Contribution of Second Trimester Sonographic Placental Morphology to Uterine Artery Doppler in the Prediction of Placenta-Mediated Pregnancy Complications
Abstract
1. Introduction
2. Methods
2.1. Study Design and Participants
2.2. Data Collection
2.3. Sonographic Placental Markers
2.4. Outcomes
2.5. Data Analysis
3. Results
3.1. Characteristics and Outcomes of the Study Groups
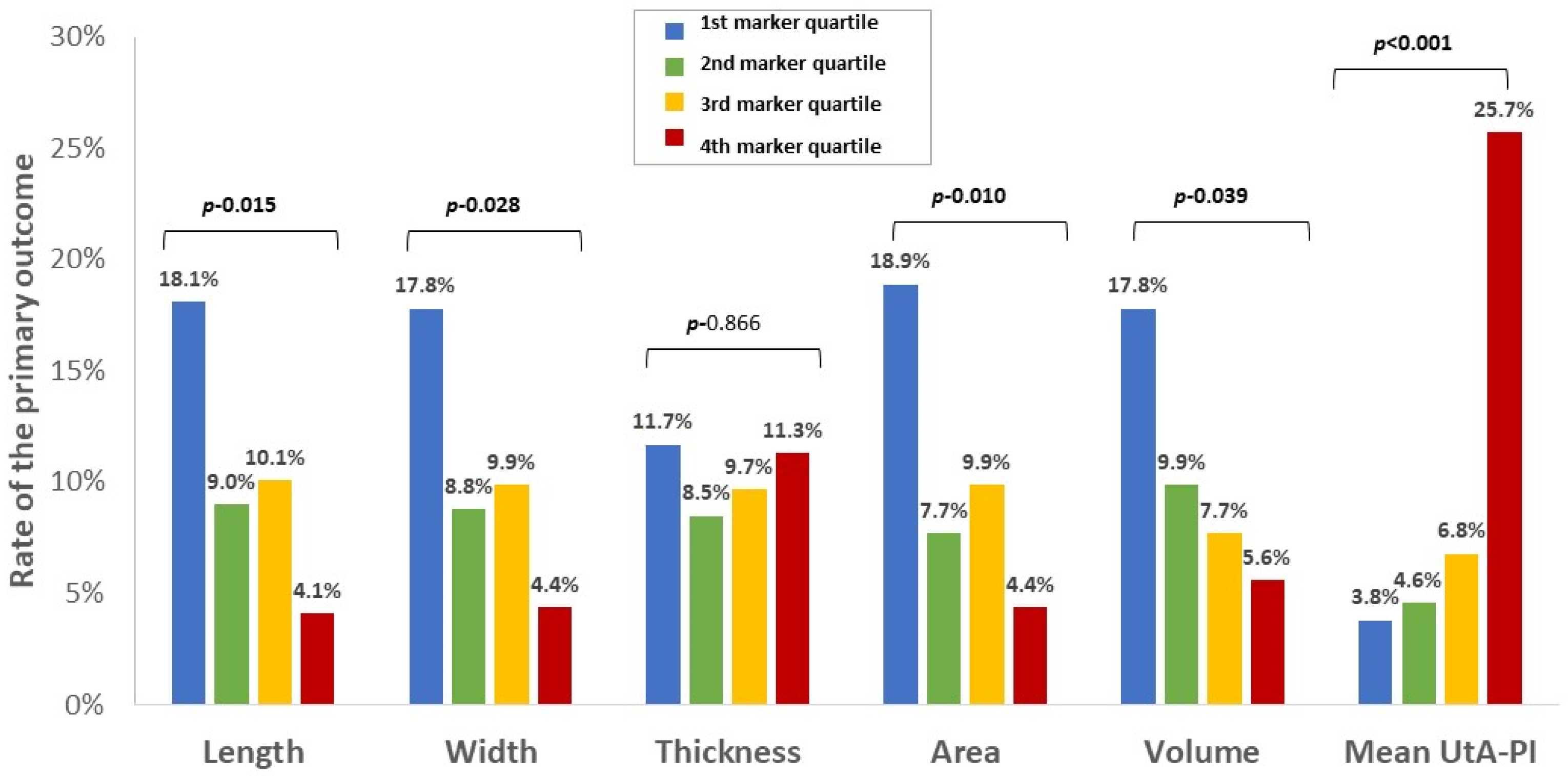
3.2. Rate and Distribution of Sonographic Placental Markers
3.3. Association of Sonographic Placental Markers with Adverse Outcomes
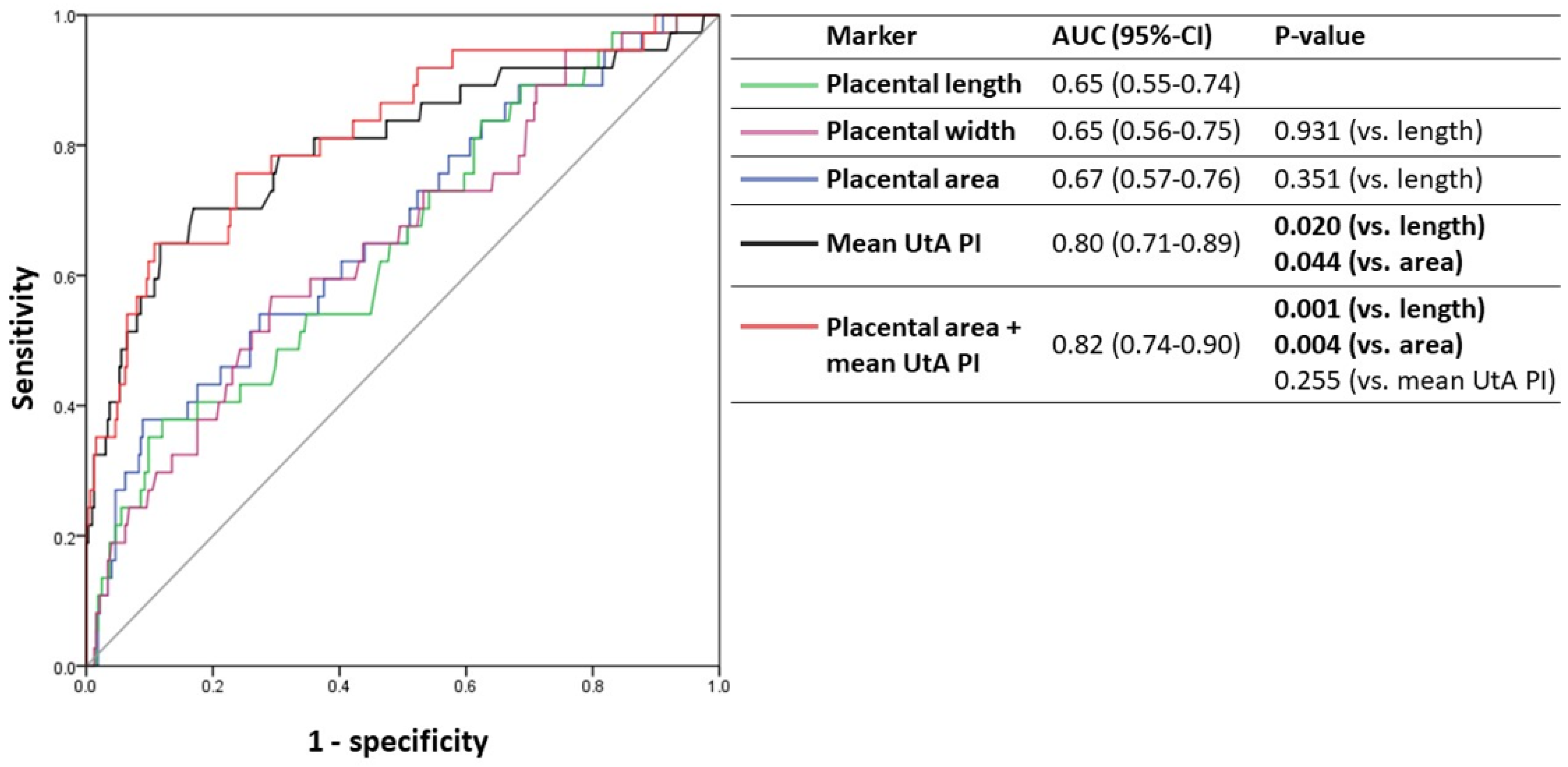
3.4. Predictive Accuracy of Sonographic Placental Markers for Adverse Outcomes
4. Discussion
4.1. Principal Findings
4.2. Interpretation of the Results in the Context of Previous Observations
4.3. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Restriction, F.G. ACOG Practice Bulletin No. 204 Summary: Fetal Growth Restriction. Obstet. Gynecol. 2019, 133, 390–392. [Google Scholar] [CrossRef]
- Lausman, A.; Kingdom, J.; Gagnon, R.; Basso, M.; Bos, H.; Crane, J.; Davies, G.; Delisle, M.-F.; Hudon, L.; Menticoglou, S.; et al. Intrauterine Growth Restriction: Screening, Diagnosis, and Management. J. Obstet. Gynaecol. Can. 2013, 35, 741–748. [Google Scholar] [CrossRef]
- Keating, S.; Melamed, N.; Walker, M.; Sebire, N.J.; Levytska, K.; Higgins, M.; Kingdom, J.C.P. Placental Pathology in Relation to Uterine Artery Doppler Findings in Pregnancies with Severe Intrauterine Growth Restriction and Abnormal Umbilical Artery Doppler Changes. Am. J. Perinatol. 2017, 34, 451–457. [Google Scholar] [CrossRef]
- Cantwell, R.; Clutton-Brock, T.; Cooper, G.; Dawson, A.; Drife, J.; Garrod, D.; Harper, A.; Hulbert, D.; Lucas, S.; McClure, J.; et al. Saving Mothers’ Lives The Eighth Report of the Confidential Enquiries into Maternal Deaths in the United Kingdom Centre for Maternal and Child Enquiries. BJOG 2011, 118, 1–203. [Google Scholar]
- Crispi, F.; Llurba, E.; Domínguez, C.; Martín-Gallán, P.; Cabero, L.; Gratacós, E. Predictive value of angiogenic factors and uterine artery Doppler for early- vs. late-onset pre-eclampsia and intrauterine growth restriction. Ultrasound Obstet. Gynecol. 2008, 31, 303–309. [Google Scholar] [CrossRef]
- De Kat, A.C.; Hirst, J.; Woodward, M.; Kennedy, S.; Peters, S.A. Prediction models for preeclampsia: A systematic review. Pregnancy Hypertens. Int. J. Women’s Cardiovasc. Health 2019, 16, 48–66. [Google Scholar] [CrossRef] [PubMed]
