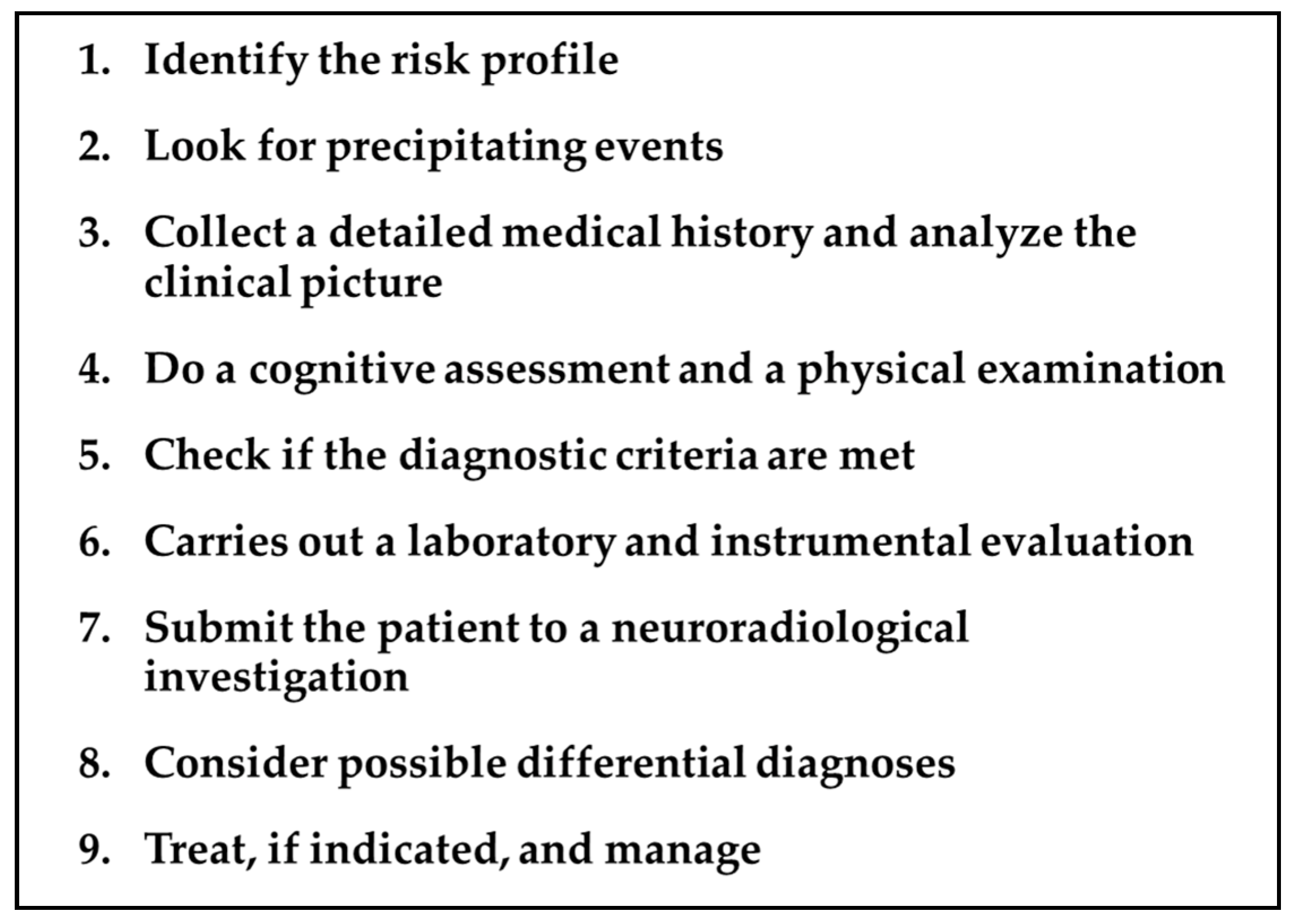
Forgetting the Unforgettable: Transient Global Amnesia Part II: A Clinical Road Map
Abstract
1. Introduction
2. TGA Roadmap
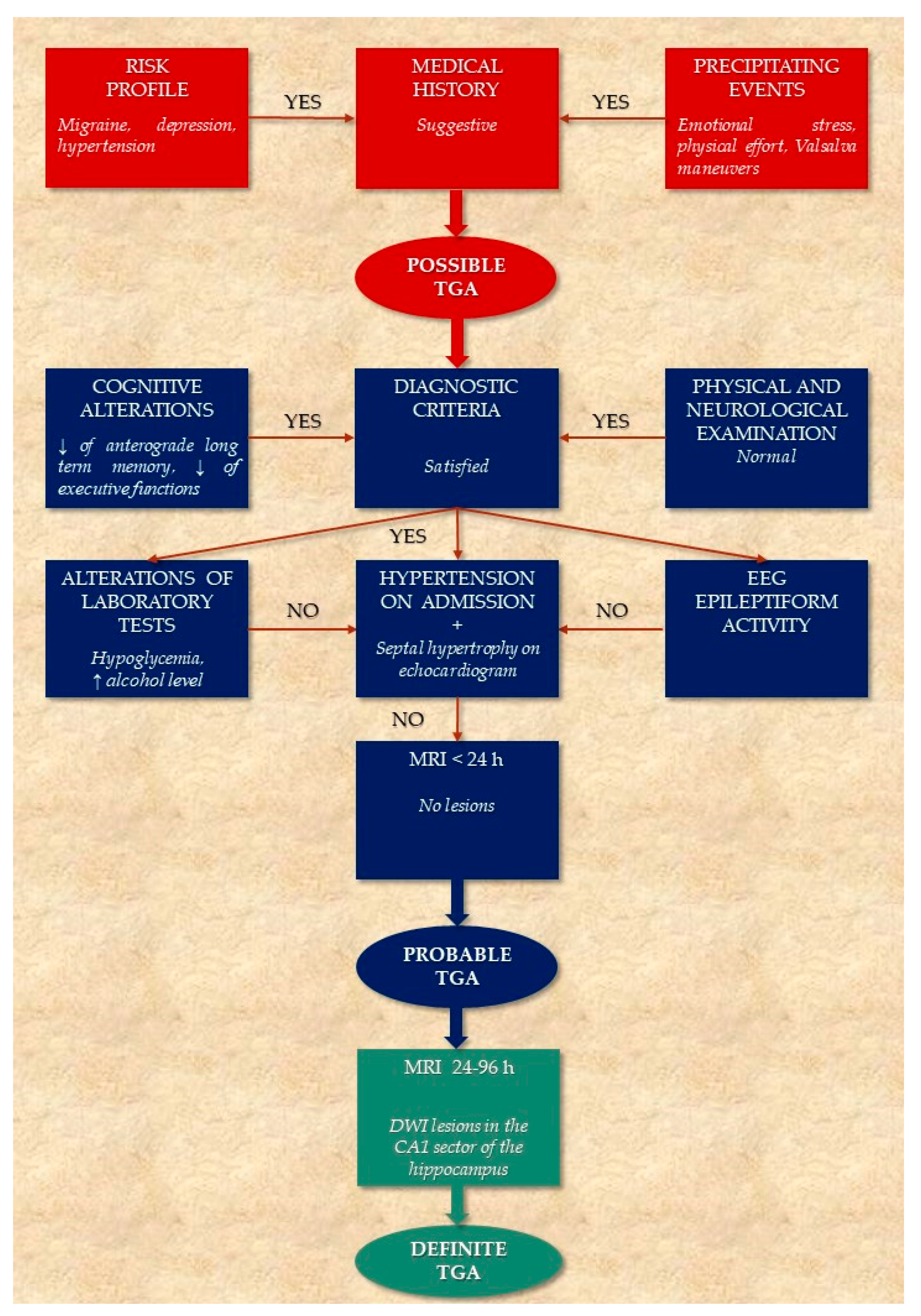
2.1. Risk Profile
- Migraine history. In 2014 a large nationwide, population-based cohort study, enrolling 158.301 migraine patients and 158.301 healthy controls (HC), demonstrated that migraines are associated with an increased risk of TGA (incidence rate ratio =2.48, p = 0.002), particularly in female patients aged 40–60 years [17]. Noteworthy, in the same study, the subjects with a history of migraines had a significantly younger age of TGA onset (56.6 years) compared to the control group (61.4 years), suggesting that migraines could lead to an earlier age of disease onset [17]. In a recent analysis of the data obtained from the Nationwide Inpatient Sample, which represents 20% of the US community hospitals for the years 1999–2008, patients with a diagnosis of migraines had 5.98 times greater odds of having TGA compared with patients without migraines [18]. In a more recent systematic review and meta-analysis, it was confirmed that there is a higher relative risk (RR) of TGA for migraine vs. non-migraine individuals [RR = 2.48, 95% confidence-interval (95% CI) = (1.32, 4.87)] [19].
