Major Adverse Cardiovascular Events in Coronary Type 2 Diabetic Patients: Identification of Associated Factors Using Electronic Health Records and Natural Language Processing
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Source
2.3. Study Subjects
2.4. Extraction of the Unstructured Free-Text from EHRs
2.5. External Validation of EHRead® Performance
2.6. Statistical Data Analyses
2.7. Ethical Considerations and Study Approval
3. Results
3.1. Study Population
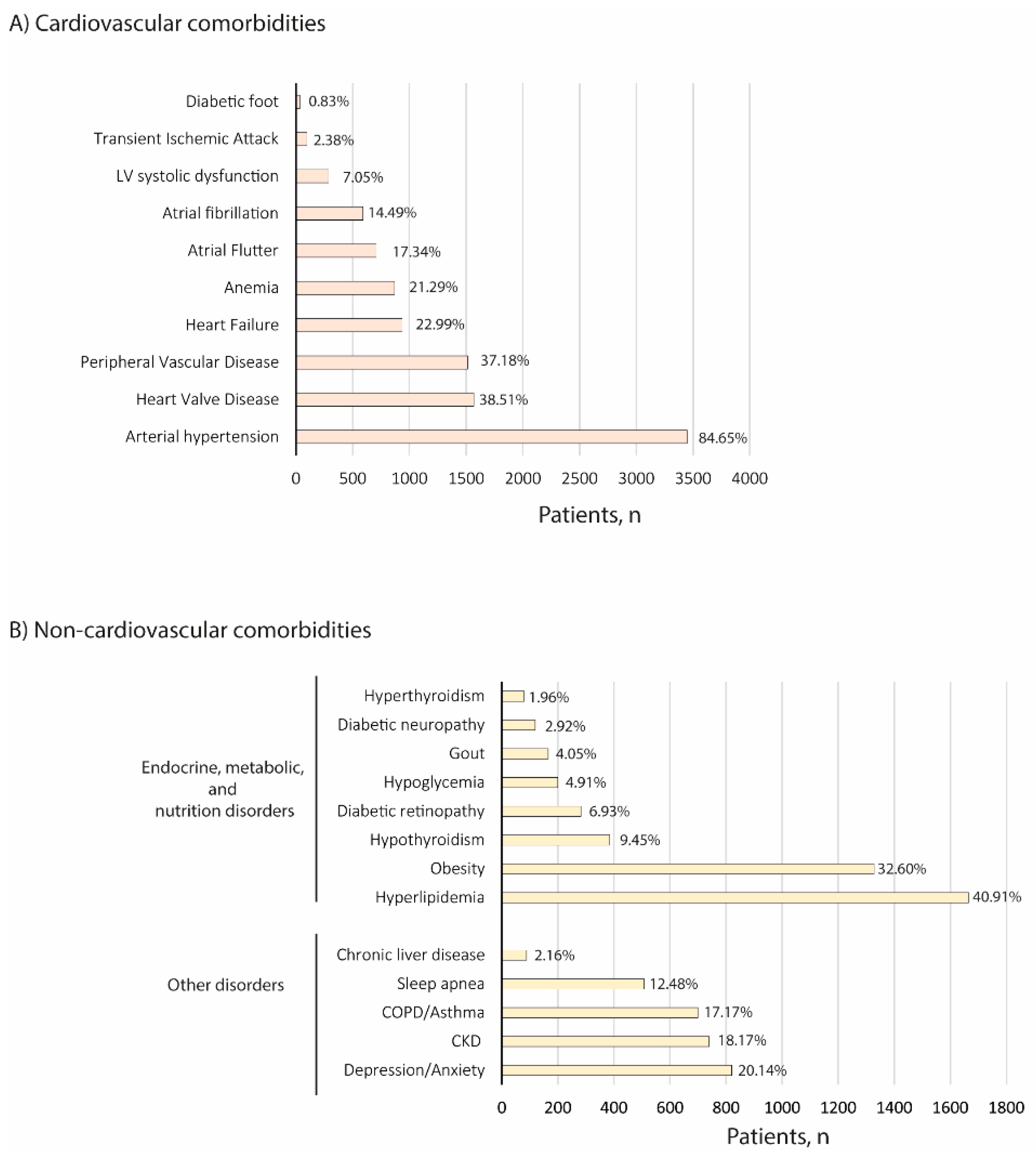
3.2. Demographic and Clinical Characteristics
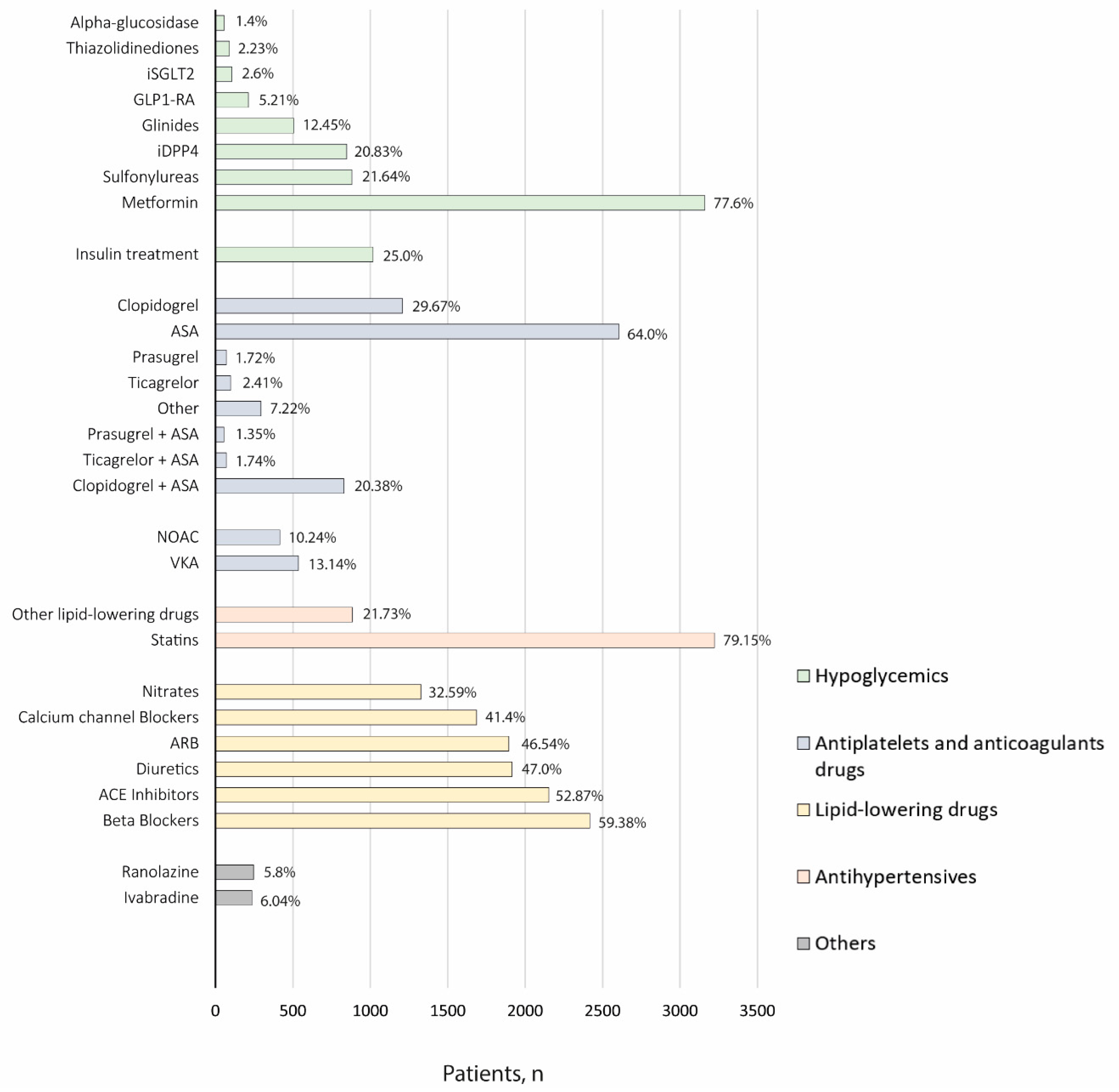
3.3. Disease Management
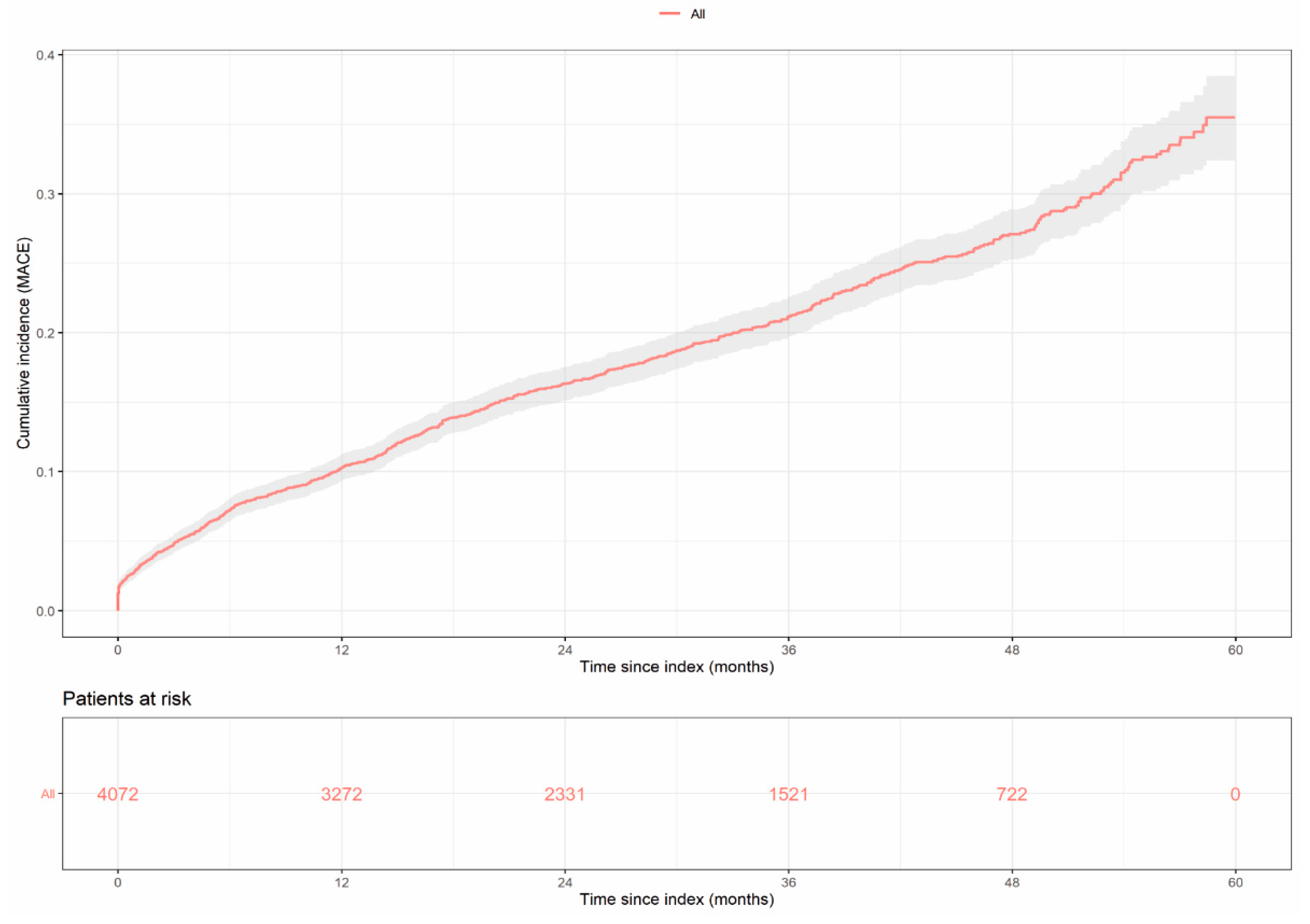
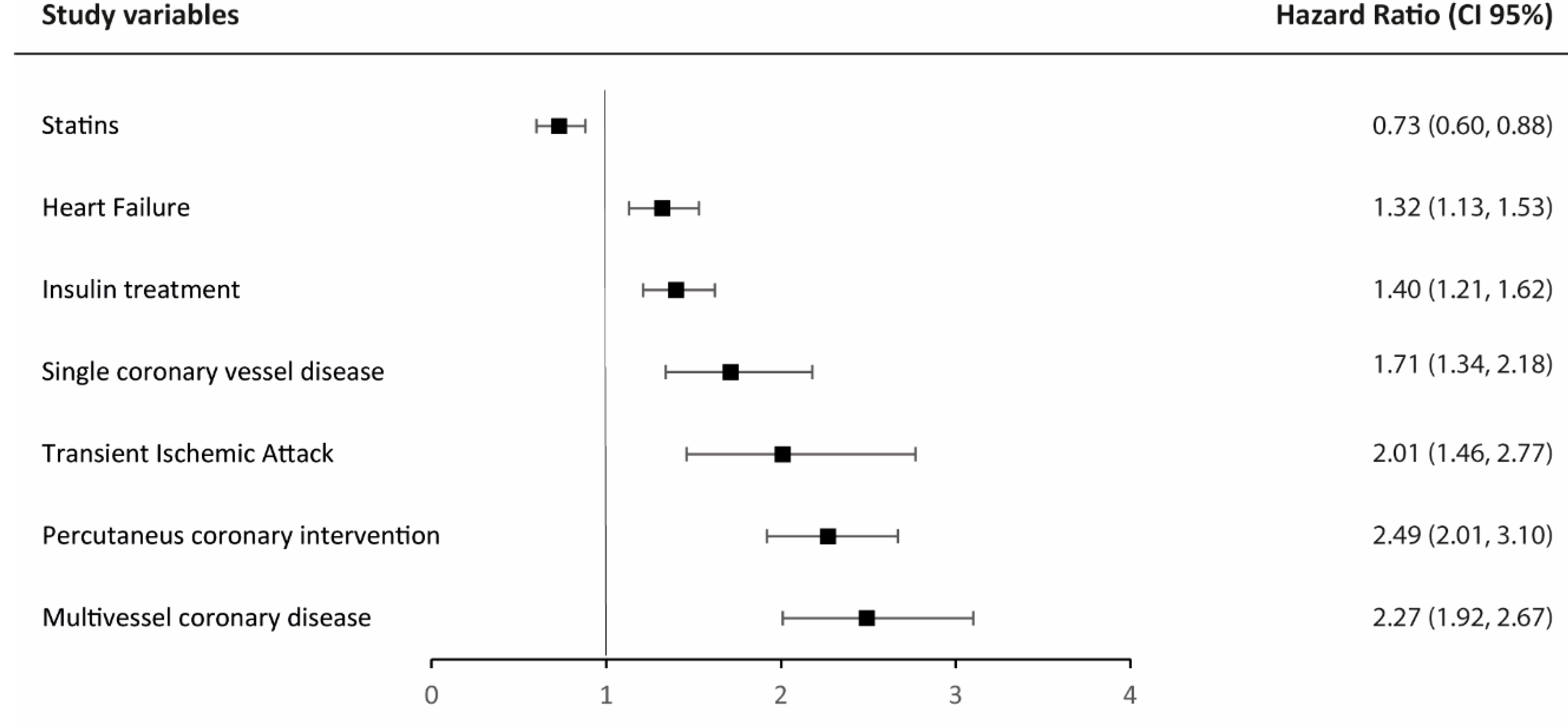
3.4. Major Adverse Cardiovascular Events (MACE)
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9(th) edition. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef] [PubMed]
- Tsao, C.W.; Aday, A.W.; Almarzooq, Z.I.; Alonso, A.; Beaton, A.Z.; Bittencourt, M.S.; Boehme, A.K.; Buxton, A.E.; Carson, A.P.; Commodore-Mensah, Y.; et al. Heart Disease and Stroke Statistics-2022 Update: A Report from the American Heart Association. Circulation 2022, 145, e153–e639. [Google Scholar] [CrossRef]
- Zamora, A.; Marrugata, J. Prognosis of Diabetic Patients with Coronary Heart Disease. Rev. Española Cardiol. 2002, 55, 751–762. [Google Scholar] [CrossRef]
- DeFronzo, R.A.; Ferrannini, E.; Groop, L.; Henry, R.R.; Herman, W.H.; Holst, J.J.; Hu, F.B.; Kahn, C.R.; Raz, I.; Shulman, G.I.; et al. Type 2 diabetes mellitus. Nat. Rev. Dis. Prim. 2015, 1, 15019. [Google Scholar] [CrossRef] [PubMed]
- Aronson, D.; Edelman, E.R. Coronary artery disease and diabetes mellitus. Cardiol. Clin. 2014, 32, 439–455. [Google Scholar] [CrossRef] [PubMed]
