Cluster Analysis of Home Polygraphic Recordings in Symptomatic Habitually-Snoring Children: A Precision Medicine Perspective
Abstract
1. Introduction
2. Materials and Methods
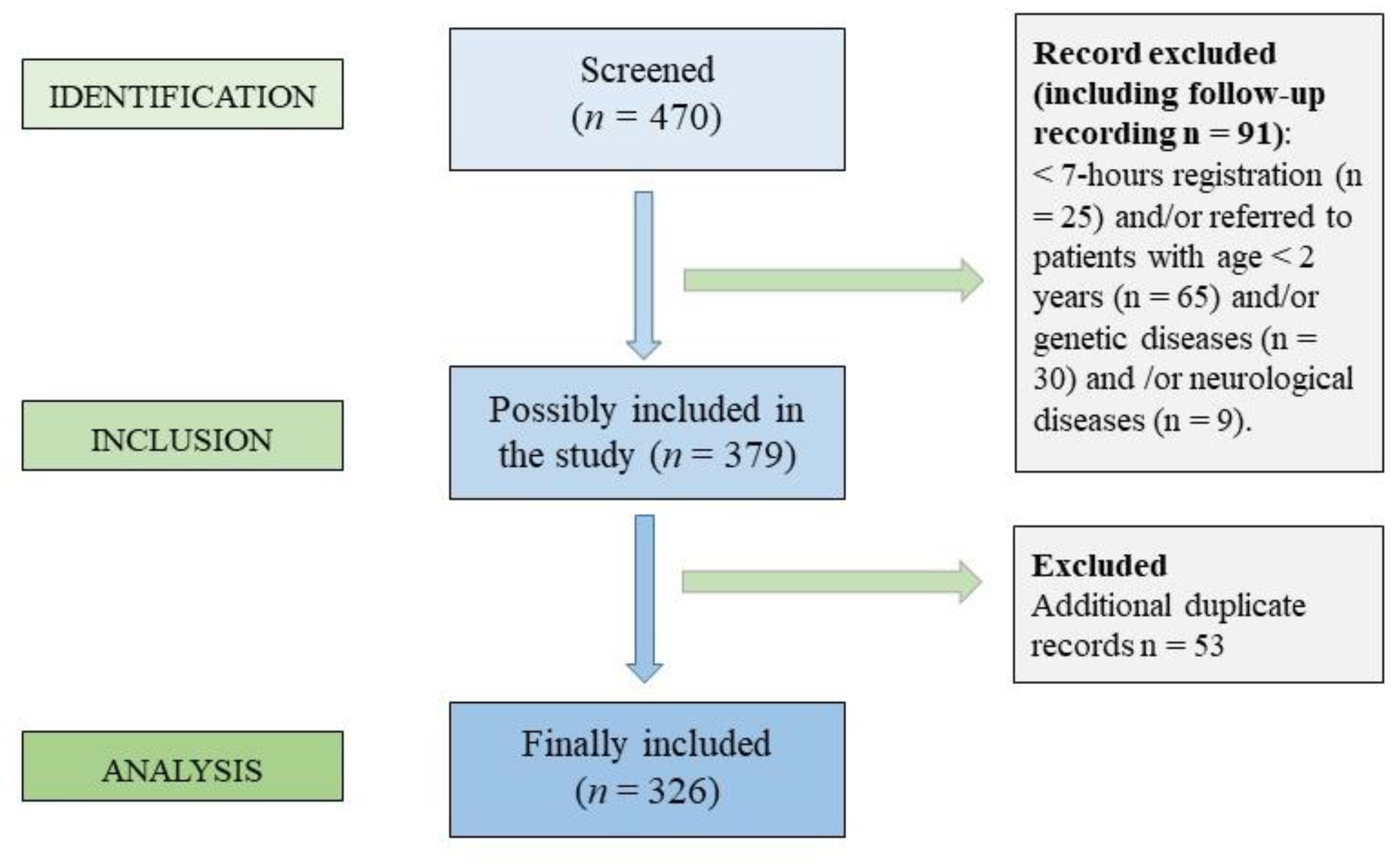
2.1. Study Design and Population
2.2. Anthropometry
2.3. Pediatric Home Respiratory Polygraphy
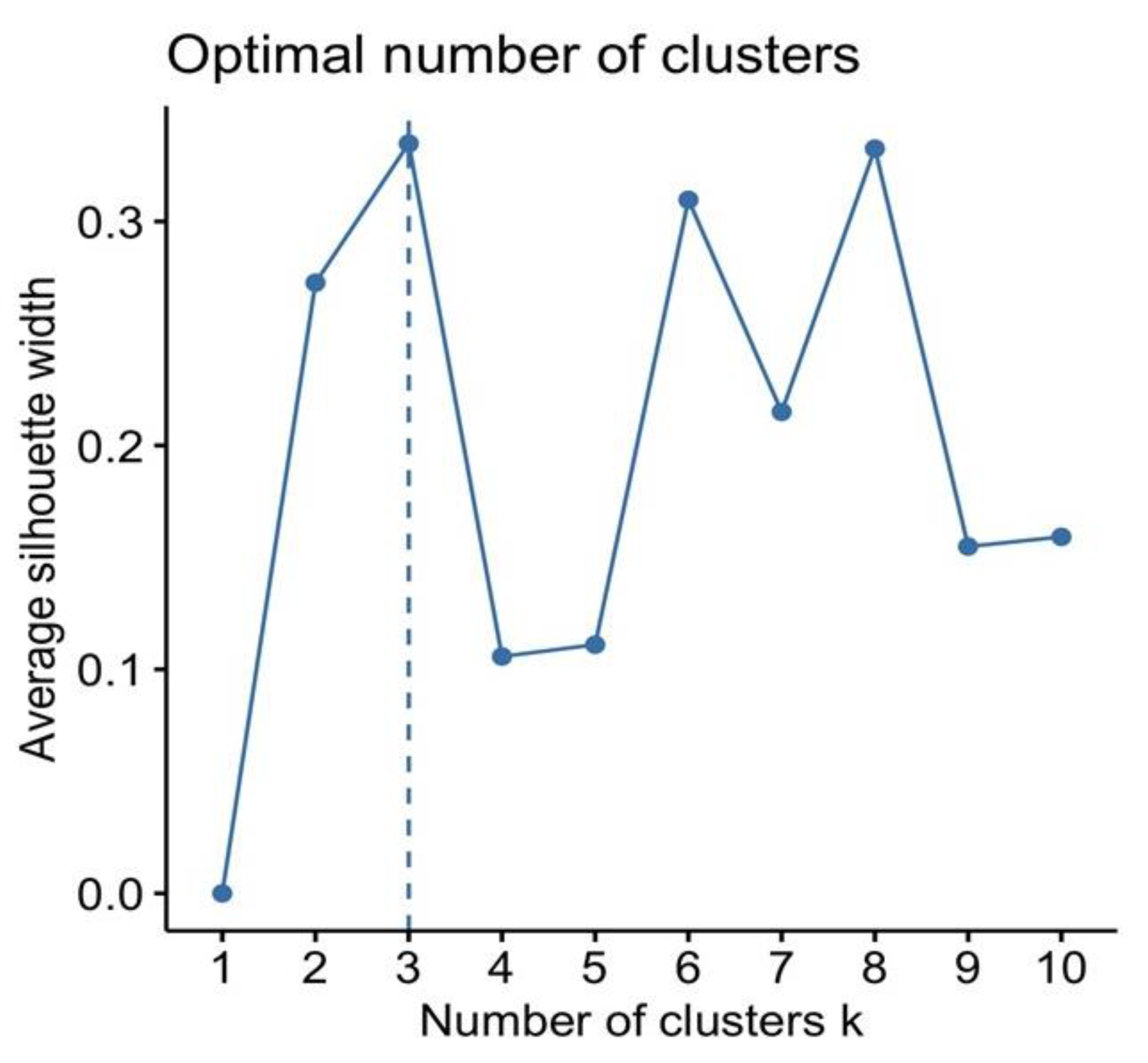
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Marcus, C.L.; Brooks, L.J.; Ward, S.D.; Draper, K.A.; Gozal, D.; Halbower, A.C.; Jones, J.; Lehmann, C.; Schechter, M.S.; Sheldon, S.; et al. Diagnosis and Management of Childhood Obstructive Sleep Apnea Syndrome. Pediatrics 2012, 130, e714–e755. [Google Scholar] [CrossRef] [PubMed]
- Arens, R.; Marcus, C.L. Pathophysiology of Upper Airway Obstruction: A Developmental Perspective. Sleep 2004, 27, 997–1019. [Google Scholar] [CrossRef]
- Schwab, R.J.; Kim, C.; Bagchi, S.; Keenan, B.T.; Comyn, F.L.; Wang, S.; Tapia, I.E.; Huang, S.; Traylor, J.; Torigian, D.A.; et al. Understanding the Anatomic Basis for Obstructive Sleep Apnea Syndrome in Adolescents. Am. J. Respir. Crit. Care Med. 2015, 191, 1295–1309. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Álvarez, M.L.; Terán-Santos, J.; Ordax Carbajo, E.; Cordero-Guevara, J.A.; Navazo-Egüia, A.I.; Kheirandish-Gozal, L.; Gozal, D. Reliability of Home Respiratory Polygraphy for the Diagnosis of Sleep Apnea in Children. Chest 2015, 147, 1020–1028. [Google Scholar] [CrossRef]
- Shamsuzzaman, A.; Amin, R.S.; Calvin, A.D.; Davison, D.; Somers, V.K. Severity of Obstructive Sleep Apnea Is Associated with Elevated Plasma Fibrinogen in Otherwise Healthy Patients. Sleep Breath. Schlaf Atm. 2014, 18, 761–766. [Google Scholar] [CrossRef] [PubMed]
- Amin, R.S.; Carroll, J.L.; Jeffries, J.L.; Grone, C.; Bean, J.A.; Chini, B.; Bokulic, R.; Daniels, S.R. Twenty-Four-Hour Ambulatory Blood Pressure in Children with Sleep-Disordered Breathing. Am. J. Respir. Crit. Care Med. 2004, 169, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Hunter, S.J.; Gozal, D.; Smith, D.L.; Philby, M.F.; Kaylegian, J.; Kheirandish-Gozal, L. Effect of Sleep-Disordered Breathing Severity on Cognitive Performance Measures in a Large Community Cohort of Young School-Aged Children. Am. J. Respir. Crit. Care Med. 2016, 194, 739–747. [Google Scholar] [CrossRef]
