CD24 Is a Prognostic Marker for Multiple Myeloma Progression and Survival
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patient Samples
2.2. Data Collection
2.3. Immunophenotyping of the Samples
2.4. Statistical Analysis
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rajkumar, S.V. Multiple myeloma: 2020 update on diagnosis, risk-stratification and management. Am. J. Hematol. 2020, 95, 548–567. [Google Scholar] [CrossRef] [Green Version]
- Rajkumar, S.V.; Dimopoulos, M.A.; Palumbo, A.; Blade, J.; Merlini, G.; Mateos, M.-V.; Kumar, S.; Hillengass, J.; Kastritis, E.; Richardson, P.; et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014, 15, e538–e548. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Rajkumar, S.V. Multiple myeloma: Every year a new standard? Hematol. Oncol. 2019, 37, 62–65. [Google Scholar] [CrossRef] [Green Version]
- Minnie, S.A.; Hill, G. Immunotherapy of multiple myeloma. J. Clin. Investig. 2020, 130, 1565–1575. [Google Scholar] [CrossRef]
- Manier, S.; Salem, K.Z.; Park, J.; Landau, D.A.; Getz, G.; Ghobrial, I.M. Genomic complexity of multiple myeloma and its clinical implications. Nat. Rev. Clin. Oncol. 2017, 14, 100–113. [Google Scholar] [CrossRef]
- Giannakoulas, N.; Ntanasis-Stathopoulos, I.; Terpos, E. The Role of Marrow Microenvironment in the Growth and Development of Malignant Plasma Cells in Multiple Myeloma. Int. J. Mol. Sci. 2021, 22, 4462. [Google Scholar] [CrossRef]
- Aigner, S.; Sthoeger, Z.M.; Fogel, M.; Weber, E.; Zarn, J.; Ruppert, M.; Zeller, Y.; Vestweber, D.; Stahel, R.; Sammar, M.; et al. CD24, a Mucin-Type Glycoprotein, Is a Ligand for P-Selectin on Human Tumor Cells. Blood 1997, 89, 3385–3395. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, W. CD24, A Review of its Role in Tumor Diagnosis, Progression and Therapy. Curr. Gene Ther. 2020, 20, 109–126. [Google Scholar] [CrossRef]
- Duex, J.E.; Owens, C.; Chauca-Diaz, A.; Dancik, G.M.; Vanderlinden, L.A.; Ghosh, D.; Leivo, M.Z.; Hansel, D.E.; Theodorescu, D. Nuclear CD24 Drives Tumor Growth and Is Predictive of Poor Patient Prognosis. Cancer Res. 2017, 77, 4858–4867. [Google Scholar] [CrossRef] [Green Version]
