Sex-Related Differences in Clinical Outcomes in Patients with Atrial Fibrillation and Coronary Artery Disease: A Sub-Study of the MISOAC-AF Randomized Controlled Trial
Abstract
1. Introduction
2. Methods
2.1. Data Sources
2.2. Study Population
2.3. Study Variables
2.4. Study Outcomes
2.5. Definition of Covariates
2.6. Statistical Analysis
3. Results
3.1. Baseline Characteristics, Demographics, and Medical History
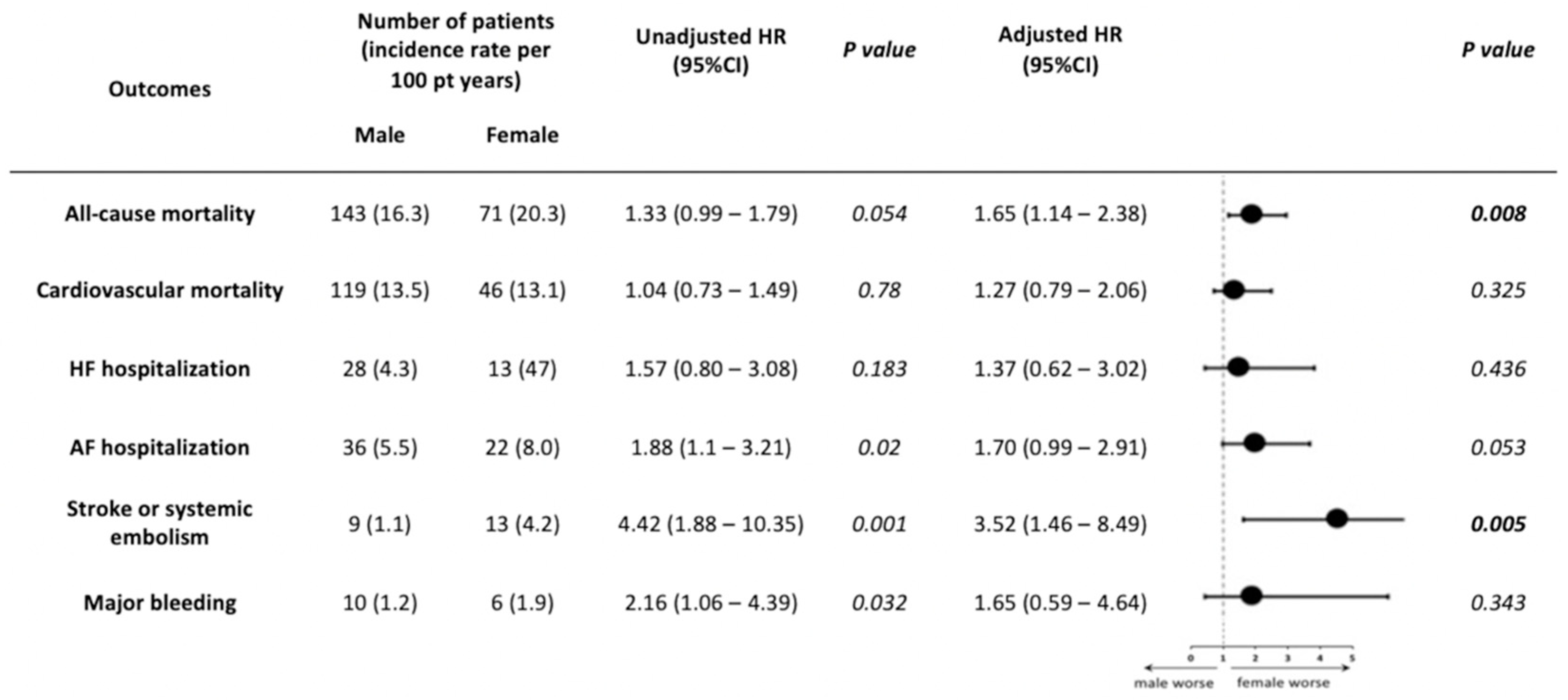
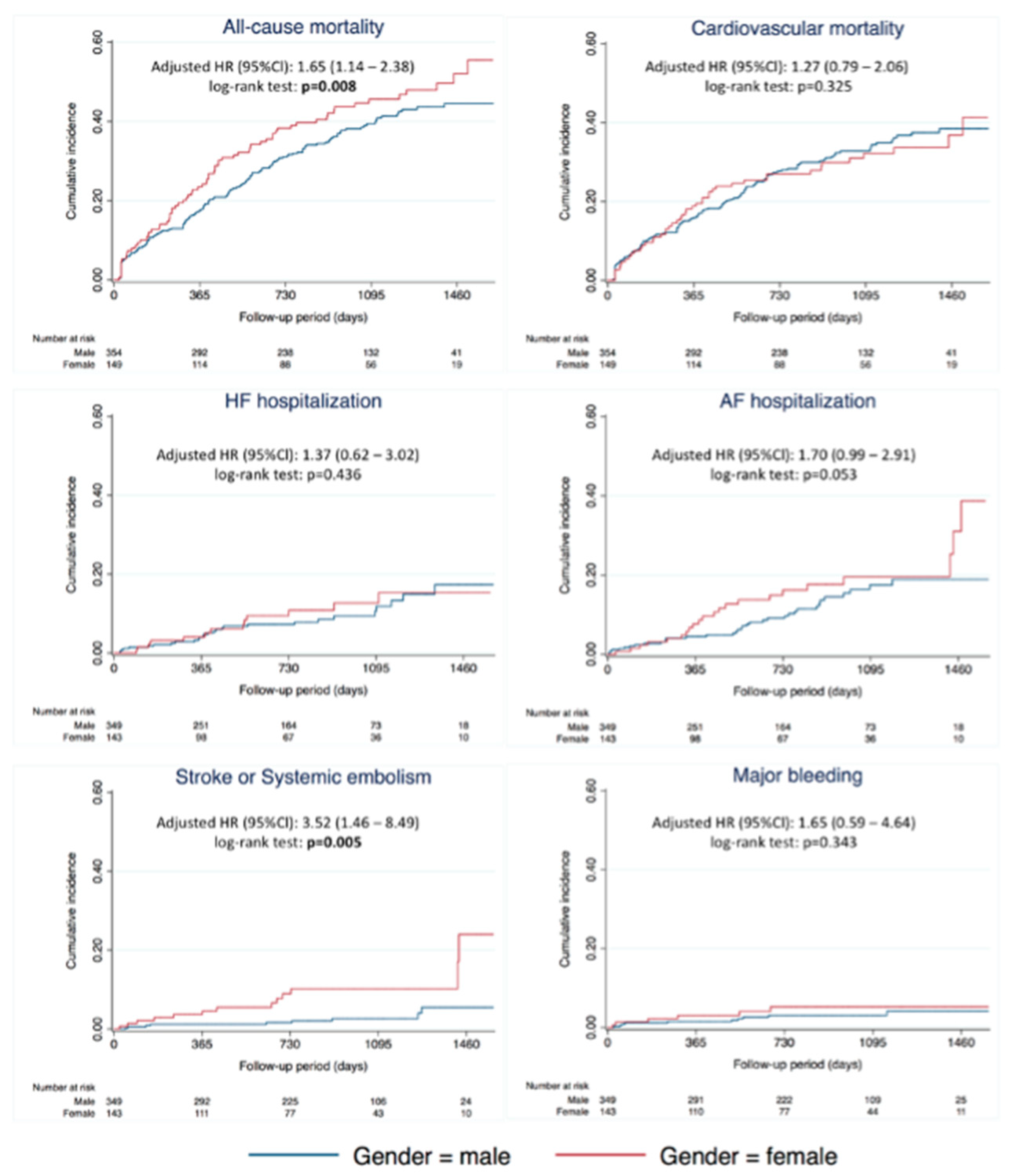
3.2. Outcomes According to Gender
3.3. Subgroup Analysis
4. Discussion
5. Strengths and Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AF | Atrial Fibrillation |
| ACS | Acute Coronary Syndrome |
| CAD | Coronary Artery Disease |
| CV | Cardiovascular |
| MINOCA | Myocardial infarction with non-obstructive coronary arteries |
References
- Zoni-Berisso, M.; Lercari, F.; Carazza, T.; Domenicucci, S. Epidemiology of atrial fbrillation: European perspective. Clin. Epidemiol. 2014, 6, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Van Gelder, I.C.; Groenveld, H.F.; Crijns, H.J.G.M.; Tuininga, Y.S.; Tijssen, J.G.P.; Alings, A.M.; Hillege, H.L.; Bergsma-Kadijk, J.A.; Cornel, J.H.; Kamp, O.; et al. Lenient versus Strict Rate Control in Patients with Atrial Fibrillation. N. Engl. J. Med. 2010, 362, 1363–1373. [Google Scholar] [CrossRef] [PubMed]
- Netzler, P.C. Rivaroxaban versus Warfarin in Nonvalvular Atrial Fibrillation. Yearb. Cardiol. 2012, 2012, 490–492. [Google Scholar] [CrossRef]
- Mostafa, A.; EL-Haddad, M.A.; Shenoy, M.; Tuliani, T. Atrial fibrillation post cardiac bypass surgery. Avicenna J. Med. 2012, 2, 65–70. [Google Scholar] [CrossRef]
- Michniewicz, E.; Mlodawska, E.; Lopatowska, P.; Tomaszuk-Kazberuk, A.; Malyszko, J. Patients with atrial fibrillation and coronary artery disease–Double trouble. Adv. Med. Sci. 2018, 63, 30–35. [Google Scholar] [CrossRef]
- Kenkre, T.S.; Malhotra, P.; Johnson, B.D.; Handberg, E.M.; Thompson, D.V.; Marroquin, O.C.; Rogers, W.J.; Pepine, C.J.; Merz, C.N.B.; Kelsey, S.F. Ten-Year Mortality in the WISE Study (Women’s Ischemia Syndrome Evaluation). Circ. Cardiovasc. Qual. Outcomes 2017, 10, e003863. [Google Scholar] [CrossRef]
