Multidimensional 3-Month Follow-Up of Severe COVID-19: Airways beyond the Parenchyma in Symptomatic Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. CT Acquisitions and Image Analysis
2.3. Statistical Analysis
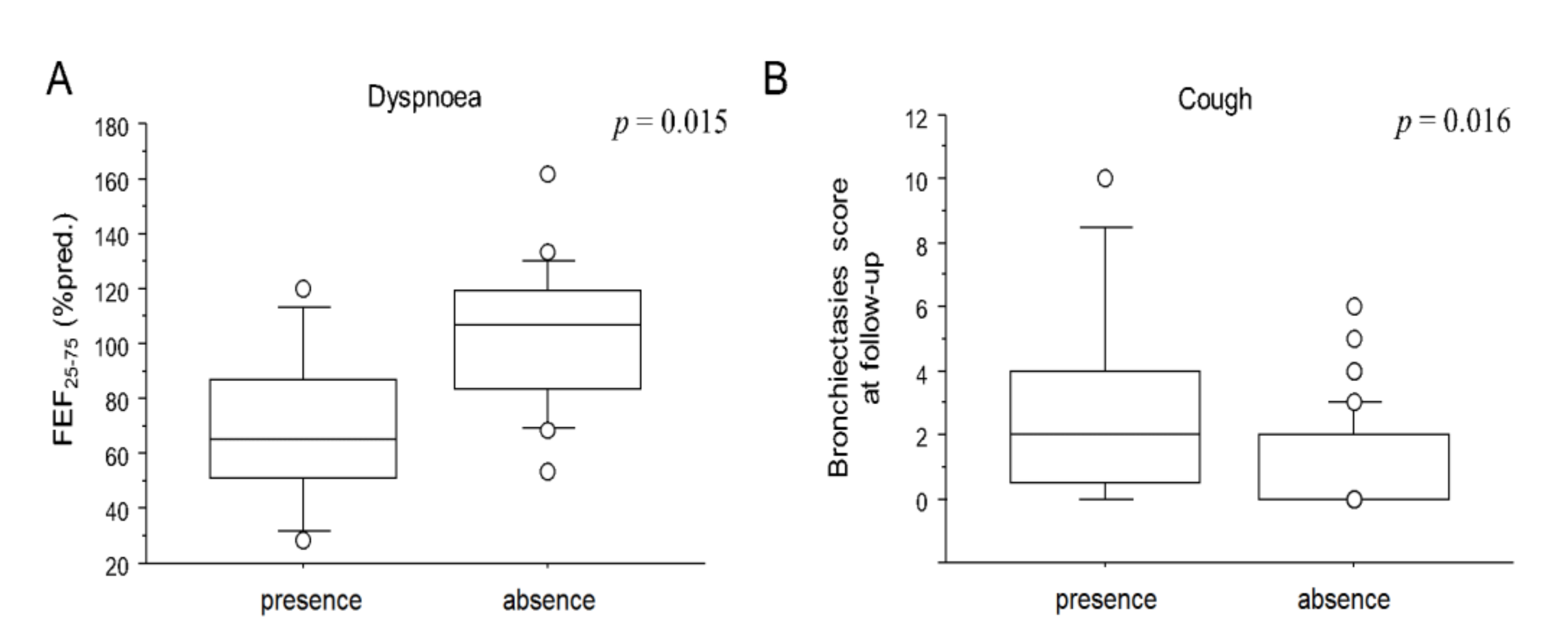
3. Results
3.1. Clinical-Radiological Characteristics at Hospital Admission and Hospitalization
3.2. Clinical-Functional-Radiological Characteristics at Follow-Up
3.3. Longitudinal Functional-Radiological Evolution from Hospitalization to Follow-Up
3.3.1. Evolution of Acute CT Abnormalities
3.3.2. Development of Chronic CT Abnormalities
3.3.3. Development of Functional Impairment
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ABG | Arterial blood gas analysis |
| ARDS | Acute respiratory distress syndrome |
| CON | Consolidation |
| CP | Crazy paving |
| CRP | C-reactive protein |
| DLCO | Diffusion of carbon monoxide |
| FIB | Fibrotic-like alterations |
| GG | Ground glass |
| HFNC | Heated humidified high-flow oxygen therapy |
| CT | Chest computed tomography |
| ICU | Intensive care unit |
| IQR | Interquartile range |
| IV | Invasive ventilation |
| FEV1 | Forced expired volume in the first second of expiration |
| FiO2 | Fraction of inspired oxygen |
| FVC | Forced vital capacity |
| K–W | Kruskal–Wallis test |
| LDH | Lactate dehydrogenase |
| mMRC | Modified medical research council |
| PaO2 | Arterial partial pressure of oxygen |
| P/F | PaO2/FiO2 ratio |
| NIV | Non-invasive ventilation |
| NV | Normal values |
| O2T | Oxygen therapy |
| RV | Residual volume |
| SD | Standard deviation |
| ALA | Acute lung alterations |
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 |
| TLC | Total lung capacity |
References
- Garg, M.; Gupta, P.; Maralakunte, M.; Praveen, K.-M.; Sinha, A.; Kang, M.; Sandhu, M.S. Diagnostic accuracy of CT and radiographic findings for novel coronavirus 2019 pneumonia: Systematic review and meta-analysis. Clin. Imaging 2021, 72, 75–82. [Google Scholar] [PubMed]
