Machine Learning Prediction of Comorbid Substance Use Disorders among People with Bipolar Disorder
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Clinical Variables Assessment
2.3. Statistical Analyses
2.3.1. Missing Data
2.3.2. Random Forest
| SUD (n = 262; 51.57%) | Non-SUD (n = 246; 48.4%) | t/Z/χ2 | p | |
|---|---|---|---|---|
| Gender (n; %) | 35.32 | <0.001 | ||
| - Female | 109; 41.6% | 167; 67.9% | ||
| Living status (n; %) | 14.27 | 0.003 | ||
| - Parents | 45; 18.8% | 83; 33.2% | ||
| - Family | 141; 59% | 114; 45.6% | ||
| - Alone | 33; 18.8% | 36; 14.4% | ||
| - Other (community) | 20; 8.4% | 17; 6.8% | ||
| Relationship status (n; %) | 27.8 | <0.001 | ||
| - Not in a relationship | 72; 29.8% | 125; 48.4% | ||
| - Married | 117; 48.3% | 84; 32.6% | ||
| - Divorced | 40; 16.5% | 47; 18.2% | ||
| - Widow | 13; 5.4% | 2; 0.8% | ||
| Working status (n; %) | 14.78 | 0.011 | ||
| - Full-time or part-time job | 127; 53.8% | 131; 52.8% | ||
| - Unemployed | 27; 11.4% | 23; 9.3% | ||
| - Retired | 33; 14% | 45; 18.1% | ||
| - Not able to work | 34; 14.4% | 17; 6.9% | ||
| Diagnosis (n; %) | 5.26 | 0.014 | ||
| - BDI | 190; 72.5% | 155; 63% | ||
| - BDII | 72; 27.5% | 91; 37% | ||
| Age and illness duration (mean ± SD) | ||||
| Age at assessment | 48.6 ± 15.08 | 43.77 ± 26.46 | 26.397 | <0.001 |
| Age at onset | 29.3 ± 13.19 | 26.4 ± 10.63 | 28.307 | <0.001 |
| Duration of illness | 19.27 ± 12.1 | 17.32 ± 12.06 | 29.959 | 0.048 |
| Number of affective episodes, lifetime (mean ± SD) | ||||
| - Depressive | 8.39 ± 11.87 | 7.13 ± 9.17 | 28.285 | 0.084 |
| - Manic | 2.25 ± 4.1 | 2.52 ± 3.69 | 35.186 | 0.066 |
| - Hypomanic | 5.51 ± 10.2 | 4.04 ± 7.76 | 28.288 | <0.001 |
| - Mixed | 0.63 ± 1.89 | 0.65 ± 1.77 | 32.958 | 0.54 |
| - Total | 16.84 ± 22.61 | 14.34 ± 16.66 | 30.338 | 0.25 |
| Polarity of the first affective episode (n; %) | 7.94 | 0.09 | ||
| - Depressive | 168; 70.9% | 66; 26.2% | ||
| - Manic | 52; 21.9% | 22; 8.7% | ||
| - Hypomanic | 9; 3.8% | 155; 61.5% | ||
| - Mixed | 6; 2.5% | 5; 2% | ||
| Number of Psychiatric admissions, lifetime (mean ± SD) | 1.59 ± 2.14 | 1.61 ± 2.01 | 30.023 | 0.61 |
| Clinical course variables, lifetime (n; %) | ||||
| - Suicide attempts | 73; 51.4% | 172; 47% | 0.702 | 0.23 |
| - Aggressive behaviours | ||||
| - Self-directed | 52; 21% | 52; 19.8% | 0.130 | 0.4 |
| - Hetero-directed | 26; 10.6% | 54; 20.6% | 9.64 | 0.001 |
| - Psychotic symptoms | 119; 49.2% | 132; 51.2% | 0.198 | 0.36 |
| - Rapid cycling | 60; 22.9% | 68; 27.6% | 1.51 | 0.130 |
| - Seasonality | 63; 25.6% | 53; 20.2% | 2.08 | 0.09 |
| - Family history of Mood Disorder | 143; 62.2% | 153; 61.4% | 0.027 | 0.47 |
| - Comorbidity with Personality Disorder | 7.31 | 0.063 | ||
| - Cluster A | 4; 1.7% | 7; 2.9% | ||
| - Cluster B | 20; 8.7% | 34; 13.9% | ||
| - Cluster C | 11; 4.8% | 4; 1.6% |
