Ongoing Use of SSRIs Does Not Alter Outcome in Hospitalized COVID-19 Patients: A Retrospective Analysis
Abstract
:1. Introduction
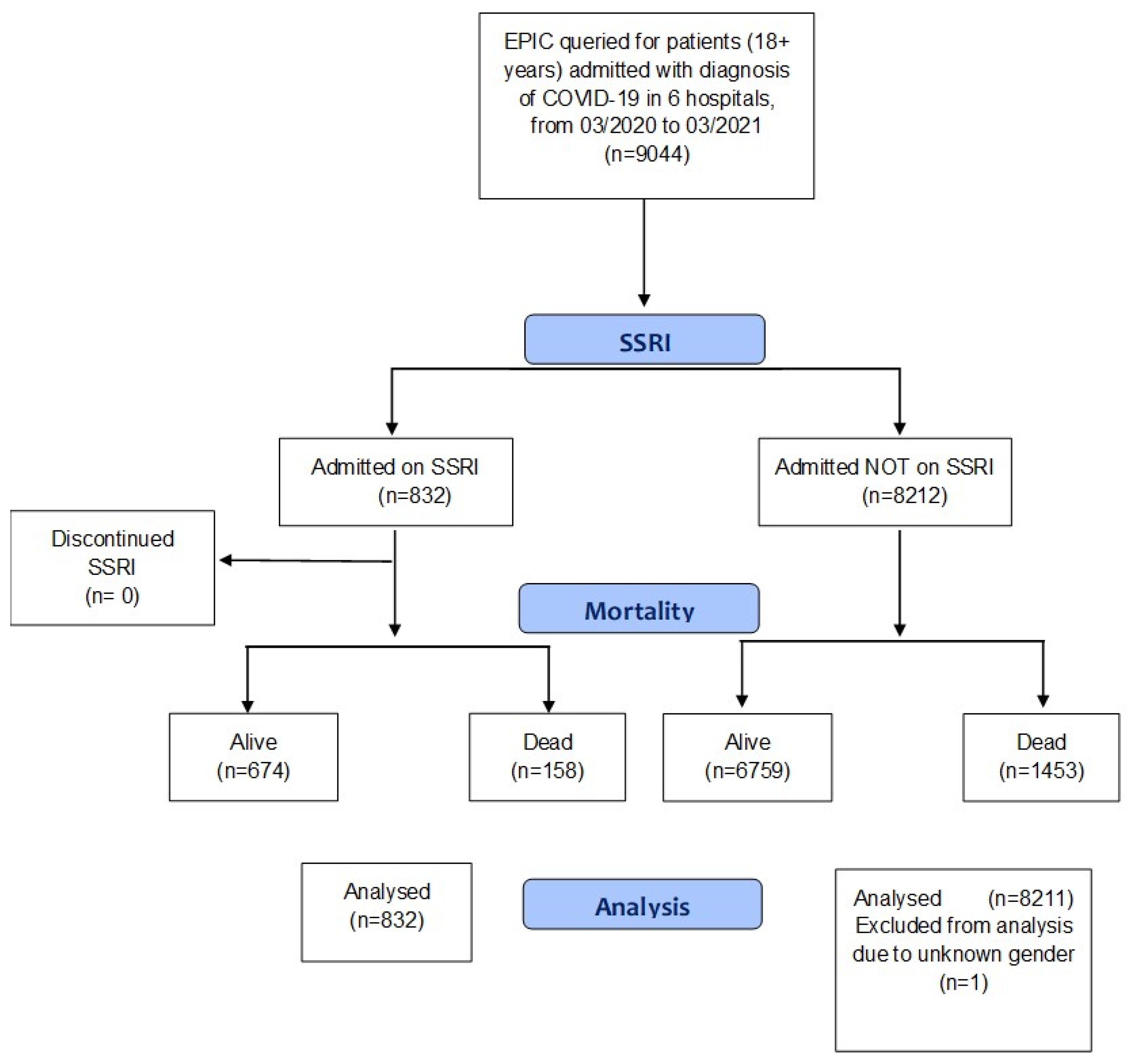
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kuriakose, S.; Singh, K.; Pau, A.K.; Daar, E.; Gandhi, R.; Tebas, P.; Evans, L.; Gulick, R.M.; Lane, H.C.; Masur, H.; et al. Developing Treatment Guidelines During a Pandemic Health Crisis: Lessons Learned From COVID-19. Ann. Int. Med. 2021, 174, 1151–1158. [Google Scholar] [CrossRef]
- Riva, L.; Yuan, S.; Yin, X.; Martin-Sancho, L.; Matsunaga, N.; Pache, L.; Burgstaller-Muehlbacher, S.; De Jesus, P.D.; Teriete, P.; Hull, M.V.; et al. Discovery of SARS-CoV-2 antiviral drugs through large-scale compound repurposing. Nature 2020, 586, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Chen, K.; Pan, J.; Lei, Y.; Zhang, D.; Fang, L.; Tang, J.; Chen, X.; Ma, Y.; Zheng, Y.; et al. Repurposing clinically approved drugs for COVID-19 treatment targeting SARS-CoV-2 papain-like protease. Int. J. Biol. Macromol. 2021, 188, 137–146. [Google Scholar] [CrossRef]
- Chiou, W.C.; Hsu, M.S.; Chen, Y.T.; Yang, J.M.; Tsay, Y.G.; Huang, H.C.; Huang, C. Repurposing existing drugs: Identification of SARS-CoV-2 3C-like protease inhibitors. J. Enzym. Inhib. Med. Chem. 2021, 36, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Pickard, A.; Calverley, B.C.; Chang, J.; Garva, R.; Gago, S.; Lu, Y.; Kadler, K.E. Discovery of re-purposed drugs that slow SARS-CoV-2 replication in human cells. PLoS Pathog. 