- Pedroso, M.A.; Palmer, K.R.; Hodges, R.J.; Costa, F.D.S.; Rolnik, D.L. Uterine Artery Doppler in Screening for Preeclampsia and Fetal Growth Restriction. Rev. Bras. Ginecol. Obstet. 2018, 40, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Coleman, M.; McCowan, L.; North, R. Mid-trimester uterine artery Doppler screening as a predictor of adverse pregnancy outcome in high-risk women. Ultrasound Obstet. Gynecol. 2000, 15, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Axt-Fliedner, R.; Schwarze, A.; Nelles, I.; Altgassen, C.; Friedrich, M.; Schmidt, W.; Diedrich, K. The value of uterine artery Doppler ultrasound in the prediction of severe complications in a risk population. Arch. Gynecol. Obstet. 2005, 271, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.; Audette, M.C.; Ye, X.Y.; Keating, S.; Hoffman, B.; Lye, S.J.; Shah, P.S.; Kingdom, J.C. Maternal Vascular Malperfusion and Adverse Perinatal Outcomes in Low-Risk Nulliparous Women. Obstet. Gynecol. 2017, 130, 1112–1120. [Google Scholar] [CrossRef]
- Kingdom, J.C.; Audette, M.C.; Hobson, S.R.; Windrim, R.C.; Morgen, E. A placenta clinic approach to the diagnosis and management of fetal growth restriction. Am. J. Obstet. Gynecol. 2018, 218, S803–S817. [Google Scholar] [CrossRef]
- Toal, M.; Chan, C.; Fallah, S.; Alkazaleh, F.; Chaddha, V.; Windrim, R.C.; Kingdom, J.C. Usefulness of a placental profile in high-risk pregnancies. Am. J. Obstet. Gynecol. 2007, 196, 363.e1–363.e7. [Google Scholar] [CrossRef]
- Toal, M.; Keating, S.; Machin, G.; Dodd, J.; Adamson, S.L.; Windrim, R.C.; Kingdom, J.C. Determinants of adverse perinatal outcome in high-risk women with abnormal uterine artery Doppler images. Am. J. Obstet. Gynecol. 2008, 198, 330.e1–330.e7. [Google Scholar] [CrossRef] [PubMed]
- Proctor, L.; Whittle, W.; Keating, S.; Viero, S.; Kingdom, J. Pathologic basis of echogenic cystic lesions in the human placenta: Role of ultrasound-guided wire localization. Placenta 2010, 31, 1111–1115. [Google Scholar] [CrossRef] [PubMed]
- Proctor, L.K.; Toal, M.; Keating, S.; Chitayat, D.; Okun, N.; Windrim, R.C.; Smith, G.; Kingdom, J.C.P. Placental size and the prediction of severe early-onset intrauterine growth restriction in women with low pregnancy-associated plasma protein-A. Ultrasound Obstet. Gynecol. 2009, 34, 274–282. [Google Scholar] [CrossRef]
