.; Cerda-Flores, R.; Torres-Munoz, I.; Gomez-Flores, M.; Salas-Alanis, J.; Ocampo-Candiani, J.; Martinez-Garza, L. Candidate gene polymorphisms and risk of psoriasis: A pilot study. Exp. Ther. Med. 2016, 11, 1217–1222. [Google Scholar] [CrossRef][Green Version]
- Fan, Z.; Zhang, Z.; Huang, Q.; Han, L.; Fang, X.; Yang, K.; Wu, S.; Zheng, Z.; Yawalkar, N.; Wang, Z.; et al. The Impact of ANxA6 Gene Polymorphism on the Efficacy of Methotrexate Treatment in Psoriasis Patients. Dermatology 2021, 237, 579–587. [Google Scholar] [CrossRef]
- Cubells, L.; Vilà de Muga, S.; Tebar, F.; Wood, P.; Evans, R.; Ingelmo-Torres, M.; Calvo, M.; Gaus, K.; Pol, A.; Grewal, T.; et al. Annexin A6-induced alterations in cholesterol transport and caveolin export from the Golgi complex. Traffic 2007, 8, 1568–1589. [Google Scholar] [CrossRef]
- Stogbauer, F.; Weigert, J.; Neumeier, M.; Wanninger, J.; Sporrer, D.; Weber, M.; Schaffler, A.; Enrich, C.; Wood, P.; Grewal, T.; et al. Annexin A6 is highly abundant in monocytes of obese and type 2 diabetic individuals and is downregulated by adiponectin in vitro. Exp. Mol. Med. 2009, 41, 501–507. [Google Scholar] [CrossRef]
- Talbot, C.P.J.; Plat, J.; Ritsch, A.; Mensink, R.P. Determinants of cholesterol efflux capacity in humans. Prog. Lipid Res. 2018, 69, 21–32. [Google Scholar] [CrossRef]
- Mehta, N.N.; Li, R.; Krishnamoorthy, P.; Yu, Y.; Farver, W.; Rodrigues, A.; Raper, A.; Wilcox, M.; Baer, A.; DerOhannesian, S.; et al. Abnormal lipoprotein particles and cholesterol efflux capacity in patients with psoriasis. Atherosclerosis 2012, 224, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Ferraz-Amaro, I.; Hernandez-Hernandez, M.V.; Armas-Gonzalez, E.; Sanchez-Perez, H.; Machado, J.D.; Diaz-Gonzalez, F. HDL cholesterol efflux capacity is related to disease activity in psoriatic arthritis patients. Clin. Rheumatol. 2020, 39, 1871–1880. [Google Scholar] [CrossRef] [PubMed]
- Grewal, T.; Heeren, J.; Mewawala, D.; Schnitgerhans, T.; Wendt, D.; Salomon, G.; Enrich, C.; Beisiegel, U.; Jackle, S. Annexin VI stimulates endocytosis and is involved in the trafficking of low density lipoprotein to the prelysosomal compartment. J. Biol. Chem. 2000, 275, 33806–33813. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Wu, Y.; Zeng, R.; Liao, K. Proteomic profiling of lipid droplet-associated proteins in primary adipocytes of normal and obese mouse. Acta Biochim. Biophys. Sin. 2012, 44, 394–406. [Google Scholar] [CrossRef] [PubMed]
- Turro, S.; Ingelmo-Torres, M.; Estanyol, J.M.; Tebar, F.; Fernandez, M.A.; Albor, C.V.; Gaus, K.; Grewal, T.; Enrich, C.; Pol, A. Identification and characterization of associated with lipid droplet protein 1: A novel membrane-associated protein that resides on hepatic lipid droplets. Traffic 2006, 7, 1254–1269. [Google Scholar] [CrossRef]
- Krautbauer, S.; Haberl, E.M.; Eisinger, K.; Pohl, R.; Rein-Fischboeck, L.; Rentero, C.; Alvarez-Guaita, A.; Enrich, C.; Grewal, T.; Buechler, C.; et al. Annexin A6 regulates adipocyte lipid storage and adiponectin release. Mol. Cell. Endocrinol. 2017, 439, 419–430. [Google Scholar] [CrossRef] [PubMed]