- Gilad, N.; Zukerman, H.; Pick, M.; Gatt, M.E. The role of CD24 in multiple myeloma tumorigenicity and effects of the microenvironment on its expression. Oncotarget 2019, 10, 5480–5491. [Google Scholar] [CrossRef] [PubMed]
- Alaterre, E.; Raimbault, S.; Goldschmidt, H.; Bouhya, S.; Requirand, G.; Robert, N.; Boireau, S.; Seckinger, A.; Hose, D.; Klein, B.; et al. CD24, CD27, CD36 and CD302 gene expression for outcome prediction in patients with multiple myeloma. Oncotarget 2017, 8, 98931–98944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jelínek, T.; Bezdekova, R.; Zátopková, M.; Burgos, L.; Simicek, M.; Sevcikova, T.; Paiva, B.; Hajek, R. Current applications of multiparameter flow cytometry in plasma cell disorders. Blood Cancer J. 2017, 7, e617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonsalves, W.I.; Timm, M.M.; Rajkumar, S.; Morice, W.G.; Dispenzieri, A.; Buadi, F.K.; Lacy, M.Q.; Dingli, D.; Leung, N.; Kapoor, P.; et al. The prognostic significance of CD45 expression by clonal bone marrow plasma cells in patients with newly diagnosed multiple myeloma. Leuk. Res. 2016, 44, 32–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- ElMenshawy, N.; Farag, N.A.; Atia, D.M.; Abousamra, N.; Shahin, D.; Fawzi, E.; Ghazi, H.; El-Kott, A.F.; Eissa, M. Prognostic Relevance of Concordant Expression CD69 and CD56 in Response to Bortezomib Combination Therapy in Multiple Myeloma Patients. Cancer Investig. 2021, 39, 777–782. [Google Scholar] [CrossRef]
- Koumpis, E.; Tassi, I.; Malea, T.; Papathanasiou, K.; Papakonstantinou, I.; Serpanou, A.; Tsolas, E.; Kapsali, E.; Vassilakopoulos, T.P.; Papoudou-Bai, A.; et al. CD56 expression in multiple myeloma: Correlation with poor prognostic markers but no effect on outcome. Pathol.-Res. Pract. 2021, 225, 153567. [Google Scholar] [CrossRef]
- Schmidt-Hieber, M.; Perez-Andres, M.; Paiva, B.; Flores-Montero, J.; Perez, J.J.; Gutierrez, N.C.; Vidriales, M.-B.; Matarraz, S.; Miguel, J.F.S.; Orfao, A. CD117 expression in gammopathies is associated with an altered maturation of the myeloid and lymphoid hematopoietic cell compartments and favorable disease features. Haematologica 2010, 96, 328–332. [Google Scholar] [CrossRef] [Green Version]
