Better Estimation of Spontaneous Preterm Birth Prediction Performance through Improved Gestational Age Dating
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Gestational Age Dating and Estimated Delivery Date
2.3. Sample Analysis
2.4. Statistical Analysis
2.5. Estimation of the Effects of Certainty of Gestational Age Dating on Prediction of Prematurity
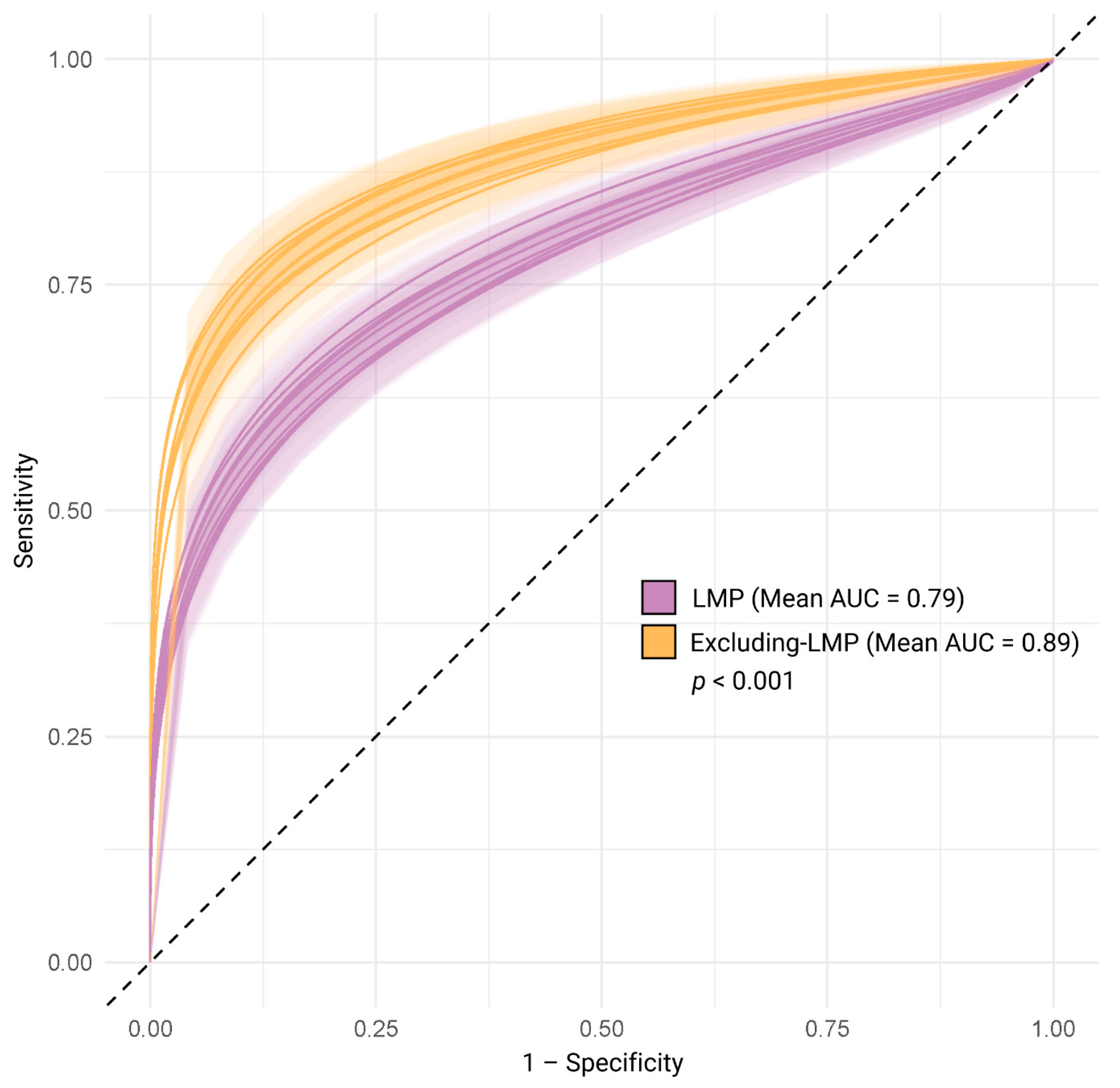
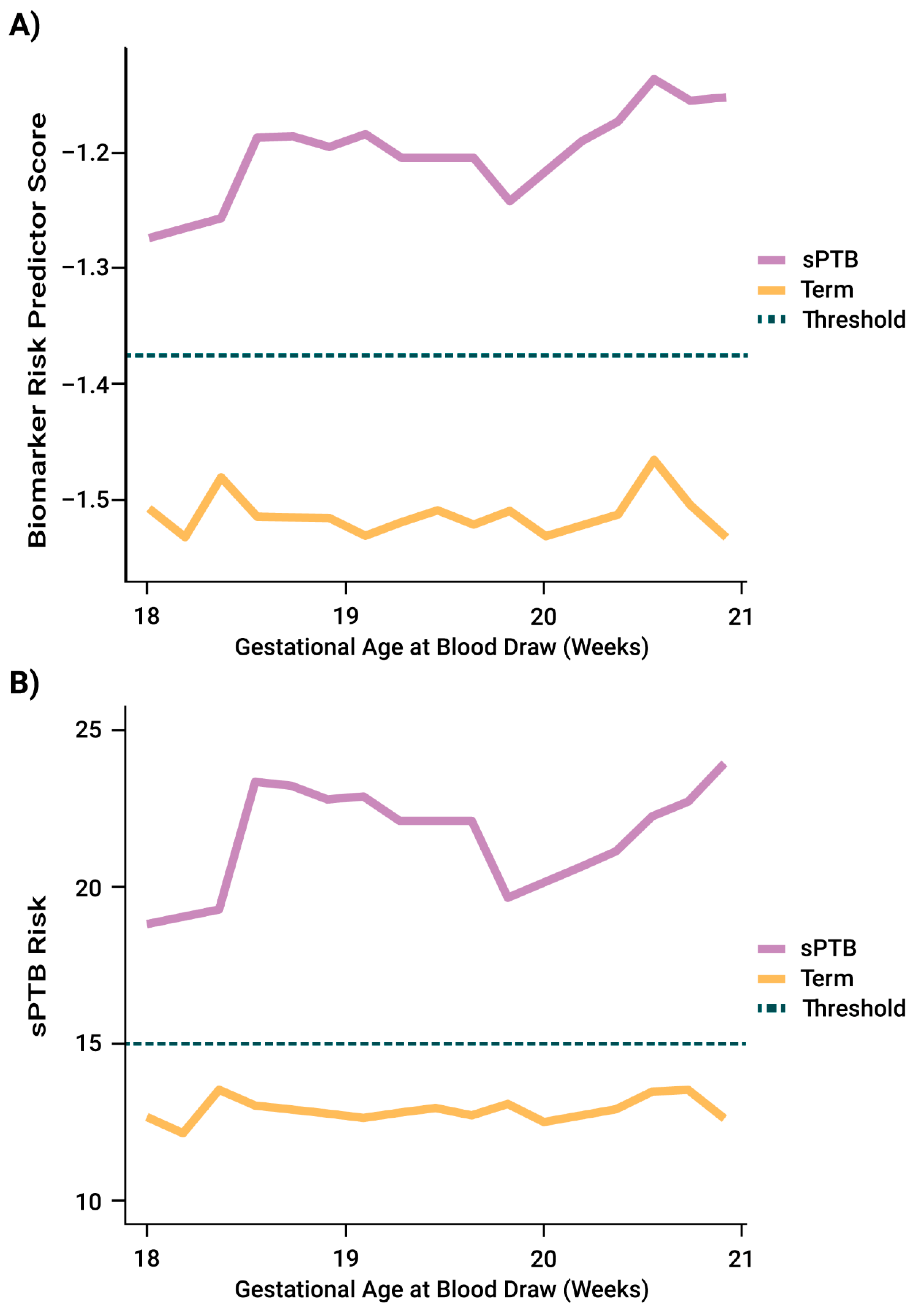
3. Results
Risk Predictor Performance
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, L.; Oza, S.; Hogan, D.; Chu, Y.; Perin, J.; Zhu, J.; Lawn, J.E.; Cousens, S.; Mathers, C.; Black, R.E. Global, regional, and national causes of under-5 mortality in 2000-15: An updated systematic analysis with implications for the Sustainable Development Goals. Lancet 2016, 388, 3027–3035. [Google Scholar] [CrossRef] [Green Version]
- Chawanpaiboon, S.; Vogel, J.P.; Moller, A.B.; Lumbiganon, P.; Petzold, M.; Hogan, D.; Landoulsi, S.; Jampathong, N.; Kongwattanakul, K.; Laopaiboon, M.; et al. Global, regional, and national estimates of levels of preterm birth in 2014: A systematic review and modelling analysis. Lancet Glob. Health 2019, 7, e37–e46. [Google Scholar] [CrossRef] [Green Version]
- Osterman, M.J.K.; Hamilton, B.E.; Martin, J.A.; Driscoll, A.K.; Valenzuela, C.P. Births: Final Data for 2020. Available online: https://www.cdc.gov/nchs/nvss/births.htm (accessed on 9 February 2022).