- Milligan, N.; Rowden, M.; Wright, E.; Melamed, N.; Lee, Y.M.; Windrim, R.C.; Kingdom, J.C. Two-dimensional sonographic assessment of maximum placental length and thickness in the second trimester: A reproducibility study. J. Matern. Neonatal Med. 2015, 28, 1653–1659. [Google Scholar] [CrossRef] [PubMed]
- McGinty, P.; Farah, N.; Dwyer, V.O.; Hogan, J.; Reilly, A.; Turner, M.J.; Stuart, B.; Kennelly, M.M. Ultrasound assessment of placental function: The effectiveness of placental biometry in a low-risk population as a predictor of a small for gestational age neonate. Prenat. Diagn. 2012, 32, 620–626. [Google Scholar] [CrossRef]
- Jain, V. Diagnosis, Evaluation, and Management of the Hypertensive Disorders of Pregnancy: Executive Summary. J. Obstet. Gynaecol. Can. 2015, 37, 774–775. [Google Scholar] [CrossRef]
- Moloney, A.; Hladunewich, M.; Manly, E.; Hui, D.; Ronzoni, S.; Kingdom, J.; Stratulat, V.; Zaltz, A.; Barrett, J.; Melamed, N. The predictive value of sonographic placental markers for adverse pregnancy outcome in women with chronic kidney disease. Pregnancy Hypertens. Int. J. Women’s Cardiovasc. Health 2020, 20, 27–35. [Google Scholar] [CrossRef]
- Hadlock, F.P.; Harrist, R.B.; Martinez-Poyer, J. In utero analysis of fetal growth: A sonographic weight standard. Radiology 1991, 181, 129–133. [Google Scholar] [CrossRef]
- Vest, A.R.; Cho, L.S. Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Obstet. Gynecol. 2013, 122, 1122–1131. [Google Scholar]
- Gómez, O.; Figueras, F.; Fernández, S.; Bennasar, M.; Martínez, J.M.; Puerto, B.; Gratacos, E. Reference ranges for uterine artery mean pulsatility index at 11-41 weeks of gestation. Ultrasound Obstet. Gynecol. 2008, 32, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Hanley, J.A.; McNeil, B.J. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 1982, 143, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Deeks, J.J.; Altman, D.G. Diagnostic tests 4: Likelihood ratios. BMJ 2004, 329, 168–169. [Google Scholar] [CrossRef] [PubMed]
- Grimes, D.A.; Schulz, K.F. Refining clinical diagnosis with likelihood ratios. Lancet 2005, 365, 1500–1505. [Google Scholar] [CrossRef]
