Effect of Third and Fourth mRNA-Based Booster Vaccinations on SARS-CoV-2 Neutralizing Antibody Titer Formation, Risk Factors for Non-Response, and Outcome after SARS-CoV-2 Omicron Breakthrough Infections in Patients on Chronic Hemodialysis: A Prospective Multicenter Cohort Study
Abstract
:1. Introduction
2. Methods
2.1. Statistical Outcome Parameter, Cohort, and Case Series Definitions
2.2. SARS-CoV-2 Antibody Test Assays
2.3. SARS-CoV-2 PCR-Test Assay
2.4. Statistical Analyses
3. Results
3.1. Effect of a Third Booster Vaccination on SARS-CoV-2 IgG Antibodies and Their Neutralizing Capacity (A)
3.2. Risk Factor Analysis for Immunological Non-Response (B)
3.3. Sensitivity Analysis
3.4. Effect of ≥4 Booster Vaccinations on SARS-CoV-2 IgG Antibodies and Their Neutralizing Capacity in Low- and/or Non-Responders (C)
3.5. Clinical Course of Breakthrough Infections in 20 Patients on Chronic Hemodialysis (D)
4. Discussion
Major Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yang, Z.H.; Pan, X.T.; Chen, Y.; Wang, L.; Chen, Q.X.; Zhu, Y.; Zhu, Y.-J.; Chen, Y.-X.; Chen, Y.-N. Psychological profiles of Chinese patients with hemodialysis during the pandemic of coronavirus disease 2019. Front. Psychiatry 2021, 12, 616016. [Google Scholar] [CrossRef] [PubMed]
- Couchoud, C.; Bayer, F.; Ayav, C.; Béchade, C.; Brunet, P.; Chantrel, F.; Frimat, L.; Galland, R.; Laurain, E.; Lobbedez, T.; et al. Low incidence of SARS-CoV-2, risk factors of mortality and the course of illness in the French national cohort of dialysis patients. Kidney Int. 2020, 98, 1519–1529. [Google Scholar] [CrossRef] [PubMed]
- Alberici, F.; Delbarba, E.; Manenti, C.; Manenti, C.; Econimo, L.; Valerio, F.; Pola, A.; Maffei, C.; Possenti, S.; Lucca, B.; et al. A report from the Brescia Renal COVID Task Force on the clinical characteristics and short-term outcome of hemodialysis patients with SARS-CoV-2 infection. Kidney Int. 2020, 98, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Hoxha, E.; Suling, A.; Turner, J.J.E.; Haubitz, M.; Floege, J.; Huber, T.B.; Galle, J.C. COVID-19 prevalence, and mortality in chronic dialysis patients. Dtsch. Arztebl. Int. 2021, 118, 195–196. [Google Scholar] [CrossRef] [PubMed]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef]
- Semenzato, L.; Botton, J.; Drouin, J.; Cuneot, F.; Dray-Spira, R.; Weill, A.; Zureik, M. Chronic diseases, health conditions and risk of COVID-19-related hospitalization and in-hospital mortality during the first wave of the epidemic in France: A cohort study of 66 million people. Lancet Reg. Health Eur. 2021, 8, 100158. [Google Scholar] [CrossRef]
- Clarke, C.; Prendecki, M.; Dhutia, A.; Ali, M.A.; Sajjad, H.; Shivakumar, O.; Lightstone, L.; Kelleher, P.; Pickering, M.C.; Thomas, D.; et al. High prevalence of asymptomatic COVID-19 infection in hemodialysis patients detected using serologic screening. J. Am. Soc. Nephrol. 2020, 31, 1969–1975. [Google Scholar] [CrossRef]
- Yau, K.; Muller, M.P.; Lin, M.; Siddiqui, N.; Neskovic, S.; Shokar, G.; Fattouh, R.; Matukas, L.M.; Beaubien-Souligny, W.; Thomas, A.; et al. COVID-19 outbreak in an urban hemodialysis unit. Am. J. Kidney Dis. 2020, 76, 690–695.e1. [Google Scholar] [CrossRef]