- Waitzman, N.J.; Jalali, A.; Grosse, S.D. Preterm birth lifetime costs in the United States in 2016: An update. Semin. Perinatol. 2021, 45, 151390. [Google Scholar] [CrossRef] [PubMed]
- Alexander, G.R.; Institute of Medicine (US) Committee on Understanding Premature Birth and Assuring Healthy Outcomes; Behrman, R.E.; Butler, A.S. Prematurity at Birth: Determinants, Consequences, and Geographic Variation (Appendix B). In Preterm Birth: Causes, Consequences, and Prevention; National Academies Press (US): Washington, DC, USA, 2007. [Google Scholar]
- American College of Obstetricians and Gynecologists, Gynecologists’ Committee on Practice, Bulletins-Obstetrics. Prediction and Prevention of Spontaneous Preterm Birth: ACOG Practice Bulletin, Number 234. Obs. Gynecol 2021, 138, e65–e90. [Google Scholar] [CrossRef] [PubMed]
- American College of Obstetricians and Gynecologists. Committee Opinion No 700: Methods for Estimating the Due Date. Obs. Gynecol. 2017, 129, e150–e154. [Google Scholar] [CrossRef] [PubMed]
- Barr, W.B.; Pecci, C.C. Last menstrual period versus ultrasound for pregnancy dating. Int. J. Gynaecol. Obs. 2004, 87, 38–39. [Google Scholar] [CrossRef] [PubMed]
- Savitz, D.A.; Terry, J.W., Jr.; Dole, N.; Thorp, J.M., Jr.; Siega-Riz, A.M.; Herring, A.H. Comparison of pregnancy dating by last menstrual period, ultrasound scanning, and their combination. Am. J. Obs. Gynecol. 2002, 187, 1660–1666. [Google Scholar] [CrossRef]
- Wegienka, G.; Baird, D.D. A comparison of recalled date of last menstrual period with prospectively recorded dates. J. Womens Health 2005, 14, 248–252. [Google Scholar] [CrossRef]
- Andersen, H.F.; Barclay, M.L. Prediction of delivery date in a computerized prenatal record system. Proc. Annu. Symp. Comput. Appl. Med. Care 1980, 1, 307–310. [Google Scholar]
- Darwish, N.A.; Thabet, S.M.; Aboul Nasr, A.L.; El Sharkawy, S.; El Tamamy, M.N. Modified Naegele’s Rule for determination of the expected date of delivery irrespective of the cycle length. Med. J. Cairo Univ. 1994, 62, 39–47. [Google Scholar]
- Caughey, A.B.; Nicholson, J.M.; Washington, A.E. First- vs second-trimester ultrasound: The effect on pregnancy dating and perinatal outcomes. Am. J. Obs. Gynecol. 2008, 198, 703.e1–703.e5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalish, R.B.; Thaler, H.T.; Chasen, S.T.; Gupta, M.; Berman, S.J.; Rosenwaks, Z.; Chervenak, F.A. First- and second-trimester ultrasound assessment of gestational age. Am. J. Obs. Gynecol. 2004, 191, 975–978. [Google Scholar] [CrossRef] [PubMed]
- Verburg, B.O.; Steegers, E.A.; De Ridder, M.; Snijders, R.J.; Smith, E.; Hofman, A.; Moll, H.A.; Jaddoe, V.W.; Witteman, J.C. New charts for ultrasound dating of pregnancy and assessment of fetal growth: Longitudinal data from a population-based cohort study. Ultrasound Obs. Gynecol. 2008, 31, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Taipale, P.; Hiilesmaa, V. Predicting delivery date by ultrasound and last menstrual period in early gestation. Obs. Gynecol. 2001, 97, 189–194. [Google Scholar] [CrossRef]