- Garcia, M.; Mulvagh, S.L.; Merz, C.N.B.; Buring, J.E.; Manson, J.A.E. Cardiovascular disease in women: Clinical perspectives. Circ. Res. 2016, 118, 1273–1293. [Google Scholar] [CrossRef]
- Kavousi, M. Differences in Epidemiology and Risk Factors for Atrial Fibrillation Between Women and Men. Front. Cardiovasc. Med. 2020, 7, 3. [Google Scholar] [CrossRef]
- Samaras, A.; Kartas, A.; Vasdeki, D.; Dividis, G.; Forozidou, E.; Fotos, G.; Kotsi, E.; Paschou, E.; Tsoukra, P.; Goulas, I.; et al. Rationale and design of a randomized study comparing Motivational Interviewing to Support Oral Anticoagulation adherence versus usual care in patients with nonvalvular atrial fibrillation: The MISOAC-AF trial. Hell. J. Cardiol. 2020, 61, 453–454. [Google Scholar] [CrossRef]
- Tzikas, A.; Samaras, A.; Kartas, A.; Vasdeki, D.; Fotos, G.; Dividis, G.; Paschou, E.; Forozidou, E.; Tsoukra, P.; Kotsi, E.; et al. Motivational Interviewing to Support Oral AntiCoagulation adherence in patients with non-valvular Atrial Fibrillation (MISOAC-AF): A randomized clinical trial. Eur. Heart J. Cardiovasc. Pharmacother. 2021, 7, f63–f71. [Google Scholar] [CrossRef]
- Hindricks, G.; Potpara, T.; Dagres, N.; Bax, J.J.; Boriani, G.; Dan, G.A.; Dilaveris, P.E.; Fauchier, L.; Arbelo, E.; Kalman, J.M.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur. Heart J. 2021, 42, 373–498. [Google Scholar] [CrossRef]
- Calkins, H.; Gliklich, R.E.; Leavy, M.B.; Piccini, J.P.; Hsu, J.C.; Mohanty, S.; Lewis, W.; Nazarian, S.; Turakhia, M.P. Harmonized outcome measures for use in atrial fibrillation patient registries and clinical practice: Endorsed by the Heart Rhythm Society Board of Trustees. Heart Rhythm 2019, 16, e3–e16. [Google Scholar] [CrossRef]
- Cannon, C.P.; Brindis, R.G.; Chaitman, B.R.; Cohen, D.J.; Cross, J.T., Jr.; Drozda, J.P., Jr.; Fesmire, F.M.; Fintel, D.J.; Fonarow, G.C.; Fox, K.A.; et al. 2013 ACCF/AHA Key data elements and definitions for measuring the clinical management and outcomes of patients with acute coronary syndromes and coronary artery disease: A report of the American college of cardiology foundation/American Heart Association. Circulation 2013, 127, 1052–1089. [Google Scholar] [CrossRef]
- Taylor, P.; Fine, J.P.; Gray, R.J.; Fine, J.P.; Gray, R.J. A Proportional Hazards Model for the Subdistribution of a Competing Risk A Proportional Hazards Model for the Subdistribution of a Competing Risk. J. Am. Stat. Assoc. 2012, 94, 37–41. [Google Scholar]
