COVID-19 and Pulmonary Thrombosis—An Unresolved Clinical Puzzle: A Single-Center Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design, Patients and Data Collection
2.2. CTPA Protocol
2.3. Statistical Analysis
3. Results
3.1. Baseline Patients’ Characteristics
3.2. Association of Pulmonary Thrombosis with COVID-19 Severity
3.3. Factors Associated with Pulmonary Thrombosis
3.4. Factors Associated with Mortality
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jiménez, D.; García-Sanchez, A.; Rali, P.; Muriel, A.; Bikdeli, B.; Ruiz-Artacho, P.; Le Mao, R.; Rodríguez, C.; Hunt, B.J.; Monreal, M. Incidence of VTE and Bleeding Among Hospitalized Patients With Coronavirus Disease 2019: A Systematic Review and Meta-analysis. Chest 2021, 159, 1182–1196. [Google Scholar] [CrossRef] [PubMed]
- Katsoularis, I.; Fonseca-Rodríguez, O.; Farrington, P.; Jerndal, H.; Lundevaller, E.H.; Sund, M.; Lindmark, K.; Connolly, A.-M.F. Risks of deep vein thrombosis, pulmonary embolism, and bleeding after COVID-19: Nationwide self-controlled cases series and matched cohort study. BMJ 2022, 377, e069590. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.-Y.; Cheng, W.-W.; Mou, Z.-W.; Xiao, D.; Li, Y.-Y.; Li, Y.-J.; Li, W.-T.; Chen, Z.-M. Risk factors for pulmonary embolism in patients with COVID-19: A systemic review and meta-analysis. Int. J. Infect. Dis. 2021, 111, 154–163. [Google Scholar] [CrossRef]
- Colling, M.E.; Kanthi, Y. COVID–19-associated coagulopathy: An exploration of mechanisms. Vasc. Med. 2020, 25, 471–478. [Google Scholar] [CrossRef]
- Varga, Z.; Flammer, A.J.; Steiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020, 395, 1417–1418. [Google Scholar] [CrossRef]
- Słomka, A.; Kowalewski, M.; Żekanowska, E. Hemostasis in Coronavirus Disease 2019—Lesson from Viscoelastic Methods: A Systematic Review. Thromb. Haemost. 2021, 121, 1181–1192. [Google Scholar] [CrossRef]
- Kirsch, B.; Aziz, M.; Kumar, S.; Burke, M.; Webster, T.; Immadi, A.; Sam, M.; Lal, A.; Estrada-Y-Martin, R.M.; Cherian, S.; et al. Wells Score to Predict Pulmonary Embolism in Patients with Coronavirus Disease 2019. Am. J. Med. 2021, 134, 688–690. [Google Scholar] [CrossRef] [PubMed]
- Kwee, R.M.; Adams, H.J.A.; Kwee, T.C. Pulmonary embolism in patients with COVID-19 and value of D-dimer assessment: A meta-analysis. Eur. Radiol. 2021, 31, 8168–8186. [Google Scholar] [CrossRef] [PubMed]
- Huda, W. Radiation Doses and Risks in Chest Computed Tomography Examinations. Proc. Am. Thorac. Soc. 2007, 4, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Raji, H.; Moosavi, S.A.J.; Dastoorpoor, M.; Mohamadipour, Z.; Ghanavati, P.M. Overuse and underuse of pulmonary CT angiography in patients with suspected pulmonary embolism. Med. J. Islam. Repub. Iran 2018, 32, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, L.; Kroft, L.; van der Wal, L.; Cannegieter, S.; Eikenboom, J.; de Jonge, E.; Huisman, M.; Klok, F. Clinical and computed tomography characteristics of COVID-19 associated acute pulmonary embolism: A different phenotype of thrombotic disease? Thromb. Res. 2020, 193, 86–89. [Google Scholar] [CrossRef]
- Mestre-Gómez, B.; Lorente-Ramos, R.M.; Rogado, J.; Franco-Moreno, A.; Obispo, B.; Salazar-Chiriboga, D.; Saez-Vaquero, T.; Torres-Macho, J.; Abad-Motos, A.; Cortina-Camarero, C.; et al. Incidence of pulmonary embolism in non-critically ill COVID-19 patients. Predicting factors for a challenging diagnosis. J. Thromb. Thrombolysis 2021, 51, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Gratz, J.; Wiegele, M.; Maleczek, M.; Herkner, H.; Schöchl, H.; Chwala, E.; Knöbl, P.; Schaden, E. Risk of Clinically Relevant Venous Thromboembolism in Critically Ill Patients With COVID-19: A Systematic Review and Meta-Analysis. Front. Med. 2021, 8, 647917. [Google Scholar] [CrossRef] [PubMed]
- Kreidieh, F.; Temraz, S. Anticoagulation for COVID-19 Patients: A Bird’s-Eye View. Clin. Appl. Thromb. 2021, 27, 10760296211039288. [Google Scholar] [CrossRef]
- Assessing SARS-CoV-2 Circulation, Variants of Concern, Non-Pharmaceutical Interventions and Vaccine Rollout in the EU/EEA, 16th update. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/covid-19-rapid-risk-assessment-16th-update-september-2021.pdf (accessed on 29 October 2022).
