Impact of Acid Suppression Therapy on Renal and Survival Outcomes in Patients with Chronic Kidney Disease: A Taiwanese Nationwide Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source
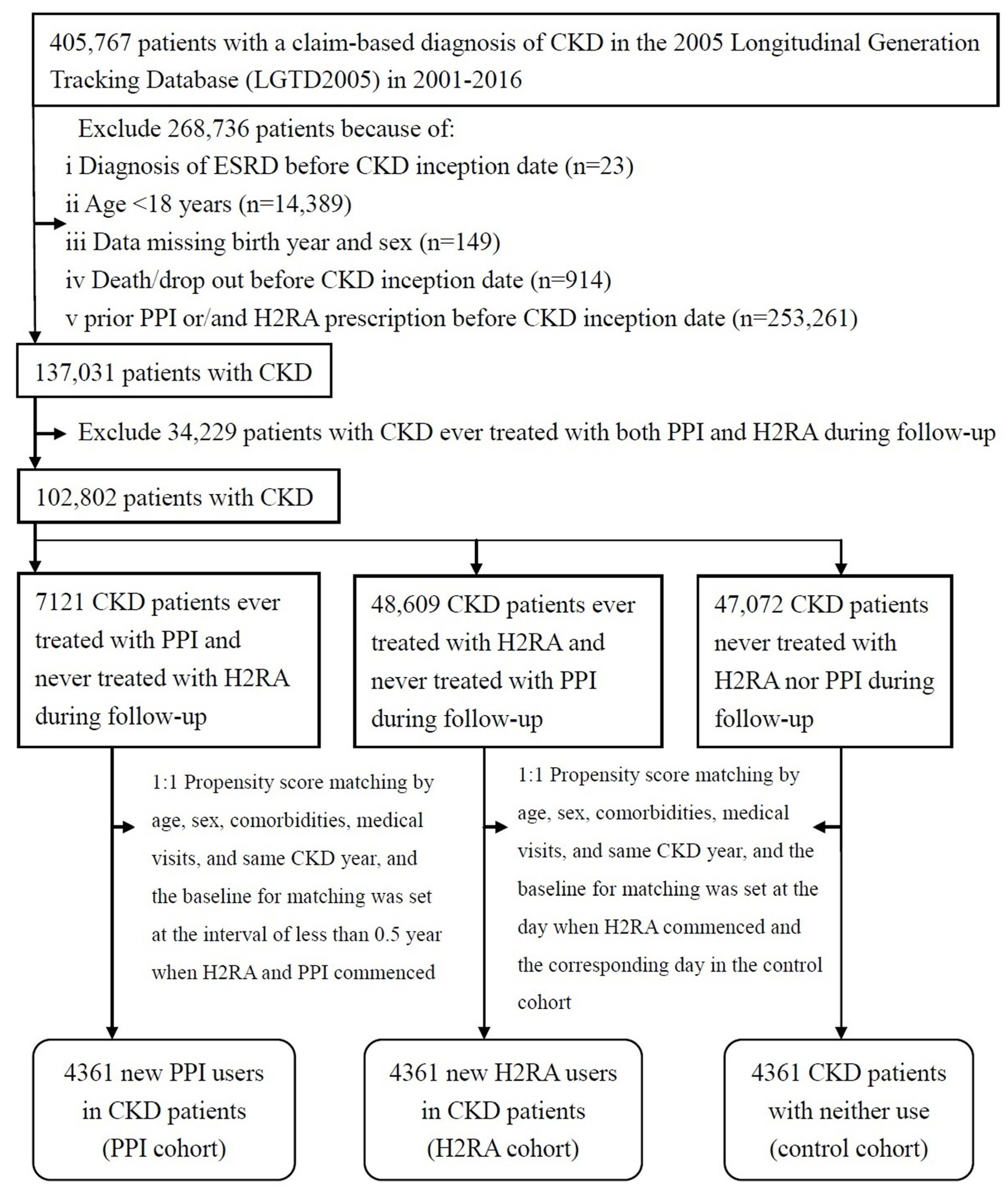
2.2. Study CKD Population
2.3. Statistical Analysis
3. Results
3.1. Demographic Data of the CKD Cohort
3.2. Association between Acid Suppression Therapy and Study Outcomes
3.3. Cumulative Incidences of ESRD and Overall Mortality
3.4. Dose–Response Relationship of Acid Suppression Therapywith ESRD, Overall, Cardiovascular, and Non-Cardiovascular Mortality
3.5. Sensitivity Analyses
3.6. Association between Acid Suppression Therapy and Study OutcomesRoughly by CKD Stages 1–4 vs. Stage 5
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Forgacs, I.; Loganayagam, A. Overprescribing proton pump inhibitors. BMJ 2008, 336, 2–3. [Google Scholar] [CrossRef] [PubMed]
- Ciz, M.; Lojek, A. Modulation of neutrophil oxidative burst via histamine receptors. Br. J. Pharmacol. 2013, 170, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Grange, C.; Gurrieri, M.; Verta, R.; Fantozzi, R.; Pini, A.; Rosa, A.C. Histamine in the kidneys: What is its role in renal pathophysiology? Br. J. Pharmacol. 2020, 177, 503–515. [Google Scholar] [CrossRef]
- Sudarikova, A.V.; Fomin, M.V.; Yankelevich, I.A.; Ilatovskaya, D.V. The implications of histamine metabolism and signaling in renal function. Physiol. Rep. 2021, 9, e14845. [Google Scholar] [CrossRef] [PubMed]
- Neumann, J.; Kirchhefer, U.; Dhein, S.; Hofmann, B.; Gergs, U. The roles of cardiovascular H2-histamine receptors under normal and pathophysiological conditions. Front. Pharmacol. 2021, 12, 732842. [Google Scholar] [CrossRef]
- Lazarus, B.; Chen, Y.; Wilson, F.P.; Sang, Y.; Chang, A.R.; Coresh, J.; Grams, M.E. Proton pump inhibitor use and the risk of chronic kidney disease. JAMA Intern. Med. 2016, 176, 238–246. [Google Scholar] [CrossRef]
- Klatte, D.C.F.; Gasparini, A.; Xu, H.; Deco, P.D.; Trevisan, M.; Johansson, A.L.V.; Wettermark, B.; Ärnlöv, J.; Janmaat, C.J.; Lindholm, B.; et al. Association between proton pump inhibitor use and risk of progression of chronic kidney disease. Gastroenterology 2017, 153, 702–710. [Google Scholar] [CrossRef]
- Xie, Y.; Bowe, B.; Li, T.; Xian, H.; Balasubramanian, S.; Al-Aly, Z. Proton pump inhibitors and risk of incident CKD and progression to ESRD. J. Am. Soc. Nephrol. 2016, 27, 3153–3163. [Google Scholar] [CrossRef]