- Goicoechea, M.; Sánchez Cámara, L.A.; Macías, N.; de Morales, A.M.; Rojas, A.G.; Basunana, A.; Arroyo, D.; Vega, A.; Abad, S.; Verde, E.; et al. COVID-19, clinical course and outcomes of 36 hemodialysis patients in Spain. Kidney Int. 2020, 98, 27–34. [Google Scholar] [CrossRef]
- Lano, G.; Braconnier, A.; Bataille, S.; Cavaille, G.; Moussi-Frances, J.; Gondouin, B.; Bindi, P.; Nakhla, M.; Mansour, J.; Halin, P.; et al. Risk factors for severity of COVID-19 in chronic dialysis patients from a multicentre French cohort. Clin. Kidney J. 2020, 13, 878–888. [Google Scholar] [CrossRef]
- Tao, K.; Tzou, P.L.; Nouhin, J.; Gupta, R.K.; de Oliveira, T.; Kosakovsky Pond, S.; Fera, D.; Shafer, R.W. The biological and clinical significance of emerging SARS-CoV-2 variants. Nat. Rev. Genet. 2021, 22, 757–773. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Nguyen, T.T.; Taitt, A.S.; Jhun, H.; Park, H.J.; Kim, S.H.; Kim, Y.G.; Song, E.Y.; Lee, Y.; Yum, H.; et al. SARS-CoV-2 Omicron Mutation Is Faster than the Chase: Multiple Mutations on Spike/ACE2 Interaction Residues. Immune Netw. 2021, 21, e3. [Google Scholar] [CrossRef] [PubMed]
- Ai, J.; Zhang, H.; Zhang, Y.; Lin, K.; Zhang, Y.; Wu, J.; Wan, Y.; Huang, Y.; Song, J.; Fu, Z.; et al. Omicron variant showed lower neutralizing sensitivity than other SARS-CoV-2 variants to immune sera elicited by vaccines after boost. Emerg. Microbes Infect. 2022, 11, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Krüger, N.; Schulz, S.; Cossmann, A.; Rocha, C.; Kempf, A.; Nehlmeier, I.; Graichen, L.; Moldenhauer, A.S.; Winkler, M.S.; et al. The Omicron variant is highly resistant against antibody-mediated neutralization: Implications for control of the COVID-19 pandemic. Cell 2022, 3, 447–456.e11. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.rki.de/DE/Content/InfAZ/N/Neuartiges_Coronavirus/Situationsbericht/Wochenbericht/Wochenbericht_2022-03-03.pdf?__blob=publicationFile (accessed on 13 March 2022).
- Carr, E.J.; Kronbichler, A.; Graham-Brown, M.; Abra, G.; Argyropoulos, C.; Harper, L.; Lerma, E.V.; Suri, R.S.; Topf, J.; Willicombe, M.; et al. Review of early immune response to SARS-CoV-2 vaccination among patients with CKD. Kidney Int. Rep. 2021, 6, 2292–2304. [Google Scholar] [CrossRef]
- Frantzen, L.; Cavaille, G.; Thibeaut, S.; El-Haik, Y. Efficacy of the BNT162b2 mRNA COVID-19 vaccine in a hemodialysis cohort. Nephrol. Dial. Transpl. 2021, 36, 1756–1757. [Google Scholar] [CrossRef]
- Carr, E.J.; Wu, M.; Harvey, R.; Billany, R.E.; Wall, E.C.; Kelly, G.; Howell, M.; Kassiotis, G.; Swanton, C.; Gandhi, S. Omicron neutralising antibodies after COVID-19 vaccination in haemodialysis patients. Lancet 2022, 399, 800–802. [Google Scholar] [CrossRef]
- Bensouna, I.; Caudwell, V.; Kubab, S.; Acquaviva, S.; Pardon, A.; Vittoz, N.; Bozman, D.F.; Hanafi, L.; Faucon, A.L.; Housset, P. SARS-CoV-2 antibody response after a third dose of the BNT162b2 vaccine in patients receiving maintenance hemodialysis or peritoneal dialysis. Am. J. Kidney Dis. 2022, 79, 185–192.e1. [Google Scholar] [CrossRef]