- Papazoglou, A.S.; Kartas, A.; Samaras, A.; Vouloagkas, I.; Vrana, E.; Moysidis, D.V.; Akrivos, E.; Kotzampasis, G.; Baroutidou, A.; Papanastasiou, A.; et al. Prognostic significance of diabetes mellitus in patients with atrial fibrillation. Cardiovasc. Diabetol. 2021, 20, 40. [Google Scholar] [CrossRef]
- Huxley, R.; Barzi, F.; Woodward, M. Excess risk of fatal coronary heart disease associated with diabetes in men and women: Meta-analysis of 37 prospective cohort studies. Br. Med. J. 2006, 332, 73–76. [Google Scholar] [CrossRef]
- Xiong, Z.; Liu, T.; Tse, G.; Gong, M.; Gladding, P.A.; Smaill, B.H.; Stiles, M.K.; Gillis, A.M.; Zhao, J. A machine learning aided systematic review and meta-analysis of the relative risk of atrial fibrillation in patients with diabetes mellitus. Front. Physiol. 2018, 9, 835. [Google Scholar] [CrossRef]
- Sato, C.; Wakabayashi, K.; Ikeda, N.; Honda, Y.; Sato, K.; Suzuki, T.; Shibata, K.; Tanno, K. Influence of sex on the incidence of potential coronary artery disease and long-term outcomes in asymptomatic patients with diabetes mellitus. IJC Heart Vasc. 2020, 27, 100504. [Google Scholar] [CrossRef]
- Matta, A.; Moussallem, N. Coronary artery disease is associated with valvular heart disease, but could it Be a predictive factor? Indian Heart J. 2019, 71, 284–287. [Google Scholar] [CrossRef]
- Fukumoto, R.; Kawai, M.; Minai, K.; Ogawa, K.; Yoshida, J.; Inoue, Y.; Morimoto, S.; Tanaka, T.; Nagoshi, T.; Ogawa, T.; et al. Conflicting relationship between age-dependent disorders, valvular heart disease and coronary artery disease by covariance structure analysis: Possible contribution of natriuretic peptide. PLoS ONE 2017, 12, e0181206. [Google Scholar] [CrossRef]
- Manfrini, O.; Yoon, J.; van der Schaar, M.; Kedev, S.; Vavlukis, M.; Stankovic, G.; Scarpone, M.; Miličić, D.; Vasiljevic, Z.; Badimon, L.; et al. Sex differences in modifiable risk factors and severity of coronary artery disease. J. Am. Heart Assoc. 2020, 9, e017235. [Google Scholar] [CrossRef]
- Jones, E.; Delia Johnson, B.; Shaw, L.J.; Bakir, M.; Wei, J.; Mehta, P.K.; Minissian, M.; Pepine, C.J.; Reis, S.E.; Kelsey, S.F.; et al. Not typical angina and mortality in women with obstructive coronary artery disease: Results from the Women’s Ischemic Syndrome Evaluation study (WISE). IJC Heart Vasc. 2020, 27, 100502. [Google Scholar] [CrossRef]
