Thickness of Biceps and Quadriceps Femoris Muscle Measured Using Point-of-Care Ultrasound as a Representation of Total Skeletal Muscle Mass
Abstract
1. Introduction
2. Materials and Methods
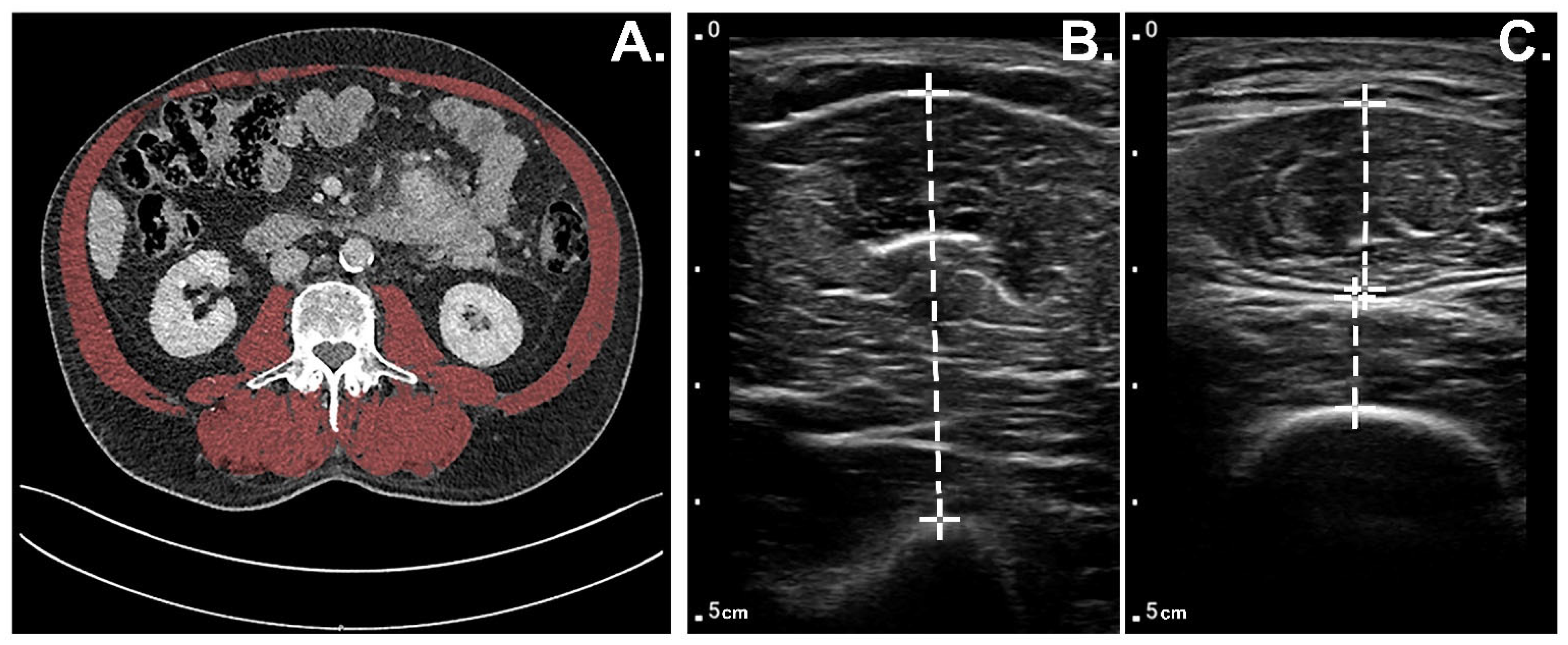
2.1. Muscle Thickness Measurements Using POCUS
2.2. Muscle Mass Measurements CT
2.3. Muscle Strength Measurements
2.4. Study Endpoints
2.5. Statistical Analysis
3. Results
3.1. Patient Body Composition Measurements
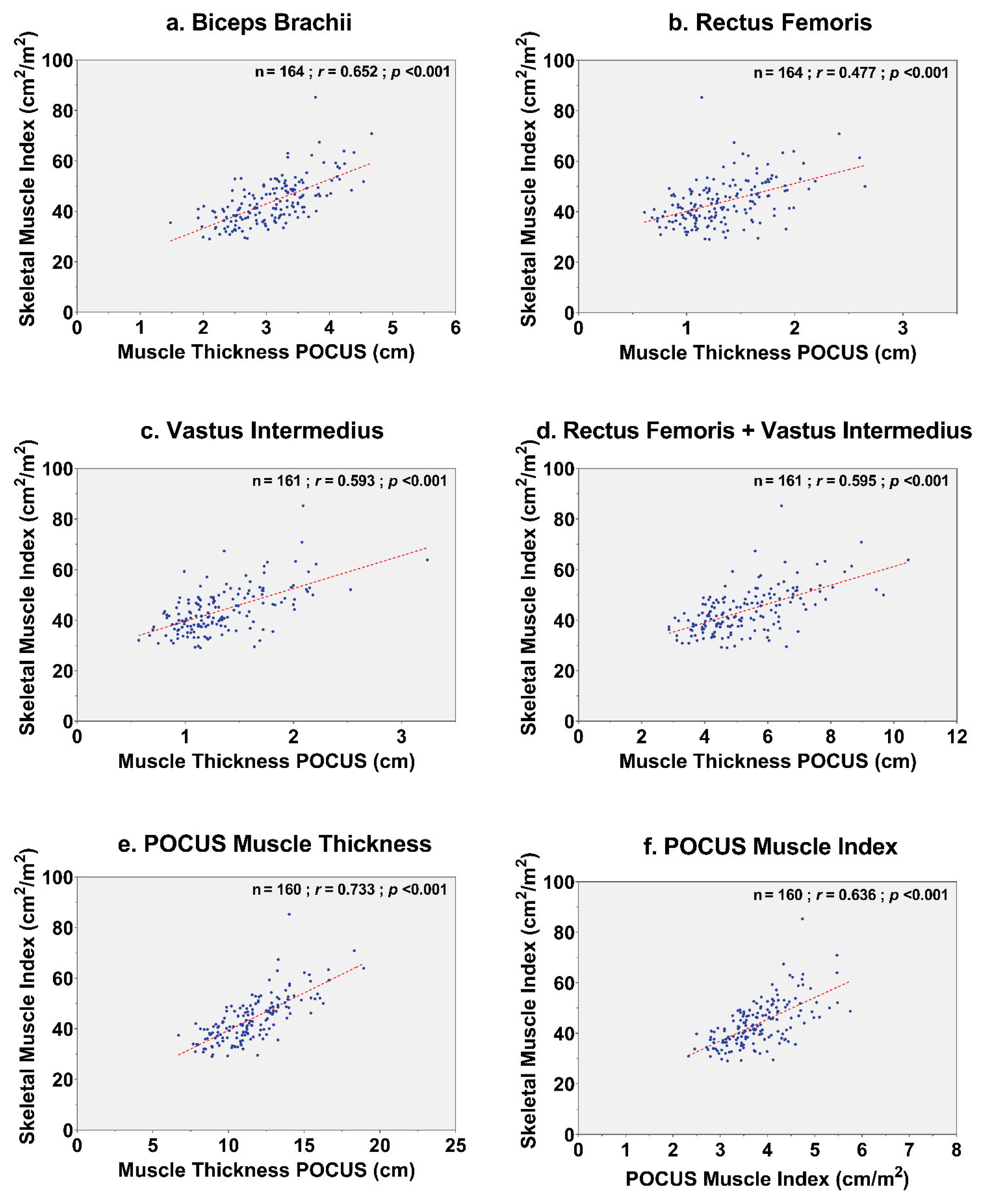
3.2. Correlation between Muscle Measurements
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McGlory, C.; van Vliet, S.; Stokes, T.; Mittendorfer, B.; Phillips, S.M. The Impact of Exercise and Nutrition on the Regulation of Skeletal Muscle Mass. J. Physiol. 2019, 597, 1251–1258. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Punyanitya, M.; Wang, Z.M.; Gallagher, D.; St-Onge, M.P.; Albu, J.; Heymsfield, S.B.; Heshka, S. Total Body Skeletal Muscle and Adipose Tissue Volumes: Estimation from a Single Abdominal Cross-Sectional Image. J. Appl. Physiol. 2004, 97, 2333–2338. [Google Scholar] [CrossRef] [PubMed]
- Boer, B.C.; de Graaff, F.; Brusse-Keizer, M.; Bouman, D.E.; Slump, C.H.; Slee-Valentijn, M.; Klaase, J.M. Skeletal Muscle Mass and Quality as Risk Factors for Postoperative Outcome after Open Colon Resection for Cancer. Int. J. Colorectal Dis. 2016, 31, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Chianca, V.; Albano, D.; Messina, C.; Gitto, S.; Ruffo, G.; Guarino, S.; Del Grande, F.; Sconfienza, L.M. Sarcopenia: Imaging Assessment and Clinical Application. Abdom. Radiol. 2021, 47, 3205–3216. [Google Scholar] [CrossRef] [PubMed]
- Bahat, G.; Turkmen, B.O.; Aliyev, S.; Catikkas, N.M.; Bakir, B.; Karan, M.A. Cut-off Values of Skeletal Muscle Index and Psoas Muscle Index at L3 Vertebra Level by Computerized Tomography to Assess Low Muscle Mass. Clin. Nutr. 2021, 40, 4360–4365. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European Consensus on Definition and Diagnosis. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef]
- Berkel, A.E.M.; Klaase, J.M.; De Graaff, F.; Brusse-Keizer, M.G.J.; Bongers, B.C.; Van Meeteren, N.L.U. Patient’s Skeletal Muscle Radiation Attenuation and Sarcopenic Obesity Are Associated with Postoperative Morbidity after Neoadjuvant Chemoradiation and Resection for Rectal Cancer. Dig. Surg. 2019, 36, 376–383. [Google Scholar] [CrossRef]