- Xie, Y.; Bowe, B.; Li, T.; Xian, H.; Yan, Y.; Al-Aly, Z. Long-term kidney outcomes among users of proton pump inhibitors without intervening acute kidney injury. Kidney Int. 2017, 9, 1482–1494. [Google Scholar] [CrossRef]
- Xie, Y.; Bowe, B.; Li, T.; Xian, H.; Yan, Y.; Al-Aly, Z. Risk of death among users of proton pump inhibitors: A longitudinal observational cohort study of United States veterans. BMJ Open 2017, 7, e015735. [Google Scholar] [CrossRef]
- Xie, Y.; Bowe, B.; Yan, Y.; Xian, H.; Li, T.; Al-Aly, Z. Estimates of all cause mortality and cause specific mortality associated with proton pump inhibitors among US veterans: Cohort study. BMJ 2019, 365, 11580. [Google Scholar] [CrossRef]
- Malfertheiner, P.; Kandulski, A.; Venerito, M. Proton-pump inhibitors: Understanding the complications and risks. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 697–710. [Google Scholar] [CrossRef] [PubMed]
- Leary, P.J.; Tedford, R.J.; Bluemke, D.A.; Bristow, M.R.; Heckbert, S.R.; Kawut, S.M.; Krieger, E.V.; Lima, J.A.; Masri, C.S.; Ralph, D.D.; et al. Histamine H2 receptor antagonists, left ventricular morphology, and heart failure risk: The MESA Study. J. Am. Coll. Cardiol. 2016, 67, 1544–1552. [Google Scholar] [CrossRef] [PubMed]
- Adelborg, K.; Sundboll, J.; Schmidt, M.; Bøtker, H.E.; Weiss, N.S.; Pedersen, L.; Sørensen, H.T. Use of histamine H2 receptor antagonists and outcomes in patients with heart failure: A nationwide population-based cohort study. Clin. Epidemiol. 2018, 10, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Cai, W.K.; Zhang, Z.; Wang, P.; Lin, X.Q.; Feng, J.; Fu, S.C.; He, G.H. Cardioprotective effect of histamine H2 antagonists in congestive heart failure: A systematic review and meta-analysis. Medicine 2018, 97, e0409. [Google Scholar] [CrossRef] [PubMed]
- Mather, J.F.; Seip, R.L.; McKay, R.G. Impact of famotidine use on clinical outcomes of hospitalized patients with COVID-19. Am. J. Gastroenterol. 2020, 115, 1617–1623. [Google Scholar] [CrossRef]
- Freedberg, D.E.; Conigliaro, J.; Wang, T.C.; Tracey, K.J.; Callahan, M.V.; Abrams, J.A.; Famotidine Research Group. Famotidine use is associated with improved clinical outcomes in hospitalized COVID-19 patients: A propensity score matched retrospective cohort study. Gastroenterology 2020, 159, 1129–1131. [Google Scholar] [CrossRef]