2.3.3. Multiple Logistic Regression
3. Results
3.1. Characteristics of the Sample
3.2. Missing Data
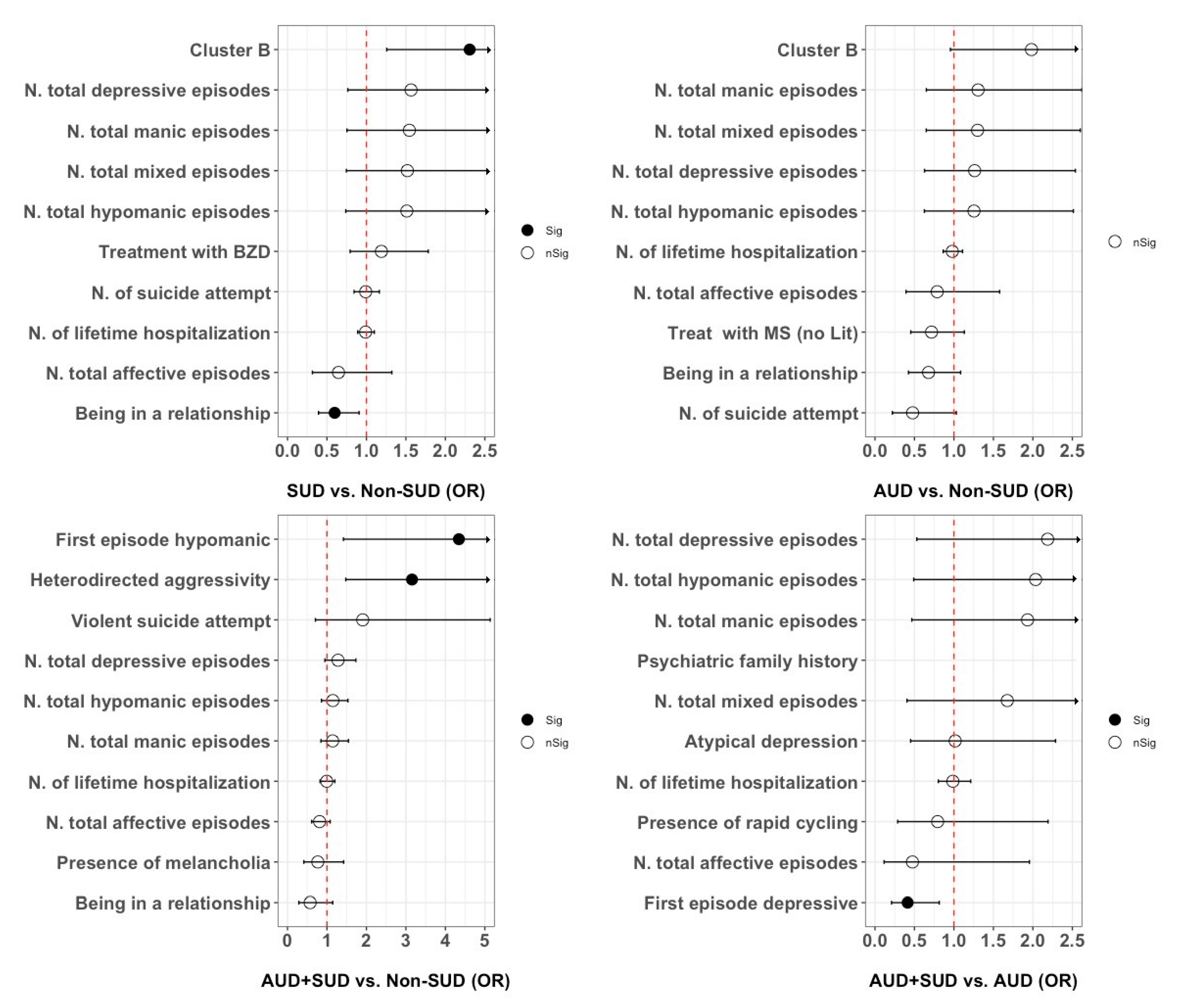
3.3. Patients with SUD vs. without SUD
3.4. Patients with AUD vs. without SUD
| Data Set | Number of Trees | Number of Features | Node Size | Accuracy% | 95% CI | p | Sensitivity | Specificity | F1-Score |
|---|---|---|---|---|---|---|---|---|---|
| SUD VS. NO-SUD | 800 | 9 | 6 | 65.3% | 54.8–74.7 | 0.004 | 69.6% | 61.2% | 0.66 |
| AUD VS. NO-SUD | 350 | 3 | 43 | 53.8% | 39.5–67.8 | 0.44 | 44% | 63% | 0.48 |
| AUD+SUD VS. NO-SUD | 500 | 26 | 47 | 75% | 56.6–88.5 | 0.003 | 75% | 75% | 0.75 |
| AUD+SUD VS. AUD | 800 | 2 | 14 | 62.5% | 43.6–78.9 | 0.107 | 43.8% | 81.2% | 0.54 |
3.5. Patients with AUD+SUD vs. without SUD or AUD
3.6. Patients with AUD+SUD vs. with AUD
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ostacher, M.J.; Perlis, R.H.; Nierenberg, A.A.; Calabrese, J.; Stange, J.P.; Salloum, I.; Weiss, R.D.; Sachs, G.S. Impact of Substance Use Disorders on Recovery From Episodes of Depression in Bipolar Disorder Patients: Prospective Data From the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD). Am. J. Psychiatry 2010, 167, 289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Menculini, G.; Steardo, L.; Verdolini, N.; Cirimbilli, F.; Moretti, P.; Tortorella, A. Substance Use Disorders in Bipolar Disorders: Clinical Correlates and Treatment Response to Mood Stabilizers. J. Affect. Disord. 2022, 300, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Simhandl, C.; Radua, J.; König, B.; Amann, B.L. Prevalence and Impact of Comorbid Alcohol Use Disorder in Bipolar Disorder: A Prospective Follow-up Study. Aust. N. Z. J. Psychiatry 2016, 50, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Pinto, J.V.; Medeiros, L.S.; Santana da Rosa, G.; Santana de Oliveira, C.E.; de Souza Crippa, J.A.; Passos, I.C.; Kauer-Sant’Anna, M. The Prevalence and Clinical Correlates of Cannabis Use and Cannabis Use Disorder among Patients with Bipolar Disorder: A Systematic Review with Meta-Analysis and Meta-Regression. Neurosci. Biobehav. Rev. 2019, 101, 78–84. [Google Scholar] [CrossRef]
- Fornaro, M.; Carvalho, A.F.; de Prisco, M.; Mondin, A.M.; Billeci, M.; Selby, P.; Iasevoli, F.; Berk, M.; Castle, D.J.; de Bartolomeis, A. The Prevalence, Odds, Predictors, and Management of Tobacco Use Disorder or Nicotine Dependence among People with Severe Mental Illness: Systematic Review and Meta-Analysis. Neurosci. Biobehav. Rev. 2022, 132, 289–303. [Google Scholar] [CrossRef]