- Mehta, P.K.; Bess, C.; Elias-Smale, S.; Vaccarino, V.; Quyyumi, A.; Pepine, C.J.; Bairey Merz, C.N. Gender in cardiovascular medicine: Chest pain and coronary artery disease. Eur. Heart J. 2019, 40, 3819–3826. [Google Scholar] [CrossRef]
- Wagstaff, A.J.; Overvad, T.F.; Lip, G.Y.H.; Lane, D.A. Is female sex a risk factor for stroke and thromboembolism in patients with atrial fibrillation? A systematic review and meta-analysis. QJM 2014, 107, 955–967. [Google Scholar] [CrossRef] [PubMed]
- Bassand, J.P.; Apenteng, P.N.; Atar, D.; Camm, A.J.; Cools, F.; Corbalan, R.; A Fitzmaurice, D.; Fox, K.A.; Goto, S.; Haas, S.; et al. GARFIELD-AF: A worldwide prospective registry of patients with atrial fibrillation at risk of stroke. Future Cardiol. 2021, 17, 19–38. [Google Scholar] [CrossRef]
- Thompson, L.E.; Maddox, T.M.; Lei, L.; Grunwald, G.K.; Bradley, S.M.; Peterson, P.N.; Masoudi, F.A.; Turchin, A.; Song, Y.; Doros, G.; et al. Sex differences in the use of oral anticoagulants for atrial fibrillation: A report from the National Cardiovascular Data Registry (NCDR®) PINNACLE registry. J. Am. Heart Assoc. 2017, 6, e005801. [Google Scholar] [CrossRef]
- Nielsen, P.B.; Skjøth, F.; Overvad, T.F.; Larsen, T.B.; Lip, G.Y.H. Female Sex Is a Risk Modifier Rather Than a Risk Factor for Stroke in Atrial Fibrillation. Circulation 2018, 137, 832–840. [Google Scholar] [CrossRef]

| Women with CAD (n = 132) | Men with CAD (n = 329) | p-Value | |
|---|---|---|---|
| Characteristics, (mean ± SD) | |||
| Age (years) | 77.21 ± 8.9 | 73.76 ± 8.9 | <0.001 |
| Body mass index (kg/m2) | 29 ± 4.7 | 28.4 ± 5.1 | 0.26 |
| Glomerular filtration rate by CKD-EPI (mL/min/1.73 m2) | 55.53 ± 27.8 | 66.03 ± 32.5 | 0.006 |
| High sensitivity Troponin-T (pg/mL) | 190.7 ± 943 | 160.7 ± 714.9 | 0.96 |
| NT-pro BNP (pg/mL) | 5222.8 ± 6546.4 | 5155.8 ± 8004.8 | 0.95 |
| LVEF | 49.92 ± 11.1 | 43.54 ± 12.9 | <0.001 |
| Length of Hospitalization (days) | 8.03 ± 8.8 | 8.69 ± 8 | 0.45 |
| Reason for hospitalization | |||
| Atrial fibrillation | 43 (32.6) | 117 (35.6) | 0.85 |
| Congestive heart failure | 16 (12.1) | 32 (9.7) | 0.78 |