- Janowitz, T.; Gablenz, E.; Pattinson, D.; Wang, T.C.; Conigliaro, J.; Tracey, K.; Tuveson, D. Famotidine use and quantitative symptom tracking for COVID-19 in non-hospitalised patients: A case series. Gut 2020, 69, 1592–1597. [Google Scholar] [CrossRef]
- Lee, H.J.; Lee, H.; Oh, S.H.; Park, J.; Park, S.; Jeon, J.S.; Noh, H.; Han, D.C.; Kwon, S.H. Chronic kidney disease (CKD) patients are exposed to more proton pump inhibitor (PPI)s compared to non-CKD patients. PLoS ONE 2018, 13, e0203878. [Google Scholar] [CrossRef]
- Green, F.W.; Norton, R.A.; Kaplan, M.M. Pharmacology and clinical use of antacids. Am. J. Hosp. Pharm. 1975, 32, 425–429. [Google Scholar]
- Cholin, L.; Ashour, T.; Mehdi, A.; Taliercio, J.J.; Daou, R.; Arrigain, S.; Schold, J.D.; Thomas, G.; Nally, J.; Nakhoul, N.L.; et al. Proton-pump inhibitor vs. H2-receptor blocker use and overall risk of CKD progression. BMC Nephrol. 2021, 22, 264. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.C.; Chen, H.T.; Yeh, C.C.; Hung, S.K.; Yu, B.H. Four prescribed Chinese herbal medicines provide renoprotection and survival benefit without hyperkalemia risk in patients with advanced chronic kidney disease: A nationwide cohort study. Phytomedicine 2022, 95, 153873. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.C.; Su, Y.C.; Li, C.Y.; Wu, C.P.; Lee, M.S. A nationwide cohort study suggests chronic hepatitis B virus infection increases the risk of end-stage renal disease among patients in Taiwan. Kidney Int. 2015, 87, 1030–1038. [Google Scholar] [CrossRef]
- Chen, Y.C.; Lin, H.Y.; Li, C.Y.; Lee, M.S.; Su, Y.C. A nationwide cohort study suggests that hepatitis C virus infection is associated with increased risk of chronic kidney disease. Kidney Int. 2014, 85, 1200–1207. [Google Scholar] [CrossRef]
- Wu, C.Y.; Chen, Y.J.; Ho, H.J.; Hsu, Y.C.; Kuo, K.N.; Wu, M.S.; Lin, J.T. Association between nucleoside analogues and risk of hepatitis B virus-related hepatocellular carcinoma recurrence following liver resection. JAMA 2012, 308, 1906–1914. [Google Scholar] [CrossRef]
- Hsu, Y.C.; Lin, J.T.; Ho, H.J.; Kao, Y.H.; Huang, Y.T.; Hsiao, N.W.; Wu, M.S.; Liu, Y.Y.; Wu, C.Y. Antiviral treatment for hepatitis C virus infection is associated with improved renal and cardiovascular outcomes in diabetic patients. Hepatology 2014, 59, 1293–1302. [Google Scholar] [CrossRef] [PubMed]
- Shariff, S.Z.; Cuerden, M.S.; Jain, A.K.; Garg, A.X. The secret of immortal time bias in epidemiologic studies. J. Am. Soc. Nephrol. 2008, 19, 841–843. [Google Scholar] [CrossRef]