- Paiva, B.; Gutiérrez, N.-C.; Chen, X.; Vídriales, M.-B.; Montalbán, M.Á.; Rosiñol, L.; Oriol, A.; Martínez-López, J.; Mateos, M.-V.; López-Corral, L.; et al. Clinical significance of CD81 expression by clonal plasma cells in high-risk smoldering and symptomatic multiple myeloma patients. Leukemia 2012, 26, 1862–1869. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, T.; Ri, M.; Kinoshita, S.; Narita, T.; Totani, H.; Ashour, R.; Ito, A.; Kusumoto, S.; Ishida, T.; Komatsu, H.; et al. Low expression of neural cell adhesion molecule, CD56, is associated with low efficacy of bortezomib plus dexamethasone therapy in multiple myeloma. PLoS ONE 2018, 13, e0196780. [Google Scholar] [CrossRef] [Green Version]
- Palumbo, A.; Avet-Loiseau, H.; Oliva, S.; Lokhorst, H.M.; Goldschmidt, H.; Rosinol, L.; Richardson, P.; Caltagirone, S.; Lahuerta, J.J.; Facon, T.; et al. Revised International Staging System for Multiple Myeloma: A Report from International Myeloma Working Group. J. Clin. Oncol. 2015, 33, 2863–2869. [Google Scholar] [CrossRef]
- Kumar, S.; Paiva, B.; Anderson, K.C.; Durie, B.; Landgren, O.; Moreau, P.; Munshi, N.; Lonial, S.; Bladé, J.; Mateos, M.-V.; et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol. 2016, 17, e328–e346. [Google Scholar] [CrossRef]
- Comenzo, R.L.; Reece, D.; Palladini, G.; Seldin, D.; Sanchorawala, V.; Landau, H.; Falk, R.; Wells, K.; Solomon, A.; Wechalekar, A.; et al. Consensus guidelines for the conduct and reporting of clinical trials in systemic light-chain amyloidosis. Leukemia 2012, 26, 2317–2325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paiva, B.; Vidriales, M.-B.; Mateo, G.; Pérez, J.J.; Montalbán, M.A.; Sureda, A.; Montejano, L.; Gutiérrez, N.C.; de Coca, A.G.; Heras, N.D.L.; et al. The persistence of immunophenotypically normal residual bone marrow plasma cells at diagnosis identifies a good prognostic subgroup of symptomatic multiple myeloma patients. Blood 2009, 114, 4369–4372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caers, J.; Garderet, L.; Kortüm, K.M.; O’Dwyer, M.E.; Van De Donk, N.W.; Binder, M.; Dold, S.M.; Gay, F.; Corre, J.; Beguin, Y.; et al. European Myeloma Network recommendations on tools for the diagnosis and monitoring of multiple myeloma: What to use and when. Haematologica 2018, 103, 1772–1784. [Google Scholar] [CrossRef]