- Nosetti, L.; Zaffanello, M.; Katz, E.S.; Vitali, M.; Agosti, M.; Ferrante, G.; Cilluffo, G.; Piacentini, G.; Grutta, S.L. Twenty-Year Follow-up of Children with Obstructive Sleep Apnea. J. Clin. Sleep Med. 2022, 18, 1573–1581. [Google Scholar] [CrossRef]
- Zhang, Z.; Abarda, A.; Contractor, A.A.; Wang, J.; Dayton, C.M. Exploring Heterogeneity in Clinical Trials with Latent Class Analysis. Ann. Transl. Med. 2018, 6, 119. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Feng, G.; Xu, Z.; Li, X.; Zheng, L.; Ge, W.; Ni, X. Identification of Different Clinical Faces of Obstructive Sleep Apnea in Children. Int. J. Pediatr. Otorhinolaryngol. 2019, 127, 109621. [Google Scholar] [CrossRef]
- Vavougios, G.D.; Natsios, G.; Pastaka, C.; Zarogiannis, S.G.; Gourgoulianis, K.I. Phenotypes of Comorbidity in OSAS Patients: Combining Categorical Principal Component Analysis with Cluster Analysis. J. Sleep Res. 2016, 25, 31–38. [Google Scholar] [CrossRef]
- Keenan, B.T.; Kim, J.; Singh, B.; Bittencourt, L.; Chen, N.H.; Cistulli, P.A.; Magalang, U.J.; McArdle, N.; Mindel, J.W.; Benediktsdottir, B.; et al. Recognizable Clinical Subtypes of Obstructive Sleep Apnea across International Sleep Centers: A Cluster Analysis. Sleep 2018, 41, zsx214. [Google Scholar] [CrossRef] [PubMed]
- Guo, K.D.; Wang, J.; Li, N.Z.; Wang, Q.J.; Han, F.; Chen, R. Cluster Analysis and Significance of Clinical Subtypes in Patients with Obstructive Sleep Apnea Hypopnea Syndrome. Zhonghua Yi Xue Za Zhi 2019, 99, 2176–2181. [Google Scholar] [CrossRef]
- Ferreira-Santos, D.; Pereira Rodrigues, P. Phenotyping obstructive sleep apnea patients: A first approach to cluster visualization. In Studies in Health Technology and Informatics; IOS Press: Amsterdam, The Netherlands, 2018; Volume 255, pp. 75–79. [Google Scholar]
