Aspirin Use Is Not Associated with the Risk of Metachronous Gastric Cancer in Patients without Helicobacter pylori Infection
Abstract
1. Introduction
2. Materials and Methods
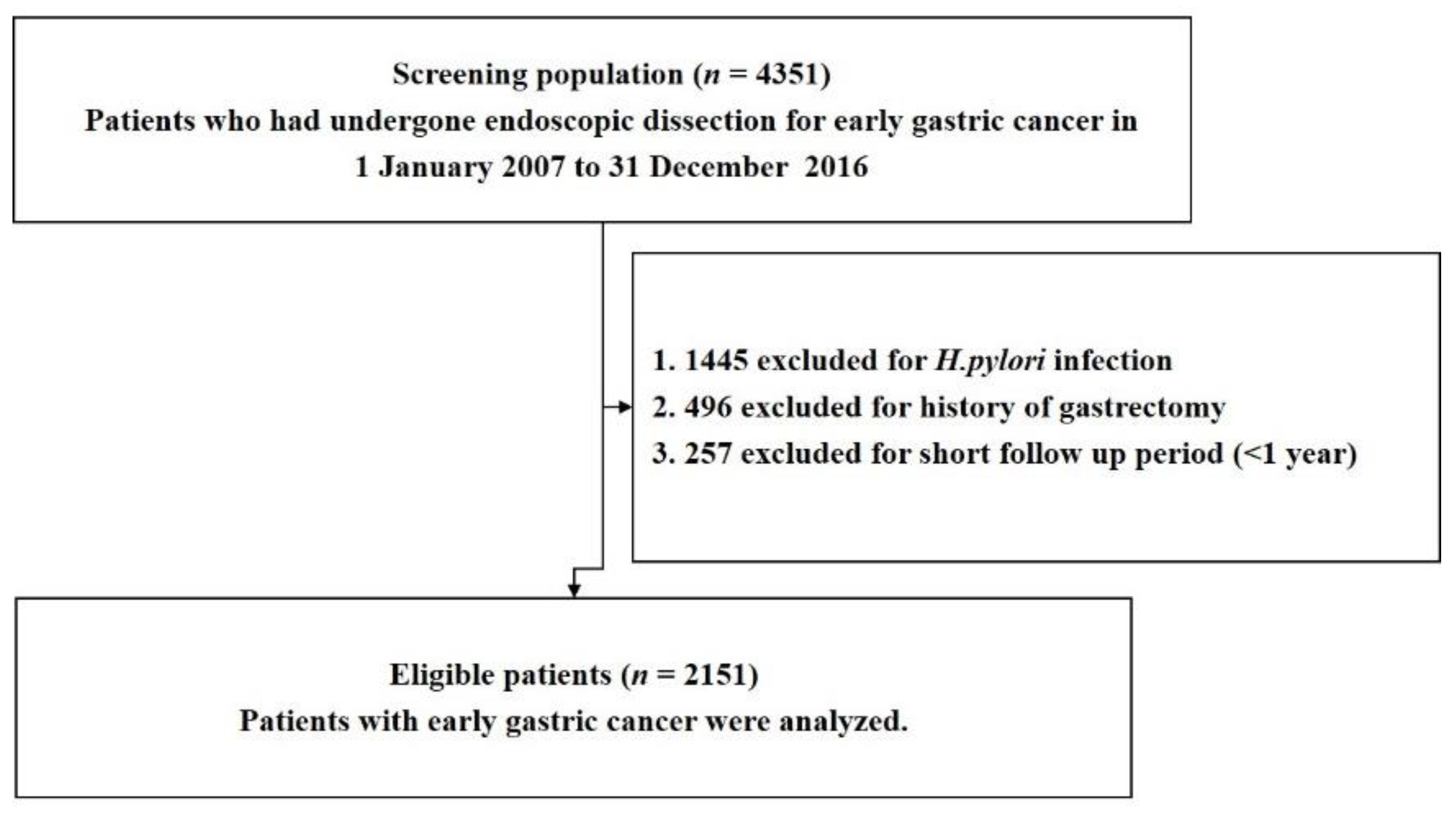
2.1. Study Design, Settings and Participants
2.2. Study Endpoints, Variables, Definitions
2.3. Statistical Analyses
3. Results
3.1. Baseline Characteristics of the Overall Cohort
3.2. Factors Associated with MGC
3.3. The Effect of the Duration of Aspirin Use on the Occurrence of MGC
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ishioka, M.; Yoshio, T.; Miyamoto, Y.; Namikawa, K.; Tokai, Y.; Yoshimizu, S.; Horiuchi, Y.; Ishiyama, A.; Hirasawa, T.; Tsuchida, T.; et al. Incidence of metachronous cancer after endoscopic submucosal dissection: A comparison between undifferentiated-type and differentiated-type early gastric cancer. Gastrointest. Endosc. 2021, 93, 557–564.e1. [Google Scholar] [CrossRef]
- Abe, S.; Oda, I.; Suzuki, H.; Nonaka, S.; Yoshinaga, S.; Nakajima, T.; Sekiguchi, M.; Mori, G.; Taniguchi, H.; Sekine, S.; et al. Long-term surveillance and treatment outcomes of metachronous gastric cancer occurring after curative endoscopic submucosal dissection. Endoscopy 2015, 47, 1113–1118. [Google Scholar] [CrossRef]
- Ikeda, Y.; Saku, M.; Kishihara, F.; Maehara, Y. Effective follow-up for recurrence or a second primary cancer in patients with early gastric cancer. Br. J. Surg. 2005, 92, 235–239. [Google Scholar] [CrossRef]
- Hosokawa, O.; Kaizaki, Y.; Watanabe, K.; Hattori, M.; Douden, K.; Hayashi, H.; Maeda, S. Endoscopic surveillance for gastric remnant cancer after early cancer surgery. Endoscopy 2002, 34, 469–473. [Google Scholar] [CrossRef] [PubMed]
- Choi, I.J.; Kook, M.C.; Kim, Y.I.; Cho, S.J.; Lee, J.Y.; Kim, C.G.; Park, B.; Nam, B.H. Helicobacter pylori therapy for the prevention of metachronous gastric cancer. N. Engl. J. Med. 2018, 378, 1085–1095. [Google Scholar] [CrossRef]
- Algra, A.M.; Rothwell, P.M. Effects of regular aspirin on long-term cancer incidence and metastasis: A systematic comparison of evidence from observational studies versus randomised trials. Lancet Oncol. 2012, 13, 518–527. [Google Scholar] [CrossRef]
- Henschke, U.K.; Luande, G.J.; Choppala, J.D. Aspirin for reducing cancer metastases? J. Natl. Med. Assoc. 1977, 69, 581–584. [Google Scholar] [PubMed]