- Dombrowski, M.P.; Wolfe, H.M.; Saleh, A.; Evans, M.I.; O’Brien, J. The sonographically thick placenta: A predictor of increased perinatal morbidity and mortality. Ultrasound Obstet. Gynecol. 1992, 2, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Toal, M.; Chaddha, V.; Windrim, R.; Kingdom, J. Ultrasound Detection of Placental Insufficiency in Women With Elevated Second Trimester Serum Alpha-Fetoprotein or Human Chorionic Gonadotropin. J. Obstet. Gynaecol. Can. 2008, 30, 198–206. [Google Scholar] [CrossRef]
- Elchalal, U.; Ezra, Y.; Levi, Y.; Bar-Oz, B.; Yanai, N.; Intrator, O.; Nadjari, M. Sonographically Thick Placenta: A Marker for Increased Perinatal Risk—A Prospective Cross-sectional Study. Placenta 2000, 21, 268–272. [Google Scholar] [CrossRef]
- Nagpal, K.; Mittal, P.; Grover, S.B. Role of Ultrasonographic Placental Thickness in Prediction of Fetal Outcome: A Prospective Indian Study. J. Obstet. Gynecol. India 2017, 68, 349–354. [Google Scholar] [CrossRef]
- Miwa, I.; Sase, M.; Torii, M.; Sanai, H.; Nakamura, Y.; Ueda, K. A thick placenta: A predictor of adverse pregnancy outcomes. SpringerPlus 2014, 3, 353. [Google Scholar] [CrossRef]
- Vachon-Marceau, C.; Demers, S.; Markey, S.; Okun, N.; Girard, M.; Kingdom, J.; Bujold, E. First-trimester placental thickness and the risk of preeclampsia or SGA. Placenta 2017, 57, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, N.; Sammel, M.D.; Leite, R.; Parry, S. First-trimester placental ultrasound and maternal serum markers as predictors of small-for-gestational-age infants. Am. J. Obstet. Gynecol. 2014, 211, 253.e1–253.e8. [Google Scholar] [CrossRef] [PubMed]
- Alanjari, A.; Wright, E.; Keating, S.; Ryan, G.; Kingdom, J. Prenatal diagnosis, clinical outcomes, and associated pathology in pregnancies complicated by massive subchorionic thrombohematoma (Breus’ mole). Prenat. Diagn. 2013, 33, 973–978. [Google Scholar] [CrossRef]
- Quant, H.S.; Sammel, M.D.; Parry, S.; Schwartz, N. Second-Trimester 3-Dimensional Placental Sonography as a Predictor of Small-for-Gestational-Age Birth Weight. J. Ultrasound Med. 2016, 35, 1693–1702. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, N.; Coletta, J.; Pessel, C.; Feng, R.; Timor-Tritsch, I.; Parry, S.; Salafia, C.N. Novel 3-Dimensional Placental Measurements in Early Pregnancy as Predictors of Adverse Pregnancy Outcomes. J. Ultrasound Med. 2010, 29, 1203–1212. [Google Scholar] [CrossRef] [PubMed]
- Arakaki, T.; Hasegawa, J.; Nakamura, M.; Hamada, S.; Muramoto, M.; Takita, H.; Ichizuka, K.; Sekizawa, A. Prediction of early- and late-onset pregnancy-induced hypertension using placental volume on three-dimensional ultrasound and uterine artery Doppler. Ultrasound Obstet. Gynecol. 2015, 45, 539–543. [Google Scholar] [CrossRef]