- Jones, K.I.; Doleman, B.; Scott, S.; Lund, J.N.; Williams, J.P. Simple Psoas Cross-Sectional Area Measurement Is a Quick and Easy Method to Assess Sarcopenia and Predicts Major Surgical Complications. Color. Dis. 2015, 17, O20–O26. [Google Scholar] [CrossRef]
- Reinders, I.; Murphy, R.A.; Brouwer, I.A.; Visser, M.; Launer, L.; Siggeirsdottir, K.; Eiriksdottir, G.; Gudnason, V.; Jonsson, P.V.; Lang, T.F.; et al. Muscle Quality and Myosteatosis: Novel Associations with Mortality Risk. Am. J. Epidemiol. 2016, 183, 53–60. [Google Scholar] [CrossRef]
- Scheerman, K.; Meskers, C.G.M.; Verlaan, S.; Maier, A.B. Sarcopenia, Low Handgrip Strength, and Low Absolute Muscle Mass Predict Long-Term Mortality in Older Hospitalized Patients: An Observational Inception Cohort Study. J. Am. Med. Dir. Assoc. 2021, 22, 816–820.e2. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European Consensus on Definition and Diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Sergi, G.; De Rui, M.; Stubbs, B.; Veronese, N.; Manzato, E. Measurement of Lean Body Mass Using Bioelectrical Impedance Analysis: A Consideration of the Pros and Cons. Aging Clin. Exp. Res. 2017, 29, 591–597. [Google Scholar] [CrossRef]
- Nijholt, W.; Scafoglieri, A.; Jager-Wittenaar, H.; Hobbelen, J.S.M.; van der Schans, C.P. The Reliability and Validity of Ultrasound to Quantify Muscles in Older Adults: A Systematic Review. J. Cachexia. Sarcopenia Muscle 2017, 8, 702–712. [Google Scholar] [CrossRef]
- Madden, K.M.; Feldman, B.; Arishenkoff, S.; Meneilly, G.S. A Rapid Point-of-Care Ultrasound Marker for Muscle Mass and Muscle Strength in Older Adults. Age Ageing 2021, 50, 505–510. [Google Scholar] [CrossRef]
- Madden, K.M.; Feldman, B.; Arishenkoff, S.; Meneilly, G.S. Point-of-Care Ultrasound Measures of Muscle and Frailty Measures. Eur. Geriatr. Med. 2021, 12, 161–166. [Google Scholar] [CrossRef]
- Li, S.; Li, H.; Hu, Y.; Zhu, S.; Xu, Z.; Zhang, Q.; Yang, Y.; Wang, Z.; Xu, J. Ultrasound for Measuring the Cross-Sectional Area of Biceps Brachii Muscle in Sarcopenia. Int. J. Med. Sci. 2020, 17, 2947–2953. [Google Scholar] [CrossRef]
- Lambell, K.J.; Tierney, A.C.; Wang, J.C.; Nanjayya, V.; Forsyth, A.; Goh, G.S.; Vicendese, D.; Ridley, E.J.; Parry, S.M.; Mourtzakis, M.; et al. Comparison of Ultrasound-Derived Muscle Thickness with Computed Tomography Muscle Cross-Sectional Area on Admission to the Intensive Care Unit: A Pilot Cross-Sectional Study. J. Parenter. Enter. Nutr. 2021, 45, 136–145. [Google Scholar] [CrossRef]