- Joosten, S.A.; Hamza, K.; Sands, S.; Turton, A.; Berger, P.; Hamilton, G. Phenotypes of Patients with Mild to Moderate Obstructive Sleep Apnoea as Confirmed by Cluster Analysis. Respirology 2012, 17, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Zaffanello, M.; Piacentini, G.; Pietrobelli, A.; Fava, C.; Lippi, G.; Maffeis, C.; Gasperi, E.; Nosetti, L.; Bonafini, S.; Tagetti, A.; et al. Ambulatory Clinical Parameters and Sleep Respiratory Events in a Group of Obese Children Unselected for Respiratory Problems. World J. Pediatr. 2017, 13, 577–583. [Google Scholar] [CrossRef]
- Berry, R.B.; Budhiraja, R.; Gottlieb, D.J.; Gozal, D.; Iber, C.; Kapur, V.K.; Marcus, C.L.; Mehra, R.; Parthasarathy, S.; Quan, S.F.; et al. Rules for Scoring Respiratory Events in Sleep: Update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. J. Clin. Sleep Med. 2012, 8, 597–619. [Google Scholar] [CrossRef]
- Moss, D.; Urschitz, M.S.; von Bodman, A.; Eitner, S.; Noehren, A.; Urschitz-Duprat, P.M.; Schlaud, M.; Poets, C.F. Reference Values for Nocturnal Home Polysomnography in Primary Schoolchildren. Pediatr. Res. 2005, 58, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Dehlink, E.; Tan, H.-L. Update on Paediatric Obstructive Sleep Apnoea. J. Thorac. Dis. 2016, 8, 224. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, A.P.; Richards, G.; De La Iglesia, B.; Rayward-Smith, V.J. Clustering Rules: A Comparison of Partitioning and Hierarchical Clustering Algorithms. J. Math. Model. Algorithms 2006, 5, 475–504. [Google Scholar] [CrossRef]
- Rousseeuw, P.J. Silhouettes: A Graphical Aid to the Interpretation and Validation of Cluster Analysis. J. Comput. Appl. Math. 1987, 20, 53–65. [Google Scholar] [CrossRef]
- Freeman, K.; Bonuck, K. Snoring, Mouth-Breathing, and Apnea Trajectories in a Population-Based Cohort Followed from Infancy to 81 Months: A Cluster Analysis. Int. J. Pediatr. Otorhinolaryngol. 2012, 76, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, H.; Kobayashi, M.; Tsuiki, S.; Yanagihara, M.; Inoue, Y. Obstructive Sleep Apnea Phenotypes in Men Based on Characteristics of Respiratory Events during Polysomnography. Sleep Breath. 2019, 23, 1087–1094. [Google Scholar] [CrossRef] [PubMed]