- Patyna, S.; Eckes, T.; Koch, B.F.; Sudowe, S.; Oftring, A.; Kohmer, N.; Rabenau, H.F.; Cisek, S.; Avaniadi, D.; Steiner, R.; et al. Impact of Moderna mRNA-1273 booster vaccine on fully vaccinated high-risk chronic dialysis patients after loss of humoral response. Vaccines 2022, 10, 585. [Google Scholar] [CrossRef]
- Broseta, J.J.; Rodríguez-Espinosa, D.; Cuadrado, E.; Rodríguez, N.; Bedini, J.L.; Maduell, F. Humoral response after three doses of mRNA-1273 or BNT162b2 SARS-CoV-2 vaccines in hemodialysis patients. Vaccines 2022, 10, 522. [Google Scholar] [CrossRef]
- Garcia, P.; Han, J.; Montez-Rath, M.; Sun, S.; Shang, T.; Parsonnet, J.; Chertow, G.M.; Anand, S.; Schiller, B.; Abrag, G. SARS-CoV-2 booster vaccine response among patients receiving dialysis. Clin. J. Am. Soc. Nephrol. 2022; ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Benning, L.; Klein, K.; Morath, C.; Bartenschlager, M.; Kim, H.; Buylaert, M.; Reineke, M.; Töllner, M.; Nusshag, C.; Kälble, F.; et al. Neutralizing antibody activity against the B.1.617.2 (delta) variant before and after a third BNT162b2 vaccine dose in hemodialysis patients. Front. Immunol. 2022, 13, 840136. [Google Scholar] [CrossRef] [PubMed]
- Shashar, M.; Nacasch, N.; Grupper, A.; Benchetrit, S.; Halperin, T.; Erez, D.; Rozenberg, I.; Shitrit, P.; Sela, Y.; Wand, O.; et al. Humoral response to Pfizer BNT162b2 vaccine booster in maintenance hemodialysis patients. Am. J. Nephrol. 2022, 53, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Notarte, K.I.; Guerrero-Arguero, I.; Velasco, J.V.; Ver, A.T.; Santos de Oliveira, M.H.; Catahay, J.A.; Khan, M.D.S.R.; Pastrana, A.; Juszczyk, G.; Torelles, J.B.; et al. Characterization of the significant decline in humoral immune response six months post-SARS-CoV-2 mRNA vaccination: A systematic review. J. Med. Virol. 2022, 94, 2939–2961. [Google Scholar] [CrossRef] [PubMed]
- Speer, C.; Schaier, M.; Husshag, C.; Töllner, M.; Buylaert, M.; Kälble, F.; Reichel, P.; Grenz, J.; Süsal, C.; Zeier, M.; et al. Longitudinal humoral responses after COVID-19 vaccination in peritoneal and hemodialysis patients over twelve weeks. Vaccines 2021, 9, 1130. [Google Scholar] [CrossRef] [PubMed]
- Tillmann, F.P.; Still, H.; von Landenberg, P. Long-term trajectories of SARS-CoV-2 neutralizing antibodies and predictive value of first dose vaccination-induced IgG-antibodies in hemodialysis patients. Int. Urol. Nephrol. 2021; ahead of print. [Google Scholar] [CrossRef]
- Clarke, C.L.; Prendecki, M.; Dhutia, A.; Gan, J.; Edwards, C.; Prout, V.; Lightstone, L.; Parker, E.; Marchesin, F.; Griffith, M.; et al. Longevity of SARS-CoV-2 immune responses in hemodialysis patients and protection against reinfection. Kidney Int. 2021, 99, 1470–1477. [Google Scholar] [CrossRef]
- Feng, S.; Phillips, D.J.; White, T.; Sayal, H.; Aley, P.K.; Bibi, S.; Dold, C.; Fuskova, M.; Gilbert, S.C.; Hirsch, I.; et al. Correlates of protection against symptomatic and asymptomatic SARS-CoV-2 infection. Nat. Med. 2021, 27, 2032–2040. [Google Scholar] [CrossRef]
- WHO/BS.2020.2403; Establishment of the WHO International Standard and Reference Panel for Anti-SARS-CoV-2 Antibody. Available online: https://www.who.int/publications/m/item/WHOBS-2020.2403 (accessed on 12 March 2022).