- Hentzen, J.E.K.R.; van Wijk, L.; Buis, C.I.; Viddeleer, A.R.; de Bock, G.H.; van der Schans, C.P.; van Dam, G.M.; Kruijff, S.; Klaase, J.M. Impact and Risk Factors for Clinically Relevant Surgery-Related Muscle Loss in Patients after Major Abdominal Cancer Surgery: Study Protocol for a Prospective Observational Cohort Study (MUSCLE POWER). Int. J. Clin. Trials 2019, 6, 138–146. [Google Scholar] [CrossRef]
- van Wijk, L.; van Duinhoven, S.; Liem, M.S.L.; Bouman, D.E.; Viddeleer, A.R.; Klaase, J.M. Risk Factors for Surgery-Related Muscle Quantity and Muscle Quality Loss and Their Impact on Outcome. Eur. J. Med. Res. 2021, 26, 36. [Google Scholar] [CrossRef]
- Banierink, H.; Bombach, J.J.C.; ten Duis, K.; IJpma, F.F.A.; Heineman, E.; van Helden, S.H.; Nijveldt, R.J.; Viddeleer, A.R.; Reininga, I.H.F. Are Sarcopenia and Myosteatosis in Elderly Patients with Pelvic Ring Injury Related to Mortality, Physical Functioning and Quality of Life? J. Clin. Med. 2021, 10, 4874. [Google Scholar] [CrossRef]
- Shen, Y.; Levolger, S.; Zaid Al-Kaylani, A.H.A.; Uyttenboogaart, M.; van Donkelaar, C.E.; Van Dijk, J.M.C.; Viddeleer, A.R.; Bokkers, R.P.H. Skeletal Muscle Atrophy and Myosteatosis Are Not Related to Long-Term Aneurysmal Subarachnoid Hemorrhage Outcome. PLoS ONE 2022, 17, e0264616. [Google Scholar] [CrossRef]
- Vedder, I.R.; Levolger, S.; Dierckx, R.A.J.O.; Zeebregts, C.J.; de Vries, J.P.P.M.; Viddeleer, A.R.; Bokkers, R.P.H. Effect of Muscle Depletion on Survival in Peripheral Arterial Occlusive Disease: Quality over Quantity. J. Vasc. Surg. 2020, 72, 2006–2016.e1. [Google Scholar] [CrossRef]
- Ticinesi, A.; Narici, M.V.; Lauretani, F.; Nouvenne, A.; Colizzi, E.; Mantovani, M.; Corsonello, A.; Landi, F.; Meschi, T.; Maggio, M. Assessing Sarcopenia with Vastus Lateralis Muscle Ultrasound: An Operative Protocol. Aging Clin. Exp. Res. 2018, 30, 1437–1443. [Google Scholar] [CrossRef]
- Paris, M.T.; Mourtzakis, M.; Day, A.; Leung, R.; Watharkar, S.; Kozar, R.; Earthman, C.; Kuchnia, A.; Dhaliwal, R.; Moisey, L.; et al. Validation of Bedside Ultrasound of Muscle Layer Thickness of the Quadriceps in the Critically Ill Patient (VALIDUM Study). J. Parenter. Enter. Nutr. 2017, 41, 171–180. [Google Scholar] [CrossRef]