- Spruyt, K.; Verleye, G.; Gozal, D. Unbiased Categorical Classification of Pediatric Sleep Disordered Breathing. Sleep 2010, 33, 1341–1347. [Google Scholar] [CrossRef]
- Spruyt, K.; Gozal, D. REM and NREM Sleep-State Distribution of Respiratory Events in Habitually Snoring School-Aged Community Children. Sleep Med. 2012, 13, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Walter, L.M.; Dassanayake, D.U.N.; Weichard, A.J.; Davey, M.J.; Nixon, G.M.; Horne, R.S.C. Back to Sleep or Not: The Effect of the Supine Position on Pediatric OSA: Sleeping Position in Children with OSA. Sleep Med. 2017, 37, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Cartwright, R.D. Effect of Sleep Position on Sleep Apnea Severity. Sleep 1984, 7, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Dayyat, E.; Maarafeya, M.M.A.; Capdevila, O.S.; Kheirandish-Gozal, L.; Montgomery-Downs, H.E.; Gozal, D. Nocturnal Body Position in Sleeping Children with and without Obstructive Sleep Apnea. Pediatr. Pulmonol. 2007, 42, 374–379. [Google Scholar] [CrossRef] [PubMed]
- Tholen, K.; Meier, M.; Kloor, J.; Friedman, N. Persistent OSA in Obese Children: Does Body Position Matter? J. Clin. Sleep Med. 2021, 17, 227–232. [Google Scholar] [CrossRef]
- Selvadurai, S.; Voutsas, G.; Massicotte, C.; Kassner, A.; Katz, S.L.; Propst, E.J.; Narang, I. Positional Obstructive Sleep Apnea in an Obese Pediatric Population. J. Clin. Sleep Med. 2020, 16, 1295–1301. [Google Scholar] [CrossRef] [PubMed]
- McLaren, A.T.; Bin-Hasan, S.; Narang, I. Diagnosis, Management and Pathophysiology of Central Sleep Apnea in Children. Paediatr. Respir. Rev. 2019, 30, 49–57. [Google Scholar] [CrossRef]
- Zaffanello, M.; Gasperi, E.; Tenero, L.; Piazza, M.; Pietrobelli, A.; Sacchetto, L.; Antoniazzi, F.; Piacentini, G. Sleep-Disordered Breathing in Children with Recurrent Wheeze/Asthma: A Single Centre Study. Children 2017, 4, 97. [Google Scholar] [CrossRef] [PubMed]