- Van Praet, J.; Reynders, M.; De Bacquer, D.; Viaene, L.; Schoutteten, M.K.; Caluwé, R.; Doubel, P.; Heylen, L.; de Bel, A.V.; van Vlem, B.; et al. Predictors and dynamics of the humoral and cellular immune response to SARS-CoV-2 mRNA vaccines in hemodialysis patients: A multicenter observational study. J. Am. Soc. Nephrol. 2021, 32, 3208–3220. [Google Scholar] [CrossRef]
- Broseta, J.J.; Rodríguez-Espinosa, D.; Rodríguez, N.; Del Mar Mosquera, M.; Marcos, M.A.; Egri, N.; Pascal, M.; Soruco, E.; Bedini, J.L.; Bayes, B.; et al. Humoral and cellular responses to mRNA-1273 and BNT162b2 SARS-CoV-2 vaccines administered to hemodialysis patients. Am. J. Kidney Dis. 2021, 78, 571–581. [Google Scholar] [CrossRef]
- Anand, S.; Montez-Rath, M.E.; Han, J.; Garcia, P.; Cadden, L.C.; Hunsader, P.; Kerschmann, R.; Beyer, P.; Dittrich, M.; Block, G.A.; et al. Antibody response to COVID-19 vaccination in patients receiving dialysis. J. Am. Soc. Nephrol. 2021, 32, 2435–2438. [Google Scholar] [CrossRef] [PubMed]
- Planas, D.; Bruel, T.; Grzelak, L.; Guivel-Benhassine, F.; Staropoli, I.; Porrot, F.; Planchais, C.; Buchrieser, J.; Rajah, M.M.; Bishop, E.; et al. Sensitivity of infectious SARS-CoV-2 B.1.1.7 and B.1.351 variants to neutralizing antibodies. Nat. Med. 2021, 27, 917–924. [Google Scholar] [CrossRef] [PubMed]
- Anand, S.; Montez-Rath, M.E.; Han, J.; Garcia, P.; Cadden, L.C.; Hunsader, P.; Morgan, C.; Kerschmann, R.; Beyer, P.; Dittrich, M.; et al. SARS-CoV-2 Vaccine Antibody Response and Breakthrough Infection in Patients Receiving Dialysis. Ann. Intern. Med. 2022, 175, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Abu-Raddad, L.J.; Chemaitelly, H.; Ayoub, H.H.; Yassine, H.M.; Al-Khatip, H.A.; Smatti, M.K.; Tang, P.; Hasan, M.R.; Coyle, P.; Al-Kanaani, Z. Effect of mRNA Vaccine Boosters against SARS-CoV-2 Omicron Infection in Qatar. N. Engl. J. Med. 2022, 386, 1804–1816. [Google Scholar] [CrossRef]
- Tan, S.H.X.; Pung, R.; Wang, L.F.; Chien Lye, D.; Ong, B.; Cook, A.R.; Tan, K.B. Association of Homologous and Heterologous Vaccine Boosters With COVID-19 Incidence and Severity in Singapore. JAMA 2022, 327, 1181–1182. [Google Scholar] [CrossRef]
- Andrews, N.; Stow, J.; Kirsebom, F.; Toffa, S.; Rickeard, T.; Gallagher, E.; Gower, C.; Kall, M.; Groves, N.; O’Connell, A.M.; et al. COVID-19 Vaccine Effectiveness against the Omicron (B.1.1.529) Variant. N. Engl. J. Med. 2022, 386, 1532–1546. [Google Scholar] [CrossRef]
- Available online: https://www.businesswire.com/news/home/20211126005595/en/Moderna-Announces-Strategy-to-Address-Omicron-B.1.1.529-SARS-CoV-2-Variant (accessed on 13 March 2022).