- Tillquist, M.; Kutsogiannis, D.J.; Wischmeyer, P.E.; Kummerlen, C.; Leung, R.; Stollery, D.; Karvellas, C.J.; Preiser, J.-C.; Bird, N.; Kozar, R.; et al. Bedside Ultrasound Is a Practical and Reliable Measurement Tool for Assessing Quadriceps Muscle Layer Thickness. J. Parenter. Enter. Nutr. 2014, 38, 886–890. [Google Scholar] [CrossRef] [PubMed]
- Paris, M.T.; Lafleur, B.; Dubin, J.A.; Mourtzakis, M. Development of a Bedside Viable Ultrasound Protocol to Quantify Appendicular Lean Tissue Mass. J. Cachexia. Sarcopenia Muscle 2017, 8, 713–726. [Google Scholar] [CrossRef] [PubMed]
- Tieland, M.; Verdijk, L.; Groot, L.; Loon, L. Handgrip Strength Does Not Represent an Appropriate Measure to Evaluate Changes in Muscle Strength during an Exercise Intervention Program in Frail Older People. Int. J. Sport Nutr. Exerc. Metab. 2014, 25, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Strasser, E.M.; Draskovits, T.; Praschak, M.; Quittan, M.; Graf, A. Association between Ultrasound Measurements of Muscle Thickness, Pennation Angle, Echogenicity and Skeletal Muscle Strength in the Elderly. Age 2013, 35, 2377–2388. [Google Scholar] [CrossRef] [PubMed]
- Looijaard, S.M.L.M.; Oudbier, S.J.; Reijnierse, E.M.; Blauw, G.J.; Meskers, C.G.M.; Maier, A.B. Single Physical Performance Measures Cannot Identify Geriatric Outpatients with Sarcopenia. J. frailty Aging 2018, 7, 262–267. [Google Scholar] [CrossRef]
- Mitchell, W.K.; Williams, J.; Atherton, P.; Larvin, M.; Lund, J.; Narici, M. Sarcopenia, Dynapenia, and the Impact of Advancing Age on Human Skeletal Muscle Size and Strength: A Quantitative Review. Front. Physiol. 2012, 3, 260. [Google Scholar] [CrossRef]
- Goodpaster, B.H.; Park, S.W.; Harris, T.B.; Kritchevsky, S.B.; Nevitt, M.; Schwartz, A.V.; Simonsick, E.M.; Tylavsky, F.A.; Visser, M.; Newman, A.B. The Loss of Skeletal Muscle Strength, Mass, and Quality in Older Adults: The Health, Aging and Body Composition Study. J. Gerontol. A. Biol. Sci. Med. Sci. 2006, 61, 1059–1064. [Google Scholar] [CrossRef]
- van Wijk, L.; van der Snee, L.; Buis, C.I.; Hentzen, J.E.K.R.; Haveman, M.E.; Klaase, J.M. A Prospective Cohort Study Evaluating Screening and Assessment of Six Modifiable Risk Factors in HPB Cancer Patients and Compliance to Recommended Prehabilitation Interventions. Perioper. Med. 2021, 10, 5. [Google Scholar] [CrossRef]