2021, 17, e1009840. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Ryan, H.; Kredo, T.; Chaplin, M.; Fletcher, T. Chloroquine or hydroxychloroquine for prevention and treatment of COVID-19. Cochrane Database Syst. Rev. 2020, 2020, CD013587. [Google Scholar] [CrossRef]
- Diez-Quevedo, C.; Iglesias-González, M.; Giralt-López, M.; Rangil, T.; Sanagustin, D.; Moreira, M.; López-Ramentol, M.; Ibáñez-Caparrós, A.; Lorán, M.E.; Bustos-Cardona, T.; et al. Mental disorders, psychopharmacological treatments, and mortality in 2150 COVID-19 Spanish inpatients. Acta Psychiatr. Scand. 2021, 143, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Hoertel, N.; Sánchez-Rico, M.; Vernet, R.; Beeker, N.; Jannot, A.S.; Neuraz, A.; Salamanca, E.; Paris, N.; Daniel, C.; Gramfort, A.; et al. Association between antidepressant use and 426 reduced risk of intubation or death in hospi-talized patients with COVID-19: Results from an 427 observational study. Mol. Psychiatry 2021, 26, 5199–5212. [Google Scholar] [CrossRef]
- Lopez, S. Can a Common Antidepressant Help in the Fight against COVID-19? LA Times. 2021. Available online: https://www.latimes.com/california/story/2021-02-03/can-common-antidepressant-help-covid-19-fight (accessed on 1 November 2021).