- Einarson, T.R.; Acs, A.; Ludwig, C.; Panton, U.H. Prevalence of cardiovascular disease in type 2 diabetes: A systematic literature review of scientific evidence from across the world in 2007–2017. Cardiovasc. Diabetol. 2018, 17, 83. [Google Scholar] [CrossRef] [PubMed]
- Maggioni, A.P.; Dondi, L.; Andreotti, F.; Ronconi, G.; Calabria, S.; Piccinni, C.; Pedrini, A.; Esposito, I.; Martini, N. Prevalence, prescriptions, outcomes and costs of type 2 diabetes patients with or without prior coronary artery disease or stroke: A longitudinal 5-year claims-data analysis of over 7 million inhabitants. Ther. Adv. Chronic Dis. 2021, 12, 20406223211026390. [Google Scholar] [CrossRef]
- Del Rio-Bermudez, C.; Medrano, I.H.; Yebes, L.; Poveda, J.L. Towards a symbiotic relationship between big data, artificial intelligence, and hospital pharmacy. J. Pharm. Policy Pract. 2020, 13, 75. [Google Scholar] [CrossRef]
- Canales, L.; Menke, S.; Marchesseau, S.; D’Agostino, A.; Del Rio-Bermudez, C.; Taberna, M.; Tello, J. Assessing the Performance of Clinical Natural Language Processing Systems: Development of an Evaluation Methodology. JMIR Med. Inform. 2021, 9, e20492. [Google Scholar] [CrossRef]
- González-Juanatey, C.; Anguita-Sáńchez, M.; Barrios, V.; Núñez-Gil, I.; Gómez-Doblas, J.J.; García-Moll, X.; Lafuente-Gormaz, C.; Rollán-Gómez, M.J.; Peral-Disdie, V.; Martínez-Dolz, L.; et al. Assessment of medical management in Coronary Type 2 Diabetic patients with previous percutaneous coronary intervention in Spain: A retrospective analysis of electronic health records using Natural Language Processing. PLoS ONE 2022, 17, e0263277. [Google Scholar] [CrossRef]