- van Wijk, L.; Bongers, B.C.; Berkel, A.E.M.; Buis, C.I.; Reudink, M.; Liem, M.S.L.; Slooter, G.D.; van Meeteren, N.L.U.; Klaase, J.M. High Effectiveness of a Home-Based Bimodal Prehabilitation Program on Preoperative Aerobic Fitness in High-Risk Patients Scheduled for Liver or Pancreatic Resection. HPB 2021, 23, S1032–S1033. [Google Scholar] [CrossRef]
- van Wijk, L.; Berkel, A.E.M.; Buis, C.I.; Bongers, B.C.; Klaase, J.M. Preoperative Home-Based Exercise Prehabilitation in Patients Scheduled for Liver or Pancreatic Resection: The First Results of the PRIOR Study. Eur. J. Surg. Oncol. 2020, 46, e128. [Google Scholar] [CrossRef]
- van Wijk, L.; Bongers, B.C.; Berkel, A.E.M.; Buis, C.I.; Reudink, M.; Liem, M.S.L.; Slooter, G.D.; van Meeteren, N.L.U.; Klaase, J.M. Improved Preoperative Aerobic Fitness Following a Home-Based Bimodal Prehabilitation Programme in High-Risk Patients Scheduled for Liver or Pancreatic Resection. Br. J. Surg. 2022, 109, 1036–1039. [Google Scholar] [CrossRef]
- Moug, S.J.; Barry, S.J.E.; Maguire, S.; Johns, N.; Dolan, D.; Steele, R.J.C.; Buchan, C.; Mackay, G.; Anderson, A.S.; Mutrie, N. Does Prehabilitation Modify Muscle Mass in Patients with Rectal Cancer Undergoing Neoadjuvant Therapy? A Subanalysis from the REx Randomised Controlled Trial. Tech. Coloproctol. 2020, 24, 959–964. [Google Scholar] [CrossRef]
- Hogenbirk, R.N.M.; Banning, L.B.D.; Visser, A.; Jager-Wittenaar, H.; Pol, R.A.; Zeebregts, C.J.; Klaase, J.M. Association between Masseter Muscle Area and Thickness and Outcome after Carotid Endarterectomy: A Retrospective Cohort Study. J. Clin. Med. 2022, 11, 3087. [Google Scholar] [CrossRef]
| Patient Characteristics | Total (n = 165) |
|---|---|
| Age, years | 65 ± 12 |
| Sex, male | 91 (55.2%) |
| ASA grade ≥3 | 41 (24.8%) |
| PG-SGA SF ≥4 | 54 (32.7%) |
| Preoperative weight loss | |
| <5% | 107 (64.8%) |
| 5–10% | 26 (15.8%) |
| ≥10% | 32 (19.4%) |
| Comorbidities | |
| Any comorbidity | 103 (62.4%) |
| Cardiac | 44 (26.7%) |
| Diabetes mellitus | 22 (13.3%) |
| Hypertension | 47 (28.5%) |
| Pulmonal | 28 (17%) |
| Renal | 7 (4.2%) |
| Type of tumor | |
| Appendix | 4 (2.4%) |
| Bile ducts | 31 (18.8%) |
| Colon | 18 (10.9%) |
| Colorectal liver metastases | 14 (8.5%) |
| Colorectal peritoneal metastases | 4 (2.4%) |
| Liver | 8 (4.8%) |
| Pancreas | 42 (25.5%) |
| Pseudomyxoma peritonei | 5 (3%) |
| Rectum | 22 (13.3%) |
| Small bowel | 5 (3.0%) |
| Other | 12 (7.3%) |