| Acute coronary syndrome | 11 (8.3) | 78 (23.7) | 0.34 |
| Valvular heart disease | 32 (24.2) | 15 (4.6) | 0.21 |
| Other cardiovascular reason | 30 (22.7) | 87 (26.4) | 0.72 |
| Clinical history, No (%) | |||
| Current smokers | 40 (30.5) | 239 (73.1) | <0.001 |
| Dyslipidemia | 91 (69.5) | 211 (64.5) | 0.31 |
| Systolic blood pressure (mean ± SD, mmHg) | 141.1 ± 26.9 | 141.5 ± 25.6 | 0.86 |
| Diastolic blood pressure (mean ± SD, mmHg) | 76.8 ± 16.5 | 81.61 ± 14 | 0.005 |
| Hypertension | 112 (85.5) | 274 (83.8) | 0.65 |
| AF duration (mean ± SD, days) | 2134.4 ± 2646.2 | 2232.1 ± 3080 | 0.85 |
| Newly-diagnosed AF | 45 (34) | 132 (40.1) | 0.31 |
| Type of existing AF | |||
| Paroxysmal | 64 (48.4) | 153 (46.5) | 0.31 |
| Persistent (>1 year) AF | 45 (34) | 102 (31) | 0.16 |
| PCI/CABG | 67 (20.5) | 260 (79.5) | <0.001 |
| Prior MI | 55 (42) | 173 (52.9) | 0.035 |
| Cardiac arrest | 5 (3.8) | 21 (6.4) | 0.27 |
| Pacemaker | 6 (4.5) | 42 (12.7) | <0.01 |
| ICD | 2 (0.4) | 6 (0.2) | 0.053 |
| Congenital heart disease | 2 (1.5) | 5 (1.5) | 0.99 |
| Diabetes mellitus | 72 (55) | 124 (37.9) | 0.001 |
| Vascular disease | 98 (74.8) | 252 (77.1) | 0.6 |
| Pulmonary disease | 26 (19.8) | 57 (17.4) | 0.54 |
| Ischemic stroke, TIA, undefined stroke | 25 (19.2) | 57 (17.5) | 0.66 |
| Heart failure | 80 (61.5) | 203 (62.1) | 0.91 |
| Valvular heart disease | 86 (72.3) | 162 (56.4) | 0.003 |
| Persistent and permanent AF | 67 (51.9) | 174 (54.4) | 0.63 |
| Paroxysmal AF | 51 (39.5) | 114 (35.6) | 0.43 |
| ACS (STEMI, NSTEMI, UA) | 24 (21.1) | 65 (23.8) | 0.55 |
| History of major bleeding | 21 (16) | 52 (15.9) | 0.97 |
| Prior cardioversion | 1 (0.8) | 15 (4.6) | 0.22 |
| Prior ablation | 0 (0) | 9 (2.8) | 0.055 |
| Treatment No (%) and risk stratification scores (mean ± SD) | |||
| Use of antiplatelets at discharge | |||
| Aspirin | 16 (14.2) | 33 (11.8) | 0.153 |
| Clopidogrel | 9 (8) | 44 (15.7) | 0.153 |
| Both | 15 (13.3) | 45 (16.1) | 0.153 |
| Use of oral anticoagulant(s) | |||
| VKA | 50 (43.5) | 99 (34.7) | 0.13 |
| NOAC | 50 (43.5) | 129 (45.3) | 0.13 |
| Dabigatran | 9 (7.8) | 24 (8.7) | 0.23 |
| Rivaroxaban | 19 (16.5) | 61 (21.4) | 0.23 |
| Apixaban | 22 (19.1) | 44 (15.4) | 0.23 |
| ACEi-ARB | 56 (42.4) | 175 (53.2) | 0.016 |