- Coyle, C.; Cafferty, F.H.; Langley, R.E. Aspirin and colorectal cancer prevention and treatment: Is it for everyone? Curr. Colorectal Cancer Rep. 2016, 12, 27–34. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Elwood, P.C.; Morgan, G.; Pickering, J.E.; Galante, J.; Weightman, A.L.; Morris, D.; Kelson, M.; Dolwani, S. Aspirin in the treatment of cancer: Reductions in metastatic spread and in mortality: A systematic review and meta-analyses of published studies. PLoS ONE 2016, 11, e0152402. [Google Scholar] [CrossRef]
- Niikura, R.; Hirata, Y.; Hayakawa, Y.; Kawahara, T.; Yamada, A.; Koike, K. Effect of aspirin use on gastric cancer incidence and survival: A systematic review and meta-analysis. JGH Open 2020, 4, 117–125. [Google Scholar] [CrossRef]
- Win, T.T.; Aye, S.N.; Lau Chui Fern, J.; Ong Fei, C. Aspirin and reducing risk of gastric cancer: Systematic review and meta-analysis of the observational studies. J. Gastrointest. Liver Dis. 2020, 29, 191–198. [Google Scholar] [CrossRef]
- Skelin, M.; Javor, E.; Lucijanic, M. Aspirin use and the risk of cancer. JAMA Oncol. 2019, 5, 912–913. [Google Scholar] [CrossRef]
- Simon, T.G.; Ma, Y.; Ludvigsson, J.F.; Chong, D.Q.; Giovannucci, E.L.; Fuchs, C.S.; Meyerhardt, J.A.; Corey, K.E.; Chung, R.T.; Zhang, X.; et al. Association between aspirin use and risk of hepatocellular carcinoma. JAMA Oncol. 2018, 4, 1683–1690. [Google Scholar] [CrossRef]
- Patrignani, P.; Patrono, C. Aspirin, platelet inhibition and cancer prevention. Platelets 2018, 29, 779–785. [Google Scholar] [CrossRef]
- Watari, J.; Tomita, T.; Tozawa, K.; Oshima, T.; Fukui, H.; Miwa, H. Preventing metachronous gastric cancer after the endoscopic resection of gastric epithelial neoplasia: Roles of helicobacter pylori eradication and aspirin. Gut Liver 2020, 14, 281–290. [Google Scholar] [CrossRef]
- Spence, A.D.; Busby, J.; Johnston, B.T.; Baron, J.A.; Hughes, C.M.; Coleman, H.G.; Cardwell, C.R. Low-dose aspirin use does not increase survival in 2 independent population-based cohorts of patients with esophageal or gastric cancer. Gastroenterology 2018, 154, 849–860.e1. [Google Scholar] [CrossRef]
- Chow, S.C.; Wang, H.; Shao, J. Sample Size Calculations in Clinical Research, 2nd ed.; Chapman & Hall/CRC: Boca Raton, FL, USA, 2008; p. 177. [Google Scholar]