| Prior oncologic treatment | 53 (32.1%) |
| Neoadjuvant chemotherapy | 46 (27.9%) |
| Neoadjuvant radiotherapy | 31 (18.8%) |
| Type of scheduled operation | |
| Liver segment resection | 39 (23.6%) |
| Colorectal resection | 36 (21.8%) |
| Pancreatic resection | 62 (37.6%) |
| CRS with HIPEC | 24 (14.5%) |
| Other | 4 (2.4%) |
| Postoperative outcomes | |
| Length of hospital stay, days | 12 (8–19) |
| Occurrence of any complication | 116 (70.3%) |
| Occurrence of severe complication (Clavien–Dindo ≥3) | 37 (22.4%) |
| Readmission <30 days | 26 (15.8%) |
| Mortality <30 days | 3 (1.8%) |
| Body Composition Measurements | Total (n = 165) |
|---|---|
| Anthropometrics | |
| Height, m | 1.74 ± 0.10 |
| Weight, kg | 79.56 ± 17.04 |
| Body mass index, kg/m2 | 26.23 ± 4.85 |
| Computed tomography | |
| Time between CT and POCUS, weeks | 5 ± 3 |
| Muscle area | |
| Total skeletal muscle index, cm2/m2 | 43.75 ± 8.87 |
| Abdominal muscle wall index, cm2/m2 | 38.17 ± 7.64 |
| Psoas muscle index, cm2/m2 | 5.58 ± 1.71 |
| Point-of-care ultrasound | |
| Muscle thickness | |
| Biceps brachii, cm | 3.08 ± 0.60 |
| Recuts femoris, cm | 1.33 ± 0.38 |
| Vastus intermedius, cm | 1.32 ± 0.41 |
| Rectus femoris + vastus intermedius, cm | 5.29 ± 1.43 |
| POCUS muscle thickness, cm | 11.49 ± 2.22 |
| POCUS muscle index, cm/m2 | 3.80 ± 0.66 |
| Muscle strength | |
| Squeeze force, kg | 34 ± 12 |
| Elbow flexion, kg | 21 ± 7 |
| Elbow extension, kg | 15 ± 5 |
| Knee flexion, kg | 21 ± 6 |
| Knee extension, kg | 25 ± 10 |
| POCUS Measurements (cm) | N | CT-Derived Skeletal Muscle Index (cm2/m2) | |
|---|---|---|---|
| Pearson’s Correlation r | p-Value | ||
| Bilateral | |||
| Biceps brachii | 164 | 0.66 | <0.001 |
| Rectus femoris | 164 | 0.48 | <0.001 |
| Vastus intermedius | 161 | 0.59 | <0.001 |
| Rectus femoris + vastus intermedius | 161 | 0.60 | <0.001 |
| POCUS muscle thickness, cm | 160 | 0.73 | <0.001 |
| POCUS muscle index, cm/m2 | 160 | 0.64 | <0.001 |
| Dominant side | |||
| Biceps brachii | 165 | 0.64 | <0.001 |
| Rectus femoris | 164 | 0.45 | <0.001 |
| Vastus intermedius | 161 | 0.57 | <0.001 |
| Rectus femoris + vastus intermedius | 161 | 0.58 | <0.001 |
| POCUS muscle thickness, cm | 161 | 0.72 | <0.001 |
| POCUS muscle index, cm/m2 | 161 | 0.62 | <0.001 |
| Nondominant side | |||
| Biceps brachii | 164 | 0.64 | <0.001 |
| Rectus femoris | 164 | 0.47 | <0.001 |