- Chinnadurai, R.; Wu, H.H.L.; Cox, E.; Moore, J.; Clough, T.; Lamerton, E.; Donne, R.; O’Riordan, E.; Poulikakos, D. Humoral Response in Hemodialysis Patients Following COVID-19 Vaccination and Breakthrough Infections during Delta and Omicron Variant Predominance. Vaccines 2022, 10, 498. [Google Scholar] [CrossRef]
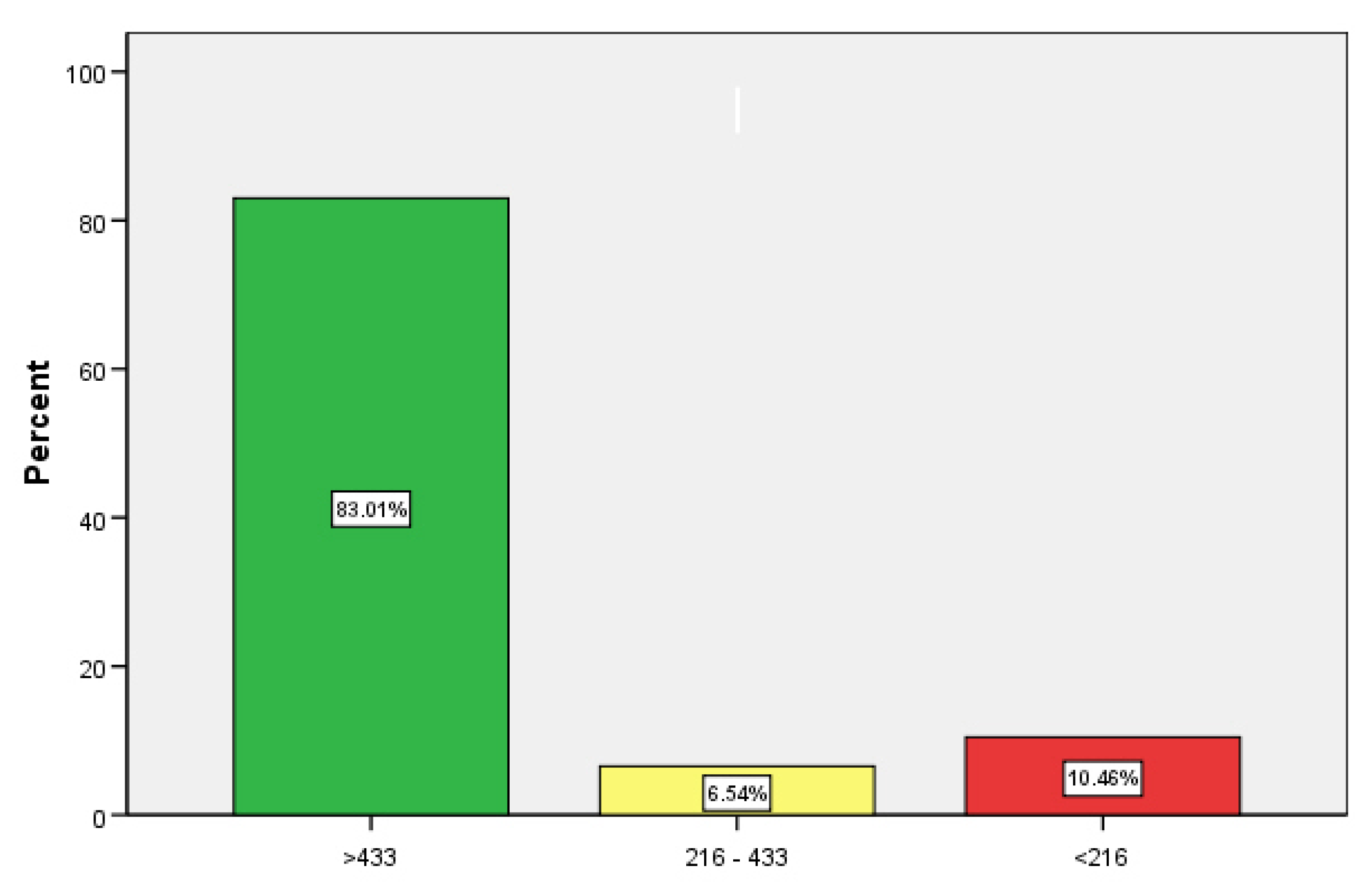
| Categories | Post Second Vaccination (No, %) | Post Third Vaccination (No, %) | Somers‘d | |||
|---|---|---|---|---|---|---|
| IgG | >433 | 84 | 54.9% | 127 | 83.0% | <0.001 |
| 216–433 | 10 | 6.5% | 10 | 6.5% | ||
| <216 | 59 | 38.6% | 16 | 10.5% | ||
| % inhibition | >66 | 99 | 64.7% | 136 | 88.9% | 0.003 |
| 33–66 | 23 | 15.0% | 7 | 4.6% | ||
| <33 | 31 | 20.3% | 10 | 6.5% | ||
| (a) | |||||
| Variable | RC-B | p-Level | Exponent-B | 95%-CI | |
| boost to lab (days) | 0.016 | 0.082 | 1.016 | 0.998 | 1.034 |
| Age (years) | 0.034 | 0.200 | 1.035 | 0.982 | 1.090 |
| Active malignancy | 1.142 | 0.300 | 3.134 | 0.362 | 27.170 |
| (b) | |||||
| Variable | RC-B | p-Level | Exponent-B | 95%-CI | |
| boost to lab (days) | 0.008 | 0.287 | 1.008 | 0.993 | 1.023 |
| Age (years) | 0.064 | 0.004 | 1.066 | 1.020 | 1.114 |
| Active malignancy | 0.458 | 0.617 | 1.581 | 0.263 | 9.494 |
| isMedication | 2.112 | 0.002 | 8.267 | 2.206 | 30.975 |
| No. | Age | BMI | Diabetes | pKTx | Vintage | Vaccination | isMeds | aTumor | Albumin | Kt/V | IgG | nTiter |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. | 50–55 | 23.0 | / | Yes | 1.59 | 5 times mRNA | ldST/Tacrolimus | / | 2843 | 1.1 | >433 | 87 |
| 2. | 85–90 | 32.4 | / | / | 0.93 | 4 times mRNA | ldST | Yes | 3116 | 1.1 | 0 | 5 |
| 3. | 65–70 | 27.7 | / | / | 3.70 | 4 times mRNA | / | / | 3624 | 1.4 | >433 | 94 |
| 4. | 95–100 | 30.6 | / | / | 1.77 | 1 vector + 3 mRNA | ldST | / | 3526 | 0.8 | 211 | 88 |
| 5. | 60–65 | 31.3 | / | / | 1.11 | 4 times mRNA | ST/Cyclophosphamide | / | 3481 | 1.1 | >433 | 98 |
| 6. | 85–90 | 25.7 | / | / | 2.38 | 4 times mRNA | / | / | 3829 | 0.9 | >433 | 100 |
| 7. | 80–85 | 23.7 | Yes | / | 5.82 | 4 times mRNA | / | / | 3069 | 1.0 | >433 | 100 |