- Psychiatric comorbidity. Epidemiological studies suggest that some personality traits might be relevant to the etiology of the disease [7,13,20,21]. Pantoni et al. found that TGA patients had a significantly higher percentage of depression or anxiety disorder, as well as phobic traits in comparison with patients who have had a transient ischemic attack (TIA) or with HC [13]. Additionally, a significantly higher percentage of TGA subjects (33.3%) reported a family history of psychiatric disease as compared with TIA subjects (13.7%) [13]. Other authors have found an increased frequency of psychological or emotional instability and a tendency to feel guilty among patients experiencing TGA events [12,22].
- Vascular risk profile. A retrospective case–control study comparing 293 TGA patients to 632 patients with TIA and 293 age- and sex-matched HC showed a significantly higher prevalence of hyperlipidemia and ischemic heart disease in TGA patients when compared to TIA patients or HC [23]. Conversely, diabetes mellitus was associated with a significantly reduced occurrence of TGA [23]. In a systematic review of observational studies examining the relationship between the conventional cardiovascular risk factors and TGA, there was evidence of a potential association between severe hypertension (defined according to a 160/95 mmHg cut-off) and TGA [24]. Diabetes mellitus (stronger evidence) and current smoking (limited evidence) were found to exert a protective effect [24]. Furthermore, the role of hypertension in TGA was extensively evaluated in a recent analysis that compared the cardiovascular risk profile of 277 patients with TGA to 216 patients with acute ischemic stroke [25]. In this study, patients with TGA had significantly higher systolic and diastolic blood pressure at admission than stroke patients, but lower signs of chronic hypertension, as measured by the extent of cerebral microangiopathy and degree of septal hypertrophy in transthoracic echocardiography [25].
2.2. Precipitating Events
- Emotional stress, (i.e., triggered by medical procedures, interpersonal conflict, birth/death announcement, and difficult/exhausting workday);
- Physical effort, (i.e., gardening, housework, and sawing wood);
- Acute pain;
- Water contact/temperature change, (i.e., hot bath/shower and cold swim);
- Sexual intercourse;
- Valsalva-associated maneuvers;
2.3. Clinical Picture
2.4. Cognitive Evaluation and Physical Examination during TGA
2.4.1. Main Cognitive Alterations
- Reduction in anterograde episodic long-term memory
- Partial loss of retrograde episodic long-term memory
- Reduction of executive function
2.4.2. Preserved Cognitive Functions in TGA
- Short-term memory
- Semantic memory
- Implicit and procedural memory
2.5. Diagnostic Criteria
2.6. Laboratory Tests and Instrumental Evaluation
2.6.1. Electroencephalography (EEG)
2.6.2. Transthoracic Echocardiography
2.7. Neuroimaging
2.7.1. Magnetic Resonance Imaging (MRI)
- Lesions can be single or multiple and vary in size from 1 to 5 mm [7].
- In a recent meta-analysis of 1732 patients with TGA the pooled incidence of right, left, and bilateral hippocampal lesions were 37% (95% CI, 29–44%), 42% (95% CI, 39–46%), and 25% (95% CI, 20–30%), respectively [9].
- In the same study, DWI with a slice thickness ≤3 mm showed a higher diagnostic yield than DWI with a slice thickness >3 mm [63% (95% CI, 53–72%) vs. 26% (95% CI, 16–40%), p < 0.01] and there was no significant difference in the diagnostic yield between 3 T and 1.5 T imaging [pooled diagnostic yield, 31% (95% CI, 25–38%) vs. 24% (95% CI, 14–37%), p = 0.31)] [9].
- Focal hippocampal DWI lesions generally resolve 7–10 days after onset of TGA, with no long-term structural changes [58]. This complete reversibility of DWI hippocampal hyperintensity without structural sequelae, as confirmed by the lack of persistent signal change on T2-weighted or FLAIR sequences, does not conform to the time course of classic ischemic lesions [9].
- T2-weighted and FLAIR sequences allow us to identify and evaluate the extent of cerebral microangiopathy in order to provide a measure of the presence and degree of chronic hypertension [25]. In this way, these sequences can provide useful indications for a more rigorous antihypertensive drug treatment in patients with chronic hypertension and can help to calculate the risk of subsequent TGA recurrence in patients without microangiopathic alterations (for details see paragraph 3.1 of this review).
- Complementary imaging studies combining MRI and focal MR spectroscopy (MRS) of CA1 DWI/T2 lesions revealed a transient lactate peak without changes of N-acetyl-aspartate (NAA) and creatine (Cr), indicating acute metabolic stress of CA-1 neurons during TGA [58]. The lactate peak was detected only in the DWI lesion and not in the perifocal tissue, suggesting that the metabolic changes in CA1 neurons were highly focal and not suggestive of a globally altered metabolic status in the hippocampus [58] (see Part I of this review for the pathogenetic implications of these neuroradiological findings).
2.7.2. Positron Emission Tomography (PET) and Single-Photon Emission Computed Tomography (SPECT)
- Many studies noted concomitant (decreased or increased) changes in cerebral blood flow in other anatomical structures, such as unilateral or bilateral thalamic, prefrontal, frontal, amygdalin, striatal, cerebellar, occipital, precentral, and postcentral areas [7,47,59,60,61,62,71,72,73,74,75,76,77,78].
2.8. Differential Diagnosis
2.8.1. Transient Epileptic Amnesia (TEA)
- In the acute phase of the seizure, the majority of patients have normal MRI findings. However, in those with detectable brain signal abnormality on the MRI, there may be diffuse signal abnormality and swelling in the hippocampi [5,93]. Additional signal abnormalities can sometimes be seen in the pulvinar and/or cortex [93].
- The response to antiepileptic therapy is clear [94].
2.8.2. Ischemic or Hypoxic Events
- At the onset of symptoms, brain MRI DWI and FLAIR imaging show acute changes in ischemic events while the same MRI sequences are usually negative in TGA [4].