- Niikura, R.; Hayakawa, Y.; Hirata, Y.; Konishi, M.; Suzuki, N.; Ihara, S.; Yamada, A.; Ushiku, T.; Fujishiro, M.; Fukayama, M.; et al. Distinct chemopreventive effects of aspirin in diffuse and intestinal-type gastric cancer. Cancer Prev. Res. 2018, 11, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Haykal, T.; Barbarawi, M.; Zayed, Y.; Yelangi, A.; Dhillon, H.; Goranta, S.; Kheiri, B.; Chahine, A.; Samji, V.; Kerbage, J.; et al. Safety and efficacy of aspirin for primary prevention of cancer: A meta-analysis of randomized controlled trials. J. Cancer Res. Clin. Oncol. 2019, 145, 1795–1809. [Google Scholar] [CrossRef] [PubMed]
- Joharatnam-Hogan, N.; Cafferty, F.; Hubner, R.; Swinson, D.; Sothi, S.; Gupta, K.; Falk, S.; Patel, K.; Warner, N.; Kunene, V.; et al. Aspirin as an adjuvant treatment for cancer: Feasibility results from the add-aspirin randomised trial. Lancet Gastroenterol. Hepatol. 2019, 4, 854–862. [Google Scholar] [CrossRef]
- Jankowski, J.A.Z.; de Caestecker, J.; Love, S.B.; Reilly, G.; Watson, P.; Sanders, S.; Ang, Y.; Morris, D.; Bhandari, P.; Brooks, C.; et al. Esomeprazole and aspirin in Barrett’s oesophagus (AspECT): A randomised factorial trial. Lancet 2018, 392, 400–408. [Google Scholar] [CrossRef]
- Sankaranarayanan, R.; Kumar, D.R.; Patel, J.; Bhat, G.J. Do aspirin and flavonoids prevent cancer through a common mechanism involving hydroxybenzoic acids?-The metabolite hypothesis. Molecules 2020, 25, 2243. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Yan, J.; Fu, X.; Pan, Q.; Sun, D.; Xu, Y.; Wang, J.; Nie, L.; Tong, L.; Shen, A.; et al. Aspirin inhibits cancer metastasis and angiogenesis via targeting heparanase. Clin. Cancer Res. 2017, 23, 6267–6278. [Google Scholar] [CrossRef] [PubMed]
- Hua, H.; Zhang, H.; Kong, Q.; Wang, J.; Jiang, Y. Complex roles of the old drug aspirin in cancer chemoprevention and therapy. Med. Res. Rev. 2019, 39, 114–145. [Google Scholar] [CrossRef]
- Drew, D.A.; Cao, Y.; Chan, A.T. Aspirin and colorectal cancer: The promise of precision chemoprevention. Nat. Rev. Cancer 2016, 16, 173–186. [Google Scholar] [CrossRef] [PubMed]
- El-Zimaity, H.M. Accurate diagnosis of Helicobacter pylori with biopsy. Gastroenterol. Clin. N. Am. 2000, 29, 863–869. [Google Scholar] [CrossRef]

| Variables | Total Patients (n = 2151) | Aspirin Users (n = 236) | Aspirin Nonusers (n = 1915) |
|---|---|---|---|
| Age, years | 63.9 ± 9.5 | 68.5 ± 8.8 | 63.4 ± 9.5 |
| Male | 1698 (78.9) | 211 (89.4) | 1487 (77.7) |