- Gomollon, F.; Gisbert, J.P.; Guerra, I.; Plaza, R.; Pajares Villarroya, R.; Moreno Almazan, L.; Lopez Martin, M.C.; Dominguez Antonaya, M.; Vera Mendoza, M.I.; Aparicio, J.; et al. Clinical characteristics and prognostic factors for Crohn's disease relapses using natural language processing and machine learning: A pilot study. Eur. J. Gastroenterol. Hepatol. 2021, 34, 389–397. [Google Scholar] [CrossRef]
- Izquierdo, J.L.; Almonacid, C.; Gonzalez, Y.; Del Rio-Bermudez, C.; Ancochea, J.; Cardenas, R.; Lumbreras, S.; Soriano, J.B. The Impact of COVID-19 on Patients with Asthma. Eur. Respir. J. 2020, 57, 2003142. [Google Scholar] [CrossRef]
- Izquierdo, J.L.; Ancochea, J.; Savana, C.-R.G.; Soriano, J.B. Clinical Characteristics and Prognostic Factors for Intensive Care Unit Admission of Patients with COVID-19: Retrospective Study Using Machine Learning and Natural Language Processing. J. Med. Internet Res. 2020, 22, e21801. [Google Scholar] [CrossRef]
- Choi, B.; Rha, S.; Yoon, S.G.; Choi, C.; Lee, M.; Kim, S. Association of Major Adverse Cardiac Events up to 5 Years in Patients with Chest Pain Without Significant Coronary Artery Disease in the Korean Population. J. Am. Heart Assoc. 2019, 8, e010541. [Google Scholar] [CrossRef]
- Sheikhalishahi, S.; Miotto, R.; Dudley, J.T.; Lavelli, A.; Rinaldi, F.; Osmani, V. Natural Language Processing of Clinical Notes on Chronic Diseases: Systematic Review. JMIR Med. Inform. 2019, 7, e12239. [Google Scholar] [CrossRef]
- Goldstein, B.A.; Navar, A.M.; Pencina, M.J.; Ioannidis, J.P. Opportunities and challenges in developing risk prediction models with electronic health records data: A systematic review. J. Am. Med. Inform. Assoc. 2017, 24, 198–208. [Google Scholar] [CrossRef]
- Luo, Y.; Thompson, W.K.; Herr, T.M.; Zeng, Z.; Berendsen, M.A.; Jonnalagadda, S.R.; Carson, M.B.; Starren, J. Natural Language Processing for EHR-Based Pharmacovigilance: A Structured Review. Drug Saf. 2017, 40, 1075–1089. [Google Scholar] [CrossRef]