- Gray, R.J. A class of K-sample tests for comparing the cumulative incidenceof a competing risk. Ann. Stat. 1988, 16, 1141–1154. [Google Scholar] [CrossRef]
- WHO Collaborating Center for Drugs Statistics Methodology. ATC Index with DDDs 2003; WHO: Oslo, Norway, 2003. [Google Scholar]
- Peng, Y.C.; Lin, C.L.; Yeh, H.Z.; Chang, C.S.; Wu, Y.L.; Kao, C.H. Association between the use of proton pump inhibitors and the risk of ESRD in renal diseases: A population-based, case-control Study. Medicine 2016, 95, e3363. [Google Scholar] [CrossRef]
- Hsu, T.W.; Liu, J.S.; Hung, S.C.; Kuo, K.L.; Chang, Y.K.; Chen, Y.C.; Hsu, C.C.; Tarng, D.C. Renoprotective effect of renin-angiotensin-aldosterone system blockade in patients with predialysis advanced chronic kidney disease, hypertension, and anemia. JAMA Intern. Med. 2014, 174, 347–354. [Google Scholar] [CrossRef]
- Hsieh, C.Y.; Su, C.C.; Shao, S.C.; Sung, S.F.; Lin, S.J.; Kao Yang, Y.H.; Lai, E.C. Taiwan’s National Health Insurance Research Database: Past and future. Clin. Epidemiol. 2019, 11, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Hattori, M.; Yamazaki, M.; Ohashi, W.; Tanaka, S.; Hattori, K.; Todoroki, K.; Fujimori, T.; Ohtsu, H.; Matsuda, N.; Hattori, Y. Critical role of endogenous histamine in promoting end-organ tissue injury in sepsis. Intensive Care Med. Exp. 2016, 4, 36. [Google Scholar] [CrossRef] [PubMed]
- Imig, J.D.; Ryan, M.J. Immune and inflammatory role in renal disease. Compr. Physiol. 2013, 3, 957–976. [Google Scholar] [CrossRef]
- Andrade-Oliveira, V.; Foresto-Neto, O.; Watanabe, I.K.M.; Zatz, R.; Câmara, N.O.S. Inflammation in Renal Diseases: New and Old Players. Front. Pharmacol. 2019, 10, 1192. [Google Scholar] [CrossRef] [PubMed]
- Estaphan, S.; Eissa, H.; Elattar, S.; Rashed, L.; Farouk, M. A study on the effect of cimetidine and L-carnitine on myoglobinuric acute kidney injury in male rats. Injury 2015, 46, 1223–1230. [Google Scholar] [CrossRef] [PubMed]
- Minai-Tehrani, D.; Khodai, S.; Aminnaseri, S.; Minoui, S.; Sobhani-Damavadifar, Z.; Alavi, S.; Osmani, R.; Ahmadi, S. Inhibition of renal alkaline phosphatase by cimetidine. Drug Metab. Lett. 2011, 5, 197–201. [Google Scholar] [CrossRef]
- Liu, J.; Lai, L.; Lin, J.; Zheng, J.; Nie, X.; Zhu, X.; Xue, J.; Liu, T. Ranitidine and finasteride inhibit the synthesis and release of trimethylamine N-oxide and mitigates its cardiovascular and renal damage through modulating gut microbiota. Int. J. Biol. Sci. 2020, 16, 790–802. [Google Scholar] [CrossRef]