2.8.3. Migraine
2.8.4. Dissociative Amnesia (DA)
2.8.5. Toxic Amnesia
2.9. Management and Treatment
- Diagnosis and management of TGA should be performed by an interprofessional team, at least composed of a neurologist, internist, radiologist, and nurse practitioner, in order to properly address the large number of differential diagnoses that need to be considered when patients present with acute amnestic syndromes.
- The patient should be examined carefully for any accompanying neurological deficits or evidence of head trauma, both of which would exclude TGA as the diagnosis.
- Since ischemic stroke treatment is a time-dependent, sometimes invasive procedure, it is extremely important in an emergency setting to rule out ischemic stroke as a cause of amnesia to avoid subjecting the patient to unnecessary or even dangerous treatments. The early use of MRI and the detection of positive DWI lesions in the first hours after the onset of symptoms is essential to recognize the ischemic origin of the event. Further investigations, including extracranial and transcranial arterial echo-color Doppler sonography as well as ultrasound examination of the extracranial venous system, can be recommended if cerebrovascular risk factors are present and the patient is younger than 50 years [7].
- If the patient has repetitive amnestic episodes, EEG is mandatory to exclude a TEA.
- Transthoracic echocardiography may be indicated to evaluate septal hypertrophy, a marker of chronic hypertension, in TGA patients with elevated blood pressure on admission.
- The patient should be observed in the hospital until the memory deficit resolves.
- It seems prudent to avoid any activity that could raise intrathoracic venous pressure until the amnesia is resolved [3].
- Since TGA is a very stressful condition, it is necessary to guarantee psychological support not only for the patient but also for the family members, who also may require reassurance [32].
- Clinicians need to reassure the caregiver and patient about the benign nature of the disorder. TGA is a self-limiting condition that resolves spontaneously and rarely recurs.
- To date, there are no established, evidence-based treatments to prevent TGA recurrence. However, the recent demonstration of a strong association between acute hypertensive peaks and TGA in patients not adapted to chronic hypertension, i.e., without microangiopathy and septal hypertrophy, suggests that avoiding blood pressure peaks would be advisable [25]. In this regard, it is worth mentioning a case report of successful prophylaxis of recurrent coital TGA with accurate blood pressure control [121].
- Possible acute treatments or secondary prevention are indicated if an alternative diagnosis, (i.e., seizures or ischemic stroke/TIA) is made.
- Patients with risks of stroke or major cardiovascular events should be treated according to primary prevention guidelines.
- Patients should undergo periodic instrumental checks, (e.g., EEG, cardiological examinations, etc.), but do not need any restrictions in daily activities once the memory deficits have been resolved.
3. Long-Term Outcome
3.1. Recurrence Rate for TGA
3.2. Cognitive Profile of TGA Patients after the Acute Phase
3.3. Long-Term Risk of Developing a Cognitive Decline
3.4. Risk of Ischemic Stroke
3.5. Risk of Epilepsy
4. Diagnostic Flow Chart
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hodges, J.R.; Warlow, C.P. Syndromes of transient amnesia: Towards a classification. A study of 153 cases. J. Neurol. Neurosurg. Psychiatry 1990, 53, 834–843. [Google Scholar] [CrossRef] [PubMed]
- Hodges, J.R.; Warlow, C.P. The aetiology of transient global amnesia. A case-control study of 114 cases with prospective follow-up. Brain 1990, 113, 639–657. [Google Scholar] [CrossRef] [PubMed]
- Arena, J.E.; Rabinstein, A.A. Transient Global Amnesia. Mayo Clin. Proc. 2015, 90, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, D.R.; Smith, J.; Wade, R.R.; Cherukuru, N.; Ursani, A.; Dobruskina, Y.; Crist, T.; Busch, R.F.; Dhanani, R.M.; Dreyer, N. Transient global amnesia: Current perspectives. Neuropsychiatr. Dis. Treat. 2017, 13, 2691–2703. [Google Scholar] [CrossRef] [PubMed]
- Alessandro, L.; Ricciardi, M.; Chaves, H.; Allegri, R.F. Acute amnestic syndromes. J. Neurol. Sci. 2020, 413, 116781. [Google Scholar] [CrossRef]
- Bartsch, T.; Alfke, K.; Stingele, R.; Rohr, A.; Freitag-Wolf, S.; Jansen, O.; Deuschl, G. Selective affection of hippocampal CA-1 neurons in patients with transient global amnesia without long-term sequelae. Brain 2006, 129, 2874–2884. [Google Scholar] [CrossRef]
- Bartsch, T.; Deuschl, G. Transient global amnesia: Functional anatomy and clinical implications. Lancet Neurol. 2010, 9, 205–214. [Google Scholar] [CrossRef]
- Sedlaczek, O.; Hirsch, J.G.; Grips, E.; Peters, C.N.; Gass, A.; Wohrle, J.; Hennerici, M. Detection of delayed focal MR changes in the lateral hippocampus in transient global amnesia. Neurology 2004, 62, 2165–2170. [Google Scholar] [CrossRef]
- Lim, S.J.; Kim, M.; Suh, C.H.; Kim, S.Y.; Shim, W.H.; Kim, S.J. Diagnostic Yield of Diffusion-Weighted Brain Magnetic Resonance Imaging in Patients with Transient Global Amnesia: A Systematic Review and Meta-Analysis. Korean J. Radiol. 2021, 22, 1680–1689. [Google Scholar] [CrossRef]
- Berli, R.; Hutter, A.; Waespe, W.; Bachli, E.B. Transient global amnesia—Not so rare after all. Swiss Med. Wkly. 2009, 139, 288–292. [Google Scholar]
- Miller, J.W.; Yanagihara, T.; Petersen, R.C.; Klass, D.W. Transient Global Amnesia and Epilepsy.Electroencephalographic distinction. Arch. Neurol. 1987, 44, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Quinette, P.; Guillery-Girard, B.; Dayan, J.; De La Sayette, V.; Marquis, S.; Viader, F.; Desgranges, B.; Eustache, F. What does transient global amnesia really mean? Review of the literature and thorough study of 142 cases. Brain 2006, 129, 1640–1658. [Google Scholar] [CrossRef] [PubMed]