| Rate control medication | 106 (80.9) | 257 (79.6) | 0.74 |
| B-blockers | 99 (75.6) | 245 (71.9) | 0.83 |
| Digoxin | 1 (0.8) | 1 (0.3) | 0.83 |
| B-blockers and digoxin | 6 (4.6) | 11 (3.4) | 0.83 |
| Rhythm control medication | 25 (20.8) | 73 (23.9) | 0.49 |
| Propafenone | 4 (3.3) | 3 (1) | 0.18 |
| Amiodarone | 20 (16.7) | 62 (20.3) | 0.18 |
| Sotalol | 1 (0.8) | 8 (2.6) | 0.18 |
| Use of statin | 74 (60.2) | 203 (66.8) | 0.195 |
| CHA2DS2-VASc (mean ± SD) | 6.08 ± 1.47 | 4.61 ± 1.56 | <0.001 |
| HAS-BLED Mean (mean ± SD) | 2.35 ± 1.09 | 2.21 ± 1.15 | 0.62 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bekiaridou, A.; Samaras, A.; Kartas, A.; Papazoglou, A.S.; Moysidis, D.V.; Patsiou, V.; Zafeiropoulos, S.; Ziakas, A.; Giannakoulas, G.; Tzikas, A. Sex-Related Differences in Clinical Outcomes in Patients with Atrial Fibrillation and Coronary Artery Disease: A Sub-Study of the MISOAC-AF Randomized Controlled Trial. J. Clin. Med. 2022, 11, 5843. https://doi.org/10.3390/jcm11195843
Bekiaridou A, Samaras A, Kartas A, Papazoglou AS, Moysidis DV, Patsiou V, Zafeiropoulos S, Ziakas A, Giannakoulas G, Tzikas A. Sex-Related Differences in Clinical Outcomes in Patients with Atrial Fibrillation and Coronary Artery Disease: A Sub-Study of the MISOAC-AF Randomized Controlled Trial. Journal of Clinical Medicine. 2022; 11(19):5843. https://doi.org/10.3390/jcm11195843
Chicago/Turabian StyleBekiaridou, Alexandra, Athanasios Samaras, Anastasios Kartas, Andreas S. Papazoglou, Dimitrios V. Moysidis, Vasiliki Patsiou, Stefanos Zafeiropoulos, Antonios Ziakas, George Giannakoulas, and Apostolos Tzikas. 2022. "Sex-Related Differences in Clinical Outcomes in Patients with Atrial Fibrillation and Coronary Artery Disease: A Sub-Study of the MISOAC-AF Randomized Controlled Trial" Journal of Clinical Medicine 11, no. 19: 5843. https://doi.org/10.3390/jcm11195843
APA StyleBekiaridou, A., Samaras, A., Kartas, A., Papazoglou, A. S., Moysidis, D. V., Patsiou, V., Zafeiropoulos, S., Ziakas, A., Giannakoulas, G., & Tzikas, A. (2022). Sex-Related Differences in Clinical Outcomes in Patients with Atrial Fibrillation and Coronary Artery Disease: A Sub-Study of the MISOAC-AF Randomized Controlled Trial. Journal of Clinical Medicine, 11(19), 5843. https://doi.org/10.3390/jcm11195843