| Tumor size, mm | 12.0 (8.0–19.0) | 12.0 (8.0–18.0) | 12.0 (8.0–19.0) |
| Tumor location | |||
| Lower | 1355 (63.0) | 150 (63.6) | 1205 (62.9) |
| Middle | 749 (34.8) | 82 (34.7) | 667 (34.8) |
| Upper | 47 (2.2) | 4 (1.7) | 43 (2.2) |
| Tumor macroscopic type | |||
| Elevated | 268 (12.5) | 33 (14.0) | 235 (12.3) |
| Flat | 310 (14.4) | 36 (15.3) | 274 (14.3) |
| Depressed | 1009 (46.9) | 123 (52.1) | 889 (46.3) |
| Mixed | 564 (26.2) | 44 (18.6) | 520 (27.2) |
| Depth of invasion | |||
| Mucosa (T1a) | 1961 (91.2) | 212 (89.8) | 1749 (91.3) |
| Submucosa (T1b) | 144 (6.7) | 16 (6.8) | 128 (6.7) |
| Histologic differentiation | |||
| Differentiated | 2146 (99.8) | 236 (100) | 1 910 (99.7) |
| Undifferentiated | 5 (0.2) | 0 | 5 (0.3) |
| Histologic heterogeneity | |||
| Absent | 2009 (93.4) | 219 (92.8) | 1790 (93.5) |
| Present | 142 (6.6) | 17 (7.2) | 125 (6.5) |
| Lymphovascular invasion | 44 (2.0) | 7 (3.0) | 37 (1.9) |
| Comorbidities | |||
| Hypertension | 378 (17.6) | 169 (71.6) | 209 (10.9) |
| Diabetes mellitus | 240 (11.2) | 92 (39.0) | 148 (7.7) |
| Myocardial infarction | 32 (1.5) | 16 (6.8) | 16 (0.8) |
| Heart failure | 66 (3.5) | 44 (32.8) | 22 (1.3) |
| Chronic kidney disease | 87 (4.0) | 29 (12.3) | 58 (3.0) |
| Liver cirrhosis | 56 (2.6) | 13 (5.5) | 43 (2.2) |
| Cerebrovascular | 206 (9.6) | 91 (38.6) | 115 (6.0) |
| Comorbidities (≥2) | 225 (12.0) | 81 (69.8) | 142 (8.2) |
| Metformin | 199 (9.3) | 52 (22.0) | 147 (7.7) |
| Factors | Univariable Analysis | Multivariable Analysis | ||
|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |
| Age (per year) | 1.04 (1.02–1.05) | <0.001 | 1.04 (1.02–1.05) | <0.001 |
| Male sex | 1.20 (0.81–1.80) | 0.36 | ||
| Tumor size, mm | 1.01 (0.99–1.03) | 0.077 | 1.01 (0.99–1.02) | 0.47 |
| Tumor location | ||||
| Lower | Reference | Reference | ||
| Middle | 1.32 (0.96–1.81) | 0.080 | 1.33 (0.96–1.82) | 0.082 |
| Upper | 1.06 (0.34–3.35) | 0.92 | 0.95 (0.30–3.00) | 0.93 |
| Tumor macroscopic type | ||||
| Elevated | Reference | Reference | ||
| Flat | 0.71 (0.40–1.25) | 0.24 | 0.76 (0.43–1.35) | 0.35 |
| Depressed | 0.64 (0.41–1.02) | 0.062 | 0.75 (0.47–1.19) | 0.23 |
| Mixed | 0.92 (0.57–1.48) | 0.72 | 0.99 (0.62–1.61) | 0.98 |
| Depth of invasion | ||||
| Mucosa (T1a) | Reference | |||
| Submucosa (T1b) | 0.94 (0.52–1.70) | 0.84 | ||
| Histologic differentiation | ||||