- Pantoni, L.; Bertini, E.; LaMassa, M.; Pracucci, G.; Inzitari, D. Clinical features, risk factors, and prognosis in transient global amnesia: A follow-up study. Eur. J. Neurol. 2005, 12, 350–356. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Lu, D.; Sveinsson, O.; Wirdefeldt, K.; Fall, K.; Piehl, F.; Valdimarsdóttir, U.; Fang, F. Is a Cancer Diagnosis Associated with Subsequent Risk of Transient Global Amnesia? PLoS ONE 2015, 10, e0122960. [Google Scholar] [CrossRef]
- Han, K.; Hu, H.-H.; Chao, A.-C.; Chang, F.-C.; Chung, C.-P.; Hsu, H.-Y.; Sheng, W.-Y.; Wu, J. Transient Global Amnesia Linked to Impairment of Brain Venous Drainage: An Ultrasound Investigation. Front. Neurol. 2019, 10, 67. [Google Scholar] [CrossRef]
- Arena, J.E.; Brown, R.D.; Mandrekar, J.; Rabinstein, A.A. Long-Term Outcome in Patients with Transient Global Amnesia: A Population-Based Study. Mayo Clin. Proc. 2017, 92, 399–405. [Google Scholar] [CrossRef]
- Lin, K.-H.; Chen, Y.-T.; Fuh, J.-L.; Li, S.-Y.; Chen, T.-J.; Tang, C.-H.; Wang, S.-J. Migraine is associated with a higher risk of transient global amnesia: A nationwide cohort study. Eur. J. Neurol. 2014, 21, 718–724. [Google Scholar] [CrossRef]
- Yi, M.; Sherzai, A.Z.; Ani, C.; Shavlik, D.; Ghamsary, M.; Lazar, E.; Sherzai, D. Strong Association Between Migraine and Transient Global Amnesia: A National Inpatient Sample Analysis. J. Neuropsychiatry Clin. Neurosci. 2019, 31, 43–48. [Google Scholar] [CrossRef]
- Liampas, I.; Siouras, A.S.; Siokas, V.; Tsouris, Z.; Rikos, D.; Brotis, A.; Aloizou, A.-M.; Dastamani, M.; Dardiotis, E. Migraine in transient global amnesia: A meta-analysis of observational studies. J. Neurol. 2021, 269, 184–196. [Google Scholar] [CrossRef]
- Inzitari, D.; Pantoni, L.; Lamassa, M.; Pallanti, S.; Pracucci, G.; Marini, P. Emotional Arousal and Phobia in Transient Global Amnesia. Arch. Neurol. 1997, 54, 866–873. [Google Scholar] [CrossRef]
- Pantoni, L.; LaMassa, M.; Inzitari, D. Transient global amnesia: A review emphasizing pathogenic aspects. Acta Neurol. Scand. 2000, 102, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Dã¶hring, J.; Schmuck, A.; Bartsch, T.; Döhring, J. Stress-related factors in the emergence of transient global amnesia with hippocampal lesions. Front. Behav. Neurosci. 2014, 8, 287. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.-W.; Park, S.Y.; Hong, J.-H.; Park, Y.H.; Kim, J.E.; Kim, S. Different Risk Factor Profiles between Transient Global Amnesia and Transient Ischemic Attack: A Large Case-Control Study. Eur. Neurol. 2013, 71, 19–24. [Google Scholar] [CrossRef]
- Liampas, I.; Raptopoulou, M.; Siokas, V.; Bakirtzis, C.; Tsouris, Z.; Aloizou, A.-M.; Dastamani, M.; Brotis, A.; Bogdanos, D.; Dardiotis, E. Conventional cardiovascular risk factors in Transient Global Amnesia: Systematic review and proposition of a novel hypothesis. Front. Neuroendocr. 2021, 61, 100909. [Google Scholar] [CrossRef] [PubMed]
- Rogalewski, A.; Beyer, A.; Friedrich, A.; Plümer, J.; Zuhorn, F.; Greeve, I.; Klingebiel, R.; Woermann, F.G.; Bien, C.G.; Schäbitz, W.-R. Transient Global Amnesia (TGA): Influence of Acute Hypertension in Patients Not Adapted to Chronic Hypertension. Front. Neurol. 2021, 12, 1003. [Google Scholar] [CrossRef]
- Lewis, S.L. Aetiology of transient global amnesia. Lancet 1998, 352, 397–399. [Google Scholar] [CrossRef]
- Rösler, A.; Mrass, G.J.; Frese, A.; Albert, I.; Schnorpfeil, F. Precipitating factors of transient global amnesia. J. Neurol. 1999, 246, 53–54. [Google Scholar] [CrossRef]
- Hoyer, C.; Ebert, A.; Sandikci, V.; Platten, M.; Szabo, K. Sex-related differences in stressful events precipitating transient global amnesia—A retrospective observational study. J. Neurol. Sci. 2021, 425, 117464. [Google Scholar] [CrossRef]
- Alessandro, L.; Calandri, I.L.; Suarez, M.F.; Heredia, M.L.; Chaves, H.; Allegri, R.F.; Farez, M.F. Transient global amnesia: Clinical features and prognostic factors suggesting recurrence. Arq. de Neuro-Psiquiatria 2019, 77, 3–9. [Google Scholar] [CrossRef]
- Agosti, C.; Borroni, B.; Akkawi, N.; Padovani, A. Cerebrovascular Risk Factors and Triggers in Transient Global Amnesia Patients with and without Jugular Valve Incompetence: Results from a Sample of 243 Patients. Eur. Neurol. 2010, 63, 291–294. [Google Scholar] [CrossRef]
- Szabo, K. Transient Global Amnesia. Front. Neurol. Neurosci. 2014, 34, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Marazzi, C.; Scoditti, U.; Ticinesi, A.; Nouvenne, A.; Pigna, F.; Guida, L.; Morelli, I.; Borghi, L.; Meschi, T. Transient global amnesia. Acta BioMed 2014, 85, 229–235. [Google Scholar] [PubMed]
- Kirshner, H.S. Transient Global Amnesia: A Brief Review and Update. Curr. Neurol. Neurosci. Rep. 2011, 11, 578–582. [Google Scholar] [CrossRef] [PubMed]
- Hoyer, C.; Higashida, K.; Fabbian, F.; De Giorgi, A.; Sandikci, V.; Ebert, A.; Platten, M.; Okazaki, S.; Manfredini, R.; Szabo, K. Chronobiology of transient global amnesia. J. Neurol. 2021, 269, 361–367. [Google Scholar] [CrossRef]
- Zeman, A.Z.; Hodges, J.R. Transient global amnesia. Br. J. Hosp. Med. 1997, 58, 257–260. [Google Scholar]
- Romoli, M.; Tuna, M.A.; Li, L.; Paciaroni, M.; Giannandrea, D.; Caprioli, F.T.; Lotti, A.; Eusebi, P.; Mosconi, M.G.; Venti, M.P.; et al. Time trends, frequency, characteristics and prognosis of short-duration transient global amnesia. Eur. J. Neurol. 2020, 27, 887–893. [Google Scholar] [CrossRef]
- Faglioni, P.; Scarpa, M.; Colombo, A.; Botti, C.; Grisanti, A. A Model-Based Study of Learning and Memory Following Transient Global Amnesia Attacks. Cortex 1992, 28, 9–22. [Google Scholar] [CrossRef]
- Kritchevsky, M.; Squire, L.R.; Zouzounis, J.A. Transient global amnesia: Characterization of anterograde and retrograde amnesia. Neurology 1988, 38, 213–219. [Google Scholar] [CrossRef]
- Kritchevsky, M.; Squire, L.R. Transient global amnesia: Evidence for extensive, temporally graded retrograde amnesia. Neurology 1989, 39, 213–218. [Google Scholar] [CrossRef]
- Quinette, P.; Guillery, B.; Desgranges, B.; De La Sayette, V.; Viader, F.; Eustache, F. Working memory and executive functions in transient global amnesia. Brain 2003, 126, 1917–1934. [Google Scholar] [CrossRef][Green Version]