- Ancochea, J.; Izquierdo, J.L.; Medrano, I.H.; Porras, A.; Serrano, M.; Lumbreras, S.; Del Rio-Bermudez, C.; Marchesseau, S.; Salcedo, I.; Zubizarreta, I.; et al. Evidence of gender differences in the diagnosis and management of COVID-19 patients: An analysis of Electronic Health Records using Natural Language Processing and machine learning. J. Womens Health 2021, 30, 393–404. [Google Scholar] [CrossRef]
- Graziani, D.; Soriano, J.B.; Del Rio-Bermudez, C.; Morena, D.; Díaz, T.; Castillo, M.; Alonso, M.; Ancochea, J.; Lumbreras, S.; Izquierdo, J.L. Characteristics and Prognosis of COVID-19 in Patients with COPD. J. Clin. Med. 2020, 9, 3259. [Google Scholar] [CrossRef]
- Espinosa-Anke, L.; Tello, J.; Pardo, A.; Medrano, I.; Ureña, A.; Salcedo, I.; Saggion, H. Savana: A Global Information Extraction and Terminology Expansion Framework in the Medical Domain. Proces. Leng. Nat. 2016, 57, 23–30. [Google Scholar]
- Benson, T. Principles of Health Interoperability HL7 and SNOMED; Springer: London, UK, 2012. [Google Scholar]
- Izquierdo, J.L.; Morena, D.; Gonzalez, Y.; Paredero, J.M.; Perez, B.; Graziani, D.; Gutierrez, M.; Rodriguez, J.M. Clinical Management of COPD in a Real-World Setting. A Big Data Analysis. Arch. Bronconeumol. 2021, 57, 94–100. [Google Scholar] [CrossRef]
- Al-Ozairi, E.; Jallo, M.K.; Hafidh, K.; Alhajeri, D.M.; Ashour, T.; Mahmoud, E.F.N.; Abd ElAal, Z.; Loulou, M. Prevalence of Cardiovascular and Renal Co-morbidities in Patients with Type 2 Diabetes in the Gulf, a Cross-sectional Observational Study. Diabetes Ther. 2021, 12, 1193–1207. [Google Scholar] [CrossRef]