- Kheirandish-Gozal, L.; Gozal, D. Obstructive Sleep Apnea and Inflammation: Proof of Concept Based on Two Illustrative Cytokines. Int. J. Mol. Sci. 2019, 20, 459. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.; Zea-Hernandez, J.A.; Sin, S.; Graw-Panzer, K.; Shifteh, K.; Isasi, C.R.; Wagshul, M.E.; Moran, E.E.; Posner, J.; Zimmerman, M.E.; et al. The Effects of Obstructive Sleep Apnea Syndrome on the Dentate Gyrus and Learning and Memory in Children. J. Neurosci. 2017, 37, 4280–4288. [Google Scholar] [CrossRef] [PubMed]
- Berry, R.B.; Brooks, R.; Gamaldo, C.; Harding, S.M.; Lloyd, R.M.; Quan, S.F.; Troester, M.T.; Vaughn, B.V. AASM Scoring Manual Updates for 2017 (Version 2.4). J. Clin. Sleep Med. JCSM Off. Publ. Am. Acad. Sleep Med. 2017, 13, 665. [Google Scholar] [CrossRef] [PubMed]
- Mukandala, G.; Tynan, R.; Lanigan, S.; O’Connor, J.J. The Effects of Hypoxia and Inflammation on Synaptic Signaling in the CNS. Brain Sci. 2016, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Boudewyns, A.; Van de Heyning, P.; Verhulst, S. Central Apneas in Children with Obstructive Sleep Apnea Syndrome: Prevalence and Effect of Upper Airway Surgery. Sleep Med. 2016, 25, 93–97. [Google Scholar] [CrossRef]
- Oceja, E.; Rodríguez, P.; Jurado, M.J.; Alonso, M.L.; Del Río, G.; Villar, M.Á.; Mediano, O.; Martínez, M.; Juarros, S.; Merino, M.; et al. Validity and Cost-Effectiveness of Pediatric Home Respiratory Polygraphy for the Diagnosis of Obstructive Sleep Apnea in Children: Rationale, Study Design, and Methodology. Methods Protoc. 2021, 4, 9. [Google Scholar] [CrossRef]
- Hornero, R.; Kheirandish-Gozal, L.; Gutiérrez-Tobal, G.C.; Philby, M.F.; Alonso-Álvarez, M.L.; Alvarez, D.; Dayyat, E.A.; Xu, Z.; Huang, Y.S.; Kakazu, M.T.; et al. Nocturnal Oximetry-Based Evaluation of Habitually Snoring Children. Am. J. Respir. Crit. Care Med. 2017, 196, 1591–1598. [Google Scholar] [CrossRef]

| Panel A | |||
| n = 326 | |||
| Age, years | 8.05 (4.09) | ||
| Sex: M, n (%) | 186 (57.06) | ||
| Height, cm | 126.88 (25.12) | ||
| Height, Percentile | 55.06 (32.78) | ||