- Rawstron, A.C.; Orfao, A.; Beksac, M.; Bezdickova, L.; Brooimans, R.A.; Bumbea, H.; Dalva, K.; Fuhler, G.; Gratama, J.; Hose, D.; et al. Report of the European Myeloma Network on multiparametric flow cytometry in multiple myeloma and related disorders. Haematologica 2008, 93, 431–438. [Google Scholar] [CrossRef] [Green Version]
- Flores-Montero, J.; De Tute, R.; Paiva, B.D.L.; Perez, J.J.; Böttcher, S.; Wind, H.; Sanoja, L.; Puig, N.; Lecrevisse, Q.; Vidriales, M.-B.; et al. Immunophenotype of normal vs. myeloma plasma cells: Toward antibody panel specifications for MRD detection in multiple myeloma. Cytom. Part B Clin. Cytom. 2015, 90, 61–72. [Google Scholar] [CrossRef]
- Paiva, B.D.L.; Vidriales, M.-B.; Pérez, J.J.; Mateo, G.; Montalbán, M.A.; Mateos, M.V.; Bladé, J.; Lahuerta, J.J.; Orfao, A.; Miguel, J.F.S. Multiparameter flow cytometry quantification of bone marrow plasma cells at diagnosis provides more prognostic information than morphological assessment in myeloma patients. Haematologica 2009, 94, 1599–1602. [Google Scholar] [CrossRef]
- Paiva, B.D.L.; Almeida, J.; Pérez-Andrés, M.; Mateo, G.; López, A.; Rasillo, A.; Vídriales, M.-B.; López-Berges, M.-C.; Miguel, J.F.S.; Orfao, A. Utility of flow cytometry immunophenotyping in multiple myeloma and other clonal plasma cell-related disorders. Cytom. Part B Clin. Cytom. 2010, 78, 239–252. [Google Scholar] [CrossRef]
- Mateo, G.; Montalbán, M.A.; Vidriales, M.-B.; Lahuerta, J.J.; Mateos, M.V.; Gutierrez, N.; Rosiñol, L.; Montejano, L.; Bladé, J.; Martínez, R.; et al. Prognostic Value of Immunophenotyping in Multiple Myeloma: A Study by the PETHEMA/GEM Cooperative Study Groups on Patients Uniformly Treated with High-Dose Therapy. J. Clin. Oncol. 2008, 26, 2737–2744. [Google Scholar] [CrossRef]
- Ngo, N.-T.; Brodie, C.; Giles, C.; Horncastle, D.; Klammer, M.; Lampert, I.A.; Rahemtulla, A.; Naresh, K.N. The significance of tumour cell immunophenotype in myeloma and its impact on clinical outcome. J. Clin. Pathol. 2009, 62, 1009–1015. [Google Scholar] [CrossRef]
- Murray, M.E.; Gavile, C.M.; Nair, J.R.; Koorella, C.; Carlson, L.M.; Buac, D.; Utley, A.; Chesi, M.; Bergsagel, P.L.; Boise, L.; et al. CD28-mediated pro-survival signaling induces chemotherapeutic resistance in multiple myeloma. Blood 2014, 123, 3770–3779. [Google Scholar] [CrossRef] [PubMed]