| Differentiated | Reference | |||
| Undifferentiated | 0.05 (0.00–44,708.22) | 0.67 | ||
| Histologic heterogeneity | ||||
| Absent | Reference | |||
| Present | 1.49 (0.89–2.50) | 0.13 | ||
| Lymphovascular invasion | 1.40 (0.52–3.78) | 0.51 | ||
| Comorbidities | ||||
| Hypertension | 0.86 (0.58–1.27) | 0.44 | ||
| Diabetes mellitus | 1.19 (0.77–1.85) | 0.43 | ||
| Myocadial infarction | 0.68 (0.17–2.73) | 0.59 | ||
| Heart failure | 1.08 (0.51–2.31) | 0.84 | ||
| Chronic kidney disease | 0.97 (0.48–1.99) | 0.94 | ||
| Liver cirrhosis | 1.16 (0.51–2.63) | 0.72 | ||
| Factors | Univariable Analysis | |
|---|---|---|
| HR (95% CI) | p-Value | |
| Duration | ||
| Non-aspirin use | Reference | |
| >6 months | 1.15 (0.73–1.79) | 0.55 |
| >1 years | 1.22 (0.78–1.93) | 0.38 |
| >2 years | 1.26 (0.78–2.01) | 0.34 |
| >3 years | 1.16 (0.69–1.95) | 0.57 |
| >4 years | 1.15 (0.66–2.00) | 0.61 |
| >5 years | 1.01 (0.54–1.86) | 0.98 |
| Duration | ||
| Non-aspirin use | Reference | |
| ≤1 years | 0.64 (0.20–2.01) | 0.45 |
| 1–4 years | 1.35 (0.66–2.76) | 0.41 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.E.; Kim, T.J.; Lee, H.; Lee, Y.C.; Chung, H.H.; Min, Y.W.; Min, B.-H.; Lee, J.H.; Kim, J.J. Aspirin Use Is Not Associated with the Risk of Metachronous Gastric Cancer in Patients without Helicobacter pylori Infection. J. Clin. Med. 2022, 11, 193. https://doi.org/10.3390/jcm11010193
Kim JE, Kim TJ, Lee H, Lee YC, Chung HH, Min YW, Min B-H, Lee JH, Kim JJ. Aspirin Use Is Not Associated with the Risk of Metachronous Gastric Cancer in Patients without Helicobacter pylori Infection. Journal of Clinical Medicine. 2022; 11(1):193. https://doi.org/10.3390/jcm11010193
Chicago/Turabian StyleKim, Ji Eun, Tae Jun Kim, Hyuk Lee, Yeong Chan Lee, Hwe Hoon Chung, Yang Won Min, Byung-Hoon Min, Jun Haeng Lee, and Jae J. Kim. 2022. "Aspirin Use Is Not Associated with the Risk of Metachronous Gastric Cancer in Patients without Helicobacter pylori Infection" Journal of Clinical Medicine 11, no. 1: 193. https://doi.org/10.3390/jcm11010193
APA StyleKim, J. E., Kim, T. J., Lee, H., Lee, Y. C., Chung, H. H., Min, Y. W., Min, B.-H., Lee, J. H., & Kim, J. J. (2022). Aspirin Use Is Not Associated with the Risk of Metachronous Gastric Cancer in Patients without Helicobacter pylori Infection. Journal of Clinical Medicine, 11(1), 193. https://doi.org/10.3390/jcm11010193