- Yanai, H.; Yoshida, H. Beneficial Effects of Adiponectin on Glucose and Lipid Metabolism and Atherosclerotic Progression: Mechanisms and Perspectives. Int. J. Mol. Sci. 2019, 20, 1190. [Google Scholar] [CrossRef]
- Mangoni, A.A.; Zinellu, A.; Sotgia, S.; Carru, C.; Erre, G.L. Methotrexate and Cardiovascular Protection: Current Evidence and Future Directions. Clin. Med. Insights Ther. 2017, 9, 1179559X17741289. [Google Scholar] [CrossRef]
- Ma, Y.; Li, L.; Shao, Y.; Bai, X.; Bai, T.; Huang, X. Methotrexate improves perivascular adipose tissue/endothelial dysfunction via activation of AMPK/eNOS pathway. Mol. Med. Rep. 2017, 15, 2353–2359. [Google Scholar] [CrossRef]
| TT (n = 40) | TC (n = 127) | CC (n = 95) | p-Value | |
|---|---|---|---|---|
| Age, mean (SD), year | 50.8 ± 16.62 | 47.94 ± 14.60 | 46.35 ± 14.96 | 0.2903 |
| Age at disease onset, mean (SD), year | 36.47 ± 16.65 | 34.34 ± 15.99 | 34.27 ± 15.12 | 0.7286 |
| Disease duration, mean (SD), year | 14.33 ± 11.17 | 13.59 ± 10.69 | 12.2 ± 10.28 | 0.4800 |
| Weight, mean (SD), kg | 69.86 ± 13.48 | 68.41 ± 11.83 | 69.03 ± 12.77 | 0.8077 |
| BMI, kg/m2 | 24.91 ± 3.67 | 24.26 ± 3.39 | 24.29 ± 3.71 | 0.5866 |
| PASI score at baseline | 12.21 ± 6.05 | 13.65 ± 7.99 | 15.4 ± 7.50 | 0.0580 |
| The mean PASI improvement at 12 week | 59.7 ± 32.05 | 60.34 ± 31.88 | 68.84 ± 31.7 | 0.1082 |
| PASI 50 response at 12 w | 25 (62.50) | 87 (68.50) | 73 (76.84) | 0.1904 |
| PASI 75 response at 12 week | 19 (47.50) | 52 (40.94) | 51 (53.68) | 0.1685 |
| PASI 90 response at 12 week | 7 (17.50) | 24 (18.90) | 26 (27.37) | 0.2474 |
| Male, n (%) | 31(77.50) | 91 (71.65) | 63 (66.320 | 0.4013 |
| Arthritis, n (%) | 24 (60.00) | 66 (51.97) | 48 (50.52) | 0.5879 |
| Smoking, n (%) | 14 (35.00) | 38 (29.92) | 29 (30.52) | 0.1795 |
| Hypertension, n (%) | 21 (52.50) | 53 (41.73) | 32 (33.68) | 0.1163 |
| Diabetes, n (%) | 10 (25.00) | 29 (22.83) | 14 (14.74) | 0.2376 |
| TC, mmol/L | 4.92 ± 0.97 | 4.88 ± 0.94 | 4.56 ± 0.87 * | 0.0215 |
| TG, mmol/L | 1.73 ± 0.98 | 1.56 ± 0.88 | 1.63 ± 1.04 | 0.6122 |
| HDL-C, mmol/L | 1.20 ± 0.26 | 1.19 ± 0.28 | 1.16 ± 0.31 | 0.5863 |
| LDL-C, mmol/L | 3.11 ± 0.76 | 3.07 ± 0.87 | 2.79 ± 0.69 * | 0.0161 |
| ApoA1, g/L | 1.06 ± 0.18 | 1.06 ± 0.18 | 1.03 ± 0.18 | 0.5198 |
| ApoB, g/L | 0.78 ± 0.15 | 0.77 ± 0.18 | 0.71 ± 0.16 * | 0.0279 |
| Lp(a), mg/L | 156.6 ± 176.5 | 165.4 ± 196.9 | 138.1 ± 133.3 | 0.5097 |
| Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|
| Predictors | OR (95%CI) | p-Value | OR (95%CI) | p-Value | |
| TC | rs11960458 | −0.209 (−0.372~−0.046) | 0.012 | −0.198 (−0.358~−0.039) | 0.015 |
| gender | −0.264 (−0.511~−0.017) | 0.036 | −0.324 (−0.563~−0.0830) | 0.008 | |
| age | 0.0137 (0.006~0.021) | 0.000 | 0.0136 (0.006~0.021) | 0.000 | |
| arthritis | 0.228 (0.002~0.454) | 0.048 | |||
| onset | 0.009 (0.002~0.0160 | 0.013 | |||
| diabetes | 0.381 (0.103~0.660) | 0.008 | |||
| ApoB | rs11960458 | −0.037 (−0.067~−0.007) | 0.014 | −0.031 (−0.061~−0.002) | 0.039 |