- Bataille, R.; Jégo, G.; Robillard, N.; Barillé-Nion, S.; Harousseau, J.-L.; Moreau, P.; Amiot, M.; Pellat-Deceunynck, C. The Phenotype of Normal, Reactive and Malignant Plasma Cells. Identification of “Many and Multiple Myelomas” and of New Targets for Myeloma Therapy. Haematologica 2006, 91, 1234–1240. [Google Scholar] [PubMed]
- Kristiansen, G.; Schlüns, K.; Yongwei, Y.; Denkert, C.; Dietel, M.; Petersen, I. CD24 is an independent prognostic marker of survival in nonsmall cell lung cancer patients. Br. J. Cancer 2003, 88, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Ria, R.; Vacca, A. Bone Marrow Stromal Cells-Induced Drug Resistance in Multiple Myeloma. Int. J. Mol. Sci. 2020, 21, 613. [Google Scholar] [CrossRef] [Green Version]
- Heider, M.; Nickel, K.; Högner, M.; Bassermann, F. Multiple Myeloma: Molecular Pathogenesis and Disease Evolution. Oncol. Res. Treat. 2021, 44, 672–681. [Google Scholar] [CrossRef]
- Kastritis, E.; Terpos, E.; Roussou, M.; Gavriatopoulou, M.; Migkou, M.; Eleutherakis-Papaiakovou, E.; Fotiou, D.; Ziogas, D.; Panagiotidis, I.; Kafantari, E.; et al. Evaluation of the Revised International Staging System in an independent cohort of unselected patients with multiple myeloma. Haematologica 2016, 102, 593–599. [Google Scholar] [CrossRef] [Green Version]
- Kapoor, R.; Kumar, R.; Dubey, A.P. Risk Stratification in Multiple Myeloma in Indian Settings. Indian J. Hematol. Blood Transfus. 2019, 36, 464–472. [Google Scholar] [CrossRef]
- Ziogas, D.C.; Dimopoulos, M.A.; Kastritis, E. Prognostic factors for multiple myeloma in the era of novel therapies. Expert Rev. Hematol. 2018, 11, 863–879. [Google Scholar] [CrossRef]
- Barwick, B.G.; Neri, P.; Bahlis, N.J.; Nooka, A.K.; Dhodapkar, M.V.; Jaye, D.L.; Hofmeister, C.C.; Kaufman, J.L.; Gupta, V.A.; Auclair, D.; et al. Multiple myeloma immunoglobulin lambda translocations portend poor prognosis. Nat. Commun. 2019, 10, 1911. [Google Scholar] [CrossRef] [Green Version]
- Gao, M.; Bai, H.; Jethava, Y.; Wu, Y.; Zhu, Y.; Yang, Y.; Xia, J.; Cao, H.; Franqui-Machin, R.; Nadiminti, K.; et al. Identification and Characterization of Tumor-Initiating Cells in Multiple Myeloma. JNCI J. Natl. Cancer Inst. 2019, 112, 507–515. [Google Scholar] [CrossRef]