- Cerullo, M.A.; Strakowski, S.M. The Prevalence and Significance of Substance Use Disorders in Bipolar Type I and II Disorder. Subst. Abus. Treat. Prev. Policy 2007, 2, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Grant, B.F.; Stinson, F.S.; Hasin, D.S.; Dawson, D.A.; Chou, S.P.; Ruan, W.J.; Huang, B. Prevalence, Correlates, and Comorbidity of Bipolar I Disorder and Axis I and II Disorders: Results from the National Epidemiologic Survery on Alcohol and Related Conditions. J. Clin. Psychiatry 2005, 66, 1205–1215. [Google Scholar] [CrossRef]
- Hunt, G.E.; Malhi, G.S.; Cleary, M.; Lai, H.M.X.; Sitharthan, T. Prevalence of Comorbid Bipolar and Substance Use Disorders in Clinical Settings, 1990–2015: Systematic Review and Meta-Analysis. J. Affect. Disord. 2016, 206, 331–349. [Google Scholar] [CrossRef]
- Lagerberg, T.V.; Icick, R.; Aminoff, S.R.; Nerhus, M.; Barrett, E.A.; Bjella, T.D.; Olsen, S.H.; Høegh, M.C.; Melle, I. Substance Misuse Trajectories and Risk of Relapse in the Early Course of Bipolar Disorder. Front. Psychiatry 2021, 12, 656962. [Google Scholar] [CrossRef]
- McIntyre, R.S.; Nguyen, H.T.; Soczynska, J.K.; Lourenco, M.T.C.; Woldeyohannes, H.O.; Konarski, J.Z. Medical and Substance-Related Comorbidity in Bipolar Disorder: Translational Research and Treatment Opportunities. Dialog. Clin. Neurosci. 2022, 10, 203–213. [Google Scholar] [CrossRef]
- Xu, N.; Huggon, B.; Saunders, K.E.A. Cognitive Impairment in Patients with Bipolar Disorder: Impact of Pharmacological Treatment. CNS Drugs 2020, 34, 29–46. [Google Scholar] [CrossRef] [PubMed]
- Dalton, E.J.; Cate-Carter, T.D.; Mundo, E.; Parikh, S.V.; Kennedy, J.L. Suicide Risk in Bipolar Patients: The Role of Co-Morbid Substance Use Disorders. Bipolar Disord. 2003, 5, 58–61. [Google Scholar] [CrossRef]
- Grunebaum, M.F.; Galfalvy, H.C.; Nichols, C.M.; Caldeira, N.A.; Sher, L.; Dervic, K.; Burke, A.K.; Mann, J.J.; Oquendo, M.A. Aggression and Substance Abuse in Bipolar Disorder. Bipolar Disord. 2006, 8, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Hjorthøj, C.; Østergaard, M.L.D.; Benros, M.E.; Toftdahl, N.G.; Erlangsen, A.; Andersen, J.T.; Nordentoft, M. Association between Alcohol and Substance Use Disorders and All-Cause and Cause-Specific Mortality in Schizophrenia, Bipolar Disorder, and Unipolar Depression: A Nationwide, Prospective, Register-Based Study. Lancet Psychiatry 2015, 2, 801–808. [Google Scholar] [CrossRef]
- Mazza, M.; Mandelli, L.; di Nicola, M.; Harnic, D.; Catalano, V.; Tedeschi, D.; Martinotti, G.; Colombo, R.; Bria, P.; Serretti, A.; et al. Clinical Features, Response to Treatment and Functional Outcome of Bipolar Disorder Patients with and without Co-Occurring Substance Use Disorder: 1-Year Follow-Up. J. Affect. Disord. 2009, 115, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Swartz, M.S.; Wagner, H.R.; Swanson, J.W.; Stroup, T.S.; McEvoy, J.P.; Reimherr, F.; Miller, D.D.; McGee, M.; Khan, A.; Canive, J.M.; et al. The Effectiveness of Antipsychotic Medications in Patients Who Use or Avoid Illicit Substances: Results from the CATIE Study. Schizophr. Res. 2008, 100, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Grant, B.F.; Goldstein, R.B.; Saha, T.D.; Patricia Chou, S.; Jung, J.; Zhang, H.; Pickering, R.P.; June Ruan, W.; Smith, S.M.; Huang, B.; et al. Epidemiology of DSM-5 Alcohol Use Disorder: Results From the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry 2015, 72, 757–766. [Google Scholar] [CrossRef] [Green Version]