- Meikle, C.K.S.; Creeden, J.F.; McCullumsmith, C.; Worth, R.G. SSRIs: Applications in inflammatory lung disease and implica-tions for COVID-19. Neuropsychopharmacol. Rep. 2021, 41, 325–335. [Google Scholar] [CrossRef]
- Pashaei, Y. Drug repurposing of selective serotonin reuptake inhibitors: Could these drugs help fight COVID-19 and save lives? J. Clin. Neurosci. 2021, 88, 163–172. [Google Scholar] [CrossRef]
- Creeden, J.F.; Imami, A.S.; Eby, H.M.; Gillman, C.; Becker, K.N.; Reigle, J.; Andari, E.; Pan, Z.K.; O’Donovan, S.M.; McCul-lumsmith, R.E.; et al. Fluoxetine as an anti-inflammatory therapy in SARS-CoV-2 infection. Biomed. Pharmacother. 2021, 138, 111437. [Google Scholar] [CrossRef]
- Sukhatme, V.P.; Reiersen, A.M.; Vayttaden, S.J.; Sukhatme, V.V. Fluvoxamine: A Review of Its Mechanism of Action and Its Role in COVID-19. Front. Pharmacol. 2021, 12, 652688. [Google Scholar] [CrossRef]
- Bialek, K.; Czarny, P.; Wigner, P.; Synowiec, E.; Barszczewska, G.; Bijak, M.; Szemraj, J.; Niemczyk, M.; Tota-Glowczyk, K.; Papp, M.; et al. Chronic Mild Stress and Venlafaxine Treatment Were Associated with Altered Expression Level and Methylation Status of New Candidate Inflammatory Genes in PBMCs and Brain Structures of Wistar Rats. Genes 2021, 12, 667. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [Green Version]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J.; HLH Across Speciality Collaboration, UK. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Lee, S.; Yu, Y.; Trimpert, J.; Benthani, F.; Mairhofer, M.; Richter-Pechanska, P.; Wyler, E.; Belenki, D.; Kaltenbrunner, S.; Pammer, M.; et al. Virus-induced senescence is a driver and therapeutic target in COVID-19. Nature 2021, 599, 283–289. [Google Scholar] [CrossRef]
- Leonard, B.E.; Myint, A. Changes in the immune system in depression and dementia: Causal or coincidental effects? Dialogues Clin. Neurosci. 2006, 8, 163–174. [Google Scholar]
- Hannestad, J.; DellaGioia, N.; Bloch, M.H. The Effect of Antidepressant Medication Treatment on Serum Levels of Inflammatory Cytokines: A Meta-Analysis. Neuropsychopharmacology 2011, 36, 2452–2459. [Google Scholar] [CrossRef]
- Hamed, M.; Hagag, R.S. The possible immunoregulatory and anti-inflammatory effects of selective serotonin reuptake inhib-itors in coronavirus disease patients. Med. Hypotheses 2020, 144, 110140. [Google Scholar] [CrossRef]
- Sen, A. Does serotonin deficiency lead to anosmia, ageusia, dysfunctional chemesthesis and increased severity of illness in COVID-19? Med. Hypotheses 2021, 153, 110627. [Google Scholar] [CrossRef]
- Taler, M.; Gil-Ad, I.; Lomnitski, L.; Korov, I.; Baharav, E.; Bar, M.; Zolokov, A.; Weizman, A. Immunomodulatory effect of selective serotonin reuptake inhibitors (SSRIs) on human T lymphocyte function and gene expression. Eur. Neuropsychopharmacol. 2007, 17, 774–780. [Google Scholar] [CrossRef] [PubMed]
- Karki, R.; Sharma, B.R.; Tuladhar, S.; Williams, E.P.; Zalduondo, L.; Samir, P.; Zheng, M.; Sundaram, B.; Banoth, B.; Malireddi, R.; et al. Synergism of TNF-α and IFN-γ Triggers In-flammatory Cell Death, Tissue Damage, and Mortality in SARS-CoV-2 Infection and Cytokine Shock Syndromes. Cell 2021, 184, 149–168. [Google Scholar] [CrossRef] [PubMed]
- Majidpoor, J.; Mortezaee, K. Interleukin-6 in SARS-CoV-2 induced disease: Interactions and therapeutic applications. Biomed. Pharmacother. 2021, 145, 112419. [Google Scholar] [CrossRef] [PubMed]