- Hafner, E.; Philipp, T.; Schuchter, K.; Dillinger-Paller, B.; Philipp, K.; Bauer, P. Second-trimester measurements of placental volume by three-dimensional ultrasound to predict small-for-gestational-age infants. Ultrasound Obstet. Gynecol. 1998, 12, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Odibo, A.O.; Goetzinger, K.R.; Huster, K.M.; Christiansen, J.K.; Odibo, L.; Tuuli, M.G. Placental volume and vascular flow assessed by 3D power Doppler and adverse pregnancy outcomes. Placenta 2011, 32, 230–234. [Google Scholar] [CrossRef] [PubMed]
- González-González, N.L.; González-Dávila, E.; Marrero, L.G.; Padrón, E.; Castro-Conde, J.R.; Plasencia, W. Value of placental volume and vascular flow indices as predictors of intrauterine growth retardation. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 212, 13–19. [Google Scholar] [CrossRef]
- Rizzo, G.; Capponi, A.; Cavicchioni, O.; Vendola, M.; Arduini, D. First trimester uterine Doppler and three-dimensional ultrasound placental volume calculation in predicting pre-eclampsia. Eur. J. Obstet. Gynecol. Reprod. Biol. 2008, 138, 147–151. [Google Scholar] [CrossRef]
- McLaughlin, K.; Snelgrove, J.W.; Audette, M.C.; Syed, A.; Hobson, S.R.; Windrim, R.C.; Melamed, N.; Carmona, S.; Kingdom, J.C. PlGF (Placental Growth Factor) Testing in Clinical Practice: Evidence From a Canadian Tertiary Maternity Referral Center. Hypertension 2021, 77, 2057–2065. [Google Scholar] [CrossRef] [PubMed]
- Bujold, E.; Chaillet, N.; Kingdom, J. Placental growth factor testing for suspected pre-eclampsia. Lancet 2019, 393, 1775–1776. [Google Scholar] [CrossRef]
- Benton, S.J.; McCowan, L.M.; Heazell, A.E.; Grynspan, D.; Hutcheon, J.A.; Senger, C.; Burke, O.; Chan, Y.; Harding, J.E.; Yockell-Lelièvre, J.; et al. Placental growth factor as a marker of fetal growth restriction caused by placental dysfunction. Placenta 2016, 42, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, S.; Parks, W.T.; Zeng, H.D.; Ravichandran, A.; Ashwal, E.; Windrim, R.C.; Hobson, S.R.; Melamed, N.; Kingdom, J.C. Diagnostic utility of serial circulating placental growth factor levels and uterine artery Doppler waveforms in diagnosing underlying placental diseases in pregnancies at high risk of placental dysfunction. Am. J. Obstet. Gynecol. 2022, 227, 618.e1–618.e16. [Google Scholar] [CrossRef]