- Blanco, C.; Flórez-Salamanca, L.; Secades-Villa, R.; Wang, S.; Hasin, D.S. Predictors of Initiation of Nicotine, Alcohol, Cannabis, and Cocaine Use: Results of the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC). Am. J. Addict. 2018, 27, 477–484. [Google Scholar] [CrossRef]
- John, W.S.; Zhu, H.; Mannelli, P.; Schwartz, R.P.; Subramaniam, G.A.; Wu, L.T. Prevalence, Patterns, and Correlates of Multiple Substance Use Disorders among Adult Primary Care Patients. Drug Alcohol Depend. 2018, 187, 79–87. [Google Scholar] [CrossRef]
- Cassidy, F.; Ahearn, E.P.; Carroll, B.J. Substance Abuse in Bipolar Disorder. Bipolar Disord. 2001, 3, 181–188. [Google Scholar] [CrossRef]
- Strakowski, S.M.; DelBello, M.P.; Fleck, D.E.; Adler, C.M.; Anthenelli, R.M.; Keck, P.E.; Arnold, L.M.; Amicone, J. Effects of Co-Occurring Alcohol Abuse on the Course of Bipolar Disorder Following a First Hospitalization for Mania. Arch. Gen. Psychiatry 2005, 62, 851–858. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strakowski, S.M.; DelBello, M.P.; Fleck, D.E.; Adler, C.M.; Anthenelli, R.M.; Keck, P.E.; Arnold, L.M.; Amicone, J. Effects of Co-Occurring Cannabis Use Disorders on the Course of Bipolar Disorder after a First Hospitalization for Mania. Arch. Gen. Psychiatry 2007, 64, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Bzdok, D.; Meyer-Lindenberg, A. Machine Learning for Precision Psychiatry: Opportunities and Challenges. Biol. Psychiatry Cogn. Neurosci. Neuroimag. 2018, 3, 223–230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radua, J.; Carvalho, A.F. Route Map for Machine Learning in Psychiatry: Absence of Bias, Reproducibility, and Utility. Eur. Neuropsychopharmacol. 2021, 50, 115–117. [Google Scholar] [CrossRef]
- Hasan, M.M.; Young, G.J.; Patel, M.R.; Modestino, A.S.; Sanchez, L.D.; Noor-E-Alam, M. A Machine Learning Framework to Predict the Risk of Opioid Use Disorder. Mach. Learn. Appl. 2021, 6, 100144. [Google Scholar] [CrossRef]
- Vieta, E. Terciarismo En Psiquiatría: El Programa de Trastornos Bipolares Del Clínic de Barcelona. J. Psychiatry Ment. Health 2011, 4, 1–4. [Google Scholar] [CrossRef]
- Goodwin, G.M.; Haddad, P.M.; Ferrier, I.N.; Aronson, J.K.; Barnes, T.; Cipriani, A.; Coghill, D.R.; Fazel, S.; Geddes, J.R.; Grunze, H.; et al. Evidence-Based Guidelines for Treating Bipolar Disorder: Revised Third Edition Recommendations from the British Association for Psychopharmacology. J. Psychopharmacol. 2016, 30, 495–553. [Google Scholar] [CrossRef]