- Golden, S.R.; Rosenstein, D.L.; Belhorn, T.; Blatt, J. Repurposing Psychotropic Agents for Viral Disorders: Beyond COVID. ASSAY Drug Dev. Technol. 2021, 19, 373–385. [Google Scholar] [CrossRef] [PubMed]
- Dechaumes, A.; Nekoua, M.P.; Belouzard, S.; Sane, F.; Engelmann, I.; Dubuisson, J.; Alidjinou, E.K.; Hober, D. Fluoxetine Can Inhibit SARS-CoV-2 In Vitro. Microorganisms 2021, 9, 339. [Google Scholar] [CrossRef] [PubMed]
- Carpinteiro, A.; Edwards, M.J.; Hoffmann, M.; Kochs, G.; Gripp, B.; Weigang, S.; Adams, C.; Carpinteiro, E.; Gulbins, A.; Keitsch, S.; et al. Pharmacological inhibition of acid sphingomyelinase prevents uptake of SARS-CoV-2 by epithelial cells. Cell Rep. Med. 2020, 1, 100142. [Google Scholar] [CrossRef]
- Hoertel, N.; Sánchez-Rico, M.; Cougoule, C.; Gulbins, E.; Kornhuber, J.; Carpinteiro, A.; Becker, K.A.; Reiersen, A.M.; Lenze, E.J.; Seftel, D.; et al. Repurposing antidepressants inhibiting the sphingomyelinase acid/ceramide system against COVID-19: Current evidence and potential mechanisms. Mol. Psychiatry 2021. [Google Scholar] [CrossRef]
- Kornhuber, J.; Hoertel, N.; Gulbins, E. The acid sphingomyelinase/ceramide system in COVID-19. Mol. Psychiatry 2021, 2021, 1–8. [Google Scholar] [CrossRef]
- Törnquist, K.; Asghar, M.Y.; Srinivasan, V.; Korhonen, L.; Lindholm, D. Sphingolipids as Modulators of SARS-CoV-2 Infection. Front. Cell Dev. Biol. 2021, 9, 689854. [Google Scholar] [CrossRef]
- Rauchman, S. COVID: Treatment trials are still urgent. Nature 2021, 594, 26. [Google Scholar] [CrossRef]
- Hsu, L.; Grüne, B.; Buess, M.; Joisten, C.; Klobucnik, J.; Nießen, J.; Patten, D.; Wolff, A.; Wiesmüller, G.A.; Kossow, A.; et al. COVID-19 Breakthrough Infections and Transmission Risk: Real-World Data Analyses from Germany’s Largest Public Health Department (Cologne). Vaccines 2021, 9, 1267. [Google Scholar] [CrossRef] [PubMed]
- Torjesen, I. COVID-19: Omicron may be more transmissible than other variants and partly resistant to existing vaccines, scientists fear. BMJ 2021, 375, n2943. [Google Scholar] [CrossRef] [PubMed]
- Lenze, E.J.; Mattar, C.; Zorumski, C.F.; Stevens, A.; Schweiger, J.; Nicol, G.E.; Miller, J.P.; Yang, L.; Yingling, M.; Avidan, M.S.; et al. Fluvoxamine vs Placebo and Clinical 429 Deterioration in Outpatients with Symptomatic COVID-19: A Randomized Clinical Trial. JAMA 2020, 324, 2292–2300. [Google Scholar] [CrossRef]
- Seftel, D.; Boulware, D.R. Prospective Cohort of Fluvoxamine for Early Treatment of Coronavirus Disease 19. Open Forum Infect. Dis. 2021, 8, ofab050. [Google Scholar] [CrossRef] [PubMed]
- Reis, G.; dos Santos Moreira-Silva, E.A.; Silva DC, M.; Thabane, L.; Milagres, A.C.; Ferreira, T.S.; dos Santos, C.V.Q.; Campos, V.H.d.S.; Nogueira, A.M.R.; de Almeida, A.P.F.G.; et al. Effect of early treatment with fluvoxamine on risk of emergency care and hospitalisation among patients with COVID-19: The TOGETHER randomised, platform clinical trial. Lancet Glob. Health 2021, 10, e42–e51. [Google Scholar] [CrossRef]
- Oskotsky, T.; Marić, I.; Tang, A.; Oskotsky, B.; Wong, R.J.; Aghaeepour, N.; Sirota, M.; Stevenson, D.K. Mortality Risk Among Patients With COVID-19 Prescribed Selective Serotonin Reuptake Inhibitor Antidepressants. JAMA Netw. Open 2021, 4, e2133090. [Google Scholar] [CrossRef]
- Brody, D.J.; Gu, Q. Antidepressant Use among Adults: United States, 2015–2018. Hyattsville. 2020. Available online: https://www.cdc.gov/nchs/data/databriefs/db377-H.pdf (accessed on 23 November 2020).