- Uttner, I.; Weber, S.; Freund, W.; Schmitz, B.; Ramspott, M.; Huber, R. Transient Global Amnesia—Full Recovery without Persistent Cognitive Impairment. Eur. Neurol. 2007, 58, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Hodges, J.R.; Ward, C.D. Observations during transient global amnesia. A behavioural and neuropsychological study of five cases. Brain 1989, 112, 595–620. [Google Scholar] [CrossRef] [PubMed]
- Hodges, J.R.; Oxbury, S.M. Persistent memory impairment following transient global amnesia. J. Clin. Exp. Neuropsychol. 1990, 12, 904–920. [Google Scholar] [CrossRef] [PubMed]
- Gallassi, R.; Stracciari, A.; Morreale, A.; Lorusso, S.; Rebucci, G.; Lugaresi, E. Transient Global Amnesia: Neuropsychological Findings after Single and Multiple Attacks. Eur. Neurol. 1993, 33, 294–298. [Google Scholar] [CrossRef]
- Borroni, B.; Agosti, C.; Brambilla, C.; Vergani, V.; Cottini, E.; Akkawi, N.; Padovani, A. Is transient global amnesia a risk factor for amnestic mild cognitive impairment? J. Neurol. 2004, 251, 1125–1127. [Google Scholar] [CrossRef] [PubMed]
- Guillery-Girard, B.; Quinette, P.; Desgranges, B.; Piolino, P.; Viader, F.; De La Sayette, V.; Eustache, F. Long-term memory following transient global amnesia: An investigation of episodic and semantic memory. Acta Neurol. Scand. 2006, 114, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Eustache, F.; Desgranges, B.; Aupée, A.M.; Guillery, B.; Baron, J.C. Functional neuroanatomy of amnesia: Positron emission tomography studies. Microsc. Res. Tech. 2000, 51, 94–100. [Google Scholar] [CrossRef]
- Jäger, T.; Bäzner, H.; Kliegel, M.; Szabo, K.; Hennerici, M.G. The transience and nature of cognitive impairments in transient global amnesia: A meta-analysis. J. Clin. Exp. Neuropsychol. 2008, 31, 8–19. [Google Scholar] [CrossRef]
- Fisher, C.M. Transient global amnesia. Precipitating activities and other observations. Arch. Neurol. 1982, 39, 605–608. [Google Scholar] [CrossRef]
- Cole, A.J.; Gloor, P.; Kaplan, R. Transient global amnesia: The electroencephalogram at onset. Ann. Neurol. 1987, 22, 771–772. [Google Scholar] [CrossRef]
- Jacome, D.E. EEG Features in Transient Global Amnesia. Clin. Electroencephalogr. 1989, 20, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Butler, C.; Graham, K.; Hodges, J.R.; Kapur, N.; Wardlaw, J.M.; Zeman, A.Z.J. The syndrome of transient epileptic amnesia. Ann. Neurol. 2007, 61, 587–598. [Google Scholar] [CrossRef] [PubMed]
- Strupp, M.; Brüning, R.; Wu, R.H.; Deimling, M.; Reiser, M.; Brandt, T. Diffusion-weighted MRI in transient global amnesia: Elevated signal intensity in the left mesial temporal lobe in 7 of 10 patients. Ann. Neurol. 1998, 43, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Gass, A.; Gaa, J.; Hirsch, J.; Schwartz, A.; Hennerici, M.G. Lack of evidence of acute ischemic tissue change in transient global amnesia on single-shot echo-planar diffusion-weighted MRI. Stroke 1999, 30, 2070–2072. [Google Scholar] [CrossRef]
- Bartsch, T.; Alfke, K.; Deuschl, G.; Jansen, O. Evolution of hippocampal CA-1 diffusion lesions in transient global amnesia. Ann. Neurol. 2007, 62, 475–480. [Google Scholar] [CrossRef]
- Lee, H.Y.; Kim, J.H.; Weon, Y.-C.; Lee, J.S.; Kim, S.Y.; Youn, S.W.; Kim, S.H. Diffusion-weighted imaging in transient global amnesia exposes the CA1 region of the hippocampus. Neuroradiology 2007, 49, 481–487. [Google Scholar] [CrossRef]
- Weon, Y.; Kim, J.; Lee, J.; Kim, S. Optimal Diffusion-Weighted Imaging Protocol for Lesion Detection in Transient Global Amnesia. Am. J. Neuroradiol. 2008, 29, 1324–1328. [Google Scholar] [CrossRef]
- Bartsch, T.; Alfke, K.; Wolff, S.; Rohr, A.; Jansen, O.; Deuschl, G. Focal MR spectroscopy of hippocampal CA-1 lesions in transient global amnesia. Neurology 2008, 70, 1030–1035. [Google Scholar] [CrossRef]
- Stillhard, G.; Landis, T.; Schiess, R.; Regard, M.; Sialer, G. Bitemporal hypoperfusion in transient global amnesia: 99m-Tc-HM-PAO SPECT and neuropsychological findings during and after an attack. J. Neurol. Neurosurg. Psychiatry 1990, 53, 339–342. [Google Scholar] [CrossRef]
- Goldenberg, G.; Podreka, I.; Pfaffelmeyer, N.; Wessely, P.; Deecke, L. Thalamic ischemia in transient global amnesia: A SPECT study. Neurology 1991, 41, 1748. [Google Scholar] [CrossRef]
- Evans, J.; Wilson, B.; Wraight, E.P.; Hodges, J.R. Neuropsychological and SPECT scan findings during and after transient global amnesia: Evidence for the differential impairment of remote episodic memory. J. Neurol. Neurosurg. Psychiatry 1993, 56, 1227–1230. [Google Scholar] [CrossRef] [PubMed]
- Jovin, T.G.; Vitti, R.A.; McCluskey, L.F. Evolution of temporal lobe hypoperfusion in transient global amnesia: A serial single photon emission computed tomography study. J. Neuroimaging 2000, 10, 238–241. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.-W.; Park, Y.H.; Park, S.Y.; Wang, M.J.; Lim, J.-S.; Kim, S.-H.; Chun, I.K.; Yang, Y.; Kim, S. Longitudinal Cerebral Perfusion Change in Transient Global Amnesia Related to Left Posterior Medial Network Disruption. PLoS ONE 2015, 10, e0145658. [Google Scholar] [CrossRef] [PubMed]
- Baron, J.-C.; Petit-Taboué, M.C.; Le Doze, F.; Desgranges, B.; Ravenel, N.; Marchal, G. Right frontal cortex hypometabolism in transient global amnesia A PET study. Brain 1994, 117, 545–552. [Google Scholar] [CrossRef]
- Eustache, F.; Desgranges, B.; Petit-Taboué, M.-C.; De La Sayette, V.; Piot, V.; Sablé, C.; Marchal, G.; Baron, J.-C. Transient global amnesia: Implicit/explicit memory dissociation and PET assessment of brain perfusion and oxygen metabolism in the acute stage. J. Neurol. Neurosurg. Psychiatry 1997, 63, 357–367. [Google Scholar] [CrossRef][Green Version]
- Tanabe, H.; Hashikawa, K.; Nakagawa, Y.; Ikeda, M.; Yamamoto, H.; Harada, K.; Tsumoto, T.; Nishimura, T.; Shiraishi, J.; Kimura, K. Memory loss due to transient hypoperfusion in the medial temporal lobes including hippocampus. Acta Neurol. Scand. 1991, 84, 22–27. [Google Scholar] [CrossRef]