- Verdolini, N.; Hidalgo-Mazzei, D.; del Matto, L.; Muscas, M.; Pacchiarotti, I.; Murru, A.; Samalin, L.; Aedo, A.; Tohen, M.; Grunze, H.; et al. Long-Term Treatment of Bipolar Disorder Type I: A Systematic and Critical Review of Clinical Guidelines with Derived Practice Algorithms. Bipolar Disord. 2020, 23, 324–340. [Google Scholar] [CrossRef]
- APA. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; American Psychiatric Association: Arlington, VA, USA, 1994. [Google Scholar]
- APA. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; Text Rev.; American Psychiatric Association: Arlington, VA, USA, 2000. [Google Scholar]
- APA. Diagnostic and Statistical Manual of Mental Disorders: DSM-5; American Psychiatric Association (APA): Arlington, VA, USA, 2013; ISBN 089042554X. [Google Scholar]
- First, M.B.; Williams, J.B.; Karg, R.S.; Spitzer, R.L. Structured Clinical Interview for DSM-5—Research Version (SCID-5 for DSM-5, Research Version SCID-5-RV); American Psychiatric Association: Arlington, VA, USA, 2017; pp. 1–94. [Google Scholar]
- Colom, F.; Vieta, E.; Daban, C.; Pacchiarotti, I.; Sanchez-Moreno, J. Clinical and Therapeutic Implications of Predominant Polarity in Bipolar Disorder. J. Affect. Disord. 2006, 93, 13–17. [Google Scholar] [CrossRef]
- R Foundation for Statistical Computing; R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2021. [Google Scholar]
- Waring, E.; Quinn, M.; McNamara, A.; de la Rubia, E.A.; Zhu, H.; Lowndes, J.; Ellis, S.; McLeod, H.; Wickham, H.; Müller, K.; et al. Skimr: Compact and Flexible Summaries of Data, R Package, Version 1.0.7. 2019. Available online: https://CRAN.R-project.org/package=skimr (accessed on 8 June 2022).
- Wright MN, Z.A. Ranger: A Fast Implementation of Random Forests for High Dimensional Data in C++ and R. J. Stat. Softw. 2017, 77, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef] [Green Version]
- Liaw, A.; Wiener, M. Classification and Regression by RandomForest. R News 2002, 2, 18–22. [Google Scholar]
- Kuhn, M. Caret: Classification and Regression Training, R Package Version 6.0-71. 2016. Available online: https://CRAN.R-project.org/package=caret (accessed on 8 June 2022).
- Lunardon, N.; Menardi, G.; Torelli, N. ROSE: A Package for Binary Imbalanced Learning. R J. 2014, 6, 82–92. [Google Scholar]
- Baethge, C.; Baldessarini, R.J.; Khalsa, H.M.K.; Hennen, J.; Salvatore, P.; Tohen, M. Substance Abuse in First-Episode Bipolar I Disorder: Indications for Early Intervention. Am. J. Psychiatry 2005, 162, 1008–1010. [Google Scholar] [CrossRef]
- Minato Nakazawa, M. Package “fmsb” Title Functions for Medical Statistics Book with Some Demographic Data Depends R (>= 2.2.0). 2022. Available online: https://cran.r-project.org/web/packages/fmsb/fmsb.pdf (accessed on 8 June 2022).