- Wei, J.; Yang, H.; Lei, P.; Fan, B.; Qiu, Y.; Zeng, B.; Yu, P.; Lv, J.; Jian, Y.; Wan, C. Analysis of thin-section CT in patients with coronavirus disease (COVID-19) after hospital discharge. J. X-ray Sci. Technol. 2020, 28, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.M.; Shang, Y.M.; Song, W.B.; Li, Q.Q.; Xie, H.; Xu, Q.F.; Jia, J.L.; Li, L.M.; Mao, H.L.; Zhou, X.M.; et al. Follow-up study of the pulmonary function and related physiological characteristics of COVID-19 survivors three months after recovery. EClinicalMedicine 2020, 25, 100463. [Google Scholar] [PubMed]
- Hui, D.S.; Joynt, G.M.; Wong, K.T.; Gomersall, C.D.; Li, T.S.; Antonio, G.; Ko, F.W.; Chan, M.C.; Chan, D.P.; Tong, M.W.; et al. Impact of severe acute respiratory syndrome (SARS) on pulmonary function, functional capacity and quality of life in a cohort of survivors. Thorax 2005, 60, 401–409. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hui, D.S.; Wong, K.T.; Ko, F.W.; Tam, L.S.; Chan, D.P.; Woo, J.; Sung, J.J. The 1-year impact of severe acute respiratory syndrome on pulmonary function, exercise capacity, and quality of life in a cohort of survivors. Chest 2005, 128, 2247–2261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Das, K.M.; Lee, E.Y.; Singh, R.; Enani, M.A.; Al Dossari, K.; Van Gorkom, K.; Larsson, S.G.; Langer, R.D. Follow-up chest radiographic findings in patients with MERS-CoV after recovery. Indian J. Radiol. Imaging 2017, 27, 342–349. [Google Scholar] [CrossRef]
- Available online: https://www.aifa.gov.it/aggiornamento-sui-farmaci-utilizzabili-per-il-trattamento-della-malattia-covid19 (accessed on 22 May 2022).
- Available online: https://www.flipsnack.com/SIAARTI/siaarti_raccomandazioni_per_la_gestione_del_paziente_criti/full-view.html (accessed on 22 May 2022).