- Bennett, K.A.; Crane, J.M.; O’Shea, P.; Lacelle, J.; Hutchens, D.; Copel, J.A. First trimester ultrasound screening is effective in reducing postterm labor induction rates: A randomized controlled trial. Am. J. Obs. Gynecol. 2004, 190, 1077–1081. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.S.; Romero, R.; Vidyadhari, D.; Fusey, S.; Baxter, J.K.; Khandelwal, M.; Vijayaraghavan, J.; Trivedi, Y.; Soma-Pillay, P.; Sambarey, P.; et al. Vaginal progesterone reduces the rate of preterm birth in women with a sonographic short cervix: A multicenter, randomized, double-blind, placebo-controlled trial. Ultrasound Obs. Gynecol. 2011, 38, 18–31. [Google Scholar] [CrossRef] [Green Version]
- Petrini, J.R.; Callaghan, W.M.; Klebanoff, M.; Green, N.S.; Lackritz, E.M.; Howse, J.L.; Schwarz, R.H.; Damus, K. Estimated effect of 17 alpha-hydroxyprogesterone caproate on preterm birth in the United States. Obs. Gynecol. 2005, 105, 267–272. [Google Scholar] [CrossRef]
- Saade, G.R.; Boggess, K.A.; Sullivan, S.A.; Markenson, G.R.; Iams, J.D.; Coonrod, D.V.; Pereira, L.M.; Esplin, M.S.; Cousins, L.M.; Lam, G.K.; et al. Development and validation of a spontaneous preterm delivery predictor in asymptomatic women. Am. J. Obs. Gynecol. 2016, 214, 633.e1–633.e24. [Google Scholar] [CrossRef] [Green Version]
- Bradford, C.; Severinsen, R.; Pugmire, T.; Rasmussen, M.; Stoddard, K.; Uemura, Y.; Wheelwright, S.; Mentinova, M.; Chelsky, D.; Hunsucker, S.W.; et al. Analytical validation of protein biomarkers for risk of spontaneous preterm birth. Clin. Mass Spectrom. 2017, 3, 25–38. [Google Scholar] [CrossRef]
- Markenson, G.R.; Saade, G.R.; Laurent, L.C.; Heyborne, K.D.; Coonrod, D.V.; Schoen, C.N.; Baxter, J.K.; Haas, D.M.; Longo, S.; Grobman, W.A.; et al. Performance of a proteomic preterm delivery predictor in a large independent prospective cohort. Am. J. Obs. Gynecol. MFM 2020, 2, 100140. [Google Scholar] [CrossRef]
- Burchard, J.; Polpitiya, A.D.; Fox, A.C.; Randolph, T.L.; Fleischer, T.C.; Dufford, M.T.; Garite, T.J.; Critchfield, G.C.; Boniface, J.J.; Saade, G.R.; et al. Clinical Validation of a Proteomic Biomarker Threshold for Increased Risk of Spontaneous Preterm Birth and Associated Clinical Outcomes: A Replication Study. J. Clin. Med. 2021, 10, 5088. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, D.M.; Fine, J.; Chappell, R. Sample size for positive and negative predictive value in diagnostic research using case–control designs. Biostatistics 2008, 10, 94–105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dowle, M.; Srinivasan, A. Data.Table: Extension of ‘Data.Frame’, Version 1.14.2. Available online: https://cran.r-project.org/web/packages/data.table/index.html (accessed on 20 December 2021).
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.C.; Müller, M. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Dorai-Raj, S. Binom: Binomial Confidence Intervals for Several Parameterizations. Available online: https://CRAN.R-project.org/package=binom (accessed on 1 December 2021).