- Anagha, K.; Shihabudheen, P.; Uvais, N.A. Side Effect Profiles of Selective Serotonin Reuptake Inhibitors: A Cross-Sectional Study in a Naturalistic Setting. Prim. Care Companion CNS Disord. 2021, 23, 20m02747. [Google Scholar] [CrossRef]
- Calusic, M.; Marcec, R.; Luksa, L.; Jurkovic, I.; Kovac, N.; Mihaljevic, S.; Likic, R. Safety and efficacy of fluvoxamine in COVID-19 ICU patients: An open label, prospective cohort trial with matched controls. Br. J. Clin. Pharmacol. 2021. Epub ahead of print. [Google Scholar] [CrossRef]
- Clelland, C.L.; Ramiah, K.; Steinberg, L.; Clelland, J.D. Analysis of the impact of antidepressants and other medications on COVID-19 infection risk in a chronic psychiatric in-patient cohort. BJPsych Open 2021, 8, e6. [Google Scholar] [CrossRef]
- Bainum, T.B.; Fike, D.S.; Mechelay, D.; Haase, K.K. Effect of Abrupt Discontinuation of Antidepressants in Critically Ill Hos-pitalized Adults. J. Hum. Pharmacol. Drug Ther. 2017, 37, 1231–1240. [Google Scholar] [CrossRef]
- Kelly, J.M.; Rubenfeld, G.D.; Masson, N.; Min, A.; Adhikari, N.K.J. Using Selective Serotonin Reuptake Inhibitors and Seroto-nin-Norepinephrine Reuptake Inhibitors in Critical Care: A Systematic Review of the Evidence for Benefit or Harm. Crit. Care Med. 2017, 45, e607–e616. [Google Scholar] [CrossRef] [PubMed]
- Halperin, D.; Reber, G. Influence of antidepressants on hemostasis. Dialogues Clin. Neurosci. 2007, 9, 47–59. [Google Scholar] [PubMed]
- Pigott, T.A.; Pato, M.T.; L’Heureux, F.; Hill, J.L.; Grover, G.N.; Bernstein, S.E.; Murphy, D.L. A controlled comparison of ad-juvant lithium carbonate or thyroid hormone in clomipramine-treated patients with obsessive-compulsive disorder. J. Clin. Psychopharmacol. 1991, 11, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Gu, S.X.; Tyagi, T.; Jain, K.; Gu, V.W.; Lee, S.H.; Hwa, J.M.; Kwan, J.M.; Krause, D.S.; Lee, A.I.; Halene, S.; et al. Thrombocytopathy and endotheliopathy: Crucial contributors to COVID-19 thromboinflammation. Nat. Rev. Cardiol. 2021, 18, 194–209. [Google Scholar] [CrossRef]
- Lohia, P.; Kapur, S.; Benjaram, S.; Cantor, Z.; Mahabadi, N.; Mir, T.; Badr, M.S. Statins and clinical outcomes in hospitalized COVID-19 patients with and without Diabetes Mellitus: A retrospective cohort study with propensity score matching. Cardiovasc. Diabetol. 2021, 20, 140. [Google Scholar] [CrossRef] [PubMed]