- Gao, M.; Kong, Y.; Yang, G.; Gao, L.; Shi, J. Multiple myeloma cancer stem cells. Oncotarget 2016, 7, 35466–35477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- García-Ortiz, A.; Rodríguez-García, Y.; Encinas, J.; Maroto-Martín, E.; Castellano, E.; Teixidó, J.; Martínez-López, J. The Role of Tumor Microenvironment in Multiple Myeloma Development and Progression. Cancers 2021, 13, 217. [Google Scholar] [CrossRef] [PubMed]
- Altevogt, P.; Sammar, M.; Hüser, L.; Kristiansen, G. Novel insights into the function of CD24: A driving force in cancer. Int. J. Cancer 2020, 148, 546–559. [Google Scholar] [CrossRef] [PubMed]
- Overdevest, J.B.; Thomas, S.; Kristiansen, G.; Hansel, D.E.; Smith, S.C.; Theodorescu, D. CD24 Offers a Therapeutic Target for Control of Bladder Cancer Metastasis Based on a Requirement for Lung Colonization. Cancer Res. 2011, 71, 3802–3811. [Google Scholar] [CrossRef] [PubMed] [Green Version]
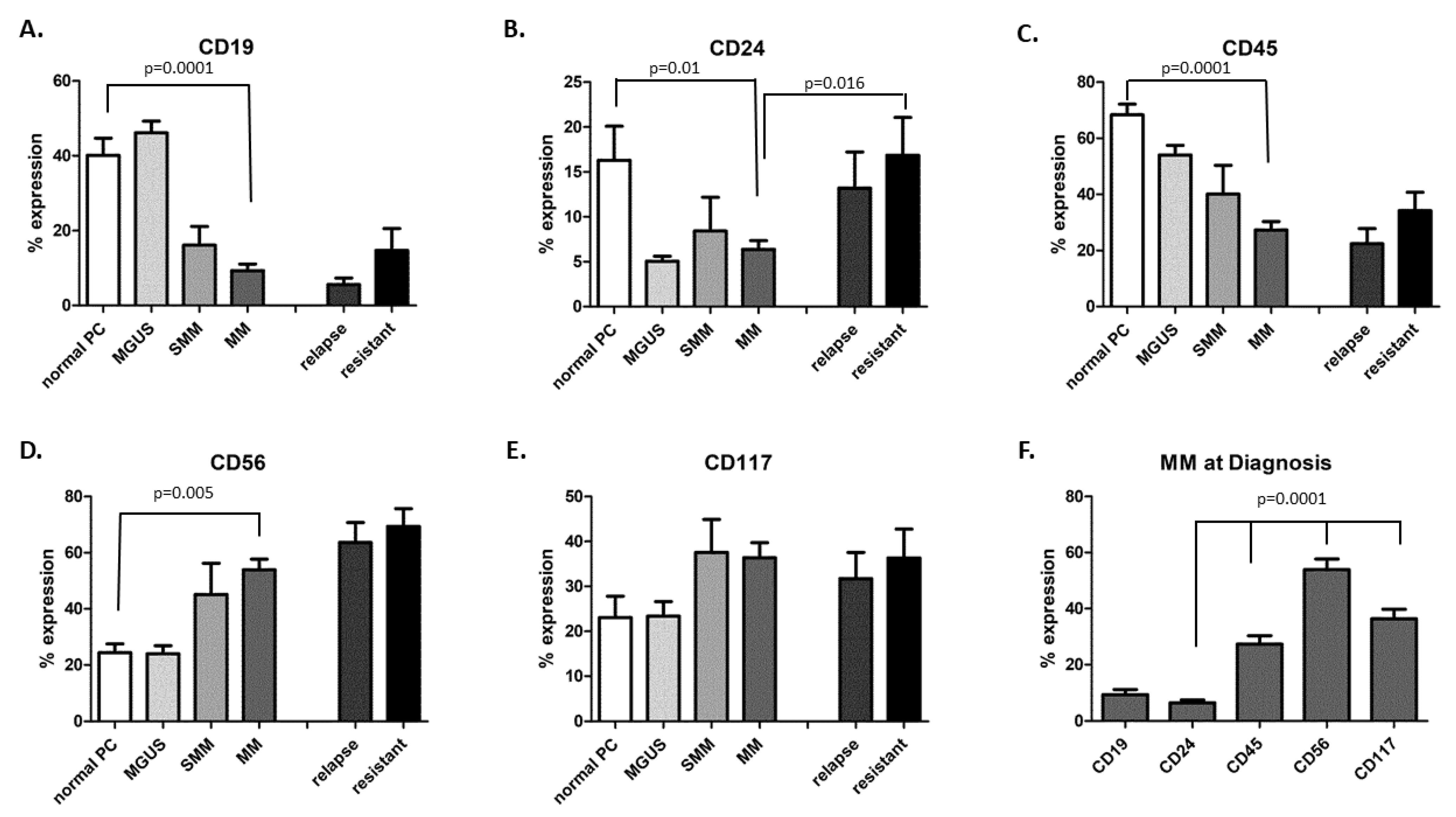
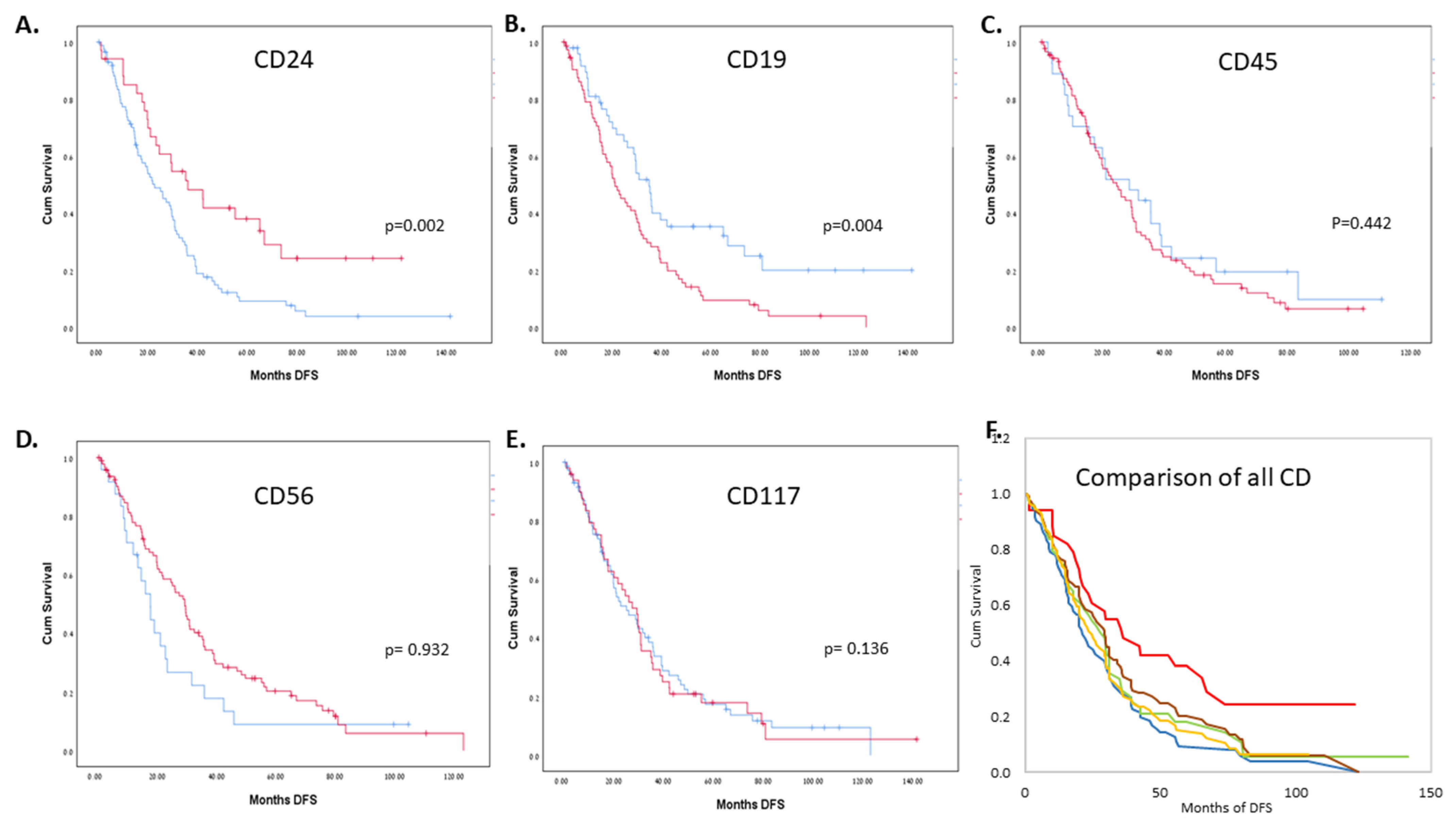
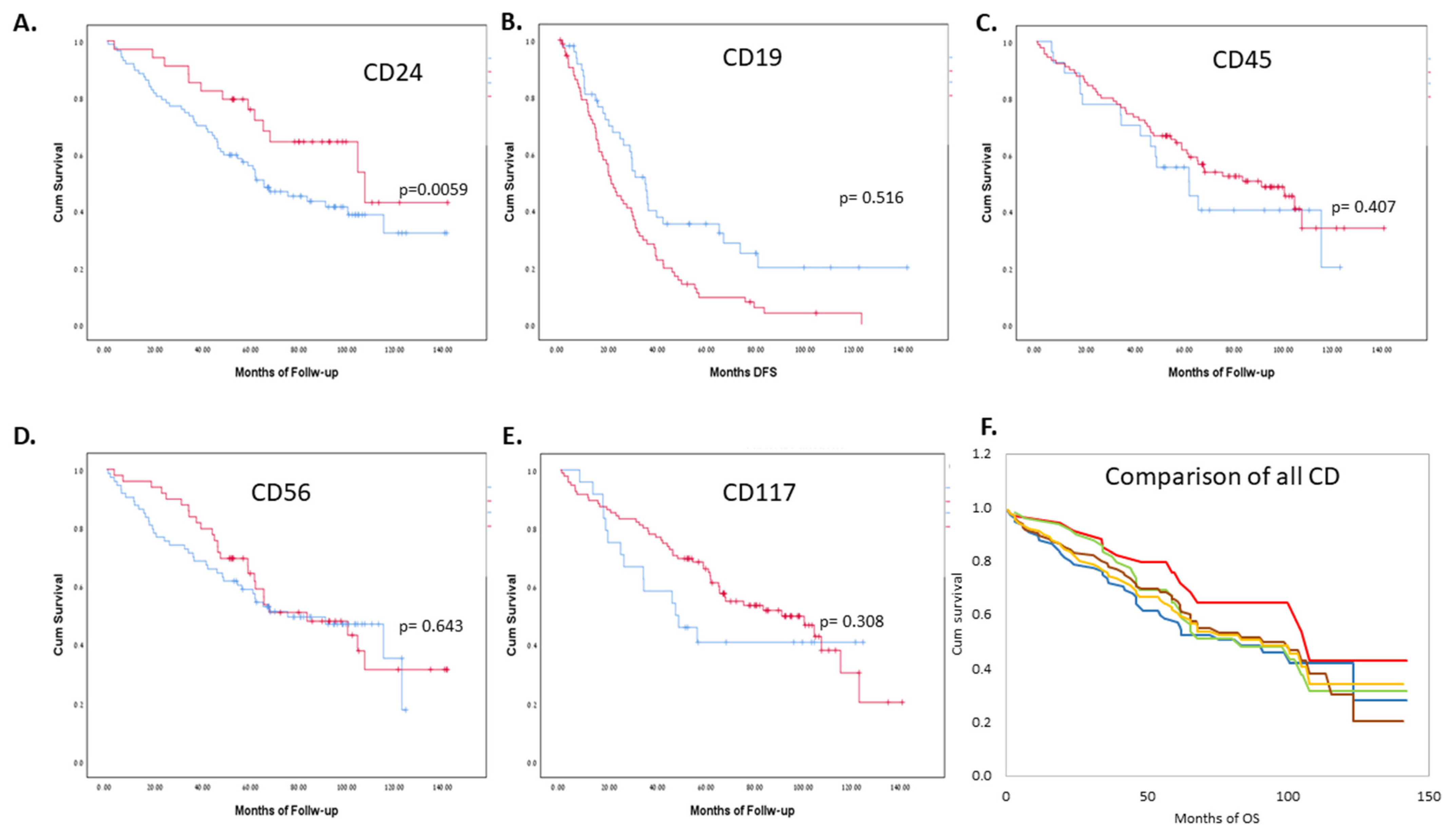
| AL | MM | CD24+ (above 5%) | CD24− (5% and below) | Total | ||
|---|---|---|---|---|---|---|
| Gender | Male | 9 (47.4%) | 66 (62.9%) | 19 (55.9%) | 54 (62.1%) | 75 (60.5%) |
| Female | 10 (52.6%) | 37.1 (37.5%) | 15 (44.1%) | 33 (37.9%) | 49 (39.5%) | |
| Median age at diagnosis (years, range) | 68.5 (48–86) | 62 (30–89) | 64.5 (30–80) | 63 (35–89) | 62.5 (30–89) | |
| M-protein (for MM patients) | IGG | 54 (51.4%) | 54 (43.5%) | 18 (52.9%) | 34 (39.1%) | |
| IGA | 19 (18.1%) | 19 (15.3%) | 3 (8.8%) | 16 (18.4%) | ||
| LC only | 30 (28.6%) | 49 (39.5%) | 13 (38.2%) | 35 (40.2%) | ||
| Non-secretory | 2 (1.9%) | 2 (1.6%) | 0 | 2 (2.3%) | ||
| Light chain | Kappa | 8 (42.1%) | 64 (61.5%) | 72 (58.0%) | 14 (41.2%) | 55 (63.2%) |
| Lambda | 11 (57.9%) | 39 (37.5%) | 50 (40.3%) | 20 (58.8%) | 30 (34.5%) | |
| Non-secretory | 0 (0%) | 2 (1.9%) | 2 (1.6%) | 0 | 2 (2.3%) | |
| R-Mayo stage (for AL patients) | 1 | 4 (21.0%) | ||||
| 2 | 1 (5.3%) | |||||
| 3 | 7 (36.8%) | |||||
| 4 | 7 (36.8%) | |||||
| R-ISS (for MM patients) | 1 | 37 (35.2%) | ||||
| 2 | 36 (34.3%) | |||||
| 3 | 24 (22.9%) | |||||
| Missing | 8 (7.6%) | |||||
| Organ involvement (for AL patients) | Heart | 14 (73.7%) | ||||
| Kidney | 13 (68.4%) | |||||