- García-Ortega, A.; Oscullo, G.; Calvillo, P.; López-Reyes, R.; Méndez, R.; Gómez-Olivas, J.D.; Bekki, A.; Fonfría, C.; Trilles-Olaso, L.; Zaldívar, E.; et al. Incidence, risk factors, and thrombotic load of pulmonary embolism in patients hospitalized for COVID-19 infection. J. Infect. 2021, 82, 261–269. [Google Scholar] [CrossRef]
- Loffi, M.; Regazzoni, V.; Toselli, M.; Cereda, A.; Palmisano, A.; Vignale, D.; Moroni, F.; Pontone, G.; Andreini, D.; Mancini, E.M.; et al. Incidence and characterization of acute pulmonary embolism in patients with SARS-CoV-2 pneumonia: A multicenter Italian experience. PLoS ONE 2021, 16, e0245565. [Google Scholar] [CrossRef] [PubMed]
- Ventura-Díaz, S.; Quintana-Pérez, J.V.; Gil-Boronat, A.; Herrero-Huertas, M.; Gorospe-Sarasúa, L.; Montilla, J.; Acosta-Batlle, J.; Blázquez-Sánchez, J.; Vicente-Bártulos, A. A higher D-dimer threshold for predicting pulmonary embolism in patients with COVID-19: A retrospective study. Emerg. Radiol. 2020, 27, 679–689. [Google Scholar] [CrossRef] [PubMed]
- Kollias, A.; Kyriakoulis, K.G.; Lagou, S.; Kontopantelis, E.; Stergiou, G.S.; Syrigos, K. Venous thromboembolism in COVID-19: A systematic review and meta-analysis. Vasc. Med. 2021, 26, 415–425. [Google Scholar] [CrossRef]
- Fauvel, C.; Weizman, O.; Trimaille, A.; Mika, D.; Pommier, T.; Pace, N.; Douair, A.; Barbin, E.; Fraix, A.; Bouchot, O.; et al. Pulmonary embolism in COVID-19 patients: A French multicentre cohort study. Eur. Heart J. 2020, 41, 3058–3068. [Google Scholar] [CrossRef] [PubMed]
- Cueto-Robledo, G.; Navarro-Vergara, D.-I.; Roldan-Valadez, E.; Garcia-Cesar, M.; Graniel-Palafox, L.-E.; Cueto-Romero, H.-D.; Perez-Calatayud, A.-A.; Enriquez-Garcia, R.; Casillas-Suarez, C. Pulmonary Embolism (PE) Prevalence in Mexican-Mestizo Patients With Severe SARS-Cov-2 (COVID-19) Pneumonia At A Tertiary-Level Hospital: A Review. Curr. Probl. Cardiol. 2022, 20, 101208. [Google Scholar] [CrossRef] [PubMed]
- Antithrombotic Therapy in Patients With COVID-19. Available online: https://www.covid19treatmentguidelines.nih.gov/therapies/antithrombotic-therapy/ (accessed on 29 October 2022).