- National Center for Health Statistics, U.S. Centers for Disease Control and Prevention. Birth Data Files. Available online: https://www.cdc.gov/nchs/data_access/vitalstatsonline.htm#Births (accessed on 1 March 2022).
| Weeks 191/7–206/7 | Weeks 180/7–206/7 | ||||||
|---|---|---|---|---|---|---|---|
| Demographic/ Clinical Variable | Value | All- Subjects | Excluding LMP | p-Value | All Subjects | Excluding LMP | p-Value |
| Maternal age | Median (IQR) | 24.5 (21.0–30.0) | 23 (21.0–28.0) | 0.72 | 24.5 (22.0–31.0) | 23.0 (21.5–28.0) | 0.7 |
| Maternal BMI | Median (IQR) | 26.5 (22.3–31.3) | 28.5 (23.8–34.6) | 0.7 | 28.0 (23.5–32.0) | 29.4 (24.4–34.6) | 0.7 |
| Gravida | Primigravida | 13 | 7 | 1 | 24 | 11 | 0.83 |
| Multigravida | 41 | 22 | 60 | 33 | |||
| Race | Black | 13 | 5 | 0.76 | 17 | 8 | 0.95 |
| White | 38 | 23 | 61 | 33 | |||
| Other | 3 | 1 | 6 | 3 | |||
| Ethnicity | Hispanic | 22 | 12 | 0.97 | 36 | 20 | 0.9 |
| Non-Hispanic | 32 | 17 | 48 | 24 | |||
| Prior PTB | No | 34 | 19 | 0.9 | 50 | 29 | 0.76 |
| Yes | 20 | 10 | 34 | 15 | |||
| GABD | Median (IQR) | 139 (135–144) | 139 (135–144) | 0.96 | 135 (130.5–142.5) | 135 (130–143) | 0.98 |
| GAB | Median (IQR) | 273 (256–281) | 273 (258–277) | 0.98 | 273 (256–281) | 273 (257–277) | 0.96 |
| Neonatal gender | Female | 22 | 21 | 0.34 | 39 | 17 | 0.46 |
| Male | 32 | 8 | 45 | 27 | |||
| Outcome | Cases | 18 | 10 | 0.95 | 28 | 15 | 0.97 |
| Controls | 36 | 19 | 56 | 29 | |||
| Total | 54 | 29 | 84 | 44 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burchard, J.; Saade, G.R.; Boggess, K.A.; Markenson, G.R.; Iams, J.D.; Coonrod, D.V.; Pereira, L.M.; Hoffman, M.K.; Polpitiya, A.D.; Treacy, R.; et al. Better Estimation of Spontaneous Preterm Birth Prediction Performance through Improved Gestational Age Dating. J. Clin. Med. 2022, 11, 2885. https://doi.org/10.3390/jcm11102885
Burchard J, Saade GR, Boggess KA, Markenson GR, Iams JD, Coonrod DV, Pereira LM, Hoffman MK, Polpitiya AD, Treacy R, et al. Better Estimation of Spontaneous Preterm Birth Prediction Performance through Improved Gestational Age Dating. Journal of Clinical Medicine. 2022; 11(10):2885. https://doi.org/10.3390/jcm11102885
Chicago/Turabian StyleBurchard, Julja, George R. Saade, Kim A. Boggess, Glenn R. Markenson, Jay D. Iams, Dean V. Coonrod, Leonardo M. Pereira, Matthew K. Hoffman, Ashoka D. Polpitiya, Ryan Treacy, and et al. 2022. "Better Estimation of Spontaneous Preterm Birth Prediction Performance through Improved Gestational Age Dating" Journal of Clinical Medicine 11, no. 10: 2885. https://doi.org/10.3390/jcm11102885
APA StyleBurchard, J., Saade, G. R., Boggess, K. A., Markenson, G. R., Iams, J. D., Coonrod, D. V., Pereira, L. M., Hoffman, M. K., Polpitiya, A. D., Treacy, R., Fox, A. C., Randolph, T. L., Fleischer, T. C., Dufford, M. T., Garite, T. J., Critchfield, G. C., Boniface, J. J., & Kearney, P. E. (2022). Better Estimation of Spontaneous Preterm Birth Prediction Performance through Improved Gestational Age Dating. Journal of Clinical Medicine, 11(10), 2885. https://doi.org/10.3390/jcm11102885