- Jelinek, H.F.; Osman, W.M.; Khandoker, A.H.; Khalaf, K.; Lee, S.; Almahmeed, W.; Alsafar, H.S. Clinical profiles, comorbidities and complications of type 2 diabetes mellitus in patients from United Arab Emirates. BMJ Open Diabetes Res. Care 2017, 5, e000427. [Google Scholar] [CrossRef]
- Wittbrodt, E.; Bhalla, N.; Andersson Sundell, K.; Gao, Q.; Dong, L.; Cavender, M.A.; Hunt, P.; Wong, N.D.; Mellstrom, C. Assessment of the high risk and unmet need in patients with CAD and type 2 diabetes (ATHENA): US healthcare resource utilization, cost and burden of illness in the Diabetes Collaborative Registry. Endocrinol. Diabetes Metab. 2020, 3, e00133. [Google Scholar] [CrossRef]
- Fihn, S.D.; Gardin, J.M.; Abrams, J.; Berra, K.; Blankenship, J.C.; Dallas, A.P.; Douglas, P.S.; Foody, J.M.; Gerber, T.C.; Hinderliter, A.L.; et al. 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS guideline for the diagnosis and management of patients with stable ischemic heart disease: A report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Circulation 2012, 126, e354–e471. [Google Scholar] [CrossRef]
- Arnold, S.V.; Bhatt, D.L.; Barsness, G.W.; Beatty, A.L.; Deedwania, P.C.; Inzucchi, S.E.; Kosiborod, M.; Leiter, L.A.; Lipska, K.J.; Newman, J.D.; et al. Clinical Management of Stable Coronary Artery Disease in Patients with Type 2 Diabetes Mellitus: A Scientific Statement from the American Heart Association. Circulation 2020, 141, e779–e806. [Google Scholar] [CrossRef]
- Gnesin, F.; Thuesen, A.C.B.; Kahler, L.K.A.; Madsbad, S.; Hemmingsen, B. Metformin monotherapy for adults with type 2 diabetes mellitus. Cochrane Database Syst. Rev. 2020, 6, CD012906. [Google Scholar] [CrossRef]
- CAPRIE Steering Committee. A Randomised, Blinded, Trial of Clopidogrel versus Aspirin in Patients at Risk of Ischaemic Events (CAPRIE); CAPRIE Steering Committee: Amsterdam, The Netherlands, 1996; pp. 1329–1339. [Google Scholar]