- Lawler, P.R.; Goligher, E.C.; Berger, J.S.; Neal, M.D.; McVerry, B.J.; Nicolau, J.C. Therapeutic Anticoagulation with Heparin in Noncritically Ill Patients with COVID-19. New Engl. J. Med. 2021, 385, 790–802. [Google Scholar] [CrossRef] [PubMed]
- Goligher, E.C.; Bradbury, C.A.; McVerry, B.J.; Lawler, P.R.; Berger, J.S.; Gong, M.N. Therapeutic Anticoagulation with Heparin in Critically Ill Patients with COVID-19. New Engl. J. Med. 2021, 385, 777–789. [Google Scholar] [CrossRef] [PubMed]
- Halmaciu, I.; Arbănași, E.M.; Kaller, R.; Mureșan, A.V.; Arbănași, E.M.; Bacalbasa, N.; Suciu, B.A.; Cojocaru, I.I.; Runcan, A.I.; Grosu, F.; et al. Chest CT Severity Score and Systemic Inflammatory Biomarkers as Predictors of the Need for Invasive Mechanical Ventilation and of COVID-19 Patients’ Mortality. Diagnostics 2022, 12, 2089. [Google Scholar] [CrossRef] [PubMed]
- Strazzulla, A.; Ben Halima, S.A.; Chouchane, I.; Rezek, M.; Stiebler, M.P.; Hamrouni, S.; Maalaoui, M.; Ghriss, N.; Guedec-Ghelfi, R.; Moini, C.; et al. The Predictive Value of Cell Blood Count Parameters to Diagnose Pulmonary Embolism in Patients with SARS-CoV-2 Infection: A Case Control Study. Antibiotics 2022, 11, 60. [Google Scholar] [CrossRef] [PubMed]
- Lax, S.F.; Skok, K.; Zechner, P.; Kessler, H.H.; Kaufmann, N.; Koelblinger, C.; Vander, K.; Bargfrieder, U.; Trauner, M. Pulmonary Arterial Thrombosis in COVID-19 With Fatal Outcome: Results From a Prospective, Single-Center, Clinicopathologic Case Series. Ann. Intern. Med. 2020, 173, 350–361. [Google Scholar] [CrossRef] [PubMed]
- Bompard, F.; Monnier, H.; Saab, I.; Tordjman, M.; Abdoul, H.; Fournier, L.; Sanchez, O.; Lorut, C.; Chassagnon, G.; Revel, M.-P. Pulmonary embolism in patients with COVID-19 pneumonia. Eur. Respir. J. 2020, 56, 2001365. [Google Scholar] [CrossRef] [PubMed]
- Cau, R.; Pacielli, A.; Fatemeh, H.; Vaudano, P.; Arru, C.; Crivelli, P.; Stranieri, G.; Suri, J.S.; Mannelli, L.; Conti, M.; et al. Complications in COVID-19 patients: Characteristics of pulmonary embolism. Clin. Imaging 2021, 77, 244–249. [Google Scholar] [CrossRef]