| Height, Z-score | 0.10 (1.39) | ||
| Weight, kg | 38.95 (28.59) | ||
| Weight, Percentile | 65.50 (34.67) | ||
| Weight, Z-score | 0.83 (1.79) | ||
| BMI, kg/m2 | 21.14 (7.98) | ||
| BMI, z-score | 1.27 (1.94) | ||
| BMI categories | |||
| Underweight | 17 (5.21) | ||
| Normal weight | 153 (46.93) | ||
| Overweight | 72 (22.09) | ||
| Obese | 84 (25.77) | ||
| Panel B | |||
| Respiratory Events | n./h (SD) | Event per Body Position | n./h (SD) |
| OA | 3.09 (6.86) | Supine OA | 4.21 (9.64) |
| MA | 0.37 (1.19) | Not supine OA | 2.30 (5.78) |
| CA | 1.83 (1.89) | Supine CA | 2.26 (5.65) |
| H | 1.79 (3.91) | Not supine CA | 1.85 (1.99) |
| RDI | 7.08 (10.10) | Supine MA | 0.49 (1.64) |
| ODI | 4.54 (9.22) | Not supine MA | 0.30 (0.98) |
| Minimum SpO2, mean (SD) % | 86.19 (10.49) | OSAS severity | n. (%) |
| Mean SpO2, mean (SD) % | 96.82 (2.68) | Mild | 191 (58.59) |
| SpO2 < 90%, mean (SD) minutes | 1.29 (6.37) | Moderate | 79 (24.23) |
| Snoring, mean (SD) %TST | 2.24 (5.73) | Severe | 56 (17.18) |
| Panel A | |||||||
| Cluster 1 | Cluster 2 | Cluster 3 | p-Value | Cluster 1 vs. Cluster 2 | Cluster 1 vs. Cluster 3 | Cluster 2 vs. Cluster 3 | |
| N = 194 | N = 93 | N = 39 | |||||
| Age, mean (SD) years | 8.69 (4.14) | 6.92 (3.43) | 7.58 (4.73) | 0.002 | 0.002 | 0.260 | 0.664 |
| Sex: M, n (%) | 112 (57.7%) | 54 (58.1%) | 20 (51.3%) | 0.739 | 1.000 | 0.900 | 0.900 |
| Weight, Percentile | 69.5 (33.2) | 60.3 (35.0) | 57.8 (38.8) | 0.036 | 0.088 | 0.128 | 0.919 |
| Weight Z-score | 1.04 (1.78) | 0.86 (1.78) | −0.14 (1.65) | 0.009 | 0.430 | 0.003 | 0.015 |
| Height, Percentile | 55.9 (32.4) | 54.4 (33.8) | 52.5 (32.7) | 0.811 | 0.925 | 0.820 | 0.950 |
| Height Z-score | 0.17 (1.36) | 0.03 (1.49) | −0.12 (1.31) | 0.406 | 0.430 | 0.310 | 0.730 |
| BMI, mean (SD) kg/m2 | 22.0 (8.14) | 18.9 (5.36) | 22.0 (11.0) | 0.006 | 0.005 | 1.000 | 0.103 |
| BMI z-score | 1.50 (1.91) | 0.97 (1.72) | 0.86 (2.42) | 0.034 | 0.044 | 0.100 | 0.620 |
| BMI categories, n. (%) | |||||||
| Underweight | 5 (2.58) | 6 (6.45) | 6 (15.4) | 0.015 | 0.259 | 0.033 | 0.259 |