- Berger, A. Incidence of Major Adverse Cardiovascular and/or Limb Events among Patients Using Aspirin for Secondary Prevention. Adv. Cardiovasc. Res. 2020, 3, 158. [Google Scholar] [CrossRef]
- Kengne, A.P. The ADVANCE cardiovascular risk model and current strategies for cardiovascular disease risk evaluation in people with diabetes. Cardiovasc. J. Afr. 2013, 24, 376–381. [Google Scholar] [CrossRef]
- Okkonen, M.; Havulinna, A.; Ukkola, O.; Huikuri, H.; Pietilä, A.; Koukkunen, H.; Lehto, S.; Mustonen, J.; Ketonen, M.; Airaksinen, J.; et al. Risk factors for major adverse cardiovascular events after the first acute coronary syndrome. Ann. Med. 2021, 53, 817–823. [Google Scholar] [CrossRef]
- Schwartz, G.G.; Nicholls, S.J.; Toth, P.P.; Sweeney, M.; Halliday, C.; Johansson, J.O.; Wong, N.C.W.; Kulikowski, E.; Kalantar-Zadeh, K.; Ginsberg, H.N.; et al. Relation of insulin treatment for type 2 diabetes to the risk of major adverse cardiovascular events after acute coronary syndrome: An analysis of the BETonMACE randomized clinical trial. Cardiovasc. Diabetol. 2021, 20, 125. [Google Scholar] [CrossRef] [PubMed]
- Van der Velde, N.; Huurman, R.; Yamasaki, Y.; Kardys, I.; Galema, T.W.; Budde, R.P.; Zijlstra, F.; Krestin, G.P.; Schinkel, A.F.; Michels, M.; et al. Frequency and Significance of Coronary Artery Disease and Myocardial Bridging in Patients with Hypertrophic Cardiomyopathy. Am. J. Cardiol. 2020, 125, 1404–1412. [Google Scholar] [CrossRef] [PubMed]
- Mascia, G.; Crotti, L.; Groppelli, A.; Canepa, M.; Merlo, A.C.; Benenati, S.; Di Donna, P.; Della Bona, R.; Soranna, D.; Zambon, A.; et al. Syncope in hypertrophic cardiomyopathy (part I): An updated systematic review and meta-analysis. Int. J. Cardiol. 