- Vannay, A.; Fekete, A.; Muller, V.; Strehlau, J.; Viklicky, O.; Veres, T.; Reusz, G.; Tulassay, T.; Szabó, A.J. Effects of histamine and the H2 receptor antagonist ranitidine on ischemia-induced acute renal failure: Involvement of IL-6 and vascular endothelial growth factor. Kidney Blood Press. Res. 2004, 27, 105–113. [Google Scholar] [CrossRef]
- Douwes, R.M.; Gomes-Neto, A.W.; Eisenga, M.F.; Loon, E.V.; Schutten, J.C.; Gans, R.O.B.; Naesens, M.; van den Berg, E.; Sprangers, B.; Berger, S.P.; et al. The association between use of proton-pump inhibitors and excess mortality after kidney transplantation: A cohort study. PLoS Med. 2020, 17, e1003140. [Google Scholar] [CrossRef]
- Grant, C.H.; Gillis, K.A.; Lees, J.S.; Traynor, J.P.; Mark, P.B.; Stevens, K.I. Proton pump inhibitor use and progression to major adverse renal events: A competing risk analysis. QJM Int. J. Med. 2019, 112, 835–840. [Google Scholar] [CrossRef]
- Liu, X.; Xu, X.; Shang, R.; Chen, Y. Asymmetric dimethylarginine (ADMA) as an important risk factor for the increased cardiovascular diseases and heart failure in chronic kidney disease. Nitric Oxide 2018, 78, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Ghebremariam, Y.T.; Cooke, J.P.; Khan, F.; Thakker, R.N.; Chang, P.; Shah, N.H.; Nead, K.T.; Leeper, N.J. Proton pump inhibitors and vascular function: A prospective cross-over pilot study. Vasc. Med. 2015, 20, 309–316. [Google Scholar] [CrossRef] [PubMed]
| Overall CKD Patients (n = 102,802) | Propensity-Matched CKD Patients (n = 13,083) | |||||||
|---|---|---|---|---|---|---|---|---|
| PPI Cohort | H2RA Cohort | Control | PPI Cohort | H2RA Cohort | Control | |||
| Variables | (n = 7121) N(%) | (n = 48,609) N(%) | (n = 47,072) N(%) | p-Value | (n = 4361) N (%) | (n = 4361) N (%) | (n = 4361) N (%) | p-Value |
| Sex | <0.0001 | 0.99 | ||||||
| Men | 4524 (63.5) | 23,825 (49.0) | 27,931 (59.3) | 2831 (64.9) | 2831 (64.9) | 2836 (65.0) | ||
| Women | 2597 (36.5) | 24,784 (51.0) | 19,141 (40.7) | 1530 (35.1) | 1530 (35.1) | 1525 (35.0) | ||
| Age (year) | <0.0001 | 1.00 | ||||||
| 18–45 | 1610 (22.6) | 21,029 (43.3) | 18,472 (39.2) | 1391(31.9) | 1386 (31.8) | 1390 (31.9) | ||
| 46–55 | 1325 (18.6) | 10,373 (21.3) | 10,075 (21.4) | 959 (22.0) | 958 (22.0) | 954 (21.9) | ||
| 56–65 | 1334 (18.7) | 7796 (16.1) | 8398 (17.8) | 804 (18.4) | 814 (18.7) | 811 (18.6) | ||