| 8. | 75–80 | 23.8 | / | / | 6.12 | 4 times mRNA | ST/Daratumumab | Yes | 3450 | 1.3 | >433 | 99 |
| 9. | 80–85 | 20.0 | Yes | / | 5.91 | 4 times mRNA | / | / | 3330 | 1.4 | >433 | 58 |
| 10. | 90–95 | 22.2 | / | / | 2.69 | 4 times mRNA | ldST | / | 3333 | 0.9 | >433 | 83 |
| No. | Age | BMI | Diabetes | pKTx | Vintage | Vaccination | isMeds | IgG | nTiter | CT | Remission/Days | Course |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. | 50–55 | 22 | No | Yes | 7.2 | 3 times mRNA | ldST | >433 | 100 | 21 | 18 | Mild |
| 2. | 55–60 | 31 | No | Yes | 10.5 | 3 times mRNA | no | >433 | 100 | 17 | 14 | Mild |
| 3. | 55–60 | 29 | No | No | 2.1 | 3 times mRNA | no | 0 | <30 | 17 | 16 | Mild |
| 4. | 30–35 | 19 | No | Yes | 5.8 | 2 times mRNA | no | >433 | 98 | 22 | 7 | Mild |
| 5. | 60–65 | 35 | Yes | No | 0.4 | 2 times mRNA | no | 70 | 48 | 9 | 29 | Mild |
| 6. | 70–75 | 28 | Yes | No | 1.6 | wild, 2 times mRNA | no | >433 | 84 | 15 | 12 | Mild |
| 7. | 50–55 | 32 | Yes | No | 4.0 | 3 times mRNA | no | >433 | 99 | 13 | 8 | Mild |
| 8. | 35–40 | 31 | Yes | Yes | 2.1 | 3 times mRNA | no | >433 | 100 | 18 | 13 | Mild |
| 9. | 80–85 | 19 | No | No | 3.4 | 3 times mRNA | no | >433 | 100 | 18 | 13 | Mild |
| 10. | 30–35 | 20 | Yes | No | 0.7 | 3 times mRNA | no | >433 | 97 | 14 | 15 | Mild |
| 11. | 80–85 | 24 | No | No | 0.9 | 4 times mRNA | no | >433 | 99 | 16 | 14 | Mild |
| 12. | 70–75 | 24 | No | No | 4.4 | 3 times mRNA | no | >433 | 100 | 16 | 12 | Mild |
| 13. | 75–80 | 20 | No | No | 7.9 | 3 times mRNA | no | >433 | 99 | 13 | 11 | Mild |
| 14. | 60–65 | 30 | Yes | No | 0.8 | 3 times mRNA | ldST | 372 | 51 | 19 | 8 | Mild |
| 15. | 60–65 | 25 | No | No | 3.8 | 3 times mRNA | No | >433 | 98 | 21 | 7 | Mild |
| 16. | 50–55 | 27 | No | Yes | 1.6 | 4 times mRNA | No | >433 | 97 | 18 | 10 | Mild |
| 17. | 70–75 | 23 | No | No | 14.7 | 3 times mRNA | No | >433 | 99 | 16 | 12 | Mild |
| 18. | 60–65 | 32 | No | No | 1.1 | 3 times mRNA | ldST | >433 | 98 | 19 | 12 | Mild |
| 19. | 75–80 | 25 | No | No | 8.7 | 3 times mRNA | No | >433 | 100 | 12 | 8 | Mild |