- Bledsoe, J.R.; Knox, D.; Peltan, I.D.; Woller, S.C.; Lloyd, J.F.; Snow, G.L.; Horne, B.D.; Connors, J.M.; Kline, J.A. D-dimer Thresholds to Exclude Pulmonary Embolism among COVID-19 Patients in the Emergency Department: Derivation with Independent Validation. Clin. Appl. Thromb. 2022, 28, 10760296221117997. [Google Scholar] [CrossRef]
- Zhou, X.; Pu, Y.; Zhang, D.; Xia, Y.; Guan, Y.; Liu, S.; Fan, L. CT findings and dynamic imaging changes of COVID-19 in 2908 patients: A systematic review and meta-analysis. Acta Radiol. 2022, 63, 291–310. [Google Scholar] [CrossRef]
- Da Silveira, W.C.; Ramos, L.E.F.; Silva, R.T.; de Paiva, B.B.M.; Pereira, P.D.; Schwarzbold, A.V.; Garbini, A.F.; Barreira, B.S.M.; de Castro, B.M.; Ramos, C.M.; et al. Predictors of venous thromboembolism in COVID-19 patients: Results of the COVID-19 Brazilian Registry. Intern. Emerg. Med. 2022, 17, 1863–1878. [Google Scholar] [CrossRef]
| Pulmonary Thrombosis (n = 178) | No Pulmonary Thrombosis (n = 397) | p-Value a | |
|---|---|---|---|
| Age, median (IQR) b | 61 (50–69) | 62 (51–70) | 0.6400 |
| Male, No. (%) b | 124 (69.7) | 258 (65.0) | 0.2941 |
| Comorbidities | |||
| Diabetes Mellitus | 37 (20.8) | 66 (16.6) | 0.2406 |
| Arterial Hypertension | 83 (46.6) | 185 (46.6) | >0.9999 |
| Gastritis/GERD | 8 (4.5) | 20 (5.0) | >0.9999 |
| Cardiovascular Disease | 37 (20.8) | 107 (27.0) | 0.1197 |
| Clinical findings on admission | |||
| Duration of illness on admission, days | 10 (8–13) | 10 (8–12) | 0.1906 |
| Peripheral oxygen saturation (spO2) | 91 (88–94) | 92 (88–94) | 0.1384 |
| MAP | 97 (90–106) | 97 (88–102) | 0.0549 |
| CHOD score | 5 (3–7) | 5 (3–5) | 0.0031 |
| Laboratory findings on admission | |||
| C-reactive Protein, mg/L | 107 (54–177) | 83 (47–148) | 0.0128 |
| Procalcitonin, µg/L | 0.15 (0.082–0.32) | 0.13 (0.084–0.27) | 0.6218 |