| Characteristic/Outcome | Overall Cohort n = 429 | Placenta Mediated Complications a n = 45 | No Placenta Mediated Complications n = 384 | p-Value |
|---|---|---|---|---|
| Maternal age (years), mean ± SD | 34.0 ± 4.9 | 33.5 ± 6.0 | 34.0 ± 4.8 | 0.601 |
| >35 years, n (%) | 160 (37.3) | 18 (40.0) | 142 (37.0) | 0.745 |
| Pre-pregnancy BMI (Kg/m2), mean ± SD | 26.3 ± 6.4 | 26.4 ± 6.1 | 26.3 ± 6.4 | 0.937 |
| Nulliparity, n (%) | 147 (34.3) | 15 (33.3) | 132 (34.4) | 1.000 |
| Maternal co-morbidity | ||||
| Pre-existing hypertension, n (%) | 37 (8.6) | 9 (20.0) | 28 (7.3) | 0.009 |
| Diabetes mellitus, n (%) | 9 (2.1) | 2 (4.4) | 7 (1.8) | 0.259 |
| Obstetrical history | ||||
| Past gestational diabetes, n (%) | 20 (4.7) | 1 (2.2) | 19 (4.9) | 0.709 |
| Past preeclampsia, n (%) | 60 (14.0) | 11 (24.4) | 49 (12.8) | 0.041 |
| Past preterm birth, n (%) | 71 (16.6) | 9 (20.0) | 62 (16.1) | 0.525 |
| Past placental abruption, n (%) | 19 (4.4) | 1 (2.2) | 18 (4.7) | 0.707 |
| Past fetal growth restriction, n (%) | 59 (13.8) | 12 (26.7) | 47 (12.2) | 0.019 |
| Past stillbirth, n (%) | 48 (11.2) | 6 (13.3) | 42 (10.9) | 0.618 |
| GA at placenta study (weeks), mean ± SD | 20.8 ± 2.3 | 20.8 ± 2.1 | 20.8 ± 2.3 | 0.292 |
| Pregnancy outcomes | ||||
| Gestational diabetes mellitus, n (%) | 49 (11.4) | 3 (6.7) | 46 (12.0) | 0.455 |
| Placental abruption, n (%) | 13 (3.0) | 3 (6.7) | 10 (2.6) | 0.146 |
| Preeclampsia, n (%) | 56 (13.1) | 15 (33.3) | 41 (10.7) | <0.001 |
| Requiring delivery < 37 weeks, n (%) | 18 (4.2) | 12 (26.7) | 6 (1.6) | <0.001 |
| Requiring delivery < 34 weeks, n (%) | 9 (2.1) | 9 (20.0) | 0 (0) | <0.001 |
| GA at birth (weeks), mean ± SD | 37.7 ± 3.3 | 33.2 ± 6.2 | 37.8 ± 2.3 | <0.001 |
| <37 weeks, n (%) | 68 (15.9) | 21 (46.7) | 47 (12.2) | <0.001 |
| <34 weeks, n (%) | 31 (7.2) | 18 (40.0) | 13 (3.4) | <0.001 |
| Birth weight (g), mean ± SD | 2956 ± 740 | 1657 ± 839 | 3104 ± 561 | <0.001 |
| Birth weight < 3rd centile, n (%) | 42 (9.8) | 42 (93.3) | 0 (0) | <0.001 |
| Female neonate, n (%) | 221 (5.1) | 27 (61.4) | 194 (50.5) | 0.203 |
| Placenta Sonographic Marker | Overall Cohort n = 429 | Placenta Mediated Complications a n = 45 | No Placenta Mediated Complications n = 384 | p-Value |
|---|---|---|---|---|
| Placental length (MoM), mean ± SD | 0.99 ± 0.20 | 0.89 ± 0.16 | 1.00 ± 0.20 | 0.001 |