| 20. | 80–85 | 19 | Yes | No | 0.6 | 1 time mRNA | ST | 0 | 0 | 14 | 12 | Mild |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tillmann, F.-P.; Figiel, L.; Ricken, J.; Still, H.; Korte, C.; Plaßmann, G.; Harth, A.; Jörres, A.; von Landenberg, P. Effect of Third and Fourth mRNA-Based Booster Vaccinations on SARS-CoV-2 Neutralizing Antibody Titer Formation, Risk Factors for Non-Response, and Outcome after SARS-CoV-2 Omicron Breakthrough Infections in Patients on Chronic Hemodialysis: A Prospective Multicenter Cohort Study. J. Clin. Med. 2022, 11, 3187. https://doi.org/10.3390/jcm11113187
Tillmann F-P, Figiel L, Ricken J, Still H, Korte C, Plaßmann G, Harth A, Jörres A, von Landenberg P. Effect of Third and Fourth mRNA-Based Booster Vaccinations on SARS-CoV-2 Neutralizing Antibody Titer Formation, Risk Factors for Non-Response, and Outcome after SARS-CoV-2 Omicron Breakthrough Infections in Patients on Chronic Hemodialysis: A Prospective Multicenter Cohort Study. Journal of Clinical Medicine. 2022; 11(11):3187. https://doi.org/10.3390/jcm11113187
Chicago/Turabian StyleTillmann, Frank-Peter, Lars Figiel, Johannes Ricken, Hermann Still, Christoph Korte, Grete Plaßmann, Ana Harth, Achim Jörres, and Philipp von Landenberg. 2022. "Effect of Third and Fourth mRNA-Based Booster Vaccinations on SARS-CoV-2 Neutralizing Antibody Titer Formation, Risk Factors for Non-Response, and Outcome after SARS-CoV-2 Omicron Breakthrough Infections in Patients on Chronic Hemodialysis: A Prospective Multicenter Cohort Study" Journal of Clinical Medicine 11, no. 11: 3187. https://doi.org/10.3390/jcm11113187
APA StyleTillmann, F.-P., Figiel, L., Ricken, J., Still, H., Korte, C., Plaßmann, G., Harth, A., Jörres, A., & von Landenberg, P. (2022). Effect of Third and Fourth mRNA-Based Booster Vaccinations on SARS-CoV-2 Neutralizing Antibody Titer Formation, Risk Factors for Non-Response, and Outcome after SARS-CoV-2 Omicron Breakthrough Infections in Patients on Chronic Hemodialysis: A Prospective Multicenter Cohort Study. Journal of Clinical Medicine, 11(11), 3187. https://doi.org/10.3390/jcm11113187