| age | 0.002 (0.001~0.004) | 0.001 | 0.002 (0.001~0.003) | 0.006 | |
| BMI | 0.011 (0.005~0.016) | 0.000 | 0.010 (0.005~0.015) | 0.000 | |
| arthritis | 0.052 (0.011~0.093) | 0.014 | |||
| weight | 0.003 (0.001~0.004) | 0.000 | |||
| onset | 0.002 (0.001~0.003) | 0.003 | |||
| hypertension | 0.067 (0.026~0.109) | 0.002 | |||
| LDL | rs11960458 | −0.187 (−0.328~0.047) | 0.009 | −0.169 (−0.312~−0.026) | 0.021 |
| diabetes | 0.380 (0.142~0.619) | 0.002 | 0.367 (0.124~0.610) | 0.003 | |
| age | 0.007 (0.000~0.013) | 0.041 | |||
| weight | 0.009 (0.001~0.016) | 0.021 | |||
| TT (n = 40) | TC (n = 127) | CC (n = 95) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 0 week | 12 week | p-Value | 0 week | 12 week | p-Value | 0 week | 12 week | p-Value | |
| TC, mmol/L | 4.92 ± 0.97 | 4.69 ± 1.02 | 0.0816 | 4.88 ± 0.94 | 4.65 ± 0.94 | <0.001 | 4.56 ± 0.87 | 4.40 ± 0.80 | 0.0111 |
| TG, mmol/L | 1.73 ± 0.98 | 1.59 ± 0.91 | 0.1344 | 1.56 ± 0.88 | 1.64 ± 1.03 | 0.2784 | 1.63 ± 1.04 | 1.41 ± 0.84 | 0.0108 |
| HDL-C, mmol/L | 1.20 ± 0.26 | 1.16 ± 0.26 | 0.4109 | 1.19 ± 0.28 | 1.15 ± 0.25 | 0.0068 | 1.16 ± 0.31 | 1.17 ± 0.32 | 0.4519 |
| LDL, mmol/L | 3.11 ± 0.76 | 2.97 ± 0.82 | 0.1501 | 3.07 ± 0.87 | 2.94 ± 0.86 | 0.0028 | 2.79 ± 0.69 | 2.68 ± 0.62 | 0.0353 |
| ApoA1, g/L | 1.06 ± 0.18 | 1.04 ± 0.15 | 0.4569 | 1.06 ± 0.18 | 1.04 ± 0.16 | 0.0434 | 1.03 ± 0.18 | 1.02 ± 0.18 | 0.2467 |
| ApoB, g/L | 0.78 ± 0.15 | 0.73 ± 0.16 | 0.0419 | 0.77 ± 0.18 | 0.72 ± 0.16 | <0.001 | 0.71 ± 0.16 | 0.67 ± 0.15 | 0.0001 |
| Lp(a), mg/L | 156.6 ± 176.5 | 133.5 ± 137.9 | 0.0503 | 165.4 ± 196.9 | 149.2 ± 176.9 | 0.0069 | 138.1 ± 133.3 | 120.5 ± 130.2 | 0.0005 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, F.; Han, L.; Wang, B.; Huang, Q.; Yawalkar, N.; Zhang, Z.; Yan, K. Annexin A6 Polymorphism Is Associated with Pro-atherogenic Lipid Profiles and with the Downregulation of Methotrexate on Anti-Atherogenic Lipid Profiles in Psoriasis. J. Clin. Med. 2022, 11, 7059. https://doi.org/10.3390/jcm11237059
Zhang F, Han L, Wang B, Huang Q, Yawalkar N, Zhang Z, Yan K. Annexin A6 Polymorphism Is Associated with Pro-atherogenic Lipid Profiles and with the Downregulation of Methotrexate on Anti-Atherogenic Lipid Profiles in Psoriasis. Journal of Clinical Medicine. 2022; 11(23):7059. https://doi.org/10.3390/jcm11237059
Chicago/Turabian StyleZhang, Fuxin, Ling Han, Bing Wang, Qiong Huang, Nikhil Yawalkar, Zhenghua Zhang, and Kexiang Yan. 2022. "Annexin A6 Polymorphism Is Associated with Pro-atherogenic Lipid Profiles and with the Downregulation of Methotrexate on Anti-Atherogenic Lipid Profiles in Psoriasis" Journal of Clinical Medicine 11, no. 23: 7059. https://doi.org/10.3390/jcm11237059
APA StyleZhang, F., Han, L., Wang, B., Huang, Q., Yawalkar, N., Zhang, Z., & Yan, K. (2022). Annexin A6 Polymorphism Is Associated with Pro-atherogenic Lipid Profiles and with the Downregulation of Methotrexate on Anti-Atherogenic Lipid Profiles in Psoriasis. Journal of Clinical Medicine, 11(23), 7059. https://doi.org/10.3390/jcm11237059