- Lopes, R.D.; Macedo, A.V.S.; de Barros E Silva, P.G.M.; Moll-Bernardes, R.J.; Dos Santos, T.M.; Mazza, L.; Feldman, A.; Arruda, G.D.S.; de Albuquerque, D.C.; Camiletti, A.S.; et al. Effect of Discontinuing vs. Continuing Angiotensin-Converting Enzyme Inhibitors and Angiotensin II Receptor Blockers on Days Alive and Out of the Hospital in Patients Admitted with COVID-19: A Randomized Clinical Trial. JAMA 2021, 325, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Davis, P.B.; Gurney, M.E.; Xu, R. COVID-19 and dementia: Analyses of risk, disparity, and outcomes from electronic health records in the US. Alzheimer Dement. 2021, 17, 1297–1306. [Google Scholar] [CrossRef]
- Hoertel, N. Do the Selective Serotonin Reuptake Inhibitor Antidepressants Fluoxetine and Fluvoxamine Reduce Mortality Among Patients With COVID-19? JAMA Netw. Open 2021, 4, e2136510. [Google Scholar] [CrossRef]
- Rudrapatna, V.A.; Glicksberg, B.S.; Butte, A.J. Utility of routinely collected electronic health records data to support effectiveness evaluations in inflammatory bowel disease: A pilot study of tofacitinib. BMJ Health Care Inform. 2021, 28, e100337. [Google Scholar] [CrossRef]
| Demographic | Patients on SSRIs/SNRIs (%) | Patients Not on SSRIs/SNRIs (%) | |
|---|---|---|---|
| n = 832 | n = 8211 | ||
| Gender | |||
| Female | 504 (60.6) | 3646 (44.4) | |
| Male | 328 (39.4) | 4565 (55.6) | |
| Age Group | |||
| >81 | 248 (29.8) | 1623 (19.8) | |
| 71–80 | 203 (24.4) | 1489 (18.1) | |
| 61–70 | 181 (21.8) | 1683 (20.5) | |
| 51–60 | 93 (11.2) | 1458 (17.8) | |
| 41–50 | 59 (7.1) | 799 (9.7) | |
| 31–40 | 35 (4.2) | 663 (8.1) | |
| 18–30 | 13 (1.6) | 455 (5.5) | |
| <18 | 0 (0.0) | 41 (0.5) | |
| Primary Race | |||
| White or Caucasian | 409 (49.2) | 2151 (26.2) | |
| Hispanic or Latino | 277 (33.3) | 4196 (51.1) | |
| Black or African American | 43 (5.2) | 493 (6.0) | |
| Asian | 28 (3.4) | 476 (5.8) | |
| Native Hawaiian or Other Pacific Islander | 2 (0.2) | 60 (0.7) | |
| American Indian or Alaskan Native | 0 (0.0.) | 5 (0.1) | |
| Other | 61 (7.3) | 683 (8.3) | |
| Unknown | 12 (1.4) | 147 (1.8) | |
| Facility Name | |||
| Providence Holy Cross Med Center | 2277 (27.7) | 186 (22.4) | |
| Providence Little Company of Mary (San Pedro) | 450 (5.5) | 41 (4.9) | |
| Providence Little Company of Mary (Torrance) | 2262 (27.5) | 137 (16.5) | |
| Providence Saint John’s Health Center | 607 (7.4) | 103 (12.4) | |
| Providence St. Joseph’s Medical Center (Burbank) | 1622 (19.8) | 209 (25.1) | |
| Providence Tarzana Medical Center | 993 (12.1) | 156 (18.8) | |
| Variables | Crude ORs (95% CI) | p-Value | Adjusted ORs (95% CI) | p-Value |
|---|---|---|---|---|
| SSRI on admission: | ||||
| No | 1 | 1 | ||
| Yes | 1.09 (0.91, 1.30) | 0.353 | 0.96 (0.79, 1.16) | 0.687 |
| Sex | ||||