- Stanojevic, S.; Kaminsky, D.A.; Miller, M.; Thompson, B.; Aliverti, A.; Barjaktarevic, I.; Cooper, B.G.; Culver, B.; Derom, E.; Hall, G.L.; et al. ERS/ATS technical standard on interpretive strategies for routine lung function tests. Eur. Respir. J. 2021, 23, 2101499. [Google Scholar] [CrossRef]
- Salahshour, F.; Mehrabinejad, M.M.; Nassiri Toosi, M.; Gity, M.; Ghanaati, H.; Shakiba, M.; Kolahi, S. Clinical and chest CT features as a predictive tool for COVID-19 clinical progress: Introducing a novel semi-quantitative scoring system. Eur. Radiol. 2021, 31, 5178–5188. [Google Scholar] [CrossRef]
- Brody, A.S.; Klein, J.S.; Molina, P.L.; Quan, J.; Bean, J.A.; Wilmott, R.W. High-resolution computed tomography in young patients with cystic fibrosis: Distribution of abnormalities and correlation with pulmonary function tests. J. Pediatr. 2004, 145, 32–38. [Google Scholar] [CrossRef]
- Hansell, D.M.; Bankier, A.A.; MacMahon, H.; McLoud, T.C.; Müller, N.L.; Remy, J. Fleischner Society: Glossary of terms for thoracic imaging. Radiology 2008, 246, 697–722. [Google Scholar] [CrossRef] [Green Version]
- Froidure, A.; Mahsouli, A.; Liistro, G.; De Greef, J.; Belkhir, L.; Gérard, L.; Bertrand, A.; Koenig, S.; Pothen, L.; Yildiz, H.; et al. Integrative respiratory follow-up of severe COVID-19 reveals common functional and lung imaging sequelae. Respir. Med. 2021, 181, 106383. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Liu, X.; Zhou, Y.; Yu, H.; Li, R.; Zhan, Q.; Ni, F.; Fang, S.; Lu, Y.; Ding, X.; et al. 3-month, 6-month, 9-month, and 12-month respiratory outcomes in patients following COVID-19-related hospitalisation: A prospective study. Lancet Respir. Med. 2021, 9, 747–754. [Google Scholar] [CrossRef]
- Cocconcelli, E.; Bernardinello, N.; Giraudo, C.; Castelli, G.; Giorgino, A.; Leoni, D.; Petrarulo, S.; Ferrari, A.; Saetta, M.; Cattelan, A.; et al. Characteristics and Prognostic Factors of Pulmonary Fibrosis after COVID-19 Pneumonia. Front. Med. 2022, 8, 823600. [Google Scholar] [CrossRef]
- Franquet, T.; Giménez, A.; Ketai, L.; Mazzini, S.; Rial, A.; Pomar, V.; Domingo, P. Air trapping in COVID-19 patients following hospital discharge: Retrospective evaluation with paired inspiratory/expiratory thin-section CT. Eur. Radiol. 2022, 32, 4427–4436. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Zhu, J.; Zhou, J.; Shang, Y.; Lin, X.; Gong, S.; Gu, L.; Dai, H.; Li, Y. Inspiratory and Expiratory Chest High-resolution CT: Small-airway Disease Evaluation in Patients with COVID-19. Curr. Med. Imaging 2021, 17, 1299–1307. [Google Scholar] [CrossRef]
- Choi, H.; Lee, H.; Lee, S.K.; Yang, B.; Chung, S.J.; Yeo, Y.; Park, T.S.; Park, D.W.; Moon, J.Y.; Kim, T.H.; et al. Impact of bronchiectasis on susceptibility to and severity of COVID-19: A nationwide cohort study. Adv. Respir. Dis. 2021, 15, 1753466621995043. [Google Scholar] [CrossRef]