| Normal weight | 87 (44.8) | 48 (51.6) | 18 (46.2) | ||||
| Overweight | 47 (24.2) | 20 (21.5) | 5 (12.8) | ||||
| Obese | 55 (28.4) | 19 (20.4) | 10 (25.6) | ||||
| Panel B | |||||||
| Polygraphy | Cluster 1 | Cluster 2 | Cluster 3 | p-Value | Cluster 1 vs. Cluster 2 | Cluster 1 vs. Cluster 3 | Cluster 2 vs. Cluster 3 |
| N = 194 | N = 93 | N = 39 | |||||
| RDI, mean (SD) n./h | 3.71 (3.23) | 6.38 (3.92) | 25.5 (19.4) | <0.001 | 0.013 | <0.001 | <0.001 |
| OA, mean (SD) n./h | 1.70 (2.24) | 2.02 (2.74) | 12.6 (15.9) | <0.001 | 0.907 | <0.001 | <0.001 |
| H, mean (SD) n./h | 0.87 (1.24) | 0.57 (0.70) | 9.29 (7.50) | <0.001 | 0.670 | <0.001 | <0.001 |
| OA + H, mean (SD) n./h | 1.29 (1.43) | 1.30 (1.47) | 10.9 (8.24) | <0.001 | 1.000 | <0.001 | <0.001 |
| CA, mean (SD) n./h | 0.91 (0.71) | 3.53 (1.91) | 2.39 (2.74) | <0.001 | <0.001 | <0.001 | <0.001 |
| MA, mean (SD) n./h | 0.22 (0.63) | 0.28 (0.34) | 1.37 (2.95) | <0.001 | 0.886 | <0.001 | <0.001 |
| ODI | 2.26 (2.71) | 2.39 (2.24) | 21.1 (19.0) | <0.001 | 0.988 | <0.001 | <0.001 |
| Minimum SpO2, mean (SD) % | 87.1 (10.5) | 87.4 (7.94) | 78.6 (12.5) | <0.001 | 0.967 | <0.001 | <0.001 |
| Mean SpO2, mean (SD) % | 97.1 (1.08) | 97.1 (1.79) | 94.7 (6.50) | <0.001 | 0.995 | <0.001 | <0.001 |
| Time SpO2 < 90% eTST, mean (SD) | 0.48 (2.29) | 1.02 (4.61) | 5.95 (15.6) | <0.001 | 0.768 | <0.001 | <0.001 |
| Snoring, mean (SD) %TST | 2.09 (5.00) | 1.68 (4.73) | 4.34 (9.72) | 0.044 | 0.839 | 0.040 | 0.064 |
| OSAS severity, n. (%) | <0.001 | <0.001 | <0.001 | <0.001 | |||
| Mild | 152 (78.4) | 39 (41.9) | 0 (0.00) | ||||
| Moderate | 33 (17.0) | 42 (45.2) | 4 (10.3) | ||||
| Severe | 9 (4.64) | 12 (12.9) | 35 (89.7) | ||||
| Position | |||||||
| Supine Sleep Time, mean (SD) % | 51.9 (23.9) | 48.6 (24.1) | 50.0 (27.6) | 0.567 | 0.547 | 0.954 | 0.900 |
| Supine OA, mean (SD) n./h | 2.19 (3.28) | 3.07 (5.47) | 17.0 (21.8) | <0.001 | 0.688 | <0.001 | <0.001 |
| Not supine OA, mean (SD) n./h | 1.21 (1.79) | 1.49 (2.07) | 9.72 (14.0) | <0.001 | 0.900 | <0.001 | <0.001 |