| Placental width (MoM), mean ± SD | 1.08 ± 0.26 | 0.96 ± 0.22 | 1.09 ± 0.26 | 0.003 |
| Placental thickness (MoM), mean ± SD | 1.05 ± 0.31 | 1.04 ± 0.31 | 1.05 ± 0.31 | 0.756 |
| Placental absolute (1-thickness [MoM]), mean ± SD | 0.24 ± 0.21 | 0.25 ± 0.18 | 0.24 ± 0.21 | 0.637 |
| Placental area (MoM), mean ± SD | 1.08 ± 0.43 | 0.87 ± 0.34 | 1.10 ± 0.43 | 0.002 |
| Placental volume (MoM), mean ± SD | 1.14 ± 0.60 | 0.89 ± 0.42 | 1.17 ± 0.61 | 0.007 |
| Abnormal placental morphology, n (%) | 67 (15.6) | 10 (22.2) | 57 (14.8) | 0.196 |
| 2-vessel cord, n (%) | 8 (1.9) | 2 (4.4) | 6 (1.6) | 0.201 |
| Marginal/velamentous cord insertion, n (%) | 40 (9.3) | 8 (17.8) | 32 (8.3) | 0.054 |
| Mean uterine artery PI (MoM), mean ± SD | 1.09 ± 0.42 | 1.56 ± 0.62 | 1.04 ± 0.30 | <0.001 |
| Mean uterine artery PI > 95th %, n (%) | 48 (11.2) | 19 (49.2) | 29 (7.6) | <0.001 |
| Bilateral uterine artery notching, n (%) | 39 (9.1) | 16 (35.6) | 23 (6.0) | <0.001 |
| Outcome | Placental Marker | Crude OR (95% CI) | Adjusted OR (95% CI)—Model 1 b | Adjusted OR (95% CI)—Model 2 c |
|---|---|---|---|---|
| Placenta-mediated complications a | Length (MoM) | 0.05 (0.01–0.31) | 0.05 (0.01–0.33) | - |
| Width (MoM) | 0.11 (0.03–0.47) | 0.09 (0.02–0.42) | - | |
| Area (MoM) | 0.19 (0.06–0.55) | 0.17 (0.06–0.54) | 0.21 (0.06–0.73) | |
| Bilateral notching | 8.66 (4.12–18.18) | 6.77 (3.09–14.83) | 1.59 (0.49–5.11) | |
| Mean UtA PI (MoM) | 14.61 (6.66–32.05) | 12.49 (5.61–27.80) | 11.71 (3.84–35.72) | |
| Preeclampsia < 34 weeks | Length (MoM) | 0.07 (0.01–1.65) | 0.13 (0.01–2.96) | - |
| Width (MoM) | 0.09 (0.01–1.28) | 0.06 (0.01–1.22) | - | |
| Area (MoM) | 0.17 (0.21–1.36) | 0.20 (0.02–1.65) | 0.59 (0.06–5.74) | |
| Bilateral notching | 23.46 (5.61–98.08) | 14.04 (2.82–70.03) | 1.15 (0.10–13.01) | |
| Mean UtA PI (MoM) | 33.79 (8.87–128.70) | 29.88 (5.90–151.32) | 22.91 (3.21–163.55) | |
| Birthweight < 3rd centile | Length (MoM) | 0.06 (0.01–0.36) | 0.06 (0.01–0.38) | - |
| Width (MoM) | 0.11 (0.03–0.52) | 0.10 (0.02–0.49) | - | |
| Area (MoM) | 0.20 (0.07–0.60) | 0.19 (0.06–0.60) | 0.26 (0.08–0.86) | |
| Bilateral notching | 6.22 (2.89–13.85) | 5.09 (2.26–11.49) | 1.32 (0.40–4.41) | |
| Mean UtA PI (MoM) | 9.38 (4.50–19.55) | 8.42 (3.95–17.98) | 7.56 (2.63–21.67) |
| Threshold Type | Marker | Threshold | Test Positive Rate [n (%)] | Sens. (%) (95%-CI) | Spec. (%) (95%-CI) | PPV (%) (95%-CI) | NPV (%) (95%-CI) | Accuracy (%) (95%-CI) | +LR (95%-CI) | −LR (95%-CI) |