| Soft tissue | 11 (57.9%) | |||||
| PNS | 11 (57.9%) | |||||
| Liver | 3 (15.8%) | |||||
| GI | 1 (5.3%) | |||||
| Cytogenetics * | Standard risk | 66 (62.9%) | 26 (76.5%) | 49 (56.3%) | 78 (62.9%) | |
| High risk | 24 (22.9%) | 4 (11.8%) | 22 (25.3%) | 26 (21.0%) | ||
| Missing | 15 (14.3%) | 4 (11.8%) | 16 (18.4%) | 20 (16.1%) | ||
| Calcium (above or below 11 mg/dL) | High | 0 (0%) | 7 (6.7%) | 0 | 7 (8.0%) | 7 (5.6%) |
| Normal | 17 (89.5%) | 96 (91.4%) | 31 (91.2%) | 79 (90.8%) | 113 (91.1%) | |
| Missing | 2 (10.5%) | 2 (1.9%) | 3 (8.2%) | 1 (1.1%) | 4 (3.2%) | |
| Creatinine (above or below 2 mg/dL) | Normal | 16 (84.2%) | 86 (81.9%) | 27 (79.4%) | 72 (82.8%) | 102 (82.3%) |
| High | 2 (10.5%) | 17 (16.2%) | 5 (14.7%) | 14 (16.1%) | 19 (15.3%) | |
| Missing | 1 (5.3%) | 2 (1.9%) | 2 (5.9%) | 1 (1.1%) | 3 (2.4%) | |
| Lytic lesions (for MM patients) ** | Yes | 66 (62.9%) | 14 (41.2%) | 41 (47.1%) | 57 (46%) | |
| No | 39 (37.1%) | 20 (58.8%) | 46 (52.9%) | 67 (54%) | ||
| Hemoglobin (above or below 10.5 g/dL) | Normal | 14 (73.7%) | 58 (55.2%) | 25 (73.5%) | 45 (51.7%) | 72 (58.0%) |
| Low | 4 (21.0%) | 45 (42.9%) | 7 (20.6%) | 41 (47.1%) | 49 (39.6%) | |
| Missing | 1 (5.3%) | 2 (1.9%) | 2 (5.9%) | 1 (1.1%) | 3 (2.4%) | |
| Induction regime | VCD | 19 (100%) | 100 (95.2%) | 31 (91.2%) | 85 (97.7%) | 119 (96.0%) |
| VTD | 0(0%) | 5 (4.8%) | 3 (8.8%) | 2 (2.3%) | 5 (4.0%) | |
| ASCT | Yes | 0 (0%) | 57 (54.3%) | 11 (32.4%) | 44 (50.6%) | 57 (46.0%) |
| No | 19 (100%) | 48 (45.7%) | 23 (67.6%) | 43 (49.4%) | 67 (54.0%) |
| AL | MM | |||
|---|---|---|---|---|
| Median | Std. Deviation | Median | Std. Deviation | |
| CD19 % | 2.00 | 2.45 | 10.08 | 16.53 |
| CD24 % | 6.65 | 11.18 | 1.21 | 19.44 |
| CD56 % | 3.55 | 5.14 | 3.40 | 14.61 |
| CD117 % | 37.25 | 36.97 | 79.24 | 40.75 |
| CD45 % | 49.08 | 33.69 | 17.05 | 35.35 |
| BM PC% | 12.06 | 13.15 | 9.79 | 34.46 |
| MM | AL | |||||
|---|---|---|---|---|---|---|
| Hazard Ratio | Confidence Interval | p-Value | Hazard Ratio | Confidence Interval | p-Value | |
| CD 24% | 0.980 | 0.959–1.002 | 0.075 | 0.900 | 0.799–1.013 | 0.082 |
| CD 19% | 1.009 | 0.993–1.025 | 0.287 | 1.199 | 0.959–1.498 | 0.110 |
| CD 56% | 0.998 | 0.980–1.016 | 0.821 | 0.911 | 0.786–1.055 | 0.212 |