| Supine H, mean (SD) n./h | 1.11 (1.83) | 0.67 (0.98) | 11.0 (10.3) | <0.001 | 0.641 | <0.001 | <0.001 |
| Not supine H, mean (SD) n./h | 0.54 (0.93) | 0.46 (0.71) | 7.93 (7.22) | <0.001 | 0.973 | <0.001 | <0.001 |
| Supine CA, mean (SD) n./h | 0.98 (1.09) | 4.37 (7.96) | 3.63 (9.61) | <0.001 | <0.001 | 0.757 | 0.016 |
| Not supine CA, mean (SD) n./h | 0.74 (0.61) | 3.89 (1.77) | 2.51 (2.75) | <0.001 | <0.001 | <0.001 | <0.00 |
| Supine MA, mean (SD) n./h | 0.25 (0.68) | 0.43 (0.75) | 1.83 (4.15) | <0.001 | 0.624 | <0.001 | <0.001 |
| Not supine MA, mean (SD) n./h | 0.16 (0.33) | 0.22 (0.30) | 1.21 (2.55) | <0.001 | 0.882 | <0.001 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zaffanello, M.; Pietrobelli, A.; Gozal, D.; Nosetti, L.; La Grutta, S.; Cilluffo, G.; Ferrante, G.; Piazza, M.; Piacentini, G. Cluster Analysis of Home Polygraphic Recordings in Symptomatic Habitually-Snoring Children: A Precision Medicine Perspective. J. Clin. Med. 2022, 11, 5960. https://doi.org/10.3390/jcm11195960
Zaffanello M, Pietrobelli A, Gozal D, Nosetti L, La Grutta S, Cilluffo G, Ferrante G, Piazza M, Piacentini G. Cluster Analysis of Home Polygraphic Recordings in Symptomatic Habitually-Snoring Children: A Precision Medicine Perspective. Journal of Clinical Medicine. 2022; 11(19):5960. https://doi.org/10.3390/jcm11195960
Chicago/Turabian StyleZaffanello, Marco, Angelo Pietrobelli, David Gozal, Luana Nosetti, Stefania La Grutta, Giovanna Cilluffo, Giuliana Ferrante, Michele Piazza, and Giorgio Piacentini. 2022. "Cluster Analysis of Home Polygraphic Recordings in Symptomatic Habitually-Snoring Children: A Precision Medicine Perspective" Journal of Clinical Medicine 11, no. 19: 5960. https://doi.org/10.3390/jcm11195960
APA StyleZaffanello, M., Pietrobelli, A., Gozal, D., Nosetti, L., La Grutta, S., Cilluffo, G., Ferrante, G., Piazza, M., & Piacentini, G. (2022). Cluster Analysis of Home Polygraphic Recordings in Symptomatic Habitually-Snoring Children: A Precision Medicine Perspective. Journal of Clinical Medicine, 11(19), 5960. https://doi.org/10.3390/jcm11195960