- Baratella, E.; Ruaro, B.; Marrocchio, C.; Starvaggi, N.; Salton, F.; Giudici, F.; Quaia, E.; Confalonieri, M.; Cova, M.A. Interstitial Lung Disease at High Resolution CT after SARS-CoV-2-Related Acute Respiratory Distress Syndrome According to Pulmonary Segmental Anatomy. J. Clin. Med. 2021, 10, 3985. [Google Scholar] [CrossRef]
- Sonnweber, T.; Sahanic, S.; Pizzini, A.; Luger, A.; Schwabl, C.; Sonnweber, B.; Kurz, K.; Koppelstätter, S.; Haschka, D.; Petzer, V.; et al. Cardiopulmonary recovery after COVID-19: An observational prospective multicentre trial. Eur. Respir. J. 2021, 57, 2003481. [Google Scholar] [CrossRef]
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef]
- Guler, S.A.; Ebner, L.; Aubry-Beigelman, C.; Bridevaux, P.O.; Brutsche, M.; Clarenbach, C.; Garzoni, C.; Geiser, T.K.; Lenoir, A.; Mancinetti, M.; et al. Pulmonary function and radiological features 4 months after COVID-19: First results from the national prospective observational Swiss COVID-19 lung study. Eur. Respir. J. 2021, 57, 2003690. [Google Scholar] [CrossRef]
- Hama Amin, B.J.; Kakamad, F.H.; Ahmed, G.S.; Ahmed, S.F.; Abdulla, B.A.; Mohammed, S.H.; Mikael, T.M.; Salih, R.Q.; Ali, R.K.; Salh, A.M.; et al. Post COVID-19 pulmonary fibrosis; A meta-analysis study. Ann. Med. Surg. 2022, 77, 103590. [Google Scholar] [CrossRef] [PubMed]
- Kyung Soo, L.; Yu, M.W. Residual Lung Lesions at 1-year CT after COVID-19. Radiology 2022, 302, 720–721. [Google Scholar]
- Garg, M.; Maralakunte, M.; Dhooria, S. Sequelae of COVID-19 pneumonia: Is it correct to label everything as post-COVID lung fibrosis? J. Postgrad. Med. 2021, 67, 224–227. [Google Scholar] [CrossRef] [PubMed]


| Subjects, n | 62 |
| Gender (male/female), n (%) | 45 (72.5)/17 (27.5) |
| Age | 68.5 ± 9.5 |
| Smoke habit, current/former/never, n (%) | 2 (3.2)/24 (38.7)/36 (58.1) |
| Comorbidities, n (%) | |
| Hypertension | 36 (58) |
| Dyslipidemia | 19 (30.6) |
| Diabetes | 17 (27.4) |
| Obesity | 11 (17.7) |
| Cardiopathy | 12 (19.3) |
| Active malignancy | 2 (3.2) |
| Chronic kidney disease | 2 (3.2) |
| P/F, ratio | 213 (143–285) |
| CRP, mg/L | 9.3 (4.3–14.3) (nv < 1) |
| Lactate, mmol/L | 1.3 (0.9–1.9) (nv < 2.3) |
| LDH, U/L | 369 (312–451) (nv < 280) |
| D-dimer, ng/mL | 813 (476–1288) (nv < 300) |
| Blood lymphocytes, cell/mcl | 830 (646–1080) (nv 1000–4800) |
| Blood neutrophyls, cell/mcl | 6540 (4310–8852) (nv 1500–8000) |
| Respiratory support, n (%) | |
| 11 (17.7) |
| 9 (14.5) |
| 25 (40.3) |
| 17 (27.5) |
| Complications during hospitaization, n (%) | |
| 5 (8.0) |
| 2 (3.2) |
| 6 (9.6) |
| 3 (4.8) |
| 2 (3.2) |
| 5 (8.0) |
| 1 (1.6) |
| 4 (6.4) |
| 12 (19.3) |
| 4 (6.4) |
| 2 (3.2) |
| 0 (0%) |
| 5 (8.0) |
| 2 (3.2) |
| 30 (48.3) |
| Steroid treatment, n (%) | 51 (83.6) |
| 39 (76.4) |
| 12 (23.6) |