- Fico, G.; Anmella, G.; Sagué-Villavella, M.; Gomez-Ramiro, M.; Hidalgo-Mazzei, D.; Vieta, E.; Murru, A. Undetermined Predominant Polarity in a Cohort of Bipolar Disorder Patients: Prevalent, Severe, and Overlooked. J. Affect. Disord. 2022, 303, 223–229. [Google Scholar] [CrossRef]
- Daban, C.; Colom, F.; Sanchez-Moreno, J.; García-Amador, M.; Vieta, E. Clinical Correlates of First-Episode Polarity in Bipolar Disorder. Compr. Psychiatry 2006, 47, 433–437. [Google Scholar] [CrossRef]
- Bezerra-Filho, S.; Galvao-de Almeida, A.; Studart, P.; Rocha, M.; Lopes, F.L.; Miranda-Scippa, A. Personality Disorders in Euthymic Bipolar Patients: A Systematic Review. Rev. Bras. Psiquiatr. 2015, 37, 162–167. [Google Scholar] [CrossRef] [Green Version]
- Verdolini, N.; Perugi, G.; Samalin, L.; Murru, A.; Angst, J.; Azorin, J.-M.; Bowden, C.L.; Mosolov, S.; Young, A.H.; Barbuti, M.; et al. Aggressiveness in Depression: A Neglected Symptom Possibly Associated with Bipolarity and Mixed Features. Acta Psychiatr. Scand. 2017, 136, 362–372. [Google Scholar] [CrossRef]
- Altamura, A.C.; Buoli, M.; Caldiroli, A.; Caron, L.; Cumerlato Melter, C.; Dobrea, C.; Cigliobianco, M.; Zanelli Quarantini, F. Misdiagnosis, Duration of Untreated Illness (DUI) and Outcome in Bipolar Patients with Psychotic Symptoms: A Naturalistic Study. J. Affect. Disord. 2015, 182, 70–75. [Google Scholar] [CrossRef]
- Salvatore, P.; Baldessarini, R.J.; Khalsa, H.M.K.; Vázquez, G.; Perez, J.; Faedda, G.L.; Amore, M.; Maggini, C.; Tohen, M. Antecedents of Manic versus Other First-Psychotic Episodes in 263 Bipolar-I Disorder Patients. Acta. Psychiatr. Scand. 2014, 129, 275. [Google Scholar] [CrossRef] [Green Version]
- Uher, R.; Pallaskorpi, S.; Suominen, K.; Mantere, O.; Pavlova, B.; Isometsä, E. Clinical Course Predicts Long-Term Outcomes in Bipolar Disorder. Psychol. Med. 2019, 49, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Najt, P.; Perez, J.; Sanches, M.; Peluso, M.A.M.; Glahn, D.; Soares, J.C. Impulsivity and Bipolar Disorder. Eur. Neuropsychopharmacol. 2007, 17, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Dervic, K.; Garcia-Amador, M.; Sudol, K.; Freed, P.; Brent, D.A.; Mann, J.J.; Harkavy-Friedman, J.M.; Oquendo, M.A. Bipolar I and II versus Unipolar Depression: Clinical Differences and Impulsivity/Aggression Traits. Eur. Psychiatry 2015, 30, 106–113. [Google Scholar] [CrossRef]
- Swann, A.C.; Dougherty, D.M.; Pazzaglia, P.J.; Pham, M.; Moeller, F.G. Impulsivity: A Link between Bipolar Disorder and Substance Abuse. Bipolar Disord. 2004, 6, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, M.; Morgan, T.A. The Relationship between Borderline Personality Disorder and Bipolar Disorder. Dialogues Clin. Neurosci. 2013, 15, 155. [Google Scholar] [CrossRef] [PubMed]
- Witt, S.H.; Streit, F.; Jungkunz, M.; Frank, J.; Awasthi, S.; Reinbold, C.S.; Treutlein, J.; Degenhardt, F.; Forstner, A.J.; Heilmann-Heimbach, S.; et al. Genome-Wide Association Study of Borderline Personality Disorder Reveals Genetic Overlap with Bipolar Disorder, Major Depression and Schizophrenia. Transl. Psychiatry 2017, 7, e1155. [Google Scholar] [CrossRef] [PubMed]
- Ruggero, C.J.; Zimmerman, M.; Chelminski, I.; Young, D. Borderline Personality Disorder and the Misdiagnosis of Bipolar Disorder. J. Psychiatr. Res. 2010, 44, 405–408. [Google Scholar] [CrossRef] [Green Version]