2022, 357, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Fumagalli, S.; Pieragnoli, P.; Haugaa, K.H.; Potpara, T.S.; Rasero, L.; Ramacciati, N.; Ricciardi, G.; Solimene, F.; Mascia, G.; Mascioli, G.; et al. The influence of age on the psychological profile of patients with cardiac implantable electronic devices: Results from the Italian population in a multicenter study conducted by the European Heart Rhythm Association. Aging Clin. Exp. Res. 2019, 31, 1219–1226. [Google Scholar] [CrossRef]
- Shahreyar, M.; Mupiddi, V.; Choudhuri, I.; Sra, J.; Tajik, A.J.; Jahangir, A. Implantable cardioverter defibrillators in diabetics: Efficacy and safety in patients at risk of sudden cardiac death. Expert Rev. Cardiovasc. Ther. 2015, 13, 897–906. [Google Scholar] [CrossRef][Green Version]
| Study Population (N = 4072) | |
|---|---|
| Age, years | |
| Mean (SD) | 70 (11) |
| Median (Q1,Q3) | 71 (63,78) |
| Gender, n (%) | |
| Female | 1533 (37.6) |
| Male | 2531 (62.2) |
| Tobacco use | 2208 (54.2) |
| Current smoker | 713 (17.5) |
| Former smoker | 1495 (36.7) |
| SD: Standard deviation. | |
| 1-Year FU | 2-Year FU | 3-Year FU | 4-Year FU | |
|---|---|---|---|---|
| MACE * | 10.28 | 16.32 | 21.18 | 27.09 |
| Myocardial infarction | 4.05 | 7.8 | 10.79 | 13.89 |
| Ischemic stroke | 1.77 | 3.41 | 5.05 | 6.94 |
| Unstable angina | 1.67 | 2.49 | 3.45 | 4.54 |
| Urgent revascularization | 4.04 | 5.57 | 6.82 | 8.69 |
| FU: Follow-up; MACE: Major adverse cardiovascular events. | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Juanatey, C.; Anguita-Sánchez, M.; Barrios, V.; Núñez-Gil, I.; Gómez-Doblas, J.J.; García-Moll, X.; Lafuente-Gormaz, C.; Rollán-Gómez, M.J.; Peral-Disdier, V.; Martínez-Dolz, L.; et al. Major Adverse Cardiovascular Events in Coronary Type 2 Diabetic Patients: Identification of Associated Factors Using Electronic Health Records and Natural Language Processing. J. Clin. Med. 2022, 11, 6004. https://doi.org/10.3390/jcm11206004