- Heiss, W.D.; Pawlik, G.; Holthoff, V.; Kessler, J.; Szelies, B. PET correlates of normal and impaired memory functions. Cerebrovasc. Brain Metab. Rev. 1992, 4, 1–27. [Google Scholar]
- Schmidtke, K.; Reinhardt, M.; Krause, T. Cerebral perfusion during transient global amnesia: Findings with HMPAO SPECT. J. Nucl. Med. 1998, 39, 155–159. [Google Scholar]
- Warren, J.D.; Chatterton, B.; Thompson, P.D. A SPECT study of the anatomy of transient global amnesia. J. Clin. Neurosci. 2000, 7, 57–59. [Google Scholar] [CrossRef]
- Guillery, B.; Desgranges, B.; de la Sayette, V.; Landeau, B.; Eustache, F.; Baron, J.-C. Transient global amnesia: Concomitant episodic memory and positron emission tomography assessment in two additional patients. Neurosci. Lett. 2002, 325, 62–66. [Google Scholar] [CrossRef]
- Zeman, A.Z. Episodic memory in transient global amnesia. J. Neurol. Neurosurg. Psychiatry 1999, 66, 135. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jung, H.H.; Baumgartner, R.W.; Burgunder, J.M.; Wielepp, J.P.; Lourens, S.; Wielepp, J.P. Reversible hyperperfusion of the right medial temporal lobe in transient global amnesia. J. Neurol. Neurosurg. Psychiatry 1996, 61, 654–655. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Laloux, P.; Brichant, C.; Cauwe, F.; Decoster, P. Technetium-99m HM-PAO Single Photon Emission Computed Tomography Imaging in Transient Global Amnesia. Arch. Neurol. 1992, 49, 543–546. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, H.; Higashi, S.; Tsuji, S.; Sumiya, H.; Miyauchi, T.; Hisada, K.; Yamashita, J. High Resolution Tc-99m HMPAO SPECT in a Patient with Transient Global Amnesia. Clin. Nucl. Med. 1993, 18, 46–49. [Google Scholar] [CrossRef]
- Sakashita, Y.; Kanai, M.; Sugimoto, T.; Taki, S.; Takamori, M. Changes in cerebral blood flow and vasoreactivity in response to acetazolamide in patients with transient global amnesia. J. Neurol. Neurosurg. Psychiatry 1997, 63, 605–610. [Google Scholar] [CrossRef][Green Version]
- Takeuchi, R.; Yonekura, Y.; Matsuda, H.; Nishimura, Y.; Tanaka, H.; Ohta, H.; Sakahara, H.; Konishi, J. Resting and aceta-zolamide-challenged technetium-99m-ECD SPECT in transient global amnesia. J. Nucl. Med. 1998, 39, 1360–1362. [Google Scholar]
- Pai, M.C.; Yang, S.S. Transient global amnesia: A retrospective study of 25 patients. Zhonghua yi xue za zhi = Chin. Med. J. Free China Ed. 1999, 62, 140–145. [Google Scholar]
- Nardone, R.; Buffone, E.C.; Matullo, M.F.; Tezzon, F. Motor cortex excitability in transient global amnesia. J. Neurol. 2004, 251, 42–46. [Google Scholar] [CrossRef]
- Yamane, Y.; Ishii, K.; Shimizu, K.; Sofue, K.; Yoshikawa, T.; Miyamoto, N.; Ohkawa, S. Global Cerebral Hypoperfusion in a Patient With Transient Global Amnesia. J. Comput. Assist. Tomogr. 2008, 32, 415–417. [Google Scholar] [CrossRef]
- Lampl, Y.; Sadeh, M.; Lorberboym, M. Transient global amnesia—Not always a benign process. Acta Neurol. Scand. 2004, 110, 75–79. [Google Scholar] [CrossRef]
- Takeuchi, R.; Matsuda, H.; Yoshioka, K.; Yonekura, Y. Cerebral blood flow SPET in transient global amnesia with automated ROI analysis by 3DSRT. Eur. J. Nucl. Med. Mol. Imaging 2004, 31, 578–589. [Google Scholar] [CrossRef] [PubMed]
- Tateno, M.; Honma, T.; Kobayashi, S.; Utsumi, K.; Fujii, K.; Morii, H. Decreased blood perfusion in right thalamus after transient global amnesia demonstrated by an automated program, 3DSRT. Psychiatry Clin. Neurosci. 2008, 62, 244. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.S.; Cho, S.S.; Choi, J.Y.; Kim, Y.H. Transient global amnesia: A study with Tc-99m ECD SPECT shortly after symptom onset and after recovery. Diagn. Interv. Radiol. 2016, 22, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Asada, T.; Matsuda, H.; Morooka, T.; Nakano, S.; Kimura, M.; Uno, M. Quantitative single photon emission tomography analysis for the diagnosis of transient global amnesia: Adaptation of statistical parametric mapping. Psychiatry Clin. Neurosci. 2000, 54, 691–694. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.A.; Jeong, J.; Yang, D.W.; Kang, B.-J.; Kim, S.H.; Chung, S.K.; Sohn, H.S.; Peterson, B.S. A Tc-99m SPECT study of regional cerebral blood flow in patients with transient global amnesia. NeuroImage 2009, 47, 50–55. [Google Scholar] [CrossRef]
- Fujii, K.; Sadoshima, S.; Ishitsuka, T.; Kusuda, K.; Kuwabara, Y.; Ichiya, Y.; Fujishima, M. Regional cerebral blood flow and metabolism in patients with transient global amnesia: A positron emission tomography study. J. Neurol. Neurosurg. Psychiatry 1989, 52, 622–630. [Google Scholar] [CrossRef]
- Zeman, A.; Boniface, S.J.; Hodges, J.R. Transient epileptic amnesia: A description of the clinical and neuropsychological features in 10 cases and a review of the literature. J. Neurol. Neurosurg. Psychiatry 1998, 64, 435–443. [Google Scholar] [CrossRef]
- Kapur, N. Transient epileptic amnesia—A clinical update and a reformulation. J. Neurol. Neurosurg. Psychiatry 1993, 56, 1184–1190. [Google Scholar] [CrossRef]
- Serafetinides, E.A. Transient epileptic amnesia--a clinical update and a reformulation. J. Neurol. Neurosurg. Psychiatry 1994, 57, 1549. [Google Scholar] [CrossRef][Green Version]
- Mosbah, A.; Tramoni, E.; Guedj, E.; Aubert, S.; Daquin, G.; Ceccaldi, M.; Félician, O.; Bartolomei, F. Clinical, neuropsychological, and metabolic characteristics of transient epileptic amnesia syndrome. Epilepsia 2014, 55, 699–706. [Google Scholar] [CrossRef]
- Milton, F.; Muhlert, N.; Pindus, D.M.; Butler, C.R.; Kapur, N.; Graham, K.S.; Zeman, A.Z.J. Remote memory deficits in transient epileptic amnesia. Brain 2010, 133, 1368–1379. [Google Scholar] [CrossRef] [PubMed]
- Felician, O.; Tramoni, E.; Bartolomei, F. Transient epileptic amnesia: Update on a slowly emerging epileptic syndrome. Rev. Neurol. 2015, 171, 289–297. [Google Scholar] [CrossRef]
- Butler, C.R.; Bhaduri, A.; Acosta-Cabronero, J.; Nestor, P.; Kapur, N.; Graham, K.; Hodges, J.R.; Zeman, A. Transient epileptic amnesia: Regional brain atrophy and its relationship to memory deficits. Brain 2008, 132, 357–368. [Google Scholar] [CrossRef]