- Post, R.M.; Leverich, G.S.; McElroy, S.L.; Kupka, R.; Suppes, T.; Altshuler, L.L.; Nolen, W.A.; Frye, M.A.; Keck, P.E.; Grunze, H.; et al. Are Personality Disorders in Bipolar Patients More Frequent in the US than Europe? Eur. Neuropsychopharmacol. 2022, 58, 47–54. [Google Scholar] [CrossRef]
- Hidalgo-Mazzei, D.; Walsh, E.; Rosenstein, L.; Zimmerman, M. Comorbid Bipolar Disorder and Borderline Personality Disorder and Substance Use Disorder. J. Nerv. Ment. Dis. 2015, 203, 54–57. [Google Scholar] [CrossRef]
- Bodkyn, C.N.; Holroyd, C.B. Neural Mechanisms of Affective Instability and Cognitive Control in Substance Use. Int. J. Psychophysiol. 2019, 146, 1–19. [Google Scholar] [CrossRef]
- Heinz, A.J.; Wu, J.; Witkiewitz, K.; Epstein, D.H.; Preston, K.L. Marriage and Relationship Closeness as Predictors of Cocaine and Heroin Use. Addict. Behav. 2009, 34, 258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sletved, K.S.O.; Ziersen, S.C.; Andersen, P.K.; Vinberg, M.; Kessing, L.V. Socio-Economic Functioning in Patients with Bipolar Disorder and Their Unaffected Siblings—Results from a Nation-Wide Population-Based Longitudinal Study. Psychol. Med. 2021, 2021, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Verdolini, N.; Amoretti, S.; Mezquida, G.; Cuesta, M.J.; Pina-Camacho, L.; García-Rizo, C.; Lobo, A.; González-Pinto, A.; Merchán-Naranjo, J.; Corripio, I.; et al. The Effect of Family Environment and Psychiatric Family History on Psychosocial Functioning in First-Episode Psychosis at Baseline and after 2 Years. Eur. Neuropsychopharmacol. 2021, 49, 54–68. [Google Scholar] [CrossRef]
- Wiström, E.D.; O’Connell, K.S.; Karadag, N.; Bahrami, S.; Hindley, G.F.L.; Lin, A.; Cheng, W.; Steen, N.E.; Shadrin, A.; Frei, O.; et al. Genome-Wide Analysis Reveals Genetic Overlap between Alcohol Use Behaviours, Schizophrenia and Bipolar Disorder and Identifies Novel Shared Risk Loci. Addiction 2022, 117, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Janiri, D.; di Nicola, M.; Martinotti, G.; Janiri, L. Who’s the Leader, Mania or Depression? Predominant Polarity and Alcohol/Polysubstance Use in Bipolar Disorders. Curr. Neuropharmacol. 2017, 15, 409–416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lagerberg, T.V.; Aminoff, S.R.; Aas, M.; Bjella, T.; Henry, C.; Leboyer, M.; Pedersen, G.; Bellivier, F.; Icick, R.; Andreassen, O.A.; et al. Alcohol Use Disorders Are Associated with Increased Affective Lability in Bipolar Disorder. J. Affect. Disord. 2017, 208, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Cremaschi, L.; Dell’Osso, B.; Vismara, M.; Dobrea, C.; Buoli, M.; Ketter, T.A.; Altamura, A.C. Onset Polarity in Bipolar Disorder: A Strong Association between First Depressive Episode and Suicide Attempts. J. Affect. Disord. 2017, 209, 182–187. [Google Scholar] [CrossRef]
- Baldessarini, R.J.; Tondo, L.; Visioli, C.; Baldessarini, R.J.; Research Center, M. First-Episode Types in Bipolar Disorder: Predictive Associations with Later Illness. Acta Psychiatr. Scand. 2014, 129, 383–392. [Google Scholar] [CrossRef]
- Pallaskorpi, S.; Suominen, K.; Rosenstrom, T.; Mantere, O.; Arvilommi, P.; Valtonen, H.; Leppamaki, S.; Garcia-Estela, A.; Grande, I.; Colom, F.; et al. Predominant Polarity in Bipolar I and II Disorders: A Five-Year Follow-up Study. J. Affect. Disord. 2019, 246, 806–813. [Google Scholar] [CrossRef] [Green Version]