| Interleukin 6, ng/L | 66 (31–108) | 59 (32–121) | 0.8638 |
| Ferritin, µg/L | 884 (536–1511) | 1042 (564–2018) | 0.3808 |
| White Blood Cells Count, ×109/L | 7.8 (5.3–11.0) | 6.8 (5.2–9.8) | 0.0517 |
| Lymphocyte count, 109/L | 0.5 (0.1–0.7) | 0.7 (0.5–0.9) | 0.1559 |
| Neutrophil/lymphocyte ratio | 8.8 (4.9–13.0) | 7.9 (5.1–13) | 0.8200 |
| Hemoglobin, g/L | 137 (129–145) | 137 (128–147) | 0.4048 |
| Platelets, ×109/L | 201 (149–274) | 181 (141–245) | 0.0131 |
| Platelets/lymphocyte ratio | 283 (187–424) | 260 (180–425) | 0.5877 |
| Aspartate Aminotransferase, IU/L | 52 (36–75) | 52 (38–80) | 0.4993 |
| Alanine Aminotransferase, IU/L | 45 (28–66) | 44 (27–72) | 0.8851 |
| Gamma-glutamyl transferase, IU/L | 55 (34–94) | 52 (32–99) | 0.7702 |
| Lactate dehydrogenase, IU/L | 410 (324–519) | 380 (294–478) | 0.0266 |
| Prothrombin time | 1.1 (0.96–1.2) | 1.1 (1.0–1.2) | 0.0030 |
| International normalized ratio | 0.97 (0.93–1.1) | 0.95 (0.91–1.0) | 0.0026 |
| Fibrinogen, g/L | 6 (5.1–7.2) | 5.9 (5.2–7.1) | 0.9713 |
| D-dimer, mg/L | 1.2 (0.74–3) | 0.94 (0.59–1.5) | <0.0001 |
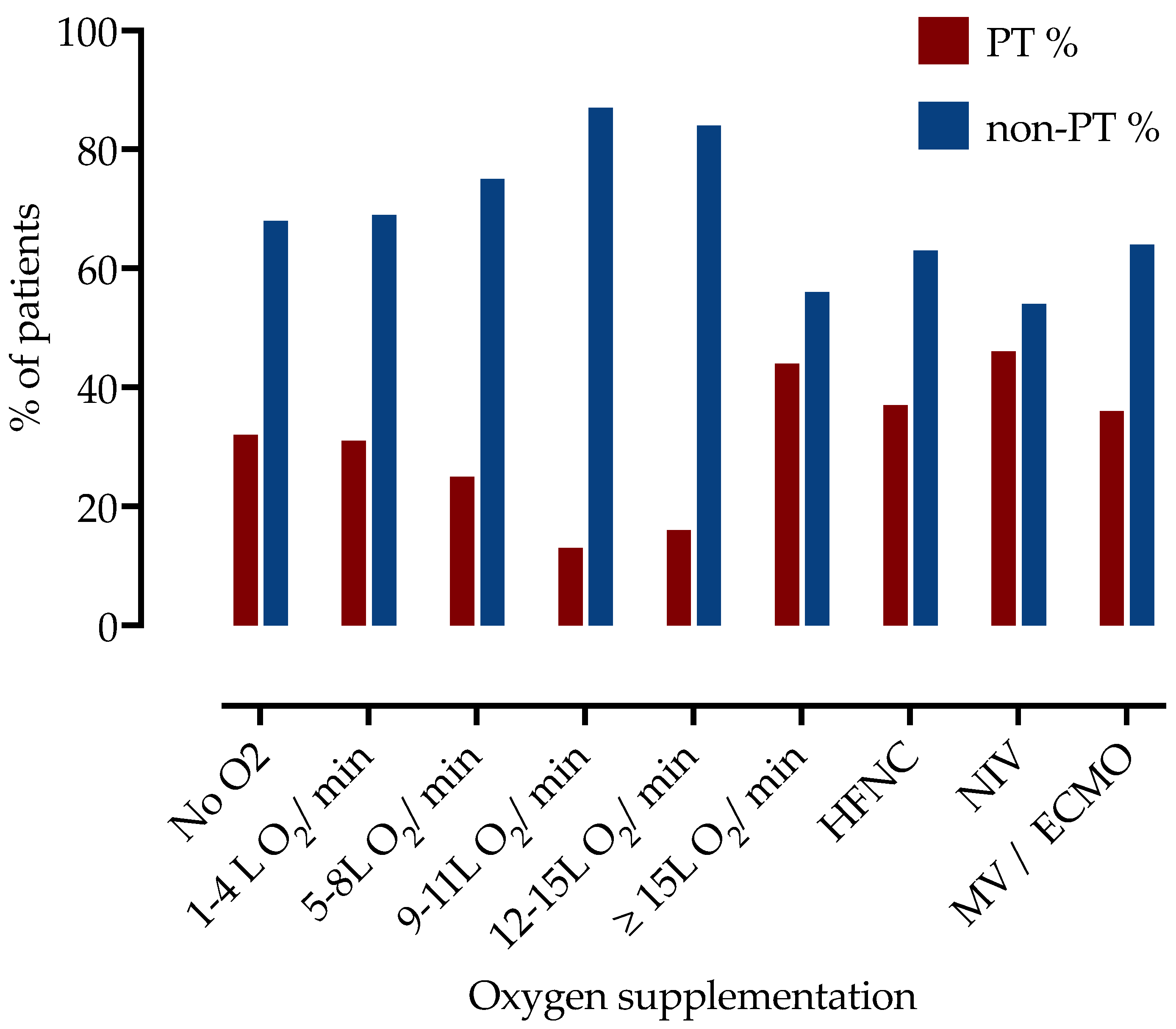
| Oxygen Supplementation | Number of Patients | Subsegmental PT | Segmental, Lobar, or Central PT | No PT |
|---|---|---|---|---|
| No oxygen | 44 | 6 (13.6%) | 8 (18.2%) | 30 (68.2%) |
| 1–4 L O2/min | 128 | 17 (13.3%) | 23 (18.0%) | 88 (68.8%) |
| 5–8 L O2/min | 65 | 5 (7.7%) | 11 (16.9%) | 49 (75.4%) |
| 9–11 L O2/min | 38 | 1 (2.6%) | 4 (10.5%) | 33 (86.8%) |
| 12–15 L O2/min | 69 | 6 (8.7%) | 5 (7.2%) | 58 (84.1%) |
| ≥15 L O2/min | 77 | 10 (13.0%) | 24 (31.2%) | 43 (55.8%) |
| HFNC | 92 | 13 (14.1%) | 21 (22.8%) | 58 (63.0%) |
| NIV | 24 | 4 (16.7%) | 7 (29.2%) | 13 (54.2%) |
| IMV and ECMO | 44 | 3 (6.8%) | 12 (27.3%) | 29 (65.9%) |
| Univariable Analysis | Multivariable Analysis | |||
|---|---|---|---|---|
| OR (95% CI) | p-Value | OR (95% CI) | p-Value | |
| Age | 0.89 (0.62–1.28) | 0.5804 | ||
| Male sex | 1.24 (0.84–1.79) | 0.2941 | ||