- Asadi-Pooya, A.A. Transient epileptic amnesia: A concise review. Epilepsy Behav. 2014, 31, 243–245. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Lázaro, C.; Santos, S.; Garcés-Redondo, M.; Piñol-Ripoll, G.; Fabre-Pi, O.; Mostacero, E.; López-Del Val, L.J.; Tejero-Juste, C.; Pascual-Millán, L.F. Ictus amnésico por infarto hipocámpo [Amnesic stroke caused by hippocampal infarction]. Rev. Neurol. 2005, 41, 27–30. [Google Scholar] [PubMed]
- Carlesimo, G.A.; Lombardi, M.G.; Caltagirone, C. Vascular thalamic amnesia: A reappraisal. Neuropsychologia 2011, 49, 777–789. [Google Scholar] [CrossRef]
- Ravindran, V.; Jain, S.; Ming, A.; Bartlett, R.J. Transient global amnesia in a patient with acute unilateral caudate nucleus ischemia. J. Clin. Neurosci. 2004, 11, 669–672. [Google Scholar] [CrossRef]
- Adler, A.C.; Warum, D.; Sapire, J.M. Transient global amnesia caused by hippocampal infarct: Case report and review of literature. Clin. Imaging 2012, 36, 584–586. [Google Scholar] [CrossRef]
- Liang, J.-F.; Shen, A.-L.; Lin, S.-K. Bilateral hippocampal abnormalities on diffusion-weighted MRI in transient global amnesia: Report of a case. Acta Neurol. Taiwanica 2009, 18, 127–129. [Google Scholar]
- Greer, D.M.; Schaefer, P.W.; Schwamm, L.H. Unilateral temporal lobe stroke causing ischemic transient global amnesia: Role for diffusion-weighted imaging in the initial evaluation. J. Neuroimaging 2001, 11, 317–319. [Google Scholar] [CrossRef]
- Ferro, J.M. Hyperacute cognitive stroke syndromes. J. Neurol. 2001, 248, 841–849. [Google Scholar] [CrossRef] [PubMed]
- Michel, P.; Beaud, V.; Eskandari, A.; Maeder, P.; Demonet, J.-F.; Eskioglou, E. Ischemic Amnesia: Causes and Outcome. Stroke 2017, 48, 2270–2273. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.; Stewart, G.; Wood, A.; Gillespie, J.E. Transient global amnesia and cortical blindness after vertebral angiography: Further evidence for the role of arterial spasm. AJNR Am. J. Neuroradiol. 1995, 16, 955–959. [Google Scholar] [PubMed]
- Yıldız, A.; Yencilek, E.; Apaydin, F.D.; Duce, M.N.; Ozer, C.; Atalay, A.; Apaydın, F.D.; Yildiz, A. Transient partial amnesia complicating cardiac and peripheral arteriography with nonionic contrast medium. Eur. Radiol. 2003, 13, L113–L115. [Google Scholar] [CrossRef]
- Benke, T.; Chemelli, A.; Lottersberger, C.; Waldenberger, P.; Karner, E.; Trinka, E. Transient global amnesia triggered by the intracarotid amobarbital procedure. Epilepsy Behav. 2005, 6, 274–278. [Google Scholar] [CrossRef]
- Donnet, A. Transient Global Amnesia Triggered by Migraine in a French Tertiary-Care Center: An 11-Year Retrospective Analysis. Headache J. Head Face Pain 2015, 55, 853–859. [Google Scholar] [CrossRef]
- Kumral, E.; Deveci, E.E.; Erdoğan, C.; Enüstün, C. Isolated hippocampal infarcts: Vascular and neuropsychological findings. J. Neurol. Sci. 2015, 356, 83–89. [Google Scholar] [CrossRef]
- Blumenfeld, A.E.; Victorio, M.C.; Berenson, F.R. Complicated Migraines. Semin. Pediatr. Neurol. 2016, 23, 18–22. [Google Scholar] [CrossRef]
- April, M.D.; Fossum, K.; Hounshell, C.; Stolper, K.; Spear, L.; Semelrath, K.; Stolper, K. A sinister cause of anterograde amnesia: Painless aortic dissection. Am. J. Emerg. Med. 2015, 33, 989.e5–989.e7. [Google Scholar] [CrossRef]
- Machado, A.; Rodrigues, A.; Ribeiro, M.; Cerqueira, J.; Soares-Fernandes, J. Tadalafil-Induced Transient Global Amnesia. J. Neuropsychiatry Clin. Neurosci. 2010, 22, 352-e28. [Google Scholar] [CrossRef][Green Version]
- Mangla, A.; Navi, B.; Layton, K.; Kamel, H. Transient Global Amnesia and the Risk of Ischemic Stroke. Stroke 2014, 45, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Tosi, L.; Righetti, C.A. Transient global amnesia and migraine in young people. Clin. Neurol. Neurosurg. 1997, 99, 63–65. [Google Scholar] [CrossRef]
- Saito, Y.; Memezawa, H. [Migraine with aura accompanied by transient global amnesia]. No hattatsu. Brain Dev. 2010, 42, 303–304. [Google Scholar]
- Maggioni, F.; Mainardi, F.; Bellamio, M.; Zanchin, G. Transient Global Amnesia Triggered by Migraine in Monozygotic Twins. Headache J. Head Face Pain 2011, 51, 1305–1308. [Google Scholar] [CrossRef]
- Volta, G.D.; Zavarise, P.; Ngonga, G.; Agosti, C.; Premi, E.; Padovani, A. Transient Global Amnesia as a Presenting Aura. Headache J. Head Face Pain 2014, 54, 551–552. [Google Scholar] [CrossRef]
- Vongvaivanich, K.; Lertakyamanee, P.; Silberstein, S.D.; Dodick, D.W. Late-life migraine accompaniments: A narrative review. Cephalalgia 2014, 35, 894–911. [Google Scholar] [CrossRef]
- Staniloiu, A.; Markowitsch, H.J. Dissociative amnesia. Lancet Psychiatry 2014, 1, 226–241. [Google Scholar] [CrossRef]
- Şar, V.; Akyüz, G.; Doğan, O. Prevalence of dissociative disorders among women in the general population. Psychiatry Res. 2007, 149, 169–176. [Google Scholar] [CrossRef]
- Brand, M.; Eggers, C.; Reinhold, N.; Fujiwara, E.; Kessler, J.; Heiss, W.-D.; Markowitsch, H.J. Functional brain imaging in 14 patients with dissociative amnesia reveals right inferolateral prefrontal hypometabolism. Psychiatry Res. Neuroimaging 2009, 174, 32–39. [Google Scholar] [CrossRef]
- Barash, J.A.; Somerville, N.; DeMaria, A. Cluster of an Unusual Amnestic Syndrome—Massachusetts, 2012–2016. MMWR Morb. Mortal Wkly. Rep. 2017, 66, 76–79. [Google Scholar] [CrossRef]
- Berlit, P. Successful prophylaxis of recurrent transient global amnesia with metoprolol. Neurology 2000, 55, 1937–1938. [Google Scholar] [CrossRef] [PubMed]
- Hinge, H.-H.; Jensen, T.S.; Kjaer, M.; Marquardsen, J.; Olivarius, B.D.F. The Prognosis of Transient Global Amnesia. Results of a multicenter study. Arch. Neurol. 1986, 43, 673–676. [Google Scholar] [CrossRef] [PubMed]
- Gandolfo, C.; Caponnetto, C.; Conti, M.; Dagnino, N.; Del Sette, M.; Primavera, A. Prognosis of Transient Global Amnesia: A Long-Term Follow-Up Study. Eur. Neurol. 1992, 32, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Melo, T.P.; Ferro, J.M.; Ferro, H. Transient global amnesia. A case control study Brain 1992, 115, 261–270. [Google Scholar] [CrossRef]