- Li, D.J.; Lin, C.H.; Wu, H.C. Factors Predicting Re-Hospitalization for Inpatients with Bipolar Mania—A Naturalistic Cohort. Psychiatry Res. 2018, 270, 749–754. [Google Scholar] [CrossRef]
- Fico, G.; Anmella, G.; Pacchiarotti, I.; Verdolini, N.; Sagué-Vilavella, M.; Corponi, F.; Manchia, M.; Vieta, E.; Murru, A. The Biology of Aggressive Behavior in Bipolar Disorder: A Systematic Review. Neurosci. Biobehav. Rev. 2020, 119, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Pacchiarotti, I.; di Marzo, S.; Colom, F.; Sánchez-Moreno, J.; Vieta, E. Bipolar Disorder Preceded by Substance Abuse: A Different Phenotype with Not so Poor Outcome? World J. Biol. Psychiatry 2009, 10, 209–216. [Google Scholar] [CrossRef]
- Stewart, C.; El-mallakh, R.S. Is Bipolar Disorder Overdiagnosed among Patients with Substance Abuse? Bipolar Disord. 2007, 9, 646–648. [Google Scholar] [CrossRef] [PubMed]
- Vieta, E.; Angst, J. Bipolar Disorder Cohort Studies: Crucial, but Underfunded. Eur. Neuropsychopharmacol. 2021, 47, 31–33. [Google Scholar] [CrossRef] [PubMed]
- González-Pinto, A.; Goikolea, J.M.; Zorrilla, I.; Bernardo, M.; Arrojo, M.; Cunill, R.; Castell, X.; Becoña, E.; López, A.; Torrens, M.; et al. Guía de Práctica Clínica Para El Tratamiento Farmacológico y Psicológico de Los Pacientes Adultos Con Trastorno Bipolar y Un Diagnóstico Comórbido de Trastorno Por Uso de Sustancias. Adicciones 2021, 34, 142–156. [Google Scholar] [CrossRef] [PubMed]
- Grunze, H.; Schaefer, M.; Scherk, H.; Born, C.; Preuss, U.W. Comorbid Bipolar and Alcohol Use Disorder—A Therapeutic Challenge. Front. Psychiatry 2021, 12, 660432. [Google Scholar] [CrossRef]
- Hansen, S.S.; Munk-Jørgensen, P.; Guldbæk, B.; Solgård, T.; Lauszus, K.S.; Albrechtsen, N.; Borg, L.; Egander, A.; Faurholdt, K.; Gilberg, A.; et al. Psychoactive Substance Use Diagnoses among Psychiatric In-Patients. Acta Psychiatr. Scand. 2000, 102, 432–438. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliva, V.; De Prisco, M.; Pons-Cabrera, M.T.; Guzmán, P.; Anmella, G.; Hidalgo-Mazzei, D.; Grande, I.; Fanelli, G.; Fabbri, C.; Serretti, A.; et al. Machine Learning Prediction of Comorbid Substance Use Disorders among People with Bipolar Disorder. J. Clin. Med. 2022, 11, 3935. https://doi.org/10.3390/jcm11143935
Oliva V, De Prisco M, Pons-Cabrera MT, Guzmán P, Anmella G, Hidalgo-Mazzei D, Grande I, Fanelli G, Fabbri C, Serretti A, et al. Machine Learning Prediction of Comorbid Substance Use Disorders among People with Bipolar Disorder. Journal of Clinical Medicine. 2022; 11(14):3935. https://doi.org/10.3390/jcm11143935
Chicago/Turabian StyleOliva, Vincenzo, Michele De Prisco, Maria Teresa Pons-Cabrera, Pablo Guzmán, Gerard Anmella, Diego Hidalgo-Mazzei, Iria Grande, Giuseppe Fanelli, Chiara Fabbri, Alessandro Serretti, and et al. 2022. "Machine Learning Prediction of Comorbid Substance Use Disorders among People with Bipolar Disorder" Journal of Clinical Medicine 11, no. 14: 3935. https://doi.org/10.3390/jcm11143935
APA StyleOliva, V., De Prisco, M., Pons-Cabrera, M. T., Guzmán, P., Anmella, G., Hidalgo-Mazzei, D., Grande, I., Fanelli, G., Fabbri, C., Serretti, A., Fornaro, M., Iasevoli, F., de Bartolomeis, A., Murru, A., Vieta, E., & Fico, G. (2022). Machine Learning Prediction of Comorbid Substance Use Disorders among People with Bipolar Disorder. Journal of Clinical Medicine, 11(14), 3935. https://doi.org/10.3390/jcm11143935