| 66–75 | 1381 (19.4) | 5944 (12.2) | 5631 (12.0) | 670 (15.4) | 665 (15.2) | 666 (15.2) | ||
| >75 | 1471 (20.7) | 3467 (7.1) | 4496 (9.6) | 537 (12.3) | 538 (12.3) | 540 (12.4) | ||
| Mean (SD) | 58.8 ± 17.3 | 48.3 ± 17.2 | 50.4 ± 17.3 | <0.0001 | 53.7 ± 17.2 | 52.8 ± 17.8 | 53.0 ± 17.5 | 0.03 |
| Comorbidities | ||||||||
| Diabetes | 1825 (25.6) | 6969 (14.3) | 9387 (19.9) | <0.0001 | 584 (13.4) | 579 (13.3) | 578 (13.3) | 0.98 |
| Coronary heart disease | 1076 (15.1) | 3926 (8.1) | 3409 (7.2) | <0.0001 | 200 (4.6) | 209 (4.8) | 200 (4.6) | 0.87 |
| Hypertension | 2779 (39.0) | 11,778 (24.3) | 12,821 (27.2) | <0.0001 | 1148 (26.3) | 1145 (26.3) | 1143 (26.2) | 0.99 |
| Acid peptic disease | 555 (7.8) | 3182 (6.6) | 1957 (4.1) | <0.0001 | 86 (2.0) | 93 (2.1) | 82 (1.9) | 0.69 |
| Chronic liver disease | 841 (11.8) | 5188 (10.7) | 4114 (8.7) | <0.0001 | 220 (5.0) | 218 (5.0) | 224 (5.1) | 0.96 |
| No. of medical visits | <0.0001 | 0.99 | ||||||
| 1–11 | 3067 (43.1) | 22,183 (45.7) | 27,460 (58.4) | 2389 (54.8) | 2379 (54.6) | 2390 (54.8) | ||
| 12–23 | 2346 (32.9) | 16,150 (33.2) | 13,295 (28.2) | 1288 (29.5) | 1295 (29.7) | 1297 (29.7) | ||
| >23 | 1708 (24.0) | 10,276 (21.1) | 6317 (13.4) | 684 (15.7) | 687 (15.7) | 674 (15.5) | ||
| Mean (SD) | 17.0 ± 14.5 | 16.0 ± 13.2 | 12.7 ± 11.6 | <0.0001 | 13.6 ± 12.6 | 13.9 ± 12.6 | 13.6 ± 12.9 | 0.30 |
| Confounding drugs | ||||||||
| NSAID | 2620 (36.8) | 11,756 (24.2) | 1220 (2.6) | <0.0001 | 1362 (31.2) | 1209 (27.7) | 85 (2.0) | <0.0001 |
| ACEI/ARB | 2149 (30.2) | 5542 (11.4) | 1251 (2.7) | <0.0001 | 920 (21.1) | 558 (12.8) | 83 (1.9) | <0.0001 |
| Outcome | Crude | Adjusted | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| ESRD * | ||||||
| Control (n = 4361) | 1.00 | Reference | 1.00 | Reference | ||
| PPI cohort (n = 4361) | 2.02 | 1.65–2.48 | <0.0001 | 1.15 | 0.91–1.45 | 0.24 |
| H2RA cohort (n = 4361) | 0.57 | 0.43–0.75 | <0.0001 | 0.40 | 0.30–0.53 | <0.0001 |
| Overall mortality # | ||||||
| Control (n = 4361) | 1.00 | Reference | 1.00 | Reference | ||
| PPI cohort (n = 4361) | 2.54 | 2.31–2.80 | <0.0001 | 1.83 | 1.65–2.03 | <0.0001 |
| H2RA cohort (n = 4361) | 0.98 | 0.88–1.09 | 0.70 | 0.64 | 0.57–0.72 | <0.0001 |
| PPI Cohort (n = 4361) | H2RA Cohort (n = 4361) | Control (n = 4361) | p-Value | |
|---|---|---|---|---|
| ESRD | ||||
| Follow-up (years), mean ± SD | 3.5 ± 3.5 | 4.8 ± 3.7 | 4.5 ± 3.6 | |
| Event number, n (%) | 270 (6.2) | 81 (1.9) | 139 (3.2) | <0.0001 |