| CHOD ≥ 5 | 1.64 (1.14–2.38) | 0.0085 | ||
| Plt ≥ 185 ×109/L | 1.46 (1.02–2.09) | 0.0381 | ||
| CRP ≥ 85 mg/L | 1.52 (1.06–2.17) | 0.0240 | ||
| D-dimer ≥ 1.0 mg/L | 1.87 (1.29–2.69) | 0.0006 | 1.78 (1.12–2.75) | 0.0082 |
| O2 ≥ 15 L | 2.03 (1.43–2.89) | 0.0001 | 1.89 (1.26–2.84) | 0.0021 |
| ICU admission | 1.81 (1.07–3.09) | 0.0320 | ||
| Low-Flow Oxygen Supplementation (<15 L/min) | High-Flow Oxygen Supplementation (≥15 L/min) | |||
|---|---|---|---|---|
| D-dimer < 1.0 mg/L (n = 167) | D-dimer ≥ 1.0 mg/L (n = 179) | D-dimer < 1.0 mg/L (n = 114) | D-dimer ≥ 1.0 mg/L (n = 115) | |
| PT incidence | 32 (19.1%) | 54 (30.2%) | 36 (31.6%) | 56 (48.7%) |
| Subsegmental PT | 17 (10.2%) | 18 (10.1%) | 12 (10.5%) | 17 (14.8%) |
| Segmental PT | 11 (6.6%) | 26 (14.5%) | 19 (16.7%) | 28 (24.3%) |
| Lobar PT | 3 (1.8%) | 7 (3.9%) | 5 (4.4%) | 7 (6.1%) |
| Central PT | 1 (0.6%) | 3 (1.7%) | 0 | 4 (3.5%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Močibob, L.; Šušak, F.; Šitum, M.; Višković, K.; Papić, N.; Vince, A. COVID-19 and Pulmonary Thrombosis—An Unresolved Clinical Puzzle: A Single-Center Cohort Study. J. Clin. Med. 2022, 11, 7049. https://doi.org/10.3390/jcm11237049
Močibob L, Šušak F, Šitum M, Višković K, Papić N, Vince A. COVID-19 and Pulmonary Thrombosis—An Unresolved Clinical Puzzle: A Single-Center Cohort Study. Journal of Clinical Medicine. 2022; 11(23):7049. https://doi.org/10.3390/jcm11237049
Chicago/Turabian StyleMočibob, Loris, Frano Šušak, Maja Šitum, Klaudija Višković, Neven Papić, and Adriana Vince. 2022. "COVID-19 and Pulmonary Thrombosis—An Unresolved Clinical Puzzle: A Single-Center Cohort Study" Journal of Clinical Medicine 11, no. 23: 7049. https://doi.org/10.3390/jcm11237049
APA StyleMočibob, L., Šušak, F., Šitum, M., Višković, K., Papić, N., & Vince, A. (2022). COVID-19 and Pulmonary Thrombosis—An Unresolved Clinical Puzzle: A Single-Center Cohort Study. Journal of Clinical Medicine, 11(23), 7049. https://doi.org/10.3390/jcm11237049