| Female | 1 | 1 | ||
| Male | 1.34 (1.20, 1.49) | <0.001 | 1.53 (1.36, 1.71) | <0.001 |
| Age category (years) a | 1.04 (1.03, 1.04) | <0.001 | 1.04 (1.04, 1.05) | <0.001 |
| Primary race: | ||||
| White or Caucasian | 1 | 1 | ||
| American Indian or Alaskan Native | 1.09 (0.06, 7.42) | 0.936 | 3.04 (0.15, 21.5) | 0.328 |
| Asian | 1.27 (1.00, 1.59) | 0.047 | 1.54 (1.20, 1.97) | <0.001 |
| Black or African American | 0.86 (0.67, 1.10) | 0.236 | 1.20 (0.91, 1.56) | 0.191 |
| Hispanic or Latino | 0.86 (0.76, 0.98) | 0.018 | 1.49 (1.28, 1.73) | <0.001 |
| Native Hawaiian or Other Pacific Islander | 0.56 (0.23, 1.15) | 0.148 | 0.98 (0.40, 2.08) | 0.968 |
| Other | 1.12 (0.91, 1.38) | 0.261 | 1.30 (1.05, 1.61) | 0.015 |
| Unknown | 1.52 (1.04, 2.18) | 0.026 | 2.04 (1.37, 2.99) | <0.001 |
| Facility Location Name: | ||||
| Providence Holy Cross Medical Center | 1 | 1 | ||
| Providence Little Company of Mary (San Pedro) | 0.80 (6.1, 1.04) | 0.107 | 0.87 (0.65, 1.14) | 0.318 |
| Providence Little Company of Mary (Torrance) | 0.95 (0.82, 1.11) | 0.516 | 0.95 (0.81, 1.12) | 0.570 |
| Providence Saint John’s Health Center | 1.68 (1.38, 2.04) | <0.001 | 1.58 (1.27, 1.95) | <0.001 |
| Providence St. Joseph Medical Center (Burbank) | 0.97 (8.3, 1.14) | 0.720 | 0.93 (0.78, 1.11) | 0.411 |
| Providence Tarzana Medical Center | 1.06 (0.88, 1.27) | 0.549 | 1.01 (0.83, 1.23) | 0.934 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rauchman, S.H.; Mendelson, S.G.; Rauchman, C.; Kasselman, L.J.; Pinkhasov, A.; Reiss, A.B. Ongoing Use of SSRIs Does Not Alter Outcome in Hospitalized COVID-19 Patients: A Retrospective Analysis. J. Clin. Med. 2022, 11, 70. https://doi.org/10.3390/jcm11010070
Rauchman SH, Mendelson SG, Rauchman C, Kasselman LJ, Pinkhasov A, Reiss AB. Ongoing Use of SSRIs Does Not Alter Outcome in Hospitalized COVID-19 Patients: A Retrospective Analysis. Journal of Clinical Medicine. 2022; 11(1):70. https://doi.org/10.3390/jcm11010070
Chicago/Turabian StyleRauchman, Steven H., Sherri G. Mendelson, Courtney Rauchman, Lora J. Kasselman, Aaron Pinkhasov, and Allison B. Reiss. 2022. "Ongoing Use of SSRIs Does Not Alter Outcome in Hospitalized COVID-19 Patients: A Retrospective Analysis" Journal of Clinical Medicine 11, no. 1: 70. https://doi.org/10.3390/jcm11010070
APA StyleRauchman, S. H., Mendelson, S. G., Rauchman, C., Kasselman, L. J., Pinkhasov, A., & Reiss, A. B. (2022). Ongoing Use of SSRIs Does Not Alter Outcome in Hospitalized COVID-19 Patients: A Retrospective Analysis. Journal of Clinical Medicine, 11(1), 70. https://doi.org/10.3390/jcm11010070