| Competing mortality, n (%) | 1155 (26.5) | 575 (13.2) | 534 (12.2) | <0.0001 |
| Cumulative incidence (%) | 8.5 (95% CI, 7.3–9.7) | 2.5 (95% CI, 2.0–3.2) | 4.6 (95% CI, 3.8–5.4) | <0.0001 |
| Overall mortality | ||||
| Follow-up (years), mean ± SD | 3.5 ± 3.5 | 4.8 ± 3.7 | 4.5 ± 3.6 | |
| Event number, n (%) | 1315 (30.2) | 631 (14.5) | 626 (14.4) | <0.0001 |
| Cumulative incidence (%) | 49.7 (95% CI, 43.8–55.3) | 31.5 (95% CI, 27.7–35.3) | 33.9 (95% CI, 24.2–43.8) | <0.0001 |
| ESRD | Overall Mortality | CV Mortality | Non-CV Mortality | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Taking Controls as the Reference | Events (%) | aHR * (95% CI) | p-Value | Events (%) | aHR # (95% CI) | p-Value | Events (%) | aHR # (95% CI) | p-Value | Events (%) | aHR # (95% CI) | p-Value |
| PPI cDDD | ||||||||||||
| <30 (n = 2198) | 157 (7.1) | 1.18 (0.86–1.62) | 0.32 | 889 (40.4) | 2.20 (1.92–2.54) | <0.0001 | 115 (5.2) | 2.01 (1.33–3.06) | 0.001 | 774 (35.2) | 2.22 (1.91–2.58) | <0.0001 |
| ≥30 (n = 2163) | 113 (5.2) | 1.24 (0.87–1.76) | 0.23 | 426 (19.7) | 1.37 (1.16–1.62) | 0.0003 | 42 (1.9) | 1.30 (0.77–2.18) | 0.33 | 384 (17.8) | 1.38 (1.15–1.65) | 0.0005 |
| H2RA cDDD | ||||||||||||
| <10 (n = 2326) | 50 (2.1) | 0.45 (0.30–0.68) | 0.0001 | 335 (14.4) | 1.04 (0.88–1.23) | 0.65 | 65 (2.8) | 1.86 (1.14–3.02) | 0.012 | 270 (11.6) | 0.95 (0.79–1.14) | 0.59 |
| ≥10 (n = 2035) | 31 (1.5) | 0.32 (0.20–0.51) | <0.0001 | 296 (14.5) | 0.50 (0.42–0.60) | <0.0001 | 29 (1.4) | 0.45 (0.27–0.75) | 0.002 | 267 (13.1) | 0.51 (0.43–0.61) | <0.0001 |
| Frequency of Prescriptions | ESRD | Overall Mortality | ||||
|---|---|---|---|---|---|---|
| Event | aHR * (95%CI) | p-Value | Event | aHR # (95%CI) | p-Value | |
| PPI | ||||||
| 0 (n = 4361) | 139 | 1 (reference) | 626 | 1 (reference) | ||
| 1 (n = 1922) | 125 | 1.22 (0.92–0.62) | 0.17 | 687 | 2.51 (2.22–2.83) | <0.0001 |
| 2 (n = 798) | 41 | 1.11 (0.78–1.60) | 0.56 | 197 | 1.61 (1.36–1.90) | <0.0001 |
| 3–4 (n = 838) | 36 | 0.92 (0.63–1.35) | 0.68 | 170 | 1.34 (1.13–1.60) | 0.001 |
| ≥5 (n = 808) | 68 | 1.46 (1.07–1.99) | 0.016 | 261 | 1.34 (1.15–1.56) | 0.0001 |
| H2RA | ||||||
| 0 (n = 4361) | 139 | 1 (reference) | 626 | 1 (reference) | ||
| 1 (n = 1595) | 38 | 0.52 (0.36–0.77) | 0.0011 | 258 | 1.25 (1.07–1.47) | 0.004 |
| 2 (n = 742) | 8 | 0.24 (0.12–0.50) | 0.0001 | 93 | 0.77 (0.62–0.96) | 0.02 |
| 3–4 (n = 771) | 18 | 0.47 (0.28–0.79) | 0.004 | 91 | 0.56 (0.45–0.70) | <0.0001 |