| Tocilizumab treatment, n (%) | 6 (9.6) |
| Antiviral treatment, n (%) | 24 (38.7) |
| 18 (75) |
| 6 (25) |
| Length of hospitalization, days | 15 (11–23) |
| Length of invasive ventilation, days | 8 (4.7–10) |
| CTH | CTFU | ΔFU-H | p-Value | |
|---|---|---|---|---|
| Total acute lung alterations (ALA) | 13.6 ± 5.5 | 7.1 ± 5.7 | −6.4 ± 6.3 | <0.0001 |
| Ground glass (GG) | 10.4 ± 4.6 | 6.9 ± 5.4 | −3.5 ± 5.9 | 0.0002 |
| Crazy paving (CP) | 3.7 ± 3.6 | 0.9 ± 2.2 | −2.2 ± 3.5 | <0.0001 |
| Consolidation (CON) | 5.9 ± 3.9 | 0.3 ± 1.1 | −5.6 ± 3.9 | <0.0001 |
| Fibrotic-like alterations (FIB) | 2.7 ± 2.4 | 4.2 ± 3.6 | +1.6 ± 3.3 | 0.0003 |
| Bronchiectasis (BRN) | 1.2 ± 2.1 | 1.4 ± 2.0 | +0.2 ± 1.8 | n.s. |
| No Symptoms, n (%) | 35 (56.4) |
| 17 (27.4) |
| 8 (12.9) |
| 6 (9.6) |
| 2 (3.2) |
| 0 |
| mMRC | 0 (0−1) |
| Respiratory failure, n (%) | 0 (0) |
| Pulmonary function test performed, n (%) | 50 (78.1) |
| Obstructive disease, n (%) | 4 (8) |
| Restrictive disease, n (%) | 7 (14) |
| Mixed disease, n (%) | 0 (0) |
| FEV1/FVC, %pred. | 79 ± 6.9 (nv > 70) |
| FEV1 %pred. | 109 ± 15.6 (nv > 80) |
| FVC %pred. | 109 ± 16.6 (nv > 80) |
| FEF 25–75%pred. | 94 ± 28.7 (nv > 80) |
| TLC %pred. | 94 ± 16.3 (nv 80–120) |
| RV %pred. | 75 ± 30.8 (nv > 80) |
| DLCO %pred. | 70 ± 14.3 (nv > 80) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonato, M.; Peditto, P.; Landini, N.; Fraccaro, A.; Catino, C.; Cuzzola, M.; Malacchini, N.; Savoia, F.; Roma, N.; Salasnich, M.; et al. Multidimensional 3-Month Follow-Up of Severe COVID-19: Airways beyond the Parenchyma in Symptomatic Patients. J. Clin. Med. 2022, 11, 4046. https://doi.org/10.3390/jcm11144046
Bonato M, Peditto P, Landini N, Fraccaro A, Catino C, Cuzzola M, Malacchini N, Savoia F, Roma N, Salasnich M, et al. Multidimensional 3-Month Follow-Up of Severe COVID-19: Airways beyond the Parenchyma in Symptomatic Patients. Journal of Clinical Medicine. 2022; 11(14):4046. https://doi.org/10.3390/jcm11144046
Chicago/Turabian StyleBonato, Matteo, Piera Peditto, Nicholas Landini, Alessia Fraccaro, Cosimo Catino, Maria Cuzzola, Nicola Malacchini, Francesca Savoia, Nicola Roma, Mauro Salasnich, and et al. 2022. "Multidimensional 3-Month Follow-Up of Severe COVID-19: Airways beyond the Parenchyma in Symptomatic Patients" Journal of Clinical Medicine 11, no. 14: 4046. https://doi.org/10.3390/jcm11144046
APA StyleBonato, M., Peditto, P., Landini, N., Fraccaro, A., Catino, C., Cuzzola, M., Malacchini, N., Savoia, F., Roma, N., Salasnich, M., Turrin, M., Zampieri, F., Zanardi, G., Zeraj, F., Rattazzi, M., Peta, M., Baraldo, S., Saetta, M., Fusaro, M., ... Romagnoli, M. (2022). Multidimensional 3-Month Follow-Up of Severe COVID-19: Airways beyond the Parenchyma in Symptomatic Patients. Journal of Clinical Medicine, 11(14), 4046. https://doi.org/10.3390/jcm11144046