| CD 45% | 0.995 | 0.987–1.003 | 0.224 | 0.995 | 0.977–1.012 | 0.552 |
| CD 117% | 0.999 | 0.992–1.006 | 0.795 | 0.994 | 0.978–1.010 | 0.442 |
| PC% | 0.984 | 0.973–0.995 | 0.003 | 0.983 | 0.924–1.047 | 0.600 |
| CD | Hazard Ratio | Confidence Interval | p-Value |
|---|---|---|---|
| CD24 | 0.975 | 0.960–0.991 | p = 0.002 |
| CD19 | 1.025 | 1.014–1.037 | p < 0.001 |
| CD117 | 1.001 | 0.996–1.006 | p = 0.770 |
| CD56 | 1.003 | 0.990–1.016 | p = 0.641 |
| CD45 | 1.004 | 0.999–1.010 | p = 0.120 |
| CD | Hazard Ratio | Confidence Interval | p-Value |
|---|---|---|---|
| CD24 | 0.979 | 0.958–0.999 | p = 0.044 |
| CD19 | 1.007 | 0.992–1.023 | p = 0.362 |
| CD117 | 0.998 | 0.992–1.005 | p = 0.601 |
| CD56 | 0.982 | 0.951–1.014 | p = 0.255 |
| CD45 | 0.995 | 0.988–1.003 | p = 0.203 |
| Hazard Ratio | Confidence Interval | p-Value | |
|---|---|---|---|
| R-ISS | 1.917 | 1.074–3.425 | p = 0.028 |
| High-risk cytogenetics * | 3.452 | 1.719–6.9333 | p < 0.001 |
| High Β2M (mg/mL) | 1.000 | 1.000–1.000 | p = 0.044 |
| Hypercalcemia ** | 3.642 | 1.637–8.105 | p = 0.002 |
| Older age | 1.047 | 1.014–1.081 | p = 0.005 |
| CD24% | 1.011 | 0.986–1.037 | p = 0.383 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gross Even-Zohar, N.; Pick, M.; Hofstetter, L.; Shaulov, A.; Nachmias, B.; Lebel, E.; Gatt, M.E. CD24 Is a Prognostic Marker for Multiple Myeloma Progression and Survival. J. Clin. Med. 2022, 11, 2913. https://doi.org/10.3390/jcm11102913
Gross Even-Zohar N, Pick M, Hofstetter L, Shaulov A, Nachmias B, Lebel E, Gatt ME. CD24 Is a Prognostic Marker for Multiple Myeloma Progression and Survival. Journal of Clinical Medicine. 2022; 11(10):2913. https://doi.org/10.3390/jcm11102913
Chicago/Turabian StyleGross Even-Zohar, Noa, Marjorie Pick, Liron Hofstetter, Adir Shaulov, Boaz Nachmias, Eyal Lebel, and Moshe E. Gatt. 2022. "CD24 Is a Prognostic Marker for Multiple Myeloma Progression and Survival" Journal of Clinical Medicine 11, no. 10: 2913. https://doi.org/10.3390/jcm11102913
APA StyleGross Even-Zohar, N., Pick, M., Hofstetter, L., Shaulov, A., Nachmias, B., Lebel, E., & Gatt, M. E. (2022). CD24 Is a Prognostic Marker for Multiple Myeloma Progression and Survival. Journal of Clinical Medicine, 11(10), 2913. https://doi.org/10.3390/jcm11102913