| ≥5 (n = 1253) | 17 | 0.28 (0.16–0.46) | <0.0001 | 189 | 0.44 (0.37–0.52) | <0.0001 |
| ESRD | Overall Mortality | ||||||
|---|---|---|---|---|---|---|---|
| N | Crude HR (95% CI) | Adjusted HR * (95% CI) | N | Crude HR (95% CI) | Adjusted HR # (95% CI) | ||
| Follow-up >30 days | Control | 4288 | 1.00 (Reference) | 1.00 (Reference) | 4292 | 1.00 (Reference) | 1.00 (Reference) |
| PPI | 3873 | 1.69 (1.35–2.11) | 1.14 (0.89–1.47) | 3914 | 1.95 (1.76–2.17) | 1.66 (1.48–1.85) | |
| H2RA | 4227 | 0.54 (0.40–0.72) | 0.40 (0.30–0.54) | 4235 | 0.87 (0.78–0.98) | 0.62 (0.55–0.70) | |
| Follow-up >60 days | Control | 4217 | 1.00 (Reference) | 1.00 (Reference) | 4226 | 1.00 (Reference) | 1.00 (Reference) |
| PPI | 3664 | 1.53 (1.21–1.93) | 1.11 (0.85–1.44) | 3717 | 1.73 (1.56–1.93) | 1.57 (1.40–1.76) | |
| H2RA | 4166 | 0.51 (0.38–0.69) | 0.39 (0.28–0.53) | 4176 | 0.86 (0.76–0.97) | 0.64 (0.57–0.73) | |
| Follow-up >90 days | Control | 4132 | 1.00 (Reference) | 1.00 (Reference) | 4141 | 1.00 (Reference) | 1.00 (Reference) |
| PPI | 3525 | 1.39 (1.09–1.78) | 1.06 (0.81–1.38) | 3586 | 1.66 (1.48–1.85) | 1.56 (1.39–1.76) | |
| H2RA | 4090 | 0.50 (0.36–0.67) | 0.38 (0.28–0.52) | 4101 | 0.85 (0.76–0.96) | 0.65 (0.57–0.74) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.-C.; Chen, Y.-C.; Chiou, W.-Y.; Yu, B.-H. Impact of Acid Suppression Therapy on Renal and Survival Outcomes in Patients with Chronic Kidney Disease: A Taiwanese Nationwide Cohort Study. J. Clin. Med. 2022, 11, 5612. https://doi.org/10.3390/jcm11195612
Chen Y-C, Chen Y-C, Chiou W-Y, Yu B-H. Impact of Acid Suppression Therapy on Renal and Survival Outcomes in Patients with Chronic Kidney Disease: A Taiwanese Nationwide Cohort Study. Journal of Clinical Medicine. 2022; 11(19):5612. https://doi.org/10.3390/jcm11195612
Chicago/Turabian StyleChen, Yi-Chun, Yen-Chun Chen, Wen-Yen Chiou, and Ben-Hui Yu. 2022. "Impact of Acid Suppression Therapy on Renal and Survival Outcomes in Patients with Chronic Kidney Disease: A Taiwanese Nationwide Cohort Study" Journal of Clinical Medicine 11, no. 19: 5612. https://doi.org/10.3390/jcm11195612
APA StyleChen, Y.-C., Chen, Y.-C., Chiou, W.-Y., & Yu, B.-H. (2022). Impact of Acid Suppression Therapy on Renal and Survival Outcomes in Patients with Chronic Kidney Disease: A Taiwanese Nationwide Cohort Study. Journal of Clinical Medicine, 11(19), 5612. https://doi.org/10.3390/jcm11195612