- Zorzon, M.; Antonutti, L.; Masè, G.; Biasutti, E.; Vitrani, B.; Cazzato, G. Transient Global Amnesia and Transient Ischemic Attack. Natural history, vascular risk factors, and associated conditions. Stroke 1995, 26, 1536–1542. [Google Scholar] [CrossRef]
- Agosti, C.; Akkawi, N.M.; Borroni, B.; Padovani, A. Recurrency in transient global amnesia: A retrospective study. Eur. J. Neurol. 2006, 13, 986–989. [Google Scholar] [CrossRef]
- Tynas, R.; Panegyres, P.K. Factors determining recurrence in transient global amnesia. BMC Neurol. 2020, 20, 1–7. [Google Scholar] [CrossRef]
- Liampas, I.; Raptopoulou, M.; Mpourlios, S.; Siokas, V.; Tsouris, Z.; Aloizou, A.-M.; Dastamani, M.; Brotis, A.; Bogdanos, D.; Xiromerisiou, G.; et al. Factors associated with recurrent transient global amnesia: Systematic review and pathophysiological insights. Rev. Neurosci. 2021, 32, 751–765. [Google Scholar] [CrossRef]
- Rogalewski, A.; Beyer, A.; Friedrich, A.; Plümer, J.; Zuhorn, F.; Klingebiel, R.; Woermann, F.G.; Bien, C.G.; Greeve, I.; Schäbitz, W.-R. Transient Global Amnesia (TGA): Younger Age and Absence of Cerebral Microangiopathy Are Potentially Predisposing Factors for TGA Recurrence. Front. Neurol. 2021, 12, 736563. [Google Scholar] [CrossRef]
- Uttner, I.; Prexl, S.; Freund, W.; Unrath, A.; Bengel, D.; Huber, R. Long-Term Outcome in Transient Global Amnesia Patients with and without Focal Hyperintensities in the CA1 Region of the Hippocampus. Eur. Neurol. 2012, 67, 155–160. [Google Scholar] [CrossRef]
- Mazzucchi, A.; Moretti, G.; Caffarra, P.; Parma, M. Neuropsychological functions in the follow-up of transient global amnesia. Brain 1980, 103, 161–178. [Google Scholar] [CrossRef] [PubMed]
- Le Pira, F.; Giuffrida, S.; Maci, T.; Reggio, E.; Zappalà, G.; Perciavalle, V. Cognitive Findings After Transient Global Amnesia: Role of Prefrontal Cortex. Appl. Neuropsychol. 2005, 12, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Jensen, T.S.; Olivarius, B.D.F. Transient global amnesia as a manifestation of transient cerebral ischemia. Acta Neurol. Scand. 1980, 61, 115–124. [Google Scholar] [CrossRef]
- Kushner, M.J.; Hauser, W.A. Transient global amnesia: A case-control study. Ann. Neurol. 1985, 18, 684–691. [Google Scholar] [CrossRef]
- Shuping, J.R.; Rollinson, R.D.; Toole, J.F. Transient global amnesia. Ann. Neurol. 1980, 7, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.W.; Petersen, R.C.; Metter, E.J.; Millikan, C.H.; Yanagihara, T. Transient global amnesia: Clinical characteristics and prognosis. Neurology 1987, 37, 733. [Google Scholar] [CrossRef]
- Romoli, M.; Tuna, M.A.; McGurgan, I.; Li, L.; Giannandrea, D.; Eusebi, P.; Caprioli, F.T.; Lotti, A.; Salvadori, N.; Sarchielli, P.; et al. Long-Term Risk of Stroke After Transient Global Amnesia in Two Prospective Cohorts. Stroke 2019, 50, 2555–2557. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.; Limaye, K.; Shaban, A.; Adams, H.P., Jr.; Leira, E.C. Transient global amnesia does not increase the risk of subsequent ischemic stroke: A propensity score-matched analysis. J. Neurol. 2021, 268, 3301–3306. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, S.-W.; Yang, Y.-H.; Ho, B.-L.; Yang, S.-T.; Chen, C.-H. The long-term risk of epilepsy after transient global amnesia: A population-based cohort study. Clin. Neurol. Neurosurg. 2020, 197, 106086. [Google Scholar] [CrossRef] [PubMed]

| Main Diagnostic Features of TGA |
|---|
| Attack must be witnessed |
| There must be anterograde amnesia during the attack |
| Cognitive impairment limited to amnesia |
| No clouding of consciousness or loss of personal identity |
| No focal neurological signs/symptoms |
| No epileptic features |
| Attack must resolve within 24 h |
| No recent head injury or active epilepsy |
| Main MRI Issues in TGA |
|---|
|
|
|
|
|
|
|
| Condition | Risk Factors | Precipitating Factors | Duration | Associated Neurologic Symptoms | MRI | EEG | Recurrence | Response to Anti-Epileptics |
|---|---|---|---|---|---|---|---|---|
| TGA | Migraine | Yes | 4–6 h | No | Transient hippocampal DWI hyperintensity | Normal | Low | No |
| TEA | No | No/yes | <60 min | No/yes (automatisms, olfactory or gustatory hallucinations) | Normal/hippocampal sclerosis or atrophy | Abnormal | High | Yes |
| Ischemic events | Vascular | No | Minutes to permanent impairment | No/yes (any) | DWI with T2-FLAIR permanent lesion | Normal | Low | No |
| Hypoxic events (i.e., aortic dissection) | Increased intrathoracic pressure | Stress reaction due to pain | 10–12 h | No | Normal | Normal | Not known | No |
| Migraine | Genetic, dietary | Yes (fasting, emotional stress, sleep problems) | 4–72 h | Auras up to 30% (visual, sensory, motor, or language abnormalities) | Normal | Normal | High | Yes |
| DA | Trauma | Yes emotional stress | Variable | No | Normal | Normal | Varies | No |
| Toxic amnesia | Substance abuse | No | Variable | Yes (disorientation, dysexecutive syndrome, etc.) | Normal or bilateral hippocampal ischemia | Normal | High | No |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sparaco, M.; Pascarella, R.; Muccio, C.F.; Zedde, M. Forgetting the Unforgettable: Transient Global Amnesia Part II: A Clinical Road Map. J. Clin. Med. 2022, 11, 3940. https://doi.org/10.3390/jcm11143940
Sparaco M, Pascarella R, Muccio CF, Zedde M. Forgetting the Unforgettable: Transient Global Amnesia Part II: A Clinical Road Map. Journal of Clinical Medicine. 2022; 11(14):3940. https://doi.org/10.3390/jcm11143940
Chicago/Turabian StyleSparaco, Marco, Rosario Pascarella, Carmine Franco Muccio, and Marialuisa Zedde. 2022. "Forgetting the Unforgettable: Transient Global Amnesia Part II: A Clinical Road Map" Journal of Clinical Medicine 11, no. 14: 3940. https://doi.org/10.3390/jcm11143940
APA StyleSparaco, M., Pascarella, R., Muccio, C. F., & Zedde, M. (2022). Forgetting the Unforgettable: Transient Global Amnesia Part II: A Clinical Road Map. Journal of Clinical Medicine, 11(14), 3940. https://doi.org/10.3390/jcm11143940