| Vastus intermedius | 161 | 0.57 | <0.001 |
| Rectus femoris + vastus intermedius | 161 | 0.58 | <0.001 |
| POCUS muscle thickness, cm | 160 | 0.72 | <0.001 |
| POCUS muscle index, cm/m2 | 160 | 0.62 | <0.001 |
| N | Total POCUS Muscle Thickness (cm) | CT-Derived Skeletal Muscle Index (cm2/m2) | |||||
|---|---|---|---|---|---|---|---|
| Pearson’s Correlation r | 95% CI | p-Value | Pearson’s Correlation r | 95% CI | p-Value | ||
| Squeeze force | 158 | 0.61 | 0.50–0.70 | <0.001 | 0.54 | 0.42–0.64 | <0.001 |
| Elbow flexion | 157 | 0.64 | 0.54–0.73 | <0.001 | 0.58 | 0.46–0.67 | <0.001 |
| Elbow extension | 157 | 0.65 | 0.53–0.72 | <0.001 | 0.57 | 0.47–0.68 | <0.001 |
| Knee flexion | 158 | 0.51 | 0.39–0.62 | <0.001 | 0.41 | 0.27–0.54 | <0.001 |
| Knee extension | 158 | 0.33 | 0.18–0.46 | <0.001 | 0.29 | 0.14–0.43 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hogenbirk, R.N.M.; Viddeleer, A.R.; Hentzen, J.E.K.R.; van der Plas, W.Y.; van der Schans, C.P.; de Bock, G.H.; Kruijff, S.; Klaase, J.M. Thickness of Biceps and Quadriceps Femoris Muscle Measured Using Point-of-Care Ultrasound as a Representation of Total Skeletal Muscle Mass. J. Clin. Med. 2022, 11, 6606. https://doi.org/10.3390/jcm11226606
Hogenbirk RNM, Viddeleer AR, Hentzen JEKR, van der Plas WY, van der Schans CP, de Bock GH, Kruijff S, Klaase JM. Thickness of Biceps and Quadriceps Femoris Muscle Measured Using Point-of-Care Ultrasound as a Representation of Total Skeletal Muscle Mass. Journal of Clinical Medicine. 2022; 11(22):6606. https://doi.org/10.3390/jcm11226606
Chicago/Turabian StyleHogenbirk, Rianne N. M., Alain R. Viddeleer, Judith E. K. R. Hentzen, Willemijn Y. van der Plas, Cees P. van der Schans, Geertruida H. de Bock, Schelto Kruijff, and Joost M. Klaase. 2022. "Thickness of Biceps and Quadriceps Femoris Muscle Measured Using Point-of-Care Ultrasound as a Representation of Total Skeletal Muscle Mass" Journal of Clinical Medicine 11, no. 22: 6606. https://doi.org/10.3390/jcm11226606
APA StyleHogenbirk, R. N. M., Viddeleer, A. R., Hentzen, J. E. K. R., van der Plas, W. Y., van der Schans, C. P., de Bock, G. H., Kruijff, S., & Klaase, J. M. (2022). Thickness of Biceps and Quadriceps Femoris Muscle Measured Using Point-of-Care Ultrasound as a Representation of Total Skeletal Muscle Mass. Journal of Clinical Medicine, 11(22), 6606. https://doi.org/10.3390/jcm11226606