|---|---|---|---|---|---|---|---|---|---|---|
| Sensitivity of 80% | Placental area (MoM) | <1.1158 | 30 (81.1) | 81 (65–92) | 39 (34–45) | 13 (11–15) | 95 (90–97) | 44 (39–49) | 1.3 (1.1–1.6) | 0.5 (0.2–0.9) |
| Mean UtA PI (MoM) | >0.9830 | 35 (81.4) | 81 (67–92) | 52 (47–57) | 16 (14–19) | 96 (93–98) | 55 (50–60) | 1.7 (1.4–2.0) | 0.4 (0.2–0.7) | |
| Placental area and mean UtA PI | >0.0596 | 30 (81.1) | 81 (65–92) | 63 (58–68) | 20 (17–24) | 97 (94–98) | 65 (60–70) | 2.2 (1.8–2.7) | 0.3 (0.1–0.6) | |
| Specificity of 80% | Placental area (MoM) | <0.7585 | 16 (43.2) | 43 (27–61) | 80 (75–84) | 20 (14–27) | 96 (90–94) | 96 (72–81) | 2.2 (1.4–3.3) | 0.7 (0.5–0.9) |
| Mean UtA PI (MoM) | >1.2386 | 27 (62.8) | 63 (47–77) | 80 (76–84) | 27 (21–33) | 95 (93–97) | 77 (74–82) | 3.2 (2.3–4.3) | 0.5 (0.3–0.7) | |
| Placental area and mean UtA PI | >0.1055 | 24 (64.9) | 65 (48–80) | 80 (75–84) | 27 (21–34) | 95 (93–97) | 79 (74–83) | 3.2 (2.4–4.5) | 0.4 (0.3–0.7) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ashwal, E.; Ali-Gami, J.; Aviram, A.; Ronzoni, S.; Mei-Dan, E.; Kingdom, J.; Melamed, N. Contribution of Second Trimester Sonographic Placental Morphology to Uterine Artery Doppler in the Prediction of Placenta-Mediated Pregnancy Complications. J. Clin. Med. 2022, 11, 6759. https://doi.org/10.3390/jcm11226759
Ashwal E, Ali-Gami J, Aviram A, Ronzoni S, Mei-Dan E, Kingdom J, Melamed N. Contribution of Second Trimester Sonographic Placental Morphology to Uterine Artery Doppler in the Prediction of Placenta-Mediated Pregnancy Complications. Journal of Clinical Medicine. 2022; 11(22):6759. https://doi.org/10.3390/jcm11226759
Chicago/Turabian StyleAshwal, Eran, Jasmine Ali-Gami, Amir Aviram, Stefania Ronzoni, Elad Mei-Dan, John Kingdom, and Nir Melamed. 2022. "Contribution of Second Trimester Sonographic Placental Morphology to Uterine Artery Doppler in the Prediction of Placenta-Mediated Pregnancy Complications" Journal of Clinical Medicine 11, no. 22: 6759. https://doi.org/10.3390/jcm11226759
APA StyleAshwal, E., Ali-Gami, J., Aviram, A., Ronzoni, S., Mei-Dan, E., Kingdom, J., & Melamed, N. (2022). Contribution of Second Trimester Sonographic Placental Morphology to Uterine Artery Doppler in the Prediction of Placenta-Mediated Pregnancy Complications. Journal of Clinical Medicine, 11(22), 6759. https://doi.org/10.3390/jcm11226759