González-Juanatey C, Anguita-Sánchez M, Barrios V, Núñez-Gil I, Gómez-Doblas JJ, García-Moll X, Lafuente-Gormaz C, Rollán-Gómez MJ, Peral-Disdier V, Martínez-Dolz L, et al. Major Adverse Cardiovascular Events in Coronary Type 2 Diabetic Patients: Identification of Associated Factors Using Electronic Health Records and Natural Language Processing. Journal of Clinical Medicine. 2022; 11(20):6004. https://doi.org/10.3390/jcm11206004
Chicago/Turabian StyleGonzález-Juanatey, Carlos, Manuel Anguita-Sánchez, Vivencio Barrios, Iván Núñez-Gil, Juan José Gómez-Doblas, Xavier García-Moll, Carlos Lafuente-Gormaz, María Jesús Rollán-Gómez, Vicente Peral-Disdier, Luis Martínez-Dolz, and et al. 2022. "Major Adverse Cardiovascular Events in Coronary Type 2 Diabetic Patients: Identification of Associated Factors Using Electronic Health Records and Natural Language Processing" Journal of Clinical Medicine 11, no. 20: 6004. https://doi.org/10.3390/jcm11206004
APA StyleGonzález-Juanatey, C., Anguita-Sánchez, M., Barrios, V., Núñez-Gil, I., Gómez-Doblas, J. J., García-Moll, X., Lafuente-Gormaz, C., Rollán-Gómez, M. J., Peral-Disdier, V., Martínez-Dolz, L., Rodríguez-Santamarta, M., Viñolas-Prat, X., Soriano-Colomé, T., Muñoz-Aguilera, R., Plaza, I., Curcio-Ruigómez, A., Orts-Soler, E., Segovia, J., Fanjul, V., ... SAVANA Research Group. (2022). Major Adverse Cardiovascular Events in Coronary Type 2 Diabetic Patients: Identification of Associated Factors Using Electronic Health Records and Natural Language Processing. Journal of Clinical Medicine, 11(20), 6004. https://doi.org/10.3390/jcm11206